A Comparative Study on the Viability of Normal and Cancerous Cells upon Irradiation with a Steady Beam of THz Rays
Abstract
:1. Introduction
2. Materials and Methods
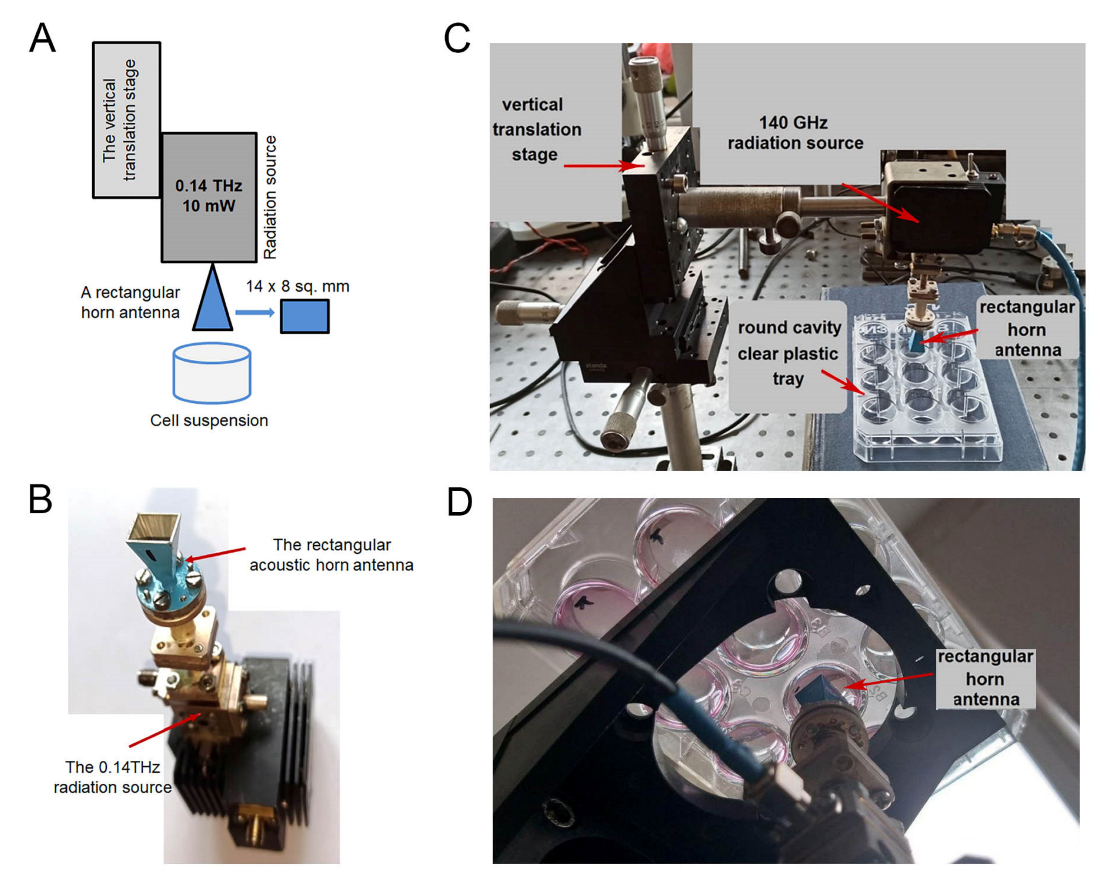
2.1. Irradiation Characteristics
2.2. Isolation of Human Peripheral Blood Mononuclear Cells
2.3. Cancer Cell Cultures
2.4. Cell Count and Metabolic Activity Assay
2.5. Fluorescent Microscopy and Cell Staining
3. Results
3.1. Mononuclear Cells Are Sensitive to THz Radiation
3.2. Effects of THz Irradiation on Cancer Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- You, B.; Chen, C.Y.; Yu, C.P.; Wang, P.H.; Lu, J.Y. Frequency-dependent skin penetration depth of terahertz radiation determined by water sorption-desorption. Opt. Express 2018, 26, 22709–22721. [Google Scholar] [CrossRef] [PubMed]
- Sizov, F. Detectors and Sources for THz and IR; Materials Research Forum LLC: Millersville, PA, USA, 2020; 325p. [Google Scholar] [CrossRef]
- Sitnikov, D.S.; Pronkin, A.A.; Ilina, I.V.; Revkova, V.A.; Konoplyannikov, M.A.; Kalsin, V.A.; Baklaushev, V.P. Numerical modelling and experimental verification of thermal effects in living cells exposed to high-power pulses of THz radiation. Sci. Rep. 2021, 11, 17916. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.; Ahn, J.; Mun, J.; Bae, S.; Jeong, Y.U.; Vinokurov, N.A.; Kim, P. In vivo analysis of THz wave irradiation induced acute inflammatory response in skin by laser-scanning confocal microscopy. Opt. Express 2014, 22, 11465–11475. [Google Scholar] [CrossRef] [PubMed]
- Hintzsche, H.; Jastrow, C.; Heinen, B.; Baaske, K.; Kleine-Ostmann, T.; Schwerdtfeger, M.; Shakfa, M.K.; Kärst, U.; Koch, M.; Schrader, T.; et al. Terahertz radiation at 0.380 THz and 2.520 THz does not lead to DNA damage in skin cells in vitro. Radiat. Res. 2013, 179, 38–45. [Google Scholar] [CrossRef] [PubMed]
- De Amicis, A.; Sanctis, S.D.; Cristofaro, S.D.; Franchini, V.; Lista, F.; Regalbuto, E.; Giovenale, E.; Gallerano, G.P.; Nenzi, P.; Bei, R.; et al. Biological effects of in vitro THz radiation exposure in human foetal fibroblasts. Mutat. Res. Genet. Toxicol. Environ. Mutagenes. 2015, 793, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Bogomazova, A.N.; Vassina, E.M.; Goryachkovskaya, T.N.; Popik, V.M.; Sokolov, A.S.; Kolchanov, N.A.; Lagarkova, M.A.; Kiselev, S.L.; Peltek, S.E. No DNA damage response and negligible genome-wide transcriptional changes in human embryonic stem cells exposed to terahertz radiation. Sci. Rep. 2015, 5, 7749. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williams, R.; Schofield, A.; Holder, G.; Downes, J.; Edgar, D.; Harrison, P.; Siggel-King, M.; Surman, M.; Dunning, D.; Hill, S.; et al. The influence of high intensity terahertz radiation on mammalian cell adhesion, proliferation and differentiation. Phys. Med. Biol. 2013, 58, 373–391. [Google Scholar] [CrossRef] [PubMed]
- Titova, L.V.; Ayesheshim, A.K.; Golubov, A.; Fogen, D.; Rodriguez-Juarez, R.; Hegmann, F.A.; Kovalchuk, O. Intense THz pulses cause H2AX phosphorylation and activate DNA damage response in human skin tissue. Biomed. Opt. Express 2013, 4, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Sitnikov, D.S.; Ilina, I.V.; Revkova, V.A.; Rodionov, S.A.; Gurova, S.A.; Shatalova, R.O.; Kovalev, A.V.; Ovchinnikov, A.V.; Chefonov, O.V.; Konoplyannikov, M.A.; et al. Effects of high intensity non-ionizing terahertz radiation on human skin fibroblasts. Biomed. Opt. Express 2021, 12, 7122–7138. [Google Scholar] [CrossRef]
- Korenstein-Ilan, A.; Barbul, A.; Hasin, P.; Eliran, A.; Gover, A.; Korenstein, R. Terahertz radiation increases genomic instability in human lymphocytes. Radiat. Res. 2008, 170, 224–234. [Google Scholar] [CrossRef] [PubMed]
- Franchini, V.; De Sanctis, S.; Marinaccio, J.; De Amicis, A.; Coluzzi, E.; Di Cristofaro, S.; Lista, F.; Regalbuto, E.; Doria, A.; Giovenale, E.; et al. Study of the effects of 0.15 terahertz radiation on genome integrity of adult fibroblasts. Environ. Mol. Mutagen. 2018, 59, 476–487. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, S.; Ueno, Y.; Hosoki, R.; Saito, T.; Idehara, T.; Yamaguchi, Y.; Otani, C.; Ogawa, Y.; Harata, M.; Hoshina, H. THz irradiation inhibits cell division by affecting actin dynamics. PLoS ONE. 2021, 16, e0248381. [Google Scholar] [CrossRef]
- IEEE Std C95.1™-2005; IEEE Standard for Safety Levels with Respect to Human Exposure to Radio Frequency Electromagnetic Fields, 3 kHz to 300 GHz. IEEE: New York, NY, USA, 2011.
- Farrell, P.J.; Allan, G.J.; Shanahan, F.; Vousden, K.H.; Crook, T. p53 is frequently mutated in Burkitt’s lymphoma cell lines. EMBO J. 1991, 10, 2879–2887. [Google Scholar] [CrossRef] [PubMed]
- van Bokhoven, A.; Varella-Garcia, M.; Korch, C.; Johannes, W.U.; Smith, E.E.; Miller, H.L.; Nordeen, S.K.; Miller, G.J.; Lucia, M.S. Molecular characterization of human prostate carcinoma cell lines. Prostate 2003, 57, 205–225. [Google Scholar] [CrossRef]
- Munshi, S.; Twining, R.C.; Dahl, R. Alamar blue reagent interacts with cell-culture media giving different fluorescence over time: Potential for false positives. J. Pharmacol. Toxicol. Methods 2014, 70, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Bock, J.; Fukuyo, Y.; Kang, S.; Phipps, M.L.; Alexandrov, L.B.; Rasmussen, K.O.; Bishop, A.R.; Rosen, E.D.; Martinez, J.S.; Chen, H.-T.; et al. Mammalian stem cells reprogramming in response to terahertz radiation. PLoS ONE. 2010, 5, e15806. [Google Scholar] [CrossRef] [PubMed]
- Shang, S.; Wu, X.; Zhang, Q.; Zhao, J.; Hu, E.; Wang, L.; Lu, X. 0.1 THz exposure affects primary hippocampus neuron gene expression via alternating transcription factor binding. Biomed. Opt. Express 2021, 12, 3729–3742. [Google Scholar] [CrossRef]
- Alexandrov, B.S.; Rasmussen, K.O.; Bishop, A.R.; Usheva, A.; Alexandrov, L.B.; Chong, S.; Dagon, Y.; Booshehri, L.G.; Mielke, C.H.; Phipps, M.L.; et al. Non-thermal effects of terahertz radiation on gene expression in mouse stem cells. Biomed. Opt. Express 2011, 2, 2679–2689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexandrov, B.S.; Phipps, M.L.; Alexandrov, L.B.; Booshehri, L.G.; Erat, A.; Zabolotny, J.; Mielke, C.H.; Chen, H.-T.; Rodriguez, G.; Rasmussen, K.Ø.; et al. Specificity and heterogeneity of terahertz radiation effect on gene expression in mouse mesenchymal stem cells. Sci. Rep. 2013, 3, 1184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perera, P.G.T.; Appadoo, D.R.T.; Cheeseman, S.; Wandiyanto, J.V.; Linklater, D.; Dekiwadia, C.; Truong, V.K.; Tobin, M.J.; Vongsvivut, J.; Bazaka, O.; et al. PC 12 Pheochromocytoma Cell Response to Super High Frequency Terahertz Radiation from Synchrotron Source. Cancers 2019, 11, 162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, E.; Wang, E.; Zhang, Q.; Li, P.; Zhang, P.; Wu, D.; Lu, X. Studying the influence of 3.1 THz irradiation on the endocytosis of neuronal cells. J. Opt. Soc. Am. B 2022, 39, 129–136. [Google Scholar] [CrossRef]
- Yamazaki, S.; Harata, M.; Idehara, T.; Konagaya, K.; Yokoyama, G.; Hoshina, H.; Ogawa, Y. Actin polymerization is activated by terahertz irradiation. Sci. Rep. 2018, 8, 9990. [Google Scholar] [CrossRef]
- Yamazaki, S.; Harata, M.; Ueno, Y.; Tsubouchi, M.; Konagaya, K.; Ogawa, Y.; Isoyama, G.; Otani, C.; Hoshina, H. Propagation of THz irradiation energy through aqueous layers: Demolition of actin filaments in living cells. Sci. Rep. 2020, 10, 9008. [Google Scholar] [CrossRef] [PubMed]
- Revet, I.; Feeney, L.; Bruguera, S.; Wilson, W.; Dong, T.K.; Oh, D.H.; Dankort, D.; Cleaver, J.E. Functional relevance of the histone gammaH2Ax in the response to DNA damaging agents. Proc. Natl. Acad. Sci. USA 2011, 108, 8663–8667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tokunaga, Y.; Tanaka, M.; Iida, H.; Kinoshita, M.; Tojima, Y.; Takeuchi, K.; Imashimizu, M. Nonthermal excitation effects mediated by subterahertz radiation on hydrogen exchange in ubiquitin. Biophys. J. 2021, 120, 2386–2393. [Google Scholar] [CrossRef]
- Nemova, E.A.; Dultseva, G.G.; Nikolaev, N.A.; Cherkasova, O.P. Effect of terahertz radiation on intermolecular interactions of albumin under aerobic and anaerobic conditions. J. Phys. Conf. Ser. 2021, 2067, 012015. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovalevska, L.; Golenkov, O.; Kulahina, Y.; Callender, T.; Sizov, F.; Kashuba, E. A Comparative Study on the Viability of Normal and Cancerous Cells upon Irradiation with a Steady Beam of THz Rays. Life 2022, 12, 376. https://doi.org/10.3390/life12030376
Kovalevska L, Golenkov O, Kulahina Y, Callender T, Sizov F, Kashuba E. A Comparative Study on the Viability of Normal and Cancerous Cells upon Irradiation with a Steady Beam of THz Rays. Life. 2022; 12(3):376. https://doi.org/10.3390/life12030376
Chicago/Turabian StyleKovalevska, Larysa, Olexandr Golenkov, Yelyzaveta Kulahina, Todd Callender, Fedir Sizov, and Elena Kashuba. 2022. "A Comparative Study on the Viability of Normal and Cancerous Cells upon Irradiation with a Steady Beam of THz Rays" Life 12, no. 3: 376. https://doi.org/10.3390/life12030376
APA StyleKovalevska, L., Golenkov, O., Kulahina, Y., Callender, T., Sizov, F., & Kashuba, E. (2022). A Comparative Study on the Viability of Normal and Cancerous Cells upon Irradiation with a Steady Beam of THz Rays. Life, 12(3), 376. https://doi.org/10.3390/life12030376





