Combination Therapy Comprising Paclitaxel and 5-Fluorouracil by Using Folic Acid Functionalized Bovine Milk Exosomes Improves the Therapeutic Efficacy against Breast Cancer
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Isolation of Exosomes
2.3. Fabrication of Drug-Loaded Exosomes and Smart Exosomes
2.4. Particle Size, Polydispersity Index, Zeta Potential, and Entrapment Efficiency
2.5. Scanning Electron Microscopy (SEM) for Morphology Analysis
2.6. In-Vitro Drug Release Study
2.7. X-ray Diffraction Analysis
2.8. Freeze Drying
2.9. Cell Culture and Conditions
2.10. In-Vitro Cytotoxicity
2.11. Qualitative Cell Uptake and Nuclear Co-Localization
2.12. Quantitative Cell Uptake Study
2.13. Intracellular Trafficking Assay
2.14. Annexin-V Apoptotic Assay
2.15. Wound Healing Assay
2.16. Cell Migration Assay
2.17. Statistical Analysis
3. Results and Discussion
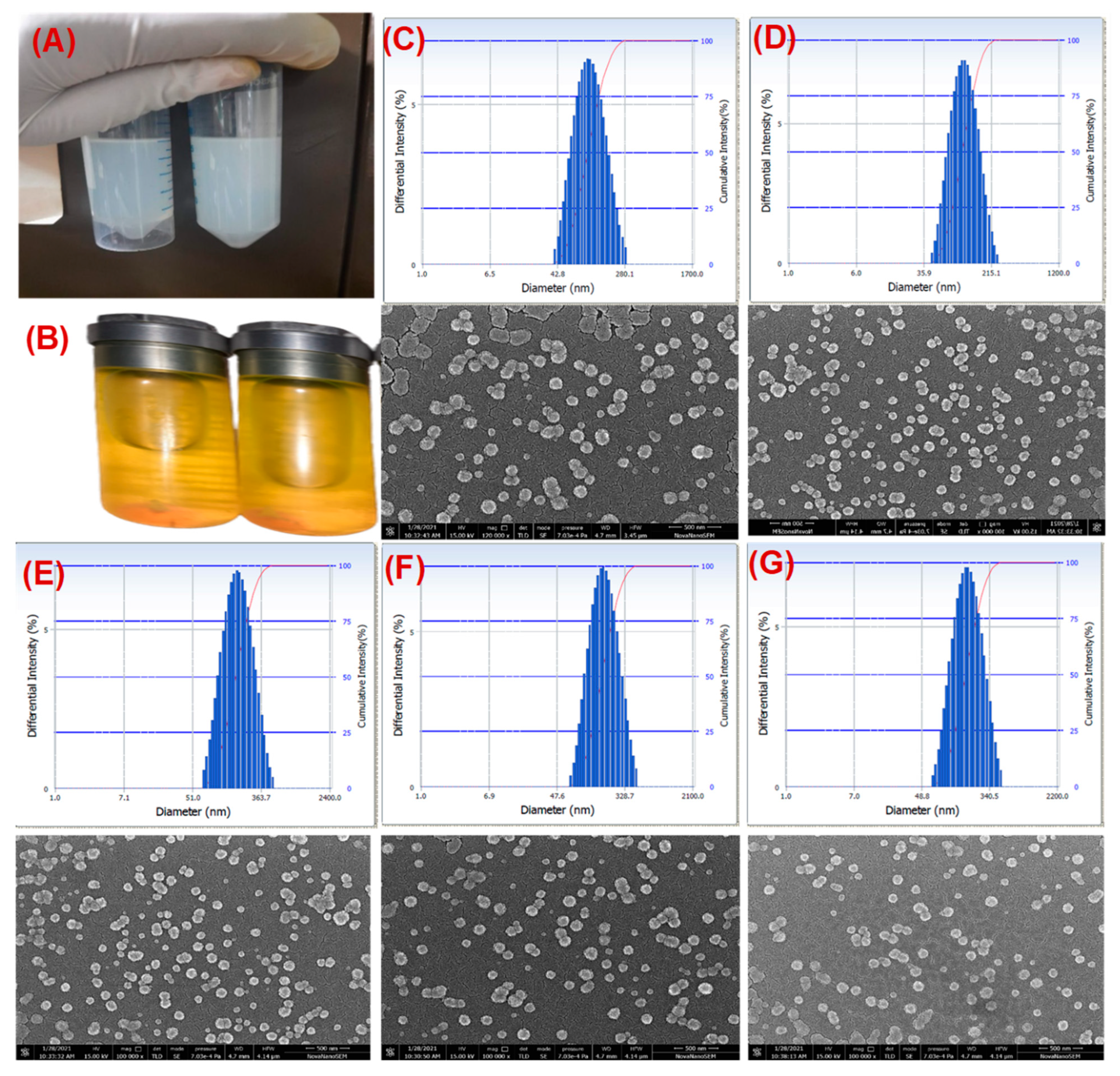
3.1. Isolation and Characterization of Exosomes and Drug-Loaded Exosomes
3.2. Powder X-ray Diffraction
3.3. Freeze Drying
3.4. In-Vitro Drug Release
3.5. Cytotoxicity Study
3.6. Cellular Uptake and Nuclear Colocalization
3.7. Quantitative Cell Uptake Assay
3.8. Intracellular Trafficking of Free Drugs and Exosomal Formulations
3.9. Annexin-V Apoptosis Assay
3.10. Wound Healing and Migration Assay
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kumar, D.N.; Chaudhuri, A.; Aqil, F.; Dehari, D.; Munagala, R.; Singh, S.; Gupta, R.C.; Agrawal, A.K. Exosomes as Emerging Drug Delivery and Diagnostic Modality for Breast Cancer: Recent Advances in Isolation and Application. Cancers 2022, 14, 1435. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, A.; Kumar, D.N.; Dehari, D.; Singh, S.; Kumar, P.; Bolla, P.K.; Kumar, D.; Agrawal, A.K. Emergence of Nanotechnology as a Powerful Cavalry against Triple-Negative Breast Cancer (TNBC). Pharmaceuticals 2022, 15, 542. [Google Scholar] [CrossRef] [PubMed]
- Negi, S.; Chaudhuri, A.; Kumar, D.N.; Dehari, D.; Singh, S.; Agrawal, A.K. Nanotherapeutics in autophagy: A paradigm shift in cancer treatment. Drug Deliv. Transl. Res. 2022, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Nunez, C.; Capelo, J.L.; Igrejas, G.; Alfonso, A.; Botana, L.M.; Lodeiro, C. An overview of the effective combination therapies for the treatment of breast cancer. Biomaterials 2016, 97, 34–50. [Google Scholar] [CrossRef]
- Lee, A.; Djamgoz, M.B.A. Triple negative breast cancer: Emerging therapeutic modalities and novel combination therapies. Cancer Treat. Rev. 2018, 62, 110–122. [Google Scholar] [CrossRef]
- Buzdar, A.U.; Valero, V.; Ibrahim, N.K.; Francis, D.; Broglio, K.R.; Theriault, R.L.; Pusztai, L.; Green, M.C.; Singletary, S.E.; Hunt, K.K.; et al. Neoadjuvant therapy with paclitaxel followed by 5-fluorouracil, epirubicin, and cyclophosphamide chemotherapy and concurrent trastuzumab in human epidermal growth factor receptor 2-positive operable breast cancer: An update of the initial randomized study population and data of additional patients treated with the same regimen. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2007, 13, 228–233. [Google Scholar] [CrossRef] [Green Version]
- Sparano, J.A.; Wang, M.; Martino, S.; Jones, V.; Perez, E.A.; Saphner, T.; Wolff, A.C.; Sledge, G.W., Jr.; Wood, W.C.; Davidson, N.E. Weekly paclitaxel in the adjuvant treatment of breast cancer. N. Engl. J. Med. 2008, 358, 1663–1671. [Google Scholar]
- Alves, R.C.; Fernandes, R.P.; Eloy, J.O.; Salgado, H.R.N.; Chorilli, M. Characteristics, Properties and Analytical Methods of Paclitaxel: A Review. Crit. Rev. Anal. Chem. 2018, 48, 110–118. [Google Scholar] [CrossRef]
- Miele, E.; Spinelli, G.P.; Miele, E.; Tomao, F.; Tomao, S. Albumin-bound formulation of paclitaxel (Abraxane® ABI-007) in the treatment of breast cancer. Int. J. Nanomed. 2009, 4, 99. [Google Scholar]
- Hamm, C.; Walter–Dilks, C. The Role of Albumin-Bound Paclitaxel (Abraxane) in the Treatment of Metastatic Breast Cancer; CED-SOS Advice Report. 2007. Available online: https://scholar.google.com/scholar?hl=en&as_sdt=0%2C5&q=10.%09Hamm%2C+C.%3B+Walter%E2%80%93Dilks%2C+C.+The+Role+of+Albumin-bound+Paclitaxel+%28Abraxane%29+in+the+Treatment+of+Metastatic+Breast+Cancer%3B+CED-SOS+Advice+Report&btnG (accessed on 14 July 2022).
- Fader, A.N.; Rose, P.G. Abraxane for the treatment of gynecologic cancer patients with severe hypersensitivity reactions to paclitaxel. Int. J. Gynecol. Cancer 2009, 19. [Google Scholar] [CrossRef]
- Longley, D.B.; Harkin, D.P.; Johnston, P.G. 5-fluorouracil: Mechanisms of action and clinical strategies. Nat. Rev. Cancer 2003, 3, 330–338. [Google Scholar] [CrossRef]
- Agrawal, A.K.; Das, M.; Jain, S. In situ gel systems as ‘smart’carriers for sustained ocular drug delivery. Expert Opin. Drug Deliv. 2012, 9, 383–402. [Google Scholar] [CrossRef]
- Cameron, D.; Gabra, H.; Leonard, R. Continuous 5-fluorouracil in the treatment of breast cancer. Br. J. Cancer 1994, 70, 120–124. [Google Scholar] [CrossRef] [Green Version]
- Thomas, S.A.; Grami, Z.; Mehta, S.; Patel, K. Adverse effects of 5-fluorouracil: Focus on rare side effects. Cancer Cell Microenviron. 2016, 3, e1266. [Google Scholar]
- Carrillo, E.; Navarro, S.A.; Ramírez, A.; García, M.Á.; Griñán-Lisón, C.; Perán, M.; Marchal, J.A. 5-Fluorouracil derivatives: A patent review (2012–2014). Expert Opin. Ther. Pat. 2015, 25, 1131–1144. [Google Scholar] [CrossRef]
- Walko, C.M.; Lindley, C. Capecitabine: A review. Clin. Ther. 2005, 27, 23–44. [Google Scholar] [CrossRef]
- Mesía, R.; Vázquez, S.; Grau, J.J.; García-Sáenz, J.A.; Lozano, A.; García, C.; Carles, J.; Irigoyen, A.; Mañós, M.; García-Paredes, B.; et al. A phase 2 open label, single-arm trial to evaluate the combination of cetuximab plus taxotere, cisplatin, and 5-flurouracil as an induction regimen in patients with unresectable squamous cell carcinoma of the head and neck. Int. J. Radiat. Oncol.*Biol.*Phys. 2016, 94, 289–296. [Google Scholar] [CrossRef]
- Herman, L.C.; Chen, L.; Garnett, A.; Feldman, L.E.; Smith, B.; Weichselbaum, R.R.; Spiotto, M.T. Comparison of carboplatin-paclitaxel to docetaxel-cisplatin-5-flurouracil induction chemotherapy followed by concurrent chemoradiation for locally advanced head and neck cancer. Oral Oncol. 2014, 50, 52–58. [Google Scholar] [CrossRef]
- Anjum, M.M.; Patel, K.K.; Dehari, D.; Pandey, N.; Tilak, R.; Agrawal, A.K.; Singh, S. Anacardic acid encapsulated solid lipid nanoparticles for Staphylococcus aureus biofilm therapy: Chitosan and DNase coating improves antimicrobial activity. Drug Deliv. Transl. Res. 2021, 11, 305–317. [Google Scholar] [CrossRef]
- Zanardi, E.; Bregni, G.; De Braud, F.; Di Cosimo, S. Better together: Targeted combination therapies in breast cancer. Semin. Oncol. 2015, 42, 887–895. [Google Scholar] [CrossRef]
- Jain, S.; Spandana, G.; Agrawal, A.K.; Kushwah, V.; Thanki, K. Enhanced antitumor efficacy and reduced toxicity of docetaxel loaded estradiol functionalized stealth polymeric nanoparticles. Mol. Pharm. 2015, 12, 3871–3884. [Google Scholar] [CrossRef]
- Munagala, R.; Aqil, F.; Jeyabalan, J.; Gupta, R.C. Bovine milk-derived exosomes for drug delivery. Cancer Lett. 2016, 371, 48–61. [Google Scholar] [CrossRef] [Green Version]
- Babaker, M.A.; Aljoud, F.A.; Alkhilaiwi, F.; Algarni, A.; Ahmed, A.; Khan, M.I.; Saadeldin, I.M.; Alzahrani, F.A. The Therapeutic Potential of Milk Extracellular Vesicles on Colorectal Cancer. Int. J. Mol. Sci. 2022, 23, 6812. [Google Scholar] [CrossRef]
- Lai, R.C.; Yeo, R.W.Y.; Tan, K.H.; Lim, S.K. Exosomes for drug delivery—a novel application for the mesenchymal stem cell. Biotechnol. Adv. 2013, 31, 543–551. [Google Scholar] [CrossRef]
- Bondhopadhyay, B.; Sisodiya, S.; Alzahrani, F.A.; Bakhrebah, M.A.; Chikara, A.; Kasherwal, V.; Khan, A.; Rani, J.; Dar, S.A.; Akhter, N. Exosomes: A forthcoming era of breast cancer therapeutics. Cancers 2021, 13, 4672. [Google Scholar] [CrossRef]
- Kandimalla, R.; Aqil, F.; Alhakeem, S.S.; Jeyabalan, J.; Tyagi, N.; Agrawal, A.; Yan, J.; Spencer, W.; Bondada, S.; Gupta, R.C. Targeted oral delivery of paclitaxel using colostrum-derived exosomes. Cancers 2021, 13, 3700. [Google Scholar] [CrossRef]
- Agrawal, A.K.; Aqil, F.; Jeyabalan, J.; Spencer, W.A.; Beck, J.; Gachuki, B.W.; Alhakeem, S.S.; Oben, K.; Munagala, R.; Bondada, S. Milk-derived exosomes for oral delivery of paclitaxel. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 1627–1636. [Google Scholar] [CrossRef]
- Aqil, F.; Kausar, H.; Agrawal, A.K.; Jeyabalan, J.; Kyakulaga, A.-H.; Munagala, R.; Gupta, R. Exosomal formulation enhances therapeutic response of celastrol against lung cancer. Exp. Mol. Pathol. 2016, 101, 12–21. [Google Scholar] [CrossRef]
- Aqil, F.; Munagala, R.; Jeyabalan, J.; Agrawal, A.K.; Kyakulaga, A.-H.; Wilcher, S.A.; Gupta, R.C. Milk exosomes-Natural nanoparticles for siRNA delivery. Cancer Lett. 2019, 449, 186–195. [Google Scholar] [CrossRef]
- Aqil, F.; Jeyabalan, J.; Agrawal, A.K.; Kyakulaga, A.-H.; Munagala, R.; Parker, L.; Gupta, R.C. Exosomal delivery of berry anthocyanidins for the management of ovarian cancer. Food Funct. 2017, 8, 4100–4107. [Google Scholar] [CrossRef]
- Jain, S.; Harde, H.; Indulkar, A.; Agrawal, A.K. Improved stability and immunological potential of tetanus toxoid containing surface engineered bilosomes following oral administration. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.; Gupta, P.; Khanna, A.; Sharma, R.; Chandrabanshi, H.; Gupta, N.; Patil, U.; Yadav, S. Development and characterization of in situ gel system for nasal insulin delivery. Die Pharm.-Int. J. Pharm. Sci. 2010, 65, 188–193. [Google Scholar]
- Yamada, T.; Inoshima, Y.; Matsuda, T.; Ishiguro, N. Comparison of methods for isolating exosomes from bovine milk. J. Vet. Med. Sci. 2012, 74, 1523–1525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, M.S.; Haney, M.J.; Zhao, Y.; Mahajan, V.; Deygen, I.; Klyachko, N.L.; Inskoe, E.; Piroyan, A.; Sokolsky, M.; Okolie, O. Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 655–664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, S.; Sharma, J.M.; Agrawal, A.K.; Mahajan, R.R. Surface stabilized efavirenz nanoparticles for oral bioavailability enhancement. J. Biomed. Nanotechnol. 2013, 9, 1862–1874. [Google Scholar] [CrossRef]
- Agrawal, A.K.; Urimi, D.; Harde, H.; Kushwah, V.; Jain, S. Folate appended chitosan nanoparticles augment the stability, bioavailability and efficacy of insulin in diabetic rats following oral administration. RSC Adv. 2015, 5, 105179–105193. [Google Scholar] [CrossRef]
- Agrawal, A.K.; Harde, H.; Thanki, K.; Jain, S. Improved stability and antidiabetic potential of insulin containing folic acid functionalized polymer stabilized multilayered liposomes following oral administration. Biomacromolecules 2014, 15, 350–360. [Google Scholar] [CrossRef]
- Jain, S.; Patil, S.R.; Swarnakar, N.K.; Agrawal, A.K. Oral delivery of doxorubicin using novel polyelectrolyte-stabilized liposomes (layersomes). Mol. Pharm. 2012, 9, 2626–2635. [Google Scholar] [CrossRef]
- Shim, M.K.; Park, J.; Yoon, H.Y.; Lee, S.; Um, W.; Kim, J.-H.; Kang, S.-W.; Seo, J.-W.; Hyun, S.-W.; Park, J.H. Carrier-free nanoparticles of cathepsin B-cleavable peptide-conjugated doxorubicin prodrug for cancer targeting therapy. J. Control. Release 2019, 294, 376–389. [Google Scholar] [CrossRef]
- Shalaby, K.S.; Soliman, M.E.; Casettari, L.; Bonacucina, G.; Cespi, M.; Palmieri, G.F.; Sammour, O.A.; El Shamy, A.A. Determination of factors controlling the particle size and entrapment efficiency of noscapine in PEG/PLA nanoparticles using artificial neural networks. Int. J. Nanomed. 2014, 9, 4953. [Google Scholar]
- Patel, K.K.; Gade, S.; Anjum, M.; Singh, S.K.; Maiti, P.; Agrawal, A.K.; Singh, S. Effect of penetration enhancers and amorphization on transdermal permeation flux of raloxifene-encapsulated solid lipid nanoparticles: An ex vivo study on human skin. Appl. Nanosci. 2019, 9, 1383–1394. [Google Scholar] [CrossRef]
- Urimi, D.; Agrawal, A.K.; Kushwah, V.; Jain, S. Polyglutamic acid functionalization of chitosan nanoparticles enhances the therapeutic efficacy of insulin following oral administration. AAPS PharmSciTech 2019, 20, 131. [Google Scholar] [CrossRef]
- Del Prado-Audelo, M.L.; Caballero-Florán, I.H.; Meza-Toledo, J.A.; Mendoza-Muñoz, N.; González-Torres, M.; Florán, B.; Cortés, H.; Leyva-Gómez, G. Formulations of curcumin nanoparticles for brain diseases. Biomolecules 2019, 9, 56. [Google Scholar] [CrossRef] [Green Version]
- Gaihre, B.; Khil, M.S.; Lee, D.R.; Kim, H.Y. Gelatin-coated magnetic iron oxide nanoparticles as carrier system: Drug loading and in vitro drug release study. Int. J. Pharm. 2009, 365, 180–189. [Google Scholar] [CrossRef]
- Patel, K.K.; Agrawal, A.K.; Anjum, M.; Tripathi, M.; Pandey, N.; Bhattacharya, S.; Tilak, R.; Singh, S. DNase-I functionalization of ciprofloxacin-loaded chitosan nanoparticles overcomes the biofilm-mediated resistance of Pseudomonas aeruginosa. Appl. Nanosci. 2020, 10, 563–575. [Google Scholar] [CrossRef]
- Harde, H.; Agrawal, A.K.; Jain, S. Trilateral‘3P’mechanics of stabilized layersomes technology for efficient oral immunization. J. Biomed. Nanotechnol. 2015, 11, 363–381. [Google Scholar] [CrossRef]
- Kushwah, V.; Jain, D.K.; Agrawal, A.K.; Jain, S. Improved antitumor efficacy and reduced toxicity of docetaxel using anacardic acid functionalized stealth liposomes. Colloids Surf. B Biointerfaces 2018, 172, 213–223. [Google Scholar] [CrossRef]
- Singh, S.; Kushwah, V.; Agrawal, A.K.; Jain, S. Insulin-and quercetin-loaded liquid crystalline nanoparticles: Implications on oral bioavailability, antidiabetic and antioxidant efficacy. Nanomedicine 2018, 13, 521–537. [Google Scholar] [CrossRef]
- Agrawal, A.K.; Kumar, K.; Swarnakar, N.K.; Kushwah, V.; Jain, S. “Liquid crystalline nanoparticles”: Rationally designed vehicle to improve stability and therapeutic efficacy of insulin following oral administration. Mol. Pharm. 2017, 14, 1874–1882. [Google Scholar] [CrossRef]
- Shilpi, D.; Kushwah, V.; Agrawal, A.K.; Jain, S. Improved stability and enhanced oral bioavailability of atorvastatin loaded stearic acid modified gelatin nanoparticles. Pharm. Res. 2017, 34, 1505–1516. [Google Scholar] [CrossRef]
- Bari, E.; Perteghella, S.; Catenacci, L.; Sorlini, M.; Croce, S.; Mantelli, M.; Avanzini, M.A.; Sorrenti, M.; Torre, M.L. Freeze-dried and GMP-compliant pharmaceuticals containing exosomes for acellular mesenchymal stromal cell immunomodulant therapy. Nanomedicine 2019, 14, 753–765. [Google Scholar] [CrossRef]
- Öğünç, Y.; Demirel, M.; Yakar, A.; İncesu, Z. Vincristine and ε-viniferine-loaded PLGA-b-PEG nanoparticles: Pharmaceutical characteristics, cellular uptake and cytotoxicity. J. Microencapsul. 2017, 34, 38–46. [Google Scholar] [CrossRef]
- Gan, C.W.; Feng, S.-S. Transferrin-conjugated nanoparticles of poly (lactide)-D-α-tocopheryl polyethylene glycol succinate diblock copolymer for targeted drug delivery across the blood–brain barrier. Biomaterials 2010, 31, 7748–7757. [Google Scholar] [CrossRef]
- Cid-Barrio, L.; Bouzas-Ramos, D.; Salinas-Castillo, A.; Ogra, Y.; Encinar, J.R.; Costa-Fernández, J.M. Quantitative assessment of cellular uptake and differential toxic effects of HgSe nanoparticles in human cells. J. Anal. At. Spectrom. 2020, 35, 1979–1988. [Google Scholar] [CrossRef]
- Zeng, X.; Zhang, Y.; Nyström, A.M. Endocytic uptake and intracellular trafficking of bis-MPA-based hyperbranched copolymer micelles in breast cancer cells. Biomacromolecules 2012, 13, 3814–3822. [Google Scholar] [CrossRef]
- Joglekar, A.V.; Dehari, D.; Anjum, M.; Dulla, N.; Chaudhuri, A.; Singh, S.; Agrawal, A.K. Therapeutic potential of venom peptides: Insights in the nanoparticle-mediated venom formulations. Future J. Pharm. Sci. 2022, 8, 34. [Google Scholar] [CrossRef]
- Kushwah, V.; Katiyar, S.S.; Dora, C.P.; Agrawal, A.K.; Lamprou, D.A.; Gupta, R.C.; Jain, S. Co-delivery of docetaxel and gemcitabine by anacardic acid modified self-assembled albumin nanoparticles for effective breast cancer management. Acta Biomater. 2018, 73, 424–436. [Google Scholar] [CrossRef] [Green Version]
- Karunanidhi, P.; Verma, N.; Kumar, D.N.; Agrawal, A.K.; Singh, S. Triphenylphosphonium functionalized Ficus religiosa L. extract loaded nanoparticles improve the mitochondrial function in oxidative stress induced diabetes. AAPS PharmSciTech 2021, 22, 158. [Google Scholar] [CrossRef]
- Kyakulaga, A.H.; Aqil, F.; Munagala, R.; Gupta, R.C. Withaferin A inhibits epithelial to mesenchymal transition in non-small cell lung cancer cells. Sci. Rep. 2018, 8, 15737. [Google Scholar] [CrossRef]
- Tay, C.Y.; Cai, P.; Setyawati, M.I.; Fang, W.; Tan, L.P.; Hong, C.H.; Chen, X.; Leong, D.T. Nanoparticles strengthen intracellular tension and retard cellular migration. Nano Lett. 2014, 14, 83–88. [Google Scholar] [CrossRef]
- Aqil, F.; Munagala, R.; Jeyabalan, J.; Agrawal, A.K.; Gupta, R. Exosomes for the enhanced tissue bioavailability and efficacy of curcumin. AAPS J. 2017, 19, 1691–1702. [Google Scholar] [CrossRef] [PubMed]
- Oosthuyzen, W.; Sime, N.E.; Ivy, J.R.; Turtle, E.J.; Street, J.M.; Pound, J.; Bath, L.E.; Webb, D.J.; Gregory, C.D.; Bailey, M.A. Quantification of human urinary exosomes by nanoparticle tracking analysis. J. Physiol. 2013, 591, 5833–5842. [Google Scholar] [CrossRef] [PubMed]
- Charoenviriyakul, C.; Takahashi, Y.; Nishikawa, M.; Takakura, Y. Preservation of exosomes at room temperature using lyophilization. Int. J. Pharm. 2018, 553, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Marshalek, J.P.; Sheeran, P.S.; Ingram, P.; Dayton, P.A.; Witte, R.S.; Matsunaga, T.O. Intracellular delivery and ultrasonic activation of folate receptor-targeted phase-change contrast agents in breast cancer cells in vitro. J. Control. Release 2016, 243, 69–77. [Google Scholar] [CrossRef] [Green Version]









| Samples | Size (nm) * | PDI * | Zeta Potential (mV) * | Entrapment Efficiency (EE%) * | Drug Loading * |
|---|---|---|---|---|---|
| Exosomes | 83.5 ± 2.6 | 0.145 ± 0.08 | −23.7 ± 0.9 | - | - |
| Exo-PAC | 86.6 ± 2.6 | 0.156 ± 0.019 | −25 ± 0.5 | 85 ± 3.8 | 28.3 ± 3.9 |
| Exo-5-FU | 88.2 ± 2.8 | 0.200 ± 0.012 | −28.2 ± 1.3 | 78 ± 2.2 | 19 ± 4.6 |
| FA-Exo-PAC | 95.3 ± 3.4 | 0.158 ± 0.016 | −27.09 ± 1.6 | 82 ± 5.6 | 26.2 ± 2.6 |
| FA-Exo-5-FU | 97.3 ± 3.4 | 0.186 ± 0.022 | −26.2 ± 1.5 | 75 ± 2.5 | 17 ± 2.9 |
| Freeze-Drying | Before Freeze-Drying | After Freeze-Drying (5% Trehalose) | ||||
|---|---|---|---|---|---|---|
| Size (nm) | PDI | Drug Content | Size (nm) | PDI | Drug Content | |
| Exo | 83.5 ± 2.6 | 0.145 ± 0.08 | - | 84.2 ± 3.1 | 0.148 ± 0.06 | - |
| Exo-PAC | 86.6 ± 2.6 | 0.156 ± 0.019 | 28.3 ± 3.9 | 87.2 ± 2.2 | 0.161 ± 0.015 | 28.2 ± 2.5 |
| Exo-5-FU | 88.2 ± 2.8 | 0.200 ± 0.012 | 19 ± 4.6 | 88.9 ± 2.6 | 0.198 ± 0.018 | 18.9 ± 3.5 |
| FA-Exo-PAC | 95.3 ± 3.4 | 0.158 ± 0.06 | 26.2 ± 2.6 | 94.3 ± 2.4 | 0.158 ± 0.016 | 26.1 ± 2.8 |
| FA-Exo-5-FU | 97 ± 3.4 | 0.186 ± 0.022 | 17 ± 2.9 | 96.9 ± 1.8 | 0.186 ± 0.021 | 16.9 ± 2.6 |
| Group | IC50 (µM) | |||||
|---|---|---|---|---|---|---|
| 24 h | 48 h | 72 h | ||||
| MCF-7 | MDA-MB-231 | MCF-7 | MDA-MB-231 | MCF-7 | MDA-MB-231 | |
| Exo * | 15.1 ± 2.2 | 18.3 ± 1.8 | 13 ± 1.6 | 15.8 ± 2.1 | 15.2 ± 1.4 | 17.8 ± 2.2 |
| Free PAC | 0.8 ± 0.2 | 1.1 ± 0.1 | 0.6 ± 0.5 | 0.9 ± 0.2 | 0.78 ± 0.2 | 1.2 ± 0.1 |
| Free 5-FU | 3.8 ± 0.2 | 15.6 ± 1.9 | 2.9 ± 0.5 | 14.6 ± 0.8 | 3.6 ± 0.3 | 14.8 ± 2.2 |
| Free-PAC/5-FU | 0.6 ± 0.3 | 0.9 ± 0.4 | 0.52 ± 0.11 | 0.78 ± 0.1 | 0.69 ± 0.21 | 0.96 ± 0.3 |
| Exo-PAC | 0.63 ± 0.28 | 0.88 ± 0.2 | 0.54 ± 0.1 | 0.76 ± 0.21 | 0.67 ± 0.1 | 0.9 ± 0.22 |
| Exo-5-FU | 3.0 ± 0.21 | 12.2 ± 0.23 | 2.2 ± 0.4 | 10.9 ± 0.54 | 2.9 ± 0.31 | 11.8 ± 0.2 |
| Exo-PAC/5-FU | 0.4 ± 0.2 | 0.6 ± 0.12 | 0.28 ± 0.12 | 0.52 ± 0.3 | 0.48 ± 0.2 | 0.78 ± 0.23 |
| FA-EXO-PAC/5-FU | 0.2 ± 0.1 | 0.50 ± 0.22 | 0.11 ± 0.08 | 0.21 ± 0.22 | 0.18 ± 0.09 | 0.48 ± 0.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, D.N.; Chaudhuri, A.; Dehari, D.; Shekher, A.; Gupta, S.C.; Majumdar, S.; Krishnamurthy, S.; Singh, S.; Kumar, D.; Agrawal, A.K. Combination Therapy Comprising Paclitaxel and 5-Fluorouracil by Using Folic Acid Functionalized Bovine Milk Exosomes Improves the Therapeutic Efficacy against Breast Cancer. Life 2022, 12, 1143. https://doi.org/10.3390/life12081143
Kumar DN, Chaudhuri A, Dehari D, Shekher A, Gupta SC, Majumdar S, Krishnamurthy S, Singh S, Kumar D, Agrawal AK. Combination Therapy Comprising Paclitaxel and 5-Fluorouracil by Using Folic Acid Functionalized Bovine Milk Exosomes Improves the Therapeutic Efficacy against Breast Cancer. Life. 2022; 12(8):1143. https://doi.org/10.3390/life12081143
Chicago/Turabian StyleKumar, Dulla Naveen, Aiswarya Chaudhuri, Deepa Dehari, Anusmita Shekher, Subash C. Gupta, Shreyasi Majumdar, Sairam Krishnamurthy, Sanjay Singh, Dinesh Kumar, and Ashish Kumar Agrawal. 2022. "Combination Therapy Comprising Paclitaxel and 5-Fluorouracil by Using Folic Acid Functionalized Bovine Milk Exosomes Improves the Therapeutic Efficacy against Breast Cancer" Life 12, no. 8: 1143. https://doi.org/10.3390/life12081143
APA StyleKumar, D. N., Chaudhuri, A., Dehari, D., Shekher, A., Gupta, S. C., Majumdar, S., Krishnamurthy, S., Singh, S., Kumar, D., & Agrawal, A. K. (2022). Combination Therapy Comprising Paclitaxel and 5-Fluorouracil by Using Folic Acid Functionalized Bovine Milk Exosomes Improves the Therapeutic Efficacy against Breast Cancer. Life, 12(8), 1143. https://doi.org/10.3390/life12081143







