Exogenous 3-Iodothyronamine (T1AM) Can Affect Phosphorylation of Proteins Involved on Signal Transduction Pathways in In Vitro Models of Brain Cell Lines, but These Effects Are Not Strengthened by Its Catabolite, 3-Iodothyroacetic Acid (TA1)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Cell Treatments
2.3. Gene Expression Analysis
2.4. Uptake of T1AM and TA1
2.5. Cell Viability
2.6. Glucose Consumption
2.7. cAMP Concentration Assay
2.8. Protein Expression Analysis
2.9. Statistical Analysis
3. Results
3.1. Expression of the Receptors of the Glutamatergic Pathway
3.2. Cellular Uptake of T1AM and TA1
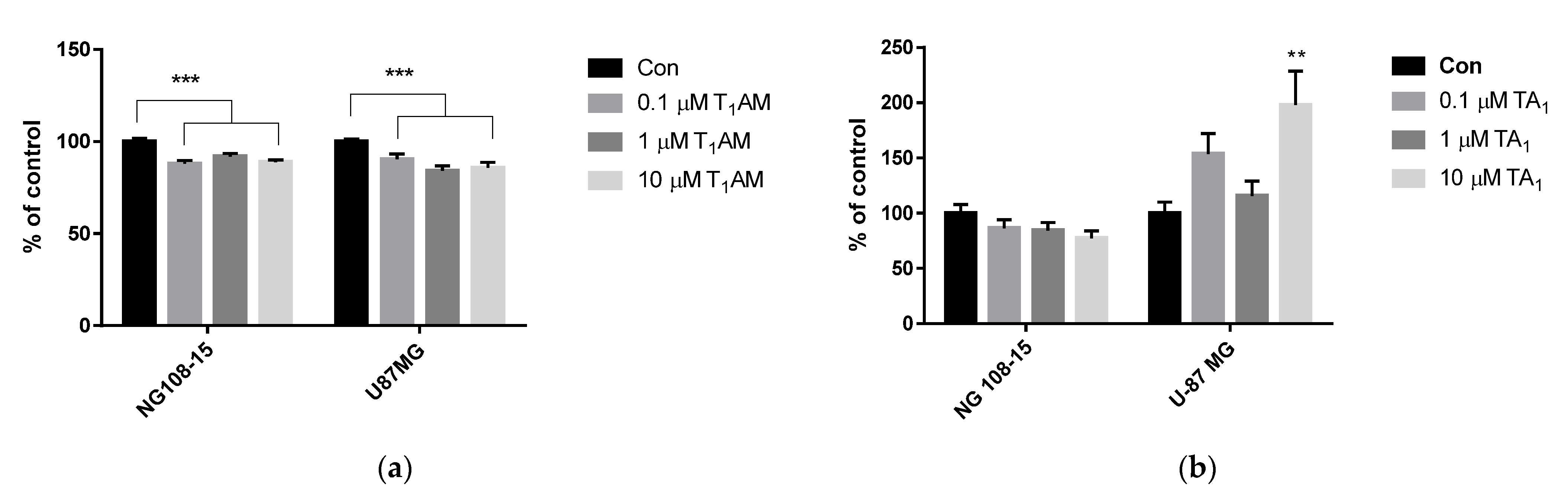
3.3. Cell Viability
3.4. Glucose Consumption
3.5. cAMP Assay
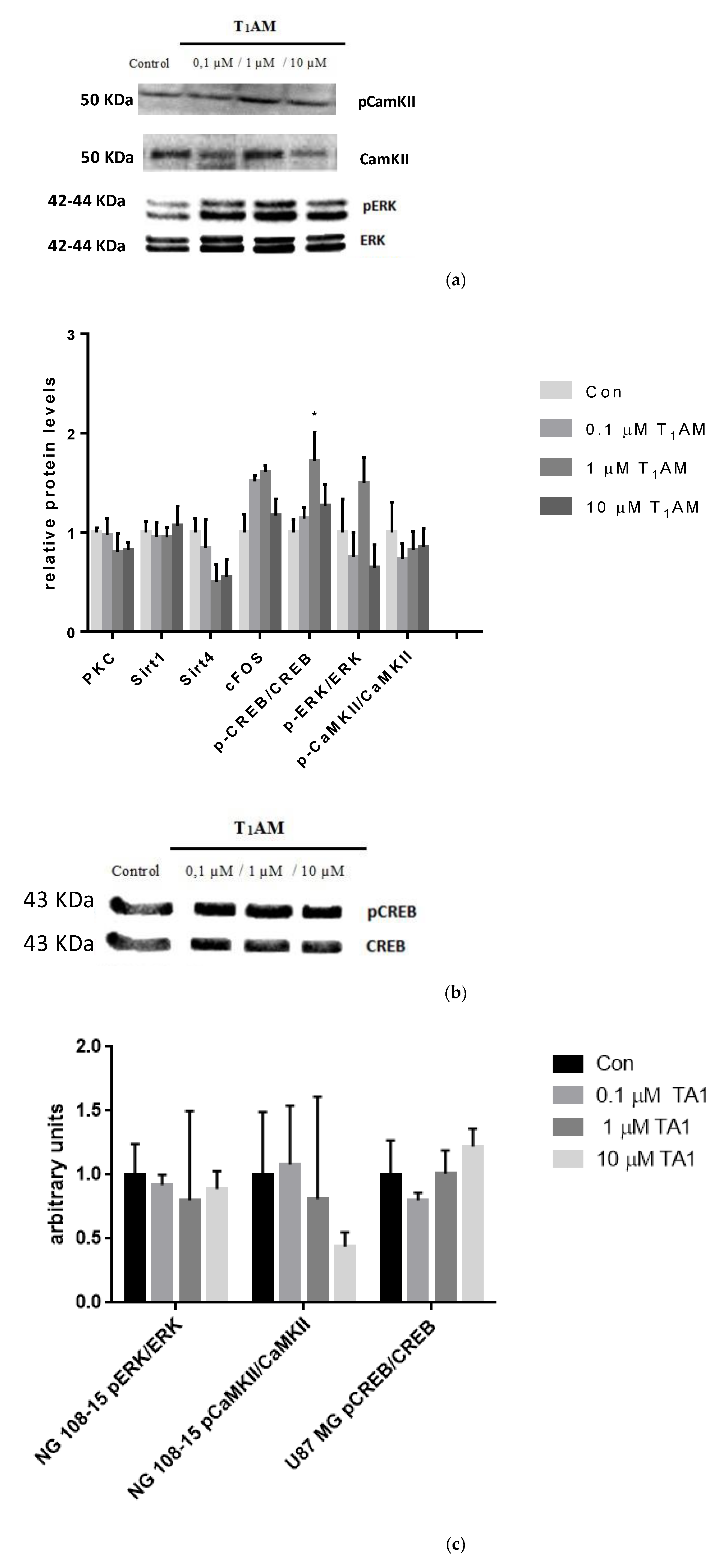
3.6. Effects of T1AM and TA1 on the Expression of Signaling Cascade
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Scanlan, T.S.; Suchland, K.L.; Hart, M.E.; Chiellini, G.; Huang, Y.; Kruzich, P.J.; Frascarelli, S.; Crossley, D.A.; Bunzow, J.R.; Ronca-Testoni, S.; et al. 3-Iodothyronamine is an e-endogenous and rapid-acting derivative of thyroid hormone. Nat. Med. 2004, 10, 638–642. [Google Scholar] [CrossRef] [PubMed]
- Köhrle, J. The Colorful Diversity of Thyroid Hormone Metabolites. Eur. Thyroid J. 2019, 8, 115–129. [Google Scholar] [CrossRef] [PubMed]
- Saba, A.; Chiellini, G.; Frascarelli, S.; Marchini, M.; Ghelardoni, S.; Raffaelli, A.; Tonacchera, M.; Vitti, P.; Scanlan, T.S.; Zucchi, R. Tissue distribution and cardiac metabolism of 3-iodothyronamine. Endocrinology 2010, 151, 5063–5073. [Google Scholar] [CrossRef]
- Hoefig, C.S.; Wuensch, T.; Rijntjes, E.; Lehmphul, I.; Daniel, H.; Schweizer, U.; Mittag, J.; Köhrle, J. Biosynthesis of 3-Iodothyronamine From T4 in Murine Intestinal Tissue. Endocrinology 2015, 156, 4356–4364. [Google Scholar] [CrossRef]
- Wood, W.J.; Geraci, T.; Nilsen, A.; DeBarber, A.E.; Scanlan, T.S. Iodothyronamines are oxidatively deaminated to iodothyroacetic acids in vivo. Chembiochem 2009, 10, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Snead, A.N.; Santos, M.S.; Seal, R.P.; Miyakawa, M.; Edwards, R.H.; Scanlan, T.S. Thyronamines inhibit plasma membrane and vesicular monoammine transport. ACS Chem Biol. 2007, 2, 390–398. [Google Scholar] [CrossRef] [PubMed]
- Regard, J.B.; Kataoka, H.; Cano, D.A.; Camerer, E.; Yin, L.; Zheng, Y.W.; Scanlan, T.S.; Hebrok, M.; Coughlin, S.R. Probing cell type-specific functions of Gi in vivo identifies GPCR regulators of insulin secretion. J. Clin. Investig. 2007, 117, 4034–4043. [Google Scholar] [CrossRef]
- Gompf, H.S.; Greenberg, J.H.; Aston-Jones, G.; Ianculescu, A.G.; Scanlan, T.S.; Dratman, M.B. 3-Monoiodothyronamine: The rationale for its action as an endogenous adrenergic-blocking neuromodulator. Brain Res. 2010, 10, 130–140. [Google Scholar] [CrossRef]
- Manni, M.E.; De Siena, G.; Saba, A.; Marchini, M.; Landucci, E.; Gerace, E.; Zazzeri, M.; Musilli, C.; Pellegrini-Giampietro, D.; Matucci, R.; et al. Pharmacological effects of 3-iodothyronamine (T1AM) in mice include facilitation of memory acquisition and retention and reduction of pain threshold. Br. J. Pharmacol. 2013, 168, 354–362. [Google Scholar] [CrossRef]
- Bellusci, L.; Laurino, A.; Sabatini, M.; Sestito, S.; Lenzi, P.; Raimondi, L.; Rapposelli, S.; Biagioni, F.; Fornai, F.; Salvetti, A.; et al. New Insights into the Potential Roles of 3-Iodothyronamine (T1AM) and Newly Developed Thyronamine-Like TAAR1 Agonists in Neuroprotection. Front Pharmacol. 2017, 8, 905. [Google Scholar] [CrossRef] [Green Version]
- Minatohara, K.; Akiyoshi, M.; Okuno, H. Role of Immediate-Early Genes in Synaptic Plasticity and Neuronal Ensembles Underlying the Memory Trace. Front Mol. Neurosci. 2016, 5, 78. [Google Scholar] [CrossRef] [PubMed]
- Giese, K.P.; Mizuno, K. The roles of protein kinases in learning and memory. Learn Mem. 2013, 20, 540–552. [Google Scholar] [CrossRef] [PubMed]
- Piehl, S.; Hoefig, C.S.; Scanlan, T.S.; Köhrle, J. Thyronamines--past, present, and future. Endocr. Rev. 2011, 32, 64–80. [Google Scholar] [CrossRef] [PubMed]
- Braulke, L.J.; Klingenspor, M.; DeBarber, A.; Tobias, S.C.; Grandy, D.K.; Scanlan, T.S.; Heldmaier, G. 3-Iodothyronamine: A novel hormone controlling the balance between glucose and lipid utilisation. J. Comp. Physiol. B 2008, 178, 167–177. [Google Scholar] [CrossRef]
- Laurino, A.; De Siena, G.; Resta, F.; Masi, A.; Musilli, C.; Zucchi, R.; Raimondi, L. 3-iodothyroacetic acid, a metabolite of thyroid hormone, induces itch and reduces threshold to noxious and to painful heat stimuli in mice. Br. J. Pharmacol. 2015, 172, 1859–1868. [Google Scholar] [CrossRef] [PubMed]
- Musilli, C.; De Siena, G.; Manni, M.E.; Logli, A.; Landucci, E.; Zucchi, R.; Saba, A.; Donzelli, R.; Passani, M.B.; Provensi, G.; et al. Histamine mediates behavioural and metabolic effects of 3-iodothyroacetic acid, an endogenous end product of thyroid hormone metabolism. Br. J. Pharmacol. 2014, 171, 3476–3484. [Google Scholar] [CrossRef]
- Ge, Y.; Wang, Y.T. Postsynaptic signaling at glutamatergic synapses as therapeutic targets. Curr. Opin. Neurobiol. 2022, 75, 102585. [Google Scholar] [CrossRef]
- Ménard, C.; Gaudreau, P.; Quirion, R. Signaling pathways relevant to cognition-enhancing drug targets. Handb. Exp. Pharmacol. 2015, 228, 59–98. [Google Scholar] [CrossRef]
- Sacripanti, G.; Lorenzini, L.; Bandini, L.; Frascarelli, S.; Zucchi, R.; Ghelardoni, S. 3-Iodothyronamine and 3,5,3’-triiodo-L-thyronine reduce SIRT1 protein expression in the HepG2 cell line. Horm. Mol. Biol. Clin. Investig. 2020, 2, 41. [Google Scholar] [CrossRef]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef] [Green Version]
- Lorenzini, L.; Ghelardoni, S.; Saba, A.; Sacripanti, G.; Chiellini, G.; Zucchi, R. Recovery of 3-Iodothyronamine and Derivatives in Biological Matrixes: Problems and Pitfalls. Thyroid 2017, 27, 1323–1331. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Sacripanti, G.; Nguyen, N.M.; Lorenzini, L.; Frascarelli, S.; Saba, A.; Zucchi, R.; Ghelardoni, S. 3,5-Diiodo-l-Thyronine Increases Glucose Consumption in Cardiomyoblasts Without Affecting the Contractile Performance in Rat Heart. Front Endocrinol. 2018, 9, 282. [Google Scholar] [CrossRef] [PubMed]
- Cull-Candy, S.G.; Leszkiewicz, D.N. Role of distinct NMDA receptor subtypes at central synapses. Sci. STKE 2004, 255, 16. [Google Scholar] [CrossRef] [PubMed]
- Flint, A.C.; Maisch, U.S.; Weishaupt, J.H.; Kriegstein, A.R.; Monyer, H. NR2A subunit expression shortens NMDA receptor synaptic currents in developing neocortex. J. Neurosci. 1997, 17, 2469–2476. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6573498/ (accessed on 1 April 1997). [CrossRef]
- Mierau, S.B.; Meredith, R.M.; Upton, A.L.; Paulsen, O. Dissociation of experience-dependent and -indipendent changes in excitatory synaptic transmission during development of barrel cortex. Proc. Natl. Acad. Sci. USA 2004, 101, 15518–15523. [Google Scholar] [CrossRef]
- Sheng, M.; Cummings, J.; Roldan, L.A.; Jan, Y.N.; Jan, L.Y. Changing subunit composition of heteromeric NMDA receptors during development of rat cortex. Nature 1994, 68, 144–147. [Google Scholar] [CrossRef]
- Böckers, T.M.; Zimmer, M.; Müller, A.; Bergmann, M.; Brose, N.; Kreutz, M.R. Expression of the NMDA R1 receptor in selected human brain regions. Neuroreport 1994, 5, 965–969. [Google Scholar] [CrossRef]
- Henderson, J.T.; Georgiou, J.; Jia, Z.; Robertson, J.; Elowe, S.; Roder, J.C.; Pawson, T. The Receptor Tyrosine Kinase EphB2 Regulates NMDA-Dependent Synaptic Function. Neuron 2001, 32, 1041–1056. [Google Scholar] [CrossRef]
- Pozniak, P.D.; White, M.K.; Khalili, K. TNF-α/NF-κB Signaling in the CNS: Possible Connection to EPHB2. J. Neuroimmune Pharmacol. 2014, 9, 133–141. [Google Scholar] [CrossRef] [Green Version]
- Saito, N.; Shirai, Y. Protein kinase C gamma (PKC gamma): Function of neuron specific isotype. J. Biochem. 2002, 132, 683–687. [Google Scholar] [CrossRef] [PubMed]
- Moriguchi, S.; Shioda, N.; Han, F.; Narahashi, T.; Fukunaga, K. CaM kinase II and protein kinase C activations mediate enhancement of long-term potentiation by nefiracetam in the rat hippocampal CA1 region. J. Neurochem. 2008, 3, 1092–1103. [Google Scholar] [CrossRef] [PubMed]
- Laurino, A.; Landucci, E.; Resta, F.; De Siena, G.; Pellegrini-Giampietro, D.E.; Masi, A.; Mannaioni, G.; Raimondi, L. Anticonvulsant and Neuroprotective Effects of the Thyroid Hormone Metabolite 3-Iodothyroacetic Acid. Thyroid 2018, 28, 1387–1397. [Google Scholar] [CrossRef] [PubMed]
- Magupalli, V.G.; Mochida, S.; Yan, J.; Jiang, X.; Westenbroek, R.E.; Nairn, A.C.; Scheuer, T.; Catterall, W.A. Ca2+-independent Activation of Ca2+/Calmodulin-dependent Protein Kinase II Bound to the C-terminal Domain of CaV2.1 Calcium Channels. J. Biol. Chem. 2013, 288, 4637–4648. [Google Scholar] [CrossRef]
- Kida, S. A Functional Role for CREB as a Positive Regulator of Memory Formation and LTP. Exp. Neurobiol. 2012, 21, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Lynch, M.A. Long-term potentiation and memory. Physiol. Rev. 2004, 84, 87–136. [Google Scholar] [CrossRef]
- Assadi-Porter, F.M.; Reiland, H.; Sabatini, M.; Lorenzini, L.; Carnicelli, V.; Rogowski, M.; Selen Alpergin, E.S.; Tonelli, M.; Ghelardoni, S.; Saba, A.; et al. Metabolic Reprogramming by 3-Iodothyronamine (T1AM): A New Perspective to Reverse Obesity through Co-Regulation of Sirtuin 4 and 6 Expression. Int. J. Mol. Sci. 2018, 19, 1535. [Google Scholar] [CrossRef] [Green Version]




| T1AM | NG 108-15 Cells | U-87 MG Cells | ||||||
|---|---|---|---|---|---|---|---|---|
| Medium | Cell Lysate | Medium | Cell Lysate | |||||
| (µM) | T1AM (nM) | TA1 (nM) | T1AM (nM) | TA1 (nM) | T1AM (nM) | TA1 (nM) | T1AM (nM) | TA1 (nM) |
| 0.1 | N.D. | 135 ± 12 | N.D. | N.D. | N.D. | 95 ± 6 | N.D. | N.D. |
| 1 | N.D. | 311 ± 161 | N.D. | 7.7 ± 0.2 | N.D. | 1165 ± 41 | N.D. | 22 ± 5 |
| 10 | 0.66 ± 0.14 | 4996 ± 97 | 10 ± 6 | 91 ± 19 | 0.36 ± 0.01 | 13,214 ± 302 | 6 ± 0.3 | 144 ± 80 |
| Cell Lines | Cytosolic Fraction | Mitochondrial Fraction | Nuclear Fraction | |||
|---|---|---|---|---|---|---|
| T1AM (µM) | TA1 (nM) | T1AM (µM) | TA1 (nM) | T1AM (µM) | TA1 (nM) | |
| NG108-15 | 2.89 ± 0.13 | 205.3 ± 27.6 | 1.66 ± 0.1 | 18.6 ± 2.9 | 2.77 ± 0.07 | 46.5 ± 9.3 |
| U-87 MG | 2.62 ± 0.24 | 20.5 ± 0.9 | 0.54 ± 0.17 | N.D. | 1.63 ± 0.34 | N.D. |
| Cell Lines/TA1 | Medium (nM) | Lysate (nM) | ||||
|---|---|---|---|---|---|---|
| 0.1 µM | 1 µM | 10 µM | 0.1 µM | 1 µM | 10 µM | |
| NG 108-15 | 233 ± 18 | 1795 ± 55 | 5355 ± 175 | 6.8 ± 1.9 | 72.4 ± 6.9 | 995 ± 95 |
| U-87 MG | 135 ± 26 | 1193 ± 140 | 1520 ± 327 | 1.6 ± 0.1 | 15.4 ± 3.9 | 120 ± 22 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bandini, L.; Sacripanti, G.; Borsò, M.; Tartaria, M.; Fogliaro, M.P.; Giannini, G.; Carnicelli, V.; Figuccia, M.E.; Verlotta, S.; De Antoni, F.; et al. Exogenous 3-Iodothyronamine (T1AM) Can Affect Phosphorylation of Proteins Involved on Signal Transduction Pathways in In Vitro Models of Brain Cell Lines, but These Effects Are Not Strengthened by Its Catabolite, 3-Iodothyroacetic Acid (TA1). Life 2022, 12, 1352. https://doi.org/10.3390/life12091352
Bandini L, Sacripanti G, Borsò M, Tartaria M, Fogliaro MP, Giannini G, Carnicelli V, Figuccia ME, Verlotta S, De Antoni F, et al. Exogenous 3-Iodothyronamine (T1AM) Can Affect Phosphorylation of Proteins Involved on Signal Transduction Pathways in In Vitro Models of Brain Cell Lines, but These Effects Are Not Strengthened by Its Catabolite, 3-Iodothyroacetic Acid (TA1). Life. 2022; 12(9):1352. https://doi.org/10.3390/life12091352
Chicago/Turabian StyleBandini, Lavinia, Ginevra Sacripanti, Marco Borsò, Maria Tartaria, Maria Pia Fogliaro, Giulia Giannini, Vittoria Carnicelli, Matteo Emanuele Figuccia, Sara Verlotta, Fiammetta De Antoni, and et al. 2022. "Exogenous 3-Iodothyronamine (T1AM) Can Affect Phosphorylation of Proteins Involved on Signal Transduction Pathways in In Vitro Models of Brain Cell Lines, but These Effects Are Not Strengthened by Its Catabolite, 3-Iodothyroacetic Acid (TA1)" Life 12, no. 9: 1352. https://doi.org/10.3390/life12091352





