Long-Term Macular Vascular Changes after Primary Rhegmatogenous Retinal Detachment Surgery Resolved with Different Tamponade or Different Surgical Techniques
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Setting of the Study
2.2. Participants
2.3. Surgical Procedure
2.4. Functional Parameter: Best-Corrected Visual Acuity
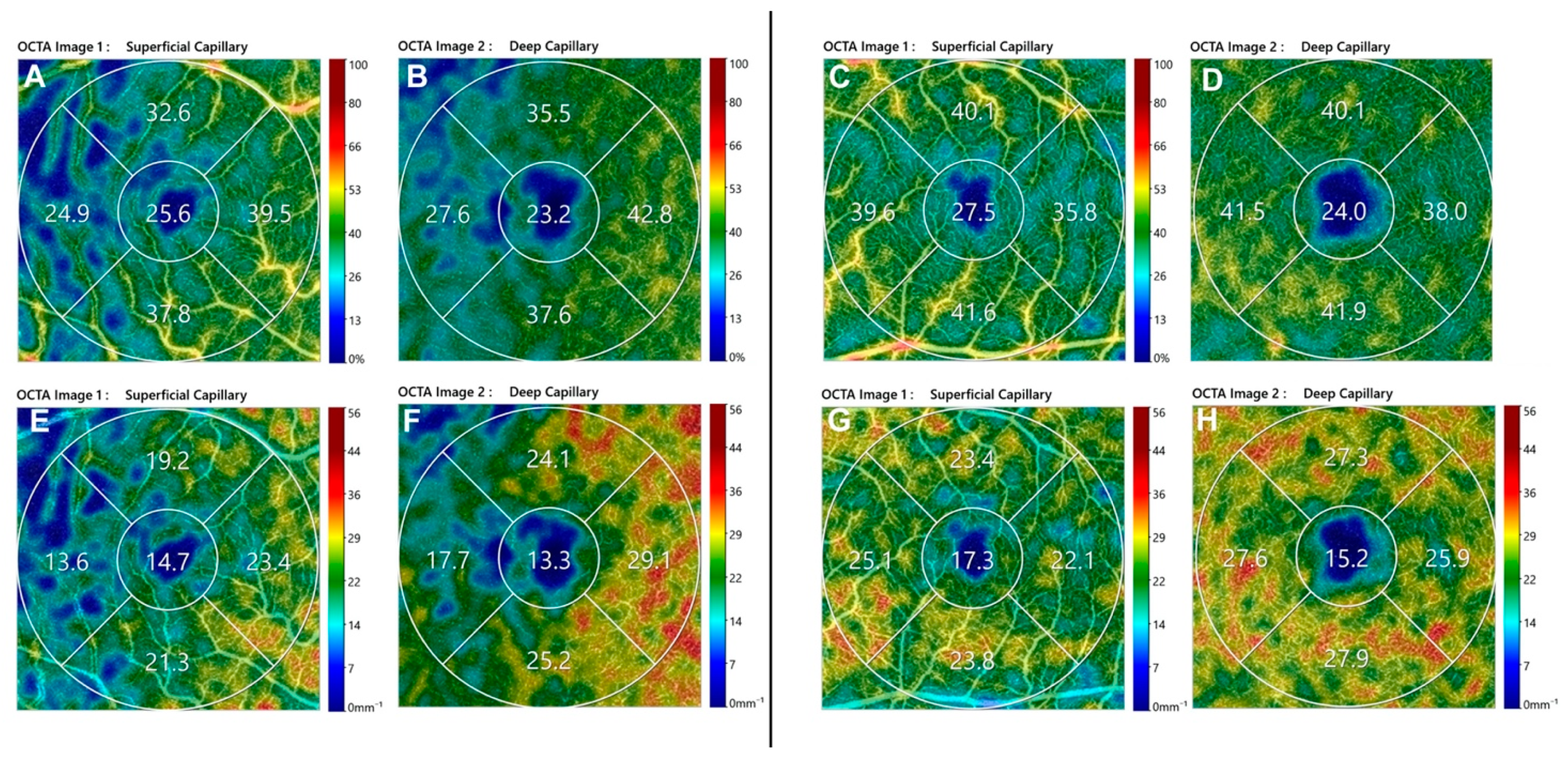
2.5. Perfusion Parameters: OCTA Image Acquisition and Analysis
2.6. Statistical Analysis
3. Results
3.1. Macula-ON Group
3.2. Macula-OFF Group
3.3. Surgery Subgroup Analysis
3.4. OCT Findings
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cheng, K.C.; Cheng, K.Y.; Cheng, K.H.; Chen, K.J.; Chen, C.H.; Wu, W.C. Using optical coherence tomography to evaluate macular changes after surgical management for rhegmatogenous retinal detachment. Kaohsiung J. Med. Sci. 2016, 32, 248–254. [Google Scholar] [CrossRef]
- Coppola, M.; Marchese, A.; Cicinelli, M.V.; Rabiolo, A.; Giuffrè, C.; Gomarasca, S.; Querques, G.; Bandello, F. Macular optical coherence tomography findings after vitreoretinal surgery for rhegmatogenous retinal detachment. Eur. J. Ophthalmol. 2020, 30, 805–816. [Google Scholar] [CrossRef]
- Gupta, R.R.; Iaboni, D.S.M.; Seamone, M.E.; Sarraf, D. Inner, outer, and full-thickness retinal folds after rhegmatogenous retinal detachment repair: A review. Surv. Ophthalmol. 2019, 64, 135–161. [Google Scholar] [CrossRef]
- Mastropasqua, R.; Toto, L.; Mattei, P.A.; di Nicola, M.; Zecca, I.A.L.; Carpineto, P.; di Antonio, L. Reproducibility and Repeatability of Foveal Avascular Zone Area Measurements Using Swept-Source Optical Coherence Tomography Angiography in Healthy Subjects. Eur. J. Ophthalmol. 2017, 27, 336–341. [Google Scholar] [CrossRef]
- de Carlo, T.E.; Romano, A.; Waheed, N.K.; Duker, J.S. A review of optical coherence tomography angiography (OCTA). Int. J. Retin. Vitr. 2015, 1, 5. [Google Scholar] [CrossRef]
- Spaide, R.F.; Klancnik, J.M., Jr.; Cooney, M.J. Retinal vascular layers imaged by fluorescein angiography and optical coherence tomography angiography. JAMA Ophthalmol. 2015, 133, 45–50. [Google Scholar] [CrossRef]
- Christou, E.E.; Stavrakas, P.; Batsos, G.; Christodoulou, E.; Stefaniotou, M. Association of OCT-A characteristics with postoperative visual acuity after rhegmatogenous retinal detachment surgery: A review of the literature. Int. Ophthalmol. 2021, 41, 2283–2292. [Google Scholar] [CrossRef]
- Cardillo Piccolino, F. Vascular changes in rhegmatogenous retinal detachment. Ophthalmologica 1983, 186, 17–24. [Google Scholar] [CrossRef]
- Satoh, Y. Retinal circulation in rhegmatogenous retinal detachment demonstrated by videofluorescence angiography and image analysis. I. The condition of retinal circulation before retinal detachment surgery. Nippon Ganka Gakkai Zasshi 1989, 93, 1002–1008. [Google Scholar]
- Sato, T.; Kanai, M.; Busch, C.; Wakabayashi, T. Foveal avascular zone area after macula-off rhegmatogenous retinal detachment repair: An optical coherence tomography angiography study. Graefes Arch. Clin. Exp. Ophthalmol. 2017, 255, 2071–2072. [Google Scholar] [CrossRef]
- Wang, H.; Xu, X.; Sun, X.; Ma, Y.; Sun, T. Macular perfusion changes assessed with optical coherence tomography angiography after vitrectomy for rhegmatogenous retinal detachment. Graefes Arch. Clin. Exp. Ophthalmol. 2019, 257, 733–740. [Google Scholar] [CrossRef]
- van Bussel, E.M.; van der Valk, R.; Bijlsma, W.R.; La Heij, E.C. Impact of duration of macula-off retinal detachment on visual outcome: A systematic review and meta-analysis of literature. Retina 2014, 34, 1917–1925. [Google Scholar] [CrossRef] [PubMed]
- Funatsu, R.; Terasaki, H.; Koriyama, C.; Yamashita, T.; Shiihara, H.; Sakamoto, T. Silicone oil versus gas tamponade for primary rhegmatogenous retinal detachment treated successfully with a propensity score analysis: Japan Retinal Detachment Registry. Br. J. Ophthalmol. 2022, 106, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Lei, B.; Jiang, R.; Huang, X.; Zhou, M.; Xu, G. Changes of macular vessel density and thickness in gas and silicone oil tamponades after vitrectomy for macula-on rhegmatogenous retinal detachment. BMC Ophthalmol. 2021, 21, 392. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.W.; Yoo, R.; Jo, Y.H.; Kim, H.C. Correlation of microvascular structures on optical coherence tomography angiography with visual acuity in retinal vein occlusion. Retina 2017, 37, 1700–1709. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.A.; Reeser, F.; O’Brien, W.J.; Fleischman, J.A. Prostacyclin and thromboxane A2 derivatives in rhegmatogenous subretinal fluid. Arch. Ophthalmol. 1983, 101, 463–464. [Google Scholar] [CrossRef] [PubMed]
- Quintyn, J.C.; Brasseur, G. Subretinal fluid in primary rhegmatogenous retinal detachment: Physiopathology and composition. Surv. Ophthalmol. 2004, 49, 96–108. [Google Scholar] [CrossRef]
- Ricker, L.J.; Kijlstra, A.; de Jager, W.; Liem, A.T.; Hendrikse, F.; La Heij, E.C. Chemokine levels in subretinal fluid obtained during scleral buckling surgery after rhegmatogenous retinal detachment. Investig. Ophthalmol. Vis. Sci. 2010, 51, 4143–4150. [Google Scholar] [CrossRef]
- Roldán-Pallarés, M.; Musa, A.S.; Hernández-Montero, J.; Bravo-Llatas, C. Preoperative duration of retinal detachment and preoperative central retinal artery hemodynamics: Repercussion on visual acuity. Graefes Arch. Clin. Exp. Ophthalmol. 2009, 247, 625–631. [Google Scholar] [CrossRef]
- Iandiev, I.; Uhlmann, S.; Pietsch, U.C.; Biedermann, B.; Reichenbach, A.; Wiedemann, P.; Bringmann, A. Endothelin receptors in the detached retina of the pig. Neurosci. Lett. 2005, 384, 72–75. [Google Scholar] [CrossRef]
- Çetinkaya Yaprak, A.; Küçük, M.F.; Yaprak, L.; Erol, M.K. Change in retinal and choroidal microvascular structures after rhegmatogenous retinal detachment surgery and effects on visual recovery. J. Fr. Ophtalmol. 2021, 44, 804–812. [Google Scholar] [CrossRef] [PubMed]
- McKay, K.M.; Vingopoulos, F.; Wang, J.C.; Papakostas, T.D.; Silverman, R.F.; Marmalidou, A.; Lains, I.; Eliott, D.; Vavvas, D.G.; Kim, L.A.; et al. Retinal Microvasculature Changes After Repair of Macula-off Retinal Detachment Assessed with Optical Coherence Tomography Angiography. Clin. Ophthalmol. 2020, 14, 1759–1767. [Google Scholar] [CrossRef]
- Chatziralli, I.; Theodossiadis, G.; Parikakis, E.; Chatzirallis, A.; Dimitriou, E.; Theodossiadis, P. Inner retinal layers’ alterations and microvasculature changes after vitrectomy for rhegmatogenous retinal detachment. Int. Ophthalmol. 2020, 40, 3349–3356. [Google Scholar] [CrossRef] [PubMed]
- Tsen, C.; Sheu, S.; Chen, S.; Wu, T. Imaging analysis with optical coherence tomography angiography after primary repair of macula-off rhegmatogenous retinal detachment. Graefes Arch. Clin. Exp. Ophthalmol. 2019, 257, 1847–1855. [Google Scholar] [CrossRef] [PubMed]
- D’Aloisio, R.; Gironi, M.; Verdina, T.; Vivarelli, C.; Leonelli, R.; Mariotti, C.; Kaleci, S.; Toto, L.; Mastropasqua, R. Early Structural and Vascular Changes after Within-24 Hours Vitrectomy for Recent Onset Rhegmatogenous Retinal Detachment Treatment: A Pilot Study Comparing Bisected Macula and Not Bisected Macula. J. Clin. Med. 2022, 11, 3498. [Google Scholar] [CrossRef]
- Hong, E.H.; Cho, H.; Kim, D.R.; Kang, M.H.; Shin, Y.U.; Seong, M. Changes in Retinal Vessel and Retinal Layer Thickness After Vitrectomy in Retinal Detachment via Swept-Source OCT Angiography. Investig. Ophthalmol. Vis. Sci. 2020, 61, 35. [Google Scholar] [CrossRef]
- Barca, F.; Bacherini, D.; Dragotto, F.; Tartaro, R.; Lenzetti, C.; Finocchio, L.; Virgili, G.; Caporossi, T.; Giansanti, F.; Savastano, A.; et al. OCT Angiography Findings in Macula-ON and Macula-OFF Rhegmatogenous Retinal Detachment: A Prospective Study. J. Clin. Med. 2020, 9, 3982. [Google Scholar] [CrossRef]
- Jiang, J.; Chen, S.; Jia, Y.D.; Li, R.; Zhou, J.X.; Li, R.M. Evaluation of macular vessel density changes after vitrectomy with silicone oil tamponade in patients with rhegmatogenous retinal detachment. Int. J. Ophthalmol. 2021, 14, 881–886. [Google Scholar] [CrossRef]
- Bonfiglio, V.; Ortisi, E.; Scollo, D.; Reibaldi, M.; Russo, A.; Pizzo, A.; Faro, G.; Macchi, I.; Fallico, M.; Toro, M.D.; et al. Vascular changes after vitrectomy for rhegmatogenous retinal detachment: Optical coherence tomography angiography study. Acta Ophthalmol. 2019, 98, e563–e569. [Google Scholar] [CrossRef]
- Hassanpoor, N.; Milani, A.E.; Kordestani, A.; Niyousha, M.R. Analysis of Retinal Layers’ Thickness and Vascular Density after Successful Scleral Buckle Surgery. J. Curr. Ophthalmol. 2021, 33, 304–309. [Google Scholar] [CrossRef]
- Nam, S.H.; Kim, K.; Kim, E.S.; Kim, D.G.; Yu, S.Y. Longitudinal Microvascular Changes on Optical Coherence Tomographic Angiography after Macula-Off Rhegmatogenous Retinal Detachment Repair Surgery. Ophthalmologica 2021, 244, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Resch, M.D.; Balogh, A.; Lászik, G.; Nagy, Z.Z.; Papp, A. Association between retinal vessel density and postoperative time after primary repair of rhegmatogenous retinal detachment. PLoS ONE 2021, 16, e0258126. [Google Scholar] [CrossRef]
- Borrelli, E.; Balasubramanian, S.; Triolo, G.; Barboni, P.; Sadda, S.R.; Sadun, A.A. Topographic Macular Microvascular Changes and Correlation With Visual Loss in Chronic Leber Hereditary Optic Neuropathy. Am. J. Ophthalmol. 2018, 192, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, E.; Toto, L.; Viggiano, P.; Evangelista, F.; Palmieri, M.; Mastropasqua, R. Widefield topographical analysis of the retinal perfusion and neuroretinal thickness in healthy eyes: A pilot study. Eye 2020, 34, 2264–2270. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Aggarwal, K.; Akella, M.; Agrawal, R.; Khandelwal, N.; Bansal, R.; Singh, R.; Gupta, V.; OCTA Study Group. Fractal dimension and optical coherence tomography angiography features of the central macula after repair of rhegmatogenous retinal detachments. Retina 2019, 39, 2167–2177. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Kim, E.S.; Yu, S.Y. Optical coherence tomography angiography analysis of foveal microvascular changes and inner retinal layer thinning in patients with diabetes. Br. J. Ophthalmol. 2018, 102, 1226–1231. [Google Scholar] [CrossRef] [PubMed]
- Menke, M.N.; Kowal, J.H.; Dufour, P.; Wolf-Schnurrbusch, U.E.; Ceklic, L.; Framme, C.; Wolf, S. Retinal layer measurements after successful macula-off retinal detachment repair using optical coherence tomography. Investig. Ophthalmol. Vis. Sci. 2014, 55, 6575–6579. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, J.Y.; Lee, S.Y.; Jeong, J.H.; Lee, E.K. Foveal Microvascular Structures in Eyes with Silicone Oil Tamponade for Rhegmatogenous Retinal Detachment: A Swept-source Optical Coherence Tomography Angiography Study. Sci. Rep. 2020, 10, 2555. [Google Scholar] [CrossRef]
- Williams, P.D.; Fuller, C.G.; Scott, I.U.; Fuller, D.G.; Flynn, H.W. Vision loss associated with the use and removal of intraocular silicone oil. Clin. Ophthalmol. 2008, 2, 955–959. [Google Scholar]
- Dogramaci, M.; Williams, K.; Lee, E.; Williamson, T.H. Foveal light exposure is increased at the time of removal of silicone oil with the potential for phototoxicity. Graefes Arch. Clin. Exp. Ophthalmol. 2013, 251, 35–39. [Google Scholar] [CrossRef]
- García-Ayuso, D.; Salinas-Navarro, M.; Agudo-Barriuso, M.; Alarcón-Martínez, L.; Vidal-Sanz, M.; Villegas-Pérez, M.P. Retinal ganglion cell axonal compression by retinal vessels in light-induced retinal degeneration. Mol. Vis. 2011, 17, 1716–1733. [Google Scholar]
- Grzybowski, A.; Pieczynski, J.; Ascaso, F.J. Neuronal complications of intravitreal silicone oil: An updated review. Acta Ophthalmol. 2014, 92, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Winter, M.; Eberhardt, W.; Scholz, C.; Reichenbach, A. Failure of potassium siphoning by Müller cells: A new hypothesis of perfluorocarbon liquid-induced retinopathy. Investig. Ophthalmol. Vis. Sci. 2000, 41, 256–261. [Google Scholar]
- Inoue, M.; Iriyama, A.; Kadonosono, K.; Tamaki, Y.; Yanagi, Y. Effects of perfluorocarbon liquids and silicone oil on human retinal pigment epithelial cells and retinal ganglion cells. Retina 2009, 29, 677–681. [Google Scholar] [CrossRef] [PubMed]
- Bambas, B.; Eckardt, C.; Vowinkel, E.; Kruse, H. Toxische Substanzen im Silikonöl nach intraokularer Injektion [Toxic substances with silicone oil after intraocular injections]. Ophthalmologe 1995, 92, 663–667. (In German) [Google Scholar] [PubMed]
- Wickham, L.; Asaria, R.H.; Alexander, R.; Luthert, P.; Charteris, D.G. Immunopathology of intraocular silicone oil: Enucleated eyes. Br. J. Ophthalmol. 2007, 91, 253–257. [Google Scholar] [CrossRef]
- Asaria, R.H.; Kon, C.H.; Bunce, C.; Sethi, C.S.; Limb, G.A.; Khaw, P.T.; Aylward, G.W.; Charteris, D.G. Silicone oil concentrates fibrogenic growth factors in the retro-oil fluid. Br. J. Ophthalmol. 2004, 88, 1439–1442. [Google Scholar] [CrossRef]


| Variables | |
|---|---|
| Patients, n | 82 |
| Age, mean yrs ± SD | 63 ± 12 |
| Male, n (%) | 50 (61) |
| Diabetes | 6 (7.3) |
| Hypertension | 38 (46.3) |
| Pre-operative factors | |
| Right eye: Left eye, % | 55: 45 |
| Macula OFF, n (%) | 49 (59.8) |
| Pseudophakic | 20 (24.4) |
| Intraoperative factors | |
| Type of surgery, n (%) | |
| PPV | 70 (85.4) |
| SB | 12 (14.6) |
| Type of surgery | |
| Gas | 65 (79.3) |
| SO | 17 (20.7) |
| Combined cataract surgery | 42 (51.2) |
| 360° laser | 12 (14.6) |
| Operation time, min (mean ± SD) | 91.27 ± 34.9 |
| Post-operative factors | |
| Duration of SO tamponade, months (mean ± SD) | 4.81 ± 1.8 |
| Hypotony (IOP < 5 mmHg) | 0 |
| Secondary glaucoma (IOP > 21 mmHg), n (%) | 8 (9.8) |
| Intraretinal cysts, % | 12 (14.6) |
| Follow-up period (months; m ± SD) | 13.1 ± 2 |
| BCVA-Post | Macula-ON Group | Macula-OFF Group | ||
|---|---|---|---|---|
| β ± SE | p-Value | β ± SE | p-Value | |
| Type of surgery | −0.009 ± 0.043 | 0.841 | −0.059 ± 0.084 | 0.487 |
| Tamponade | 0.273 ± 0.088 | 0.004 * | −0.007 ± 0.119 | 0.955 |
| Follow-up period | 0.002 ± 0.005 | 0.640 | −0.001 ± 0.007 | 0.878 |
| Macula-ON Group | Macula-OFF Group | |||||
|---|---|---|---|---|---|---|
| Study Eyes | Control Eyes | p-Value | Study Eyes | Control Eyes | p-Value | |
| CENTRAL SCP VD | 30.20 ± 5.70 | 32.22 ± 5.66 | 0.002 * | 28.31 ± 5.79 | 30.32 ± 5.78 | 0.002 * |
| INF SCP VD | 40.78 ± 4.51 | 41.80 ± 3.52 | 0.275 | 40.33 ± 3.64 | 42.24 ± 2.38 | 0.002 * |
| NAS SCP VD | 39.45 ± 4.48 | 40.90 ± 3.77 | 0.038 * | 38.51 ± 3.73 | 40.19 ± 3.18 | 0.010 * |
| SUP SCP VD | 40.32 ± 4.34 | 42.14 ± 2.78 | 0.030 * | 39.65 ± 3.31 | 41.42 ± 2.88 | 0.002 * |
| TEMP SCP VD | 38.42 ± 3.78 | 39.87 ± 3.35 | 0.056 | 38.06 ± 2.85 | 39.76 ± 2.56 | 0.000 * |
| CENTRAL DCP VD | 30.30 ± 8.90 | 30.67 ± 8.15 | 0.752 | 28.76 ± 7.44 | 29.05 ± 8.15 | 0.784 |
| INF DCP VD | 42.76 ± 3.41 | 42.61 ± 2.65 | 0.829 | 42.08 ± 3.08 | 42.82 ± 2.99 | 0.185 |
| NAS DCP VD | 42.74 ± 3.94 | 43.00 ± 2.36 | 0.698 | 41.54 ± 2.79 | 42.37 ± 2.93 | 0.117 |
| SUP DCP VD | 42.28 ± 4.06 | 43.03 ± 2.47 | 0.307 | 41.85 ± 2.83 | 42.40 ± 2.63 | 0.176 |
| TEMP DCP VD | 41.83 ± 3.69 | 41.96 ± 2.61 | 0.842 | 41.43 ± 2.97 | 41.58 ± 2.70 | 0.731 |
| CENTRAL SCP VLD | 16.17 ± 2.83 | 17.65 ± 2.86 | 0.001 * | 15.23 ± 2.87 | 16.53 ±2.96 | 0.001 * |
| INF SCP VLD | 21.01 ± 3.52 | 21.70 ± 3.69 | 0.221 | 21.11 ± 3.66 | 22.24 ± 3.35 | 0.004 * |
| NAS SCP VLD | 20.88 ± 3.33 | 21.83 ± 3.25 | 0.086 | 21.04 ± 3.29 | 21.92 ± 2.99 | 0.032 * |
| SUP SCP VLD | 20.55 ± 3.48 | 21.59 ± 4.04 | 0.090 | 20.73 ± 3.27 | 22.08 ± 3.65 | 0.001 * |
| TEMP SCP VLD | 20.18 ± 2.98 | 20.78 ± 3.55 | 0.259 | 20.46 ± 2.77 | 21.50 ± 2.85 | 0.002 * |
| CENTRAL DCP VLD | 16.54 ± 4.40 | 16.80 ± 4.58 | 0.739 | 16.10 ± 4.25 | 16.30 ± 4.72 | 0.766 |
| INF DCP VLD | 25.29 ± 3.89 | 24.66 ± 4.04 | 0.288 | 25.06 ± 3.95 | 25.28 ± 3.90 | 0.598 |
| NAS DCP VLD | 24.66 ± 3.64 | 25.06 ± 3.72 | 0.537 | 24.82 ± 3.60 | 25.01 ± 3.46 | 0.655 |
| SUP DCP VLD | 24.78 ± 3.74 | 25.10 ± 4.46 | 0.600 | 24.90 ± 3.65 | 25.41 ± 3.79 | 0.089 |
| TEMP DCP VLD | 24.30 ± 3.17 | 24.12 ± 3.62 | 0.747 | 24.34 ± 3.34 | 24.26 ± 3.40 | 0.796 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gironi, M.; D’Aloisio, R.; Verdina, T.; Vivarelli, C.; Leonelli, R.; Kaleci, S.; Toto, L.; Mastropasqua, R. Long-Term Macular Vascular Changes after Primary Rhegmatogenous Retinal Detachment Surgery Resolved with Different Tamponade or Different Surgical Techniques. Life 2022, 12, 1525. https://doi.org/10.3390/life12101525
Gironi M, D’Aloisio R, Verdina T, Vivarelli C, Leonelli R, Kaleci S, Toto L, Mastropasqua R. Long-Term Macular Vascular Changes after Primary Rhegmatogenous Retinal Detachment Surgery Resolved with Different Tamponade or Different Surgical Techniques. Life. 2022; 12(10):1525. https://doi.org/10.3390/life12101525
Chicago/Turabian StyleGironi, Matteo, Rossella D’Aloisio, Tommaso Verdina, Chiara Vivarelli, Riccardo Leonelli, Shaniko Kaleci, Lisa Toto, and Rodolfo Mastropasqua. 2022. "Long-Term Macular Vascular Changes after Primary Rhegmatogenous Retinal Detachment Surgery Resolved with Different Tamponade or Different Surgical Techniques" Life 12, no. 10: 1525. https://doi.org/10.3390/life12101525
APA StyleGironi, M., D’Aloisio, R., Verdina, T., Vivarelli, C., Leonelli, R., Kaleci, S., Toto, L., & Mastropasqua, R. (2022). Long-Term Macular Vascular Changes after Primary Rhegmatogenous Retinal Detachment Surgery Resolved with Different Tamponade or Different Surgical Techniques. Life, 12(10), 1525. https://doi.org/10.3390/life12101525











