1. Introduction
Infections can cause moderate to severe damages and are a burden to global economies and public health [
1]. These infections are increasing due to the emergence of higher virulence, such as the multidrug-resistant bacteria caused by multiple antibiotic treatments [
2,
3]. Some of the most problematic multidrug resistant organisms that are encountered currently include
Pseudomonas aeruginosa,
Acinetobacter baumannii, extended-spectrum beta-lactamases (ESBL)-producing
Escherichia coli and
Klebsiella pneumoniae, vancomycin-resistant enterococci, methicillin-resistant
Staphylococcus aureus and extensively drug-resistant
Mycobacterium tuberculosis [
2,
4]. In addition to increasing resistance to existing agents, there is a lack of new antibiotics in development [
5].
In order to find solutions for this important issue, more and more research is now made on medicinal plants trying to find new natural sources of antibiotics. Indeed, traditional phytotherapy has been the main source of remedy for various diseases in the past, and several studies have proved its efficacy. Researchers in this field are giving importance to the use of aromatic and medicinal plants as alternatives to antibiotics or as therapeutic complements. Because of their multiple modes of action, plant extracts could be as efficient as antibiotics, with lower risks of causing resistance or side effects. Plants contain several active compounds (secondary metabolites) belonging mainly to three classes: phenolic compound, terpenes and alkaloids [
6,
7]. In Morocco, 4200 plant species were identified, of which 600 are recognized for their aromatic and medicinal properties [
8].
Among aromatic and medicinal plants, lavender is famous for the quality of its essential oils and aqueous extracts that have long been used in traditional medicine, perfumes, cosmetics, hygiene products, food industry and pharmacy [
9,
10]. In Morocco, lavender is one of the most used plants by the population for the treatment of several diseases [
11,
12]. Researchers have been investigating its properties and have proven its antidiabetic, antirheumatic, anti-inflammatory, antioxidant, antibacterial, antifungal, antidepressant and antispasmodic activities. It is also efficient in the treatment of respiratory and digestive diseases [
13,
14,
15,
16,
17,
18,
19,
20]. However, lavender is usually investigated for the activity of its essential oils, regardless of the large use of its aqueous extracts in traditional medicine. Moreover, Moroccan people tend to use lavender in combination with other plants for better results [
12,
21,
22].
L. pedunculata is one of the lavender species widely used in traditional Moroccan medicine to treat several diseases [
12,
21]. However, few research studies have been performed to investigate and prove its efficacy. For these reasons, the aim of this study is to investigate the antibacterial activity of the aqueous extract of Moroccan
L. pedunculata towards several microbial strains. Moreover, its combinations with
S. rosmarinus,
S. lavandulifolia and
O. compactum from Morocco, known for their antibacterial activities [
23,
24,
25], will also be tested. In addition, the chemical composition of the aqueous extracts of all the chosen plants will be determined using UHPLC-MS.
2. Materials and Methods
2.1. Chemicals and Reagents
The used standards are luteolin, herniarin and myricetin (purchased from Sarsyntex, Merignac, France), apigenin (obtained from Carl Roth, Karlsruhe, Germany), coumarin (Behringer, Willich, Germany), cinnamic acid (Rhône-Poulenc, Paris, France), protocatechuic acid (Koch-Light Laboratories LTD, Bucks, UK), vanillic acid (Merck, Darmstadt, Germany), chlorogenic acid (Sigma-Aldrich, St. Louis, MO, USA), rosmarinic acid (Extrasynthèse, Genay, France) and gallic acid (Prolabo, Paris, France), as well as ferulic acid and caffeic acid purchased from Sigma (St. Louis, MO, USA). Formic acid (Carlo Erba ReagentsTM, Cornaredo, Italy) and methanol (Carlo Erba Reagents, Val-de-Reuil, France) were of HPLC grades. All other chemicals used were also of analytical grade.
2.2. Plant Material
L. pedunculata,
S. rosmarinus,
S. lavandulifolia and
O. compactum were collected from different regions in Morocco (
Table 1). The used organs were chosen according to the bibliographic data [
22,
23,
24,
26], and they were dried for thirteen days in the open air and in the shade.
Plant identification was carried out at the Scientific Institute of Rabat (Rabat, Morocco) by Dr. Hamid Khamar.
2.3. Aqueous Extraction
L. pedunculata flowering tops, S. rosmarinus leaves, S. lavandulifolia leaves and O. compactum leaves and flowers were each mixed and heated with distilled water (1:20; w/v) for one hour at 75 ± 2 °C using a hot plate. The temperature was monitored using an electronic laboratory thermometer. The mixtures were then filtered, and the obtained filtrates were dried in the oven at 70 °C until obtaining dry extract powders. The latter were put in closed flasks away from light and humidity until further use.
2.4. Determination of Total Polyphenol Content
Total polyphenol concentrations in the different plant aqueous extracts were determined by the method described by Zhang and his collaborators using the Folin-Ciocalteu reagent with some modifications [
27]. In a 96-well microplate, 25 μL of the extract at 0.5 mg/mL were introduced, then 125 μL of the Folin-Ciocalteu reagent (10%) were added, followed by 100 μL of sodium carbonate at 145 mg/mL. After 5 min of orbital stirring and 2 h incubation in the dark at 25 °C, the reading was made at 760 nm by a SPECTROstarNano spectrophotometer. The negative control was prepared according to the same protocol using 25 µL of distilled water instead of the extract.
A standard calibration curve was made from different concentrations of gallic acid. It was used for the calculation of polyphenol concentrations in the extracts. The total phenolic content is therefore expressed in milligrams equivalent of gallic acid per gram of the dry extract (mg GAE/gExt).
2.5. Determination of Total Tannin Content
Total tannin concentrations in the aqueous extracts were determined using the hide-powder method. It consists of the determination of polyphenol contents in the extracts after their contact with the hide-powder, which precipitates tannins [
28].
1 mL of the 0.5 mg/mL extract was stirred with 10 mg of the hide-powder for 1 h. The mixture was then centrifuged at 2500 rpm for 5 min. The polyphenol content in the supernatant was then determined using the method described above (
Section 2.4). The total tannin content in an extract corresponds, therefore, to the difference between the total polyphenol content in the extract and the polyphenol content after precipitating tannins with the hide-powder. This content is expressed in mg GAE/gExt.
2.6. UHPLC Analysis of Aqueous Extracts
Chromatographic analysis of the aqueous extracts was carried out on an AQUITY UPLC H-Class System (Waters Corporation, Manchester, UK) equipped with two independent pumps, an automatic injector, a controller, a diode array UV detector (DAD), a mass spectrometer with ESI ionization source and a quadrupole as an analyzer. The stationary phase is a reverse phase Waters® Acquity BEH C18 column (2.1 × 50 mm, 1.7 µm) connected to a 0.2 µm in-line filter. The mobile phase is composed of two solvents: (A) ultrapure water (Milli-Q® Integral 5, MerckTM, Darmstadt, Germany) + 0.1% formic acid; (B) methanol + 0.1% formic acid. The elution gradient established was 0–5% B (1 min), 5–20% B (0.5 min), 20% B (3.5 min), 20–100% B (4 min), rinsing of the column 100% B (2 min) and re-equilibration 100–0% B (0.5 min), 0% B (2.5 min). Methanol (70%) was required for washing the system.
The aqueous extracts were solubilized in a methanol/water mixture (1:1, v/v) to obtain a concentration of 1 mg/mL, and then they were filtered through 0.2 μm PTFE filter. For each analysis, 0.004 mL of the extract was injected. The temperature was set at 30 °C, and the flow rate was set at 0.3 mL/min.
This analysis was carried out on few standards chosen according to bibliographic data. They were injected under the same conditions as those of the extracts. These standards are cinnamic acid, luteolin, apigenin, myricetin, ferulic acid, protocatechuic acid, vanillic acid, chlorogenic acid, caffeic acid, rosmarinic acid, gallic acid, herniarin and coumarin.
Chemical compounds of the extracts were identified by matching their retention time, UV spectrum and molecular weight to those of the used standards.
2.7. Antibacterial Activity of Aqueous Extracts
2.7.1. Preparation of Bacterial Suspensions
The chosen microorganisms, some of which are resistant to antibiotics, are involved in various opportunistic or nosocomial infections. They were cultured from suspensions of the strains contained in a liquid Brain Heart Infusion Agar medium in tubes containing a sloping Mueller-Hinton Agar (MHA) culture medium (MHB OxoidTM, Basingstoke, UK; BactoTM Agar, Le Pont de Claix, France). The latter were incubated for 24 h at 37 °C, and then 10 mL of Ringer Cysteine (RC) liquid (MerckTM, Darmstadt, Germany) was added into the tubes. A good mixing is necessary in order to suspend the cultured microorganisms. A drop of each suspension was collected to be added in a dilution tube containing 10 mL of RC solution. The turbidity of the obtained suspension that was used for the test was therefore estimated at 0.5 McFarland. An amount of 1 mL of suspension from each dilution tube was withdrawn to fill the wells of the inoculum replicator plate.
2.7.2. Activity of Individual Extracts
Antibacterial activity of the plants aqueous extracts was evaluated using the agar dilution method [
29]. This method allows for determining the MIC of each extract towards 34 microorganisms in in vitro culture. The aqueous extracts were primarily dissolved in water/ethanol (7:3), and then they were mixed with MHA in Petri dishes. The final concentrations tested were 1.2, 0.6, 0.3, 0.15 and 0.075 mg/mL. The Petri dishes containing the MHA-extract mixture were inoculated with the microorganisms using an inoculum replicator, and they were incubated for 24 h at 37 °C. Activity was assessed visually by the presence or absence of culture. MIC values were recorded as the lowest concentrations of extracts that completely inhibit the growth of a specific microbe. A negative control was tested using the solvent. Three antibiotics were used as positive controls: gentamicin, vancomycin and amoxicillin. The studied strains are considered to be susceptible to the used antibiotics when MIC ≤ 4 mg/L, and they are considered to be resistant to gentamicin when MIC > 8 mg/L and resistant to vancomycin and amoxicillin when MIC > 16 mg/L [
29,
30].
2.7.3. Activity of Extracts in Mixtures
In order to investigate the antibacterial activity of extracts mixtures, the checkerboard assay was used. This method consists in studying all the possible combinations in the range of the chosen concentrations (1.2, 0.6, 0.3, 0.15 and 0.075 mg/mL).
The MIC of an extract mixture is also determined by the agar dilution procedure. It corresponds to the lowest concentrations of mixtures inhibiting the growth of a microbe.
In order to determine the effect of a combination, the FIC is calculated. It is an indicator of the activity of a plant mixture against microbial strains [
31]. It is calculated as follows for the combination of extracts A and B:
FIC value tells the effect of the combination:
FIC < 1: synergistic effect;
FIC = 1: commutative effect;
1 < FIC ≤ 2: indifferent effect;
2 < FIC: antagonistic effect.
2.8. Statistical Analysis
Data are presented as means ± standard deviations. Their statistical analysis was performed using GraphPad Prism version 5.00 for Windows, GraphPad Software, San Diego, California, USA. Multiple-group comparisons were analyzed using the one-way analysis of variance (ANOVA). Statistical significance was defined as p < 0.05.
4. Discussion
Lavender species are widely used for their antibacterial activity demonstrated in several studies [
32,
33]. However, their essential oils are usually studied regardless of the large use of their aqueous extracts in the traditional medicine [
34,
35,
36]. Besides, the use of essential oil is very limited in human and animal pharmacy due to the potential occurrence of side effects and toxicity [
37].
In the present study, we tested the antibacterial effect of
L. pedunculata aqueous extract that showed a strong antibacterial activity mainly against Gram-positive strains. Lopes and his collaborators investigated the antibacterial activity of
L. pedunculata aqueous extract from Portugal using the microdilution method. They found MIC values ranging from 0.10 to 0.45 mg/mL for
E. coli, and from 0.15 to 0.45 mg/mL for
S. aureus,
P. aeruginosa and S. Typhimurium. Some of these data are close to the results of our study, where
L. pedunculata was active against
S. aureus with MIC values ranging from 0.4 to 0.6 mg/mL and against
P. aeruginosa with MIC values ranging from 0.5 to 1.2 mg/mL. However, our extract was not active against
Salmonella sp. and
E. coli [
26].
In addition to
L. pedunculata, the aqueous extracts of three other plant species,
S. rosmarinus,
S. lavandulifolia and
O. compactum, were also tested in this study and were active against several bacterial strains, especially the Gram-positive ones. In a study carried out by Ramdan et al. (2018), the antibacterial activity of the hydroethanolic extracts of
S. rosmarinus and
O. compactum from Marrakech (Morocco) was evaluated by the broth microdilution method. The respective MIC values obtained for
S. rosmarinus and
O. compactum were 25 and 12.5 mg/mL for
Salmonella enterica, 50 and 25 mg/mL for
E. coli and
P. aeruginosa, and 12.5 mg/mL for both extracts against
S. aureus. These values are very high in comparison with our study [
38]. Giner and his collaborators conducted a study on the hydroalcoholic extract mixture of
S. lavandulifolia with
S. rosmarinus and
Thymus mastichina. They showed that it is active against
E. coli and
E. aerogenes with a MIC value of 12.8 mg/mL. This mixture was also active against
S. enterica and
S. aureus, with respective MIC values of 6.4 and 0.4 mg/mL. These results are very different from the ones obtained in this work, except for
S. aureus which is quite similar [
39].
The three species were not only tested individually, but also in association with
L. pedunculata, thus giving promising activities that were found for the first time. In fact, some plant mixtures, such as the mixture of
L. pedunculata with
S. lavandulifolia, were active against the majority of the Gram-negative strains, often multidrug-resistant, while the individual extracts were not active. Moreover, activities of the plant mixtures against the Gram-positive strains were boosted. One of the most problematic Gram-positive strains was
S. aureus. It is a human pathogen that possesses a high adaptability and tenacity, making it abundant in the environment. It is capable of colonizing various human organs, and it is a source of a variety of virulence factors. The multidrug-resistant form of this microorganism, especially the methicillin-resistant
S. aureus, is one of the major microorganisms responsible for bloodstream infections that cause high levels of mortality worldwide. Since
S. aureus has succeeded in developing resistance against practically all antibiotics, finding a new alternative is urgently needed, of which includes the importance of plant extracts such as the ones tested in this study and that showed strong activities [
40,
41].
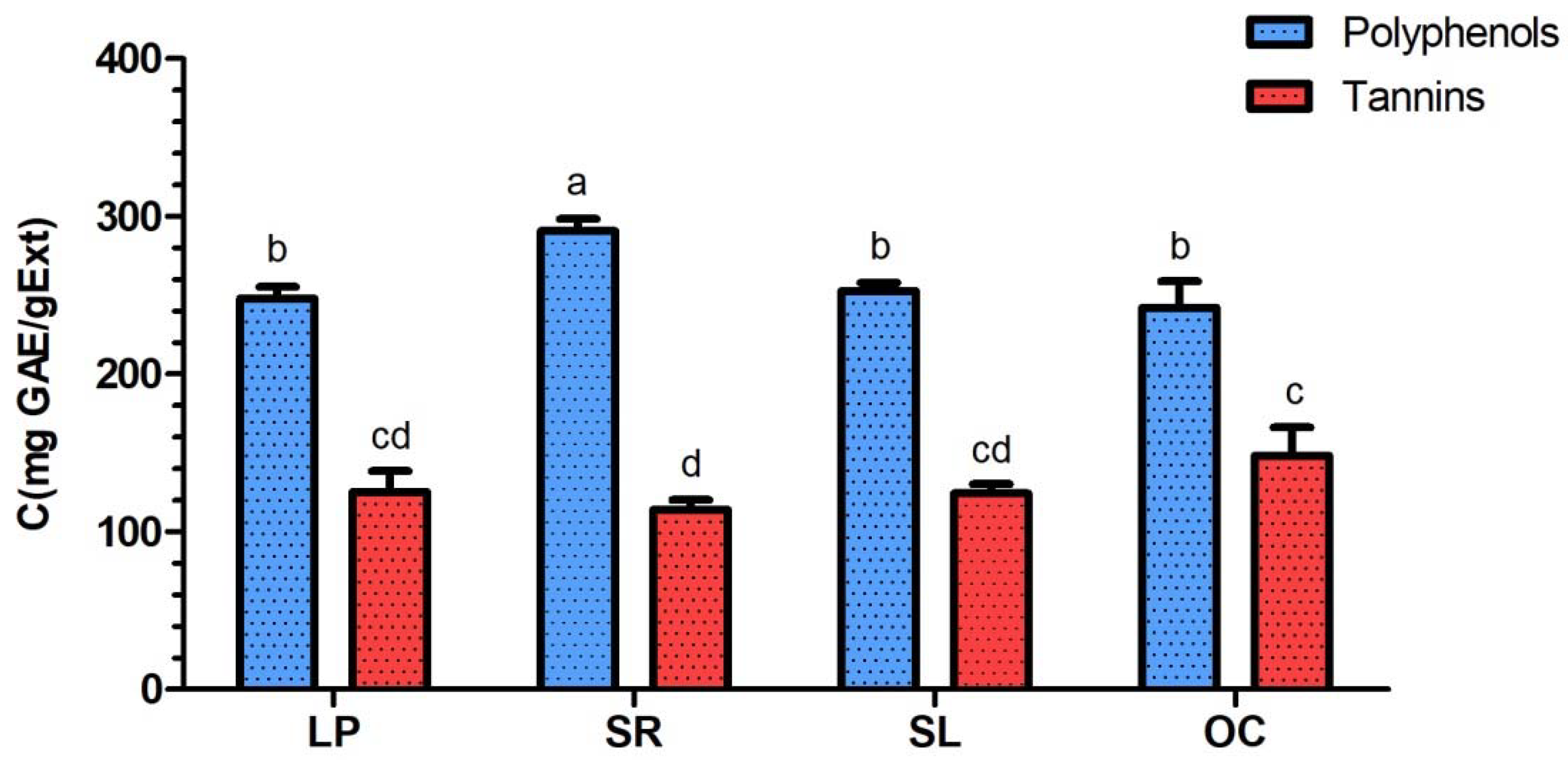
Polyphenol and tannin contents of
L. pedunculata,
S. rosmarinus,
S. lavandulifolia and
O. compactum aqueous extracts were determined during the present study using the Folin-Ciocalteu method. The highest polyphenol content corresponded to
S. rosmarinus extract (290.63 ± 7.70 mg GAE/gExt). This result was significantly high while compared to the ethanolic extract of the same plant species from Taiwan that has a polyphenol content of 161.07 ± 3.12 mg GAE/gExt [
42]. Moreover, ethanolic extract of
S. rosmarinus from different regions in Morocco also had lower polyphenol contents ranging from 74.15 to 146.63 mg GAE/gExt [
43]. As for
L. pedunculata,
S. lavandulifolia and
O. compactum aqueous extracts studied in the present work, their respective polyphenol contents are 248.03 ± 7.30, 252.67 ± 5.41 and 241.90 ± 16.96 mg GAE/gExt. The polyphenol content of
Salvia officinalis aqueous extract from Portugal was determined by Afonso et al. (2019), and it is similar to
S. lavandulifolia studied in this work (229.0 ± 44.0 mg GAE/gExt) [
44]. Furthermore, a study was conducted on the ethanolic extracts of
O. compactum aerial parts from Ouezzane and Taounate (Morocco), and their polyphenol contents were found to be lower than in our extract, with respective values of 117.60 ± 1.12 and 117.56 ± 2.74 mg GAE/gExt [
23,
45].
As for the tannin contents, the obtained values represent almost the half of the polyphenol contents, with respective values of 125.13 ± 13.24, 113.93 ± 6.15, 124.23 ± 6.08 and 148.20 ± 17.97 mg GAE/gExt for
L. pedunculata,
S. rosmarinus,
S. lavandulifolia and
O. compactum. Indeed, tannins are known for their antibacterial properties [
46,
47]; therefore, they might be the compounds that are responsible for the observed activities of the studied plant extracts.
UHPLC analysis of the plant extracts demonstrated their high content in rosmarinic acid. In fact, rosmarinic acid is found to be a good antibacterial agent [
48]. This compound has the ability of damaging the cell membrane [
49]. Other compounds were also detected in the studied extracts that are known for their antibacterial and antifungal activities such as coumarin, apigenin and caffeic acid [
49,
50,
51].
Lopes et al. (2018) analyzed the phenolic compounds of
L. pedunculata aqueous extract from 13 different natural populations in Portuguese regions using HPLC-DAD-ESI/MSn. They found that phenolic acids represent the major phenolic compounds present in these extracts. Salvianolic acid B and rosmarinic acid were present in large concentrations and caffeic acid in smaller ones. Concerning flavonoids, the main present compound was luteolin-7-O-glucuronide [
26]. In another study also conducted on
L. pedunculata from Portugal, different extracts were analyzed using HPLC/DAD. The obtained results showed that these extracts contain high concentrations of rosmarinic acid and smaller ones of luteolin. Moreover, apigenin was not quantified in the aqueous extracts, but it was present in the ethanolic and hydroethanolic ones [
52]. Some studies conducted on the methanolic and ethanolic extracts of
S. rosmarinus showed the presence of caffeic acid, rosmarinic acid, vanillic acid and ferulic acid [
24,
53]. Furthermore, in a review concerning polyphenolic compounds of
Salvia species, it was mentioned that
S. lavandulifolia contains rosmarinic acid, apigenin and luteolin [
54]. All of these studies are in concordance with the results we found in this research work.