Effectiveness of the Inactivated SARS-CoV-2 (Vero Cell) Vaccine in Peruvian Health Workers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Data Sources
2.2. Study Population
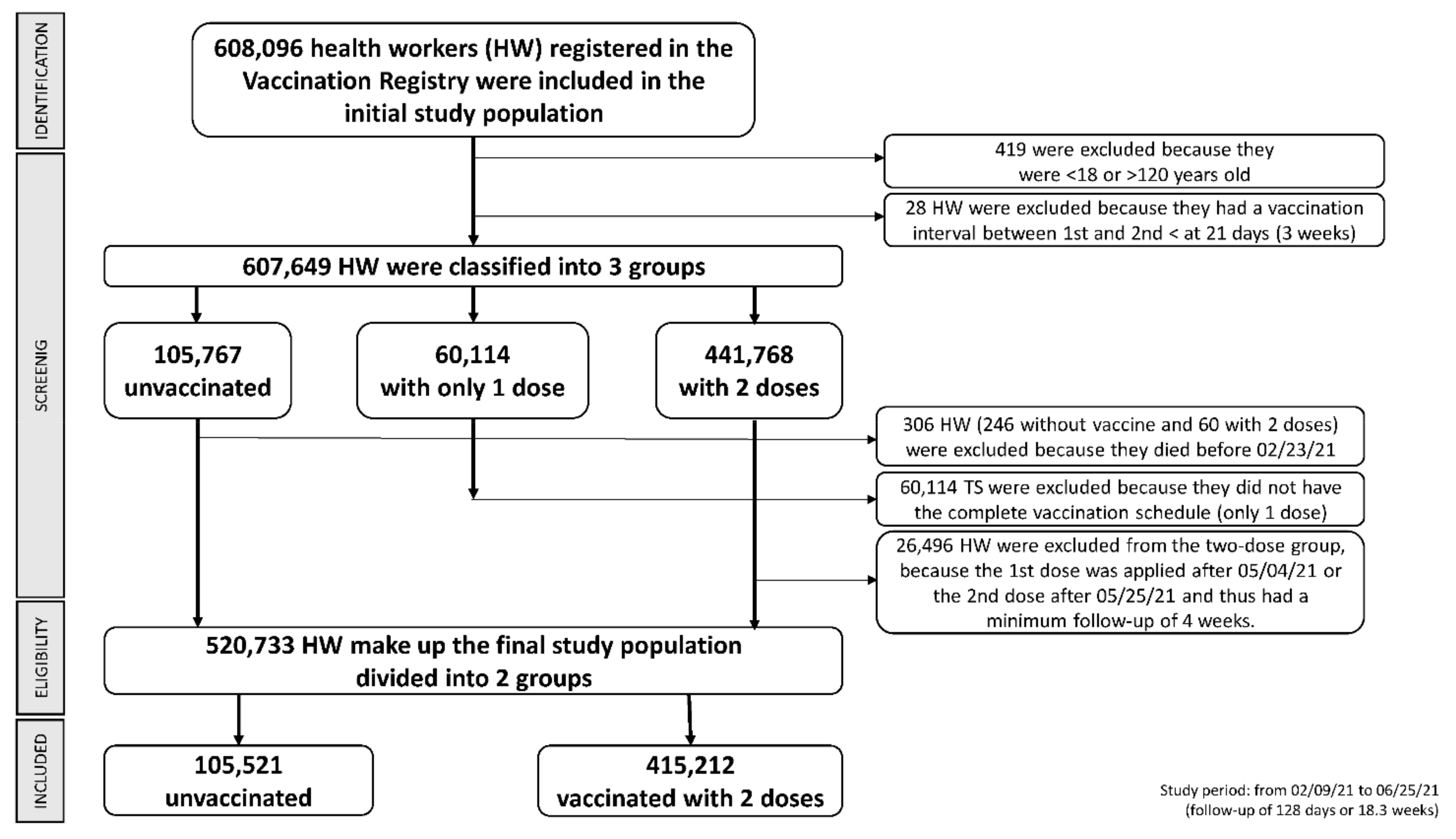
2.3. Criteria for Analysis
2.4. Statistical Analysis
3. Results
3.1. Vaccine Effectiveness in Preventing SARS-CoV-2 Infection
3.2. Vaccine Effectiveness in Hospitalized for SARS-CoV-2
3.3. Vaccine Effectiveness in Deaths from SARS-Co-V-2
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Santos-Sánchez, N.F.; Salas-Coronado, R. Origen, Características Estructurales, Medidas de Prevención, Diagnóstico y Fármacos Potenciales Para Prevenir y Controlar COVID-19. Medwave. 2020. Available online: /link.cgi/Medwave/Revisiones/RevisionClinica/8037.act (accessed on 28 March 2021).
- Asahi, K.; Undurraga, E.A.; Valdés, R.; Wagner, R. The Effect of COVID-19 on the Economy: Evidence from an Early Adopter of Localized Lockdowns. J. Glob. Health 2021, 11, 05002. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7897430/ (accessed on 7 March 2021). [CrossRef]
- Nguyen, L.H.; Drew, D.A.; Joshi, A.D.; Guo, C.-G.; Ma, W.; Mehta, R.S.; Sikavi, D.R.; Lo, C.H.; Kwon, S.; Song, M.; et al. Risk of COVID-19 among frontline healthcare workers and the general community: A prospective cohort study. medRxiv 2020. medRxiv:2020.04.29.20084111. [Google Scholar]
- Graham, B.S. Rapid COVID-19 vaccine development. Science 2020, 368, 945–946. [Google Scholar] [CrossRef]
- Shrotri, S.; Kampmann, P. Rastreador de Vacunas COVID-19. 2021. Available online: https://vac-lshtm.shinyapps.io/ncov_vaccine_landscape/ (accessed on 28 March 2021).
- Creech, C.B.; Walker, S.C.; Samuels, R.J. SARS-CoV-2 Vaccines. JAMA 2021, 325, 1318–1320. [Google Scholar] [CrossRef]
- Abu Jabal, K.; Ben-Amram, H.; Beiruti, K.; Batheesh, Y.; Sussan, C.; Zarka, S.; Edelstein, M. Impact of age, ethnicity, sex and prior infection status on immunogenicity following a single dose of the BNT162b2 mRNA COVID-19 vaccine: Real-world evidence from healthcare workers, Israel, December 2020 to January 2021. Eurosurveillance 2021, 26, 5. [Google Scholar] [CrossRef]
- Aran, D. Estimating real-world COVID-19 vaccine effectiveness in Israel using aggregated counts. medRxiv 2021. [Google Scholar] [CrossRef]
- Planas, D.; Veyer, D.; Baidaliuk, A.; Staropoli, I.; Guivel-Benhassine, F.; Rajah, M.M.; Planchais, C.; Porrot, F.; Robillard, N.; Puech, J.; et al. Reduced sensitivity of SARS-CoV-2 variant Delta to antibody neutralization. Nature 2021, 596, 276–280. [Google Scholar] [CrossRef]
- DIGEMID/MINSA. Resolución Directoral No. 486–2021/digemid/dpf/ufpb/minsa. Resolución que Autoriza Excepcionalmente la Importación y uso por Situaciones de Salud Pública la Vacuna SARS-CoV-2 (Vero Cell) Inactivada × 0.5 mL del Fabricante Beijing Institute of Biological Products Co. Ltd. (BIBP), China. DIGEMID. 2021. Available online: http://www.digemid.minsa.gob.pe/Resoluciones/ResolucionesArchivos/DPF/2021/RD_DPF_0000486_2021.pdf (accessed on 9 March 2021).
- Ministerio de Salud del Perú. Resolución Ministerial No. 848-2020-MINSA que aprueba el Documento Técnico «Plan Nacional de Vacunación contra la COVID-19. Ministerio de Salud. 2020. Available online: https://cdn.www.gob.pe/uploads/document/file/1394145/RM%20N%C2%B0848-2020-MINSA.PDF.PDF (accessed on 9 March 2021).
- Xia, S.; Zhang, Y.; Wang, Y.; Wang, H.; Yang, Y.; Gao, G.F.; Tan, W.; Wu, G.; Xu, M.; Lou, Z.; et al. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBIBP-CorV: A randomised, double-blind, placebo-controlled, phase 1/2 trial. Lancet Infect Dis. 2021, 21, 39–51. [Google Scholar] [CrossRef]
- Organización Mundial de la Salud. Peru: WHO Coronavirus Disease (COVID-19) Dashboard. Global Peru. 2021. Available online: https://covid19.who.int/region/amro/country/pe (accessed on 7 March 2021).
- Médicos Fallecidos por COVID-19 en Iberoamérica. Colegio Médico del Perú—Consejo Nacional. Available online: https://www.cmp.org.pe/medicos-fallecidos-por-covid-19-en-iberoamerica/ (accessed on 28 March 2021).
- MINSA. Resolución Ministerial No. 848-2020-MINSA. 2020. Available online: https://www.gob.pe/institucion/minsa/normas-legales/1293043-848-2020-minsa (accessed on 9 March 2021).
- Ministerio de Salud de Perú. Resolución Ministerial No. 161-2021-MINSA. 2021. Available online: https://www.gob.pe/institucion/minsa/normas-legales/1635499-161-2021-minsa (accessed on 27 March 2021).
- Halloran, M.E.; Struchiner, C.J.; Longini, I.M. Study designs for evaluating different efficacy and effectiveness aspects of vaccines. Am. J. Epidemiol. 1997, 146, 789–803. [Google Scholar] [CrossRef]
- Al Kaabi, N.; Zhang, Y.; Xia, S.; Yang, Y.; Al Qahtani, M.M.; Abdulrazzaq, N.; Al Nusair, M.; Hassany, M.; Jawad, J.S.; Abdalla, J.; et al. Effect of 2 Inactivated SARS-CoV-2 Vaccines on Symptomatic COVID-19 Infection in Adults: A Randomized Clinical Trial. JAMA 2021, 326, 35–45. Available online: https://jamanetwork.com/journals/jama/fullarticle/2780562 (accessed on 28 June 2021). [CrossRef]
- WHO Lists Additional COVID-19 Vaccine for Emergency Use and Issues Interim Policy Recommendations. Available online: https://www.who.int/news/item/07-05-2021-who-lists-additional-covid-19-vaccine-for-emergency-use-and-issues-interim-policy-recommendations (accessed on 28 June 2021).
- Dagan, N.; Barda, N.; Kepten, E.; Miron, O.; Perchik, S.; Katz, M.A.; Hernán, M.A.; Lipsitch, M.; Reis, B.; Balicer, R.D. BNT162b2 mRNA Covid-19 Vaccine in a Nationwide Mass Vaccination Setting. N. Engl. J. Med. 2021, 384, 1412–1423. [Google Scholar] [CrossRef]
- Haas, E.J.; Angulo, F.J.; McLaughlin, J.M.; Anis, E.; Singer, S.R.; Khan, F.; Brooks, N.; Smaja, M.; Mircus, G.; Pan, K. Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: An observational study using national surveillance data. Lancet 2021, 397, 1819–1829. [Google Scholar] [CrossRef]
- Chodick, G.; Tene, L.; Patalon, T.; Gazit, S.; Tov, A.B.; Cohen, D.; Muhsen, K. The effectiveness of the first dose of BNT162b2 vaccine in reducing SARS-CoV-2 infection 13–24 days after immunization: Real-world evidence. medRxiv 2021. medRxiv:2021.01.27.21250612. [Google Scholar]
- Hall, V.J.; Foulkes, S.; Saei, A.; Andrews, N.; Oguti, B.; Charlett, A.; Wellington, E.; Stowe, J.; Gillson, N.; Atti, A.; et al. COVID-19 vaccine coverage in health-care workers in England and effectiveness of BNT162b2 mRNA vaccine against infection (SIREN): A prospective, multicentre, cohort study. Lancet 2021, 397, 1725–1735. [Google Scholar] [CrossRef]
- Sadoff, J.; Gray, G.; Vandebosch, A.; Cárdenas, V.; Shukarev, G.; Grinsztejn, B.; Goepfert, P.A.; Truyers, C.; Fennema, H.; Spiessens, B. Safety and Efficacy of Single-Dose Ad26.COV2.S Vaccine against Covid-19. N. Engl. J. Med. 2021, 384, 2187–2201. [Google Scholar] [CrossRef]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- Voysey, M.; Costa Clemens, S.A.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhoratm, Q.E.; et al. Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: A pooled analysis of four randomised trials. Lancet 2021, 397, 881–891. [Google Scholar] [CrossRef]
- AZD1222 Vaccine met Primary Efficacy Endpoint in Preventing COVID-19. 2021. Available online: https://www.astrazeneca.com/media-centre/press-releases/2020/azd1222hlr.html (accessed on 25 January 2021).
- Evidence Assessment: Sinopharm/BBIBP COVID-19 Vaccine. Available online: https://cdn.who.int/media/docs/default-source/immunization/sage/2021/april/2_sage29apr2021_critical-evidence_sinopharm.pdf (accessed on 28 June 2021).
- Keehner, J.; Horton, L.E.; Pfeffer, M.A.; Longhurst, C.A.; Schooley, R.T.; Currier, J.S.; Abeles, S.R.; Torriani, F.J. SARS-CoV-2 Infection after Vaccination in Health Care Workers in California. N. Engl. J. Med. 2021, 384, 1774–1775. [Google Scholar] [CrossRef]
- Daniel, W.; Nivet, M.; Warner, J.; Podolsky, D.K. Early Evidence of the Effect of SARS-CoV-2 Vaccine at One Medical Center. N. Engl. J. Med. 2021, 384, 1962–1963. [Google Scholar] [CrossRef]
- Benenson, S.; Oster, Y.; Cohen, M.J.; Nir-Paz, R. BNT162b2 mRNA Covid-19 Vaccine Effectiveness among Health Care Workers. N. Engl. J. Med. 2021, 384, 1775–1777. [Google Scholar] [CrossRef]
- Angel, Y.; Spitzer, A.; Henig, O.; Saiag, E.; Sprecher, E.; Padova, H.; Ben-Ami, R. Association between Vaccination with BNT162b2 and Incidence of Symptomatic and Asymptomatic SARS-CoV-2 Infections among Health Care Workers. JAMA 2021, 325, 2457–2465. Available online: https://jamanetwork.com/journals/jama/fullarticle/2779853 (accessed on 16 June 2021). [CrossRef] [PubMed]
- Khoury, D.S.; Cromer, D.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Subbarao, K.; Kent, S.J.; Triccas, J.A.; Davenport, M.P. Neutralizing Antibody Levels are Highly Predictive.pdf. 2021. Available online: https://www.nature.com/articles/s41591-021-01377-8.pdf (accessed on 24 February 2022).
- Zollner, A.; Watschingera, C.; Rossler, A.; Farcet, M.R.; Pennerg, A.; Bohm, V.; Kiechlc, S.J.; Stampfel, G.; Hintenbergerg, T.; Tilg, H.; et al. B and T Cell Response to SARS-CoV-2 Vaccination in.pdf. 2021. Available online: https://www.thelancet.com/action/showPdf?pii=S2352-3964%2821%2900332-7 (accessed on 24 February 2022).
- Kalimuddin, S.; Tham, C.Y.L.; Qui, M.; de Alwis, R.; Sim, J.X.Y.; Lim, J.M.E.; Tan, H.C.; Syenina, A.; Zhang, S.L.; Le Bert, N.; et al. Early T cell and binding antibody responses are associated with COVID-19 RNA vaccine efficacy onset. Med 2021, 2, 682–688.e4. [Google Scholar] [CrossRef] [PubMed]
- Reduced T Cell and Antibody Responses to Inactivated Coronavirus Vaccine among Males and Individuals above 55 Years Old | medRxiv. Available online: https://www.medrxiv.org/content/10.1101/2021.08.16.21262069v1 (accessed on 4 April 2022).
- Dinnes, J.; Deeks, J.J.; Adriano, A.; Berhane, S.; Davenport, C.; Dittrich, S.; Emperador, D.; Takwoingi, Y.; Cunningham, J.; Beese, S. Rapid, point-of-care antigen and molecular-based tests for diagnosis of SARS-CoV-2 infection. Cochrane Database Syst. Rev. 2020, 2021, CD013705. Available online: http://doi.wiley.com/10.1002/14651858.CD013705 (accessed on 24 February 2021).
- INS: Variante Brasilera Tiene una Amplia Circulación en Varios Distritos de Lima. Instituto Nacional de Salud. Available online: http://web.ins.gob.pe/es/prensa/noticia/ins-variante-brasilera-tiene-una-amplia-circulacion-en-varios-distritos-de-lima (accessed on 29 March 2022).
- Lopez Bernal, J.; Andrews, N.; Gower, C.; Gallagher, E.; Simmons, R.; Thelwall, S.; Stowe, J.; Tessier, E.; Groves, N.; Dabrera, G.; et al. Effectiveness of Covid-19 Vaccines against the B.1.617.2 (Delta) Variant. N. Engl. J. Med. 2021, 85, 585–594. [Google Scholar] [CrossRef]
- PubMed Central Full Text PDF. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8462339/pdf/21-1427.pdf (accessed on 24 February 2022).
- Amit, S.; Beni, S.A.; Biber, A.; Grinberg, A.; Leshem, E.; Regev-Yochay, G. Post-Vaccination COVID-19 among Healthcare Workers, Israel. Emerg. Infect. Dis. 2021, 27, 1220. [Google Scholar] [CrossRef]
- Secuenciación Genómica del virus SARS-CoV-2 en el Perú. Instituto Nacional de Salud. Available online: https://web.ins.gob.pe/es/covid19/secuenciamiento-sars-cov2 (accessed on 4 April 2022).
- Ranzani, O.T.; Hitchings, M.; Dorion, M.; D’Agostini, T.L.; de Paula, R.C.; de Paula, O.F.P.; de Moura Villela, E.F.; Torres, M.S.; de Oliveira, S.B.; Schulz, W.; et al. Effectiveness of the CoronaVac vaccine in the elderly population during a P.1 variant-associated epidemic of COVID-19 in Brazil: A test-negative case-control study. BMJ 2021, 374, b2015. [Google Scholar] [CrossRef]
- Abdool Karim, S.S.; de Oliveira, T. New SARS-CoV-2 Variants—Clinical, Public Health, and Vaccine Implications. N. Engl. J. Med. 2021, 384, 1866–1888. [Google Scholar] [CrossRef]
- Patel, M.K.; Bergeri, I.; Bresee, J.S.; Cowling, B.J.; Crowcroft, N.S.; Fahmy, K.; Hirve, S.; Kang, G.; Katz, M.A.; Lanata, C.F.; et al. Evaluation of Post-Introduction COVID-19 Vaccine Effectiveness: Summary of Interim Guidance of the World Health Organization. Vaccine 2021, 39, 4013–4024. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8166525/ (accessed on 28 June 2021). [CrossRef]
- Femeba Home|Fundación Femeba. Available online: https://www.fundacionfemeba.org.ar/home (accessed on 28 June 2021).






| Variable | Unvaccinated | Cohort Vaccinated | Total | |||
|---|---|---|---|---|---|---|
| Cohort | with 2 Doses | |||||
| n0 | % | n2 | % | N | % | |
| Gender | ||||||
| Female | 72,746 | 68.9% | 268,855 | 64.8% | 341,601 | 65.6% |
| Male | 32,775 | 31.1% | 146,357 | 35.2% | 179,132 | 34.4% |
| Median age (range) | 37 | (31–46) * | 40 | (32–51) * | 40 | (31–51) * |
| Mean age (SD) | 39.75 | 11.89 | 42.66 | 12.98 | 42.04 | 12.79 |
| Age group | ||||||
| 18–29 | 20,771 | 19.7% | 65,914 | 15.9% | 86,685 | 16.6% |
| 30–59 | 77,569 | 73.5% | 297,624 | 71.7% | 375,193 | 72.1% |
| 60 or more | 7181 | 6.8% | 51,674 | 12.4% | 58,855 | 11.3% |
| Previous history of infection | ||||||
| Present | 19,802 | 18.8% | 130,889 | 31.5% | 150,691 | 28.9% |
| Absent | 85,719 | 81.2% | 284,323 | 68.5% | 370,042 | 71.1% |
| Population | 105,521 | 100% | 415,212 | 100% | 520,733 | 100% |
| Categories, Outcomes, and Vaccination Status | Events | Persons-Days of Follow-Up | Incidence Density per 1000 Days of Follow-Up | Crude Incidence Density Ratio (CI 95%) | z | p-Value | Effectiveness of the 2 Doses of the Vaccine (CI 95%) ** |
|---|---|---|---|---|---|---|---|
| Gender | |||||||
| Female | |||||||
| Infection | |||||||
| Unvaccinated | 4263 | 8,684,163 | 0.49089 | 0.667 | −20.78 | <0.0001 | 33.3% |
| Vaccinated with 2 doses | 6975 | 21,290,825 | 0.32761 | (0.64–0.69) | (30.7–35.8) | ||
| Hospitalization | |||||||
| Unvaccinated | 156 | 8,390,243 | 0.01859 | 0.224 | −14.56 | <0.0001 | 77.6% |
| Vaccinated with 2 doses | 81 | 19,483,688 | 0.00416 | (0.16–0.24) | (70.8–82.9) | ||
| Death | |||||||
| Unvaccinated | 68 | 8,972,580 | 0.00758 | 0.061 | −8.08 | <0.0001 | 93.9% |
| Vaccinated with 2 doses | 10 | 21,657,884 | 0.00046 | (0.034–0.075) | (89.9–97.7) | ||
| Male | |||||||
| Infection | |||||||
| Unvaccinated | 2041 | 4,109,548 | 0.49665 | 0.628 | −16.73 | <0.0001 | 37.2% |
| Vaccinated with 2 doses | 3585 | 11,492,768 | 0.31194 | (0.60–0.66) | (33.6–40.5) | ||
| Hospitalization | |||||||
| Unvaccinated | 187 | 3,972,196 | 0.04708 | 0.180 | −13.30 | <0.0001 | 82.0% |
| Vaccinated with 2 doses | 89 | 10,486,394 | 0.00849 | (0.14–0.23) | (76.8–86.0) | ||
| Death | |||||||
| Unvaccinated | 137 | 4,241,419 | 0.03230 | 0.056 | −12.17 | <0.0001 | 94.4% |
| Vaccinated with 2 doses | 21 | 11,679,587 | 0.00180 | (0.031–0.082) | (91.8–96.9) | ||
| Age group | |||||||
| 18–59 years | |||||||
| Infection | |||||||
| Unvaccinated | 5903 | 11,550,681 | 0.51105 | 0.658 | −25.28 | <0.0001 | 34.2% |
| Vaccinated with 2 doses | 9698 | 28,831,895 | 0.33636 | (0.64–0.68) | (32.0–36.2) | ||
| Hospitalization | |||||||
| Unvaccinated | 254 | 11,178,574 | 0.02272 | 0.203 | −14.47 | <0.0001 | 79.7% |
| Vaccinated with 2 doses | 122 | 26,418,502 | 0.00462 | (0.16–0.25) | (74.8–83.6) | ||
| Death | |||||||
| Unvaccinated | 101 | 11,962,139 | 0.00844 | 0.044 | −9.50 | <0.0001 | 95.6% |
| Vaccinated with 2 doses | 11 | 29,340,246 | 0.00037 | (0.019–0.073) | (92.7–98.1) | ||
| 60 or more years | |||||||
| Infection | |||||||
| Unvaccinated | 401 | 823,716 | 0.48682 | 0.449 | −13.30 | <0.0001 | 55.1% |
| Vaccinated with 2 doses | 862 | 3,943,266 | 0.21860 | (0.40–0.50) | (49.6–60.3) | ||
| Hospitalization | |||||||
| Unvaccinated | 89 | 792,047 | 0.11237 | 0.121 | −11.82 | <0.0001 | 87.9% |
| Vaccinated with 2 doses | 48 | 3,544,451 | 0.01354 | (0.08–0.17) | (82.9–91.5) | ||
| Death | |||||||
| Unvaccinated | 104 | 832,546 | 0.12492 | 0.040 | −13.01 | <0.0001 | 96.0% |
| Vaccinated with 2 doses | 20 | 3,988,793 | 0.00501 | (0.022–0.060) | (94.0–97.8) | ||
| Previous history of infection | |||||||
| Absent | |||||||
| Infection | |||||||
| Unvaccinated | 5636 | 10,025,416 | 0.56217 | 0.716 | −19.57 | <0.0001 | 28.4% |
| Vaccinated with 2 doses | 8923 | 22,156,861 | 0.40272 | (0.693–0.741) | (25.9–30.7) | ||
| Hospitalization | |||||||
| Unvaccinated | 319 | 2,242,740 | 0.14224 | 0.113 | −13.23 | <0.0001 | 88.7% |
| Vaccinated with 2 doses | 155 | 9,653,245 | 0.01606 | (0.093–0.137) | (86.3–90.7) | ||
| Death | |||||||
| Unvaccinated | 191 | 10,398,828 | 0.01837 | 0.065 | −13.19 | <0.0001 | 93.5% |
| Vaccinated with 2 doses | 27 | 22,621,886 | 0.00119 | (0.040–0.092) | (90.8–96.0) | ||
| Present | |||||||
| Infection | |||||||
| Unvaccinated | 668 | 2,348,981 | 0.28438 | 0.542 | −13.32 | <0.0001 | 45.8% |
| Vaccinated with 2 doses | 1637 | 10,618,300 | 0.15417 | (0.496–0.593) | (40.7–50.4) | ||
| Hospitalization | |||||||
| Unvaccinated | 24 | 2,242,740 | 0.01070 | 0.145 | −5.86 | <0.0001 | 85.5% |
| Vaccinated with 2 doses | 15 | 9,653,245 | 0.00155 | (0.076–0.277) | (72.3–92.4) | ||
| Death | |||||||
| Unvaccinated | 14 | 2,395,857 | 0.00584 | 0.032 | −4.55 | <0.0001 | 96.8% |
| Vaccinated with 2 doses | 4 | 10,707,153 | 0.00037 | (0.007–0.141) | (85.9–99.3) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solis-Castro, M.E.; Jaramillo-Corrales, A.; Gonzalez Seminario, R.V.; Janampa Grados, N.; Mamani Pilco, I.E.; Vargas Quispe, K.E.; La Torre Rosillo, L.Y.; Vásquez Dominguez, M.N.; Enriquez Cusi, D.T.; Minaya, P.; et al. Effectiveness of the Inactivated SARS-CoV-2 (Vero Cell) Vaccine in Peruvian Health Workers. Life 2022, 12, 1318. https://doi.org/10.3390/life12091318
Solis-Castro ME, Jaramillo-Corrales A, Gonzalez Seminario RV, Janampa Grados N, Mamani Pilco IE, Vargas Quispe KE, La Torre Rosillo LY, Vásquez Dominguez MN, Enriquez Cusi DT, Minaya P, et al. Effectiveness of the Inactivated SARS-CoV-2 (Vero Cell) Vaccine in Peruvian Health Workers. Life. 2022; 12(9):1318. https://doi.org/10.3390/life12091318
Chicago/Turabian StyleSolis-Castro, Maria Edith, Alex Jaramillo-Corrales, Rommell Veintimilla Gonzalez Seminario, Noemi Janampa Grados, Idania Edith Mamani Pilco, Karina Elizabeth Vargas Quispe, Lenin Yonel La Torre Rosillo, Mario Neyser Vásquez Dominguez, David Teodoro Enriquez Cusi, Percy Minaya, and et al. 2022. "Effectiveness of the Inactivated SARS-CoV-2 (Vero Cell) Vaccine in Peruvian Health Workers" Life 12, no. 9: 1318. https://doi.org/10.3390/life12091318








