Plant Extracts and SARS-CoV-2: Research and Applications
Abstract
:1. Origin and Evolution of Pathogenic Coronaviruses
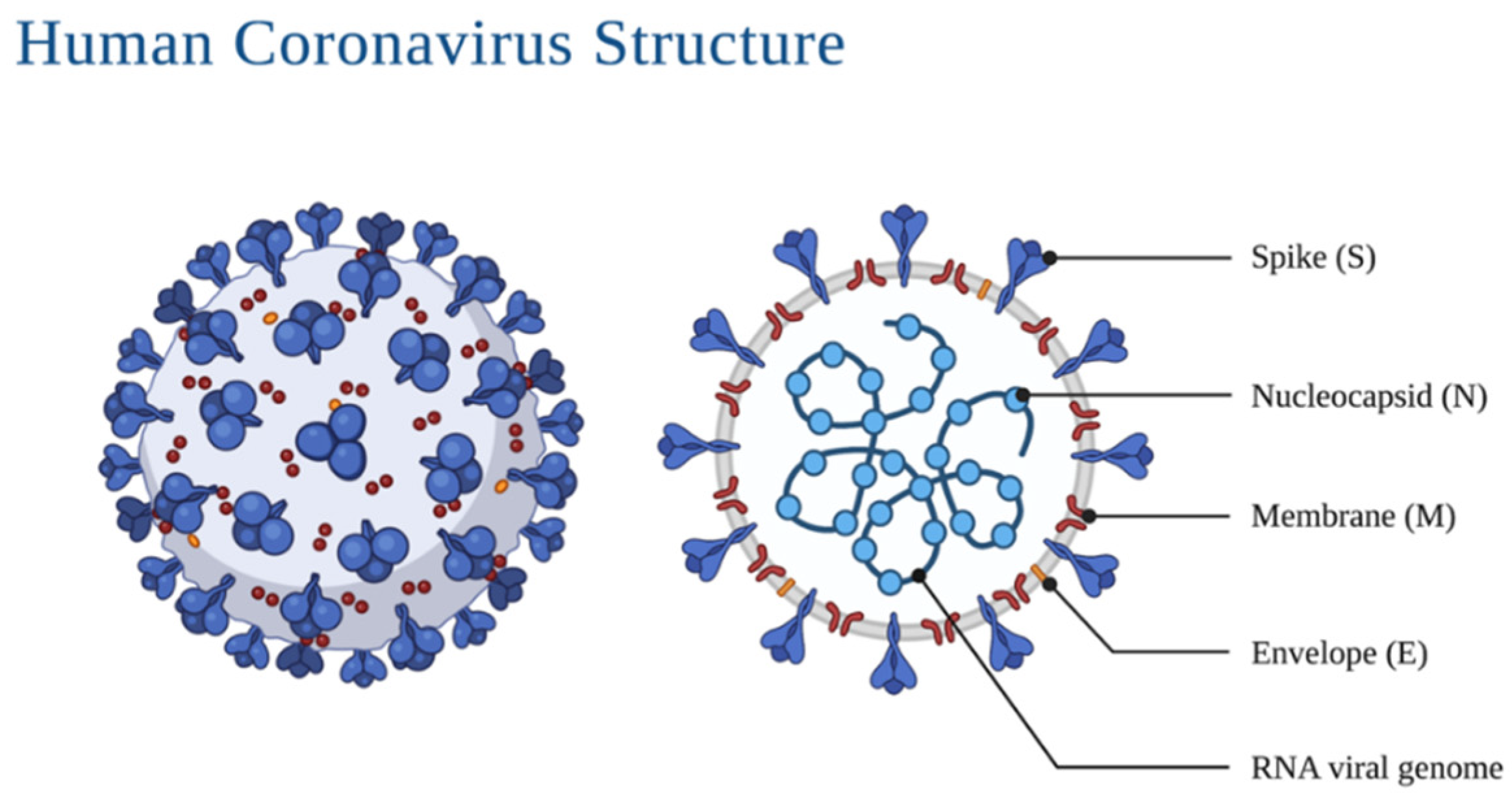
2. Morphology and Infection Route of Coronavirus
3. Antiviral Therapies and Vaccine Strategies
| Rug Name | Main Function | Permission Sought | Reference |
|---|---|---|---|
| Veklury® (remdesivir) | After conversion to remdesivir monophosphate, this compound stalls viral RNA-polymerase | EU and US | [16] |
| Olumiant® (baricitinib) | This tyrosine kinase regulates the immune response of the body, avoiding a cytokine storm | EU and US | [17] |
| Kineret® (anakinra) | This recombinant interleukin (IL)-1 receptor antagonist helps control the hyperinflammatory syndrome of COVID-19 | EU | [18] |
| RoActemra® Actemra® (tocilizumab) | Recombinant humanized monoclonal antibody that prevents the binding of IL-6 to its receptor, inhibiting the inflammatory cascade | EU and FDA | [19] |
| Bamlanivimab and etesevimab | Neutralizing monoclonal antibodies that target the spike glycoprotein of the virus rendering it impossible to enter the host cell | EU and US | [20] |
| Regdanvimab | Monoclonal antibodies which also reduce viral entry in host cells | EU | [21] |
| REGN-COV-2® Casirivimab/imdevimab | Monoclonal antibodies that also target the spike glycoprotein | US | [22] |
| Sotrovimab® (VIR-7831) | Monoclonal antibodies that reduce viral entry into host cells and help clear infected cells | EU and US | [23] |
4. Plant Metabolites: Debunking Misconceptions
5. Antiviral Activity of Natural Sources
| Phenolic Compounds/Class | Type of Compound/Extract | Extraction Methods | Virus | Reference |
|---|---|---|---|---|
| Phenolic glucosides | Ethanol extract from the leaves of the poplar tree cultivar Beaupré (Populus trichocarpa) | Ethanol extraction of the hot water-soluble portion followed by polyamide chromatography employing step-gradient elution with water and dilutions of ethanol | Poliomyelitis virusSemliki forest virus | [46] |
| p-Quinone monooximes derived from 3-methoxyphenol - 8-hydroxyquinoline | Synthetic aromatic nitro compounds | Extract washed with brine, dried over magnesium sulfate and filtered. Filtrate evaporated to dryness under reduced pressure. i-AmNO2 added to a stirred solution of a phenol in DMF in the presence of K2CO3 at 0 °C under argon. Recrystallization of the crude product from an appropriate solvent gave a p-quinone monooxime | Herpes simplex virus type 1 (HSV-1) | [51] |
| Alkyl-esters of gallic acid 3,4,5-trihydroxy derivatives of gallic acid 3,4,5-trimethoxy derivatives of gallic acid Catechin Epicatechin Quercetin Epigalocatechin | Commercial standard and lab synthesis | - | Rabies virus | [47] |
| Apigenin Naringin Atropine Genistein Gallic acid Chlorogenic acid Quinic acid | Commercial standard | Compounds dissolved in dimethyl sulfoxide to prepare a final concentration of 256 μg/mL | Herpes simplex virus type 1 (HSV-1) Parainfluenza virus type 3 (PI-3) | [49] |
| Apigenin Acacetin 7-O-[4′′′-O-acetyl-β-d-apiofuransyl-(1→3)]-β-d-xylopyranoside | Origanum vulgare L. plant extracted with 95% (v/v) ethanol | Air-dried plants percolated with 95% ethanol solution. Ethanol extract concentrated in vacuum to yield a residue, which was suspended in water and partitioned with petroleum ether and ethyl acetate, respectively | Herpes simplex virus type 1 (HSV-1) Respiratory syncytial virus (RSV) | [50] |
| Kaempferol Kaempferol-7-O-glucoside | Hydromethanolic extracts (98%) from Securigera securidaca seeds | Dried seeds extracted methanol (98%) at 40 °C. The methanol extract was eluted with n-hexane: acetone and then with 100% methanol | HIV-1 | [48] |
| Quercetin Kaempferol-3-O-(6″-O-E-p-coumaroyl)-β-d-glucopyranoside Mangiferin | Bombax ceiba L. Flowers extracted with 95% (v/v) ethanol | Extraction by reflux with 95% ethanol | Respiratory syncytial virus (RSV) | [52] |
| Xanthopurpurin (1,3-dihydroxy-9,10-anthracenedione) Vanillic acid (4-hydroxy-3-methoxybenzoic acid) | Aqueous extract from Rubia cordifolia L. aerial parts | Plant aerial parts boiled in distilled water for 1 h, the aqueous solution collected and the residual part re-extracted several times | Rotavirus | [47] |
| Catechin Gallic acid | Commercial standards | - | H1N1 Influenza virus | [55] |
| Gallic acid Ellagic acid | Aqueous and organic-solvent extracts of Rhodiola rosea L. plant | Extracts dried in vacuum at 50 °C and dissolved in DMSO | Ebola virus | [1] |
| Quercetin 3-glucoside | Hydromethanolic (70% methanol) and methanol (100%) extracts from Dianthus superbus L. leaves | Dried leaves extracted using 70% and 100% methanol | H1N1 Influenza | [53] |
| Hibiscus acid Protocatechuic acid | Acidic hibiscus tea extract | Hibiscus tea powder soaked in ultrapure water at 24 °C for 24 h, repeating the process several times | H1N1 Influenza virus | [54] |
| Phenolic Compounds/Class | Type of Extract | Extraction Methods | Concentration | Reference |
|---|---|---|---|---|
| Tetra-O-galloyl-beta-D-glucose (TGG) Luteolin | Hydroethanolic extract | Herbs extracted by maceration with 85% ethanol at room temperature for 2 weeks | 4.5 µM 83.4 µM | [59] |
| Aloe emodin Hesperetin | Aqueous extract from Isatis indigotica roots | Plant roots extracted twice with 10 volumes of distilled boiling water for 1 h | 132 μM, 366 μM and 911.592 μM 60 μM, 8.3 μM and 2718 μM | [60] |
| Saikosaponins (A, B2, C and D) | Commercial standards | Saikosaponins dissolved in DMSO and further diluted with RPMI 1640 medium | 25 µmmol | [70] |
| A. emodin | Aqueous extract of the root tuber from Rheum officinale Baill. and Polygonum multiflorum Thunb. | Deionized water | IC50 values ranged from 1 to 10 μg/mL. | [61] |
| Flavonoids from a Chinese multiherb remedy constituted by Herba Houttuyniae, Flos Chrysanthemi Indici, Herba Artemisiae Scopariae, Herba Eupatorii and Fructus Tsaoko | Hydroethanolic extract | Herb mixture extracted with 95% EtOH at room temperature | From the nine flavonoids with inhibitory effects, luteolin was the most potent with a CH50 (50% inhibitory concentration) of 0.19 mM | [71] |
| n.a. | Hydroethanolic extracts of Rheum palmatum L. (ethanol (75%)) | Petroleum ether and chloroform, and ethyl acetate | Among the extracts, RH121 has the highest activity, with an IC50 of 13.76 μg/mL | [67] |
| Tanshinones | Hydroethanolic extract (95%) of the dried roots of Salvia miltiorrhiza Bunge | The crude extract was filtered and evaporated under reduced pressure. The obtained residue was suspended with distilled water | IC50 value of 0.7µM | [65] |
| Phlorotannins | Ethanol extract of brown alga Ecklonia cava Kjellman | Ethanol at room temperature | Of the nine phlorotannins tested, two eckol groups with a diphenyl ether linked dieckol showed the most potent SARS-CoV 3CLpro trans/cis-cleavage inhibitory effects; IC50 = 2.7 and 68.1 μM, respectively) | [62] |
| Flavonoids (bavachinin, neobavaisoflavone, isobavachalcone, 4′-O-methylbavachalcone, psoralidin and corylifol A) | Fractionation of hydroethanolic extract from the seeds of Psoralea corylifolia L. | Ethanol, water and n-hexane | IC50 ranging between 4.2 and 38.4 µM | [68] |
| Juglanin Kaempferol-3-O-α-rhamnopyranosyl(1→2) [α-rhamnopyranosyl(1→6)]-β-glucopyranoside | Kaempferol acylated glucosides were previously isolated from polar extracts from the leaves of the plant Quercus ilex L.; the kaempferol triglycoside was an isolate from Viola odorata L (decoction and infusion) | Cyclohexane, Et2O, MeOH and MeOH–H2O | The most effective one was the glycoside juglanin with an IC50 of 2.3 µM | [69,72] |
| Alkylated chalcones | Hydroethanolic extract from the leaves of Angelica keiskei Ito | 95% ethanol for a week at room temperature | IC50 values of 11.4 (3CLpro) and 1.2 µM (PLpro) | [63] |
| Broussochalcone B, Broussochalcone A, 4-hydroxyisolonchocarpin, Papyriflavonol A, 3′-(3-methylbut-2-enyl)-3′,4,7-trihydroxyflavane, Kazinol A, Kazinol B, Broussoflavan A, Kazinol F, Kazinol J | Ethanolic extracts from the roots of Broussonetia papyrifera (L.) Vent. | Ethanol at room temperature and evaporated using a rotary evaporator at temperatures below 45 °C to obtain the total extract. | Papyriflavonol A was the most potent inhibitor of PLpro with an IC50 of 3.7 μM. | [64] |
| Epicatechin 5-O-beta-D-glucopyranoside-3-benzoate Neohesperidin Kaempferol-3,7-O-dirhamnoside (Kaempferitrin) Quercetin-3-O-neohesperidoside Oleic acid 3,4,5-Trimethoxyphenol Epicatechin Oxypeucedanin hydrate 3-Feruloylquinic acid Eriodictyol Apigenin Luteolin C-Methyl flavone β-amyrin Isovitexin-2″-O-rhamnoside | Methanolic extracts from Fragaria ananassa Duch. | 80% methanol at room temperature. | Strawberry methanolic extract showed the highest antiviral activity against SARS-CoV-2 with an IC50 value to 0.0062 µg/mL | [2] |
| Sinapic acid | Ethanolic extract of broccoli (obtained from a local market) | 95% ethanol | Potent SARS CoV-2 inhibition with a half-maximal inhibitory concentration (IC50) value of 2.69 µg/mL | [73] |
| Derivatives of luteolin, kaempferol, apigenin, isorhamnetin, myricetin, chrysoeriol, biochanin, isookanin and scutellarein | V. vinifera (var. Paulsen 1103) leaf extract | 75% (v/v) methanol/0.05% (v/v) trifluoroacetic acid | Leaf extract was able to inhibit both HSV-1 and SARS-CoV-2 replication in the early stages of infection by directly blocking the proteins enriched on the viral surface, at a very low concentration of 10 µg/mL | [74] |
| TFC of sampled wild S. nigra was 9.57 ± 0.65 mg RE g−1 DW of plant material for berry extracts and 77.59 ± 10.23 mg RE g−1 DW of plant material for flower extracts. TPC of sampled wild S. nigra was 41.31 ± 9.44 mg gallic acid equivalent (GAE) g−1 DW for berry extracts and 451.72 ± 25.31 mg GAE g−1 DW for flower extracts | Ethanolic Extract of Sambucus nigra L. berry and flowers | 80 % ethanol at 60 °C | Concentration-dependent inhibition of ACE2-SARS-CoV2 S-protein RBD binding was demonstrated in vitro for elderberry fruits and flowers extracts (IC50 of 1.66 mg DW ml−1 and 0.532 mg DW ml−1, respectively) | [75] |
| Caffeic acid, caftaric acid, chlorogenic acid, cichoric acid, cynarin, echinacoside | Echinacea purpurea (echinaforce) | 65% ethanol | 50 µg/mL inactivation of SARS-CoV1 and 2 | [76] |
| Glyzyrrhizin | Glycyrrhiza glabra | Acquired compound | 300 mg/L | [77] |
| Baicalein | Scutellaria baicalensis | 70% ethanol (crude extract of plant) and commercial baicalein | IC50 8.52 µg/mL (crude extract) and 0.39 µg/mL (baicalein) | [78] |
| Epigallocatechin, genistein, sulforaphane, chlorogenic acid, resveratrol and quercetin | - | Acquired compounds | IC50—33.9 µg/mL (epigallocatechin) | [79] |
| Whole plant extract | Taraxacum officinale | Aqueous extractions for 1 h | EC50—14.9 mg/mL | [80] |
6. Coronavirus Inhibition Assays
7. Future and Trends
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Adedeji, A.O.; Severson, W.; Jonsson, C.; Singh, K.; Weiss, S.R.; Sarafianos, S.G. Novel inhibitors of severe acute respiratory syndrome coronavirus entry that act by three distinct mechanisms. J. Virol. 2013, 87, 8017–8028. [Google Scholar] [CrossRef] [Green Version]
- Al-Sanea, M.M.; Abelyan, N.; Abdelgawad, M.A.; Musa, A.; Ghoneim, M.M.; Al-Warhi, T.; Aljaeed, N.; Alotaibi, O.J.; Alnusaire, T.S.; Abdelwahab, S.F.; et al. Strawberry and ginger silver nanoparticles as potential inhibitors for SARS-CoV-2 sssisted by in silico modeling and metabolic profiling. Antibiotics 2021, 10, 824. [Google Scholar] [CrossRef]
- Artika, I.M.; Dewantari, A.K.; Wiyatno, A. Molecular biology of coronaviruses: Current knowledge. Heliyon 2020, 6, e04743. [Google Scholar] [CrossRef] [PubMed]
- Berger, A.; Drosten, C.; Doerr, H.; Stürmer, M.; Preiser, W. Severe acute respiratory syndrome (SARS)-paradigm of an emerging viral infection. J. Clin. Virol. 2004, 29, 13–22. [Google Scholar] [CrossRef]
- Raj, V.S.; Osterhaus, A.D.; Fouchier, R.A.; Haagmans, B.L. MERS: Emergence of a novel human coronavirus. Curr. Opin. Virol. 2014, 5, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Harrison, A.G.; Lin, T.; Wang, P. Mechanisms of SARS-CoV-2 transmission and pathogenesis. Trends Immunol. 2020, 41, 1100–1115. [Google Scholar] [CrossRef]
- World Health Organization—WHO. Middle East Respiratory Syndrome Coronavirus (MERS-CoV). 2020. Available online: https://www.who.int/news/item/27-07-2017-countries-agree-next-steps-to-combat-global-health-threat-by-mers-cov/ (accessed on 26 July 2021).
- Chen, Z.; Boon, S.S.; Wang, M.H.; Chan, R.W.Y.; Chan, P.K.S. Genomic and evolutionary comparison between SARS-CoV-2 and other human coronaviruses. J. Virol. Methods 2021, 289, 114032. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Nyodu, R.; Maurya, V.K.; Saxena, S.K. Chapter 3—Morphology, genome organization, replication, and pathogenesis of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). In Coronavirus Disease 2019 (COVID-19); Saxena, S.K., Ed.; Springer: Singapore, 2020. [Google Scholar]
- Schoeman, D.; Fielding, B.C. Coronavirus envelope protein: Current knowledge. Virol. J. 2019, 16, 69. [Google Scholar] [CrossRef] [Green Version]
- De Wit, E.; Doremalen, N.; Falzarano Munster, V.J. SARS and MERS: Recent insights into emerging coronaviruses. Nat. Rev. Microbiol. 2016, 14, 523–534. [Google Scholar] [CrossRef]
- Low, J.S.; Jerak, J.; Tortorici, M.A.; McCallum, M.; Pinto, D.; Cassotta, A.; Foglierini, M.; Mele, F.; Abdelnabi, R.; Weynand, B.; et al. ACE-2-biinding exposes the SARS-CoV-2 fusion peptide to broadly neutralizing coronavisus antibodies. Science 2022, 377, 735–742. [Google Scholar] [CrossRef]
- Munir, M.; Tandiabang, P.; Setyawati, T.; Basry, A.; Cyio, A.; Rahman, N. Bioethical perspective of convalescent plasma therapy for COVID-19: A systematic review. Transfus. Clin. Et Biol. 2021, 28, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Alba, E.; Nuzzolo-Shihadeh, L.; Aguirre-García, G.M.; Espinosa-Mora, J.; Lecona-Garcia, J.D.; Flores-Pérez, R.O.; Mendoza-Garza, M.; Camacho-Ortiz, A. Baricitinib plus dexamethasone compared to dexamethasone for the treatment of severe COVID-19 pneumonia: A retrospective analysis. J. Microbiol. Immunol. Infect. 2021, 54, 787–793. [Google Scholar] [CrossRef] [PubMed]
- Snow, T.A.C.; Saleem, N.; Ambler, G.; Nastouli, E.; McCoy, L.E.; Singer, M.; Arulkumaran, N. Convalescent plasma for COVID-19: A meta-analysis, trial sequential analysis, and meta-regression. Br. J. Anaesth. 2021, 127, 834–844. [Google Scholar] [CrossRef] [PubMed]
- Bravo, J.P.K.; Dangerfield, T.L.; Taylor, D.W.; Johnson, K.A. Remdesivir is a delayed translocation inhibitor of SARS-CoV-2 replication. Mol. Cell 2021, 81, 1548–1552. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.R.; Zhang, Y.N.; Li, X.D.; Zhang, H.Q.; Xiao, S.Q.; Deng, F.; Yuan, Z.M.; Ye, H.Q.; Zhang, B. A cell-based large-scale screening of natural compounds for inhibitors of SARS-CoV-2. Signal Transduct. Target. Ther. 2020, 5, 218. [Google Scholar] [CrossRef]
- Kyriazopoulou, E.; Huet, T.; Cavalli, G.; Gori, A.; Kyprianou, M.; Pickkers, P.; Eugen-Olsen, J.; Clerici, M.; Veas, F.; Chatellier, G.; et al. Effect of anakinra on mortality in patients with COVID-19: A systematic review and patient-level meta-analysis. Lancet Rheumatol. 2021, 10, E690–E697. [Google Scholar] [CrossRef]
- Taramasso, L.; Magnasco, L.; Portunato, F.; Briano, F.; Vena, A.; Giacobbe, D.R.; Dentone, C.; Robba, C.; Ball, L.; Loconte, M.; et al. Clinical presentation of secondary infectious complications in COVID-19 patients with intensive care unit treated with tocilizumab or standard of care. Eur. J. Intern. Med. 2021, 94, 39–44. [Google Scholar] [CrossRef]
- Dougan, M.; Nirula, A.; Azizad, M.; Mocherla, B.; Gottlieb, R.L.; Chen, P.; Hebert, C.; Perry, R.; Boscia, J.; Heller, B.; et al. Bamlanivimab plus etesevimab in mild or moderate Covid-19. N. Engl. J. Med. 2021, 385, 1382–1392. [Google Scholar] [CrossRef]
- Kim, J.Y.; Jang, Y.R.; Hong, J.H.; Jung, J.G.; Park, J.; Streinu-Cercel, A.; Streinu-Cercel, A.; Săndulescu, O.; Lee, S.J.; Kim, S.H.; et al. Safety, virologic efficacy, and pharmacokinetics of CT-P59, a neutralizing monoclonal antibody against SARS-CoV-2 spike receptor-binding protein: Two randomized, placebo-controlled phase 1 studies in healthy subjects and patients with mild SARS-CoV-2 infection. Clin. Ther. 2021, 43, 1706–1727. [Google Scholar]
- Razonable, R.R.; Pawlowski, C.; O'Horo, J.C.; Arndt, L.L.; Arndt, R.; Bierle, D.M.; Borgen, M.D.; Hanson, S.N.; Hedin, M.C.; Lenehan, P.; et al. Casirivimab-Imdevimab treatment is associated with reduced rates of hospitalization among high-risk patients with mild to moderate coronavirus disease-19. EClinicalMecdicine 2021, 40, 101102. [Google Scholar] [CrossRef]
- Baral, P.K.; Yin, J.; James, M.N.G. Treatment and prevention strategies for the COVID 19 pandemic: A review of immunotherapeutics approaches for neutralizing SARS-CoV-2. Int. J. Biol. Macromol. 2021, 186, 490–500. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Hofmann-Winkler, H.; Krüger, N.; Kempf, A.; Nehlmeier, I.; Graichen, L.; Arora, P.; Sidarovich, A.; Moldenhauer, A.-S.; Winkler, M.S.; et al. SARS-CoV-2 variant B.1.617 is resistant to bamlanivimab and evades antibodies induced by infection and vaccination. Cell Rep. 2021, 36, 109415. [Google Scholar] [CrossRef] [PubMed]
- Rosa, S.S.; Prazeres, D.M.F.; Azevedo, A.M.; Marques, M.P.C. mRNA vaccines manufacturing: Challenges and bottlenecks. Vaccine 2021, 39, 2190–2200. [Google Scholar] [CrossRef] [PubMed]
- Nagy, A.; Alhatlani, B. An overview of current COVID-19 vaccine platforms. Comput. Struct. Biotechnol. J. 2021, 19, 2508–2517. [Google Scholar] [CrossRef]
- Ceron, W.; De-Lima-Santos, M.-F.; Quiles, M.G. Fake news agendas in the era of COVID-19: Identifying trends through fact-checking content. Online Soc. Netw. Media 2021, 21, 100116. [Google Scholar] [CrossRef]
- Gautam, S.; Gautam, A.; Chhetri, S.; Bhattarai, U. Immunity against COVID-19: Potential role of Ayush Kwath. J. Ayurveda Integr. Med. 2020, 13, 100350. [Google Scholar] [CrossRef]
- Hu, H.; Ji, Z.; Feng, C.; Pang, W.; Chen, Z.; Zhang, J.; Wang, H. PROSPERO’s systematic review protocols of traditional Chinese medicine for COVID-19: An overview. Integr. Med. Res. 2021, 10, 100774. [Google Scholar] [CrossRef]
- Liang, S.-B.; Fang, M.; Liang, C.-H.; Lan, H.-D.; Shen, C.; Yan, L.-J.; Hu, X.-Y.; Han, M.; Robinson, N.; Liu, J.-P. Therapeutic effects and safety of oral Chinese patent medicine for COVID-19: A rapid systematic review and meta-analysis of randomized controlled trials. Complement. Ther. Med. 2021, 60, 102744. [Google Scholar] [CrossRef]
- Orisakwe, O.E.; Orish, C.N.; Nwanaforo, E.O. Coronavirus disease (COVID-19) and Africa: Acclaimed home remedies. Sci. Afr. 2020, 10, e00620. [Google Scholar] [CrossRef]
- Ren, W.; Liang, P.; Ma, Y.; Sun, Q.; Pu, Q.; Dong, L.; Luo, G.; Mazhar, M.; Liu, J.; Wang, R.; et al. Research progress of traditional Chinese medicine against COVID-19. Biomed. Pharmacother. 2021, 137, 111310. [Google Scholar] [CrossRef]
- Yin, X.; Cai, S.-B.; Tao, L.-T.; Chen, L.-M.; Zhang, Z.-D.; Xiao, S.-H.; Fan, A.Y.; Zou, X. Recovery of a patient with severe COVID-19 by acupuncture and Chinese herbal medicine adjuvant to standard care. J. Integr. Med. 2021, 19, 460–466. [Google Scholar] [PubMed]
- Zhu, W.; Xu, M.; Chen, C.Z.; Guo, H.; Shen, M.; Hu, X.; Shinn, P.; Klumpp-Thomas, C.; Michael, S.G.; Zheng, W. Identification of SARS-CoV-2 3CL protease inhibitors by a quantitative high-throughput screening. ACS Pharmacol. Transl. Sci. 2020, 3, 1008–1016. [Google Scholar] [CrossRef] [PubMed]
- Feyaerts, A.F.; Luyten, W. Vitamin C as prophylaxis and adjunctive medical treatment for COVID-19? Nutrition 2020, 79, 110948. [Google Scholar] [CrossRef] [PubMed]
- Islam, A.T.M.R.; Ferdousi, J.; Shahinozzaman, M. Previously published ethnopharmacological reports reveal the potentiality of plants and plant-derived products used as traditional home remedies by Bangladeshi COVID-19 patients to combat SARS-CoV-2. Saudi J. Biol. Sci. 2021, 28, 6653–6673. [Google Scholar] [CrossRef] [PubMed]
- Lyu, M.; Fan, G.; Xiao, G.; Wang, T.; Xu, D.; Gao, J.; Ge, S.; Li, Q.; Ma, Y.; Zhang, H.; et al. Traditional chinese medicine in COVID-19. Acta Pharm. Sin. B 2021, 11, 3337–3363. [Google Scholar] [CrossRef] [PubMed]
- Shakoor, H.; Feehan, J.; Dhaheri, A.S.A.; Ali, H.I.; Platat, C.; Ismail, L.C.; Apostopoulos, V.; Stojanovska, L. Immune-boosting role of vitamins D, C, E, zinc, selenium and moega-3 fatty acids: Could they help against COVID-19? Maturitas 2021, 143, 1–9. [Google Scholar] [CrossRef]
- Gasmi, A.; Peana, M.; Noor, S.; Lysiuk, R.; Menzel, A.; Benahmed, A.G.; Bjørklund, G. Chloroquine and hydroxychloroquine in the treatment of COVID-19: The never-ending story. Appl. Microbiol. Biotechnol. 2021, 105, 1333–1343. [Google Scholar] [CrossRef]
- Di Stefano, L.; Ogburn, E.L.; Ram, M.; Scharfstein, D.O.; Li, T.; Khanal, P.; Baksh, S.N.; McBee, N.; Gruber, J.; Gildea, M.R.; et al. Hydroxychlorooquine/chloroquine for the treatment of hospitalized patients with COVID-19: An individual participant data meta-analysis. PLoS ONE 2022, 17, e0273526. [Google Scholar] [CrossRef]
- Réa-Neto, Á.; Bernardelli, R.S.; Câmara, B.M.D.; Reese, F.B.; Queiroga, M.V.O.; Oliveira, M.C. An open-label randomized controlled trial evaluating the efficacy of cholorquine/hydroxychloroquine in severe COVID-19 patients. Sci. Rep. 2021, 11, 9023. [Google Scholar] [CrossRef]
- Reis, G.; Silva, E.A.S.M.; Silva, D.C.M.; Thabane, L.; Milagres, A.C.; Ferreira, T.S.; Santos, C.V.Q.; Campos, V.H.S.; Nogueira, A.M.R.; Almeida, A.P.F.G.; et al. Effect of early treatment with ivermectin among patients with Codid-19. N. Engl. J. Med. 2022, 386, 1721–1731. [Google Scholar] [CrossRef]
- Bramante, C.T.; Huling, J.D.; Tignanelli, C.J.; Buse, J.B.; Liebovitz, D.M.; Nicklas, J.M.; Kenneth Cohen, K.; Puskarich, M.A.; Belani, H.K.; Proper, J.L.; et al. Randomized trial of metformin, ivermectin, and fluvoxamine for Covid-19. N. Engl. J. Med. 2022, 387, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Firn, R. Are NP’S different from synthetic chemicals? In Nature’s Chemicals: The Natural Products That Shaped Our World; Oxford University Press: Oxford, UK, 2010. [Google Scholar]
- Islam, M.T.; Sarkar, C.; El-Kersh, D.M.; Jamaddar, S.; Uddin, S.J.; Shilpi, J.A.; Mubarak, M.S. Natural products and their derivatives against coronavirus: A review of the non-clinical and pre-clinical data. Phytother. Res. 2020, 34, 2471–2492. [Google Scholar] [CrossRef] [PubMed]
- Van Hoof, L.; Totté, J.; Corthout, J.; Pieters, L.; Mertens, F.; Berghe, D.A.V.; Vlietinck, A.J.; Dommisse, R.; Esmans, E. Plant Antiviral Agents, VI. Plant Antiviral Agents, VI. Isolation of Antiviral Phenolic Glucosides from Populus Cultivar Beaupre by Droplet Counter-Current Chromatography. J. Nat. Prod. 1989, 52, 875–878. [Google Scholar]
- Chávez, J.H.; Leal, P.C.; Yunes, R.A.; Nunes, R.J.; Barardi, C.R.; Pinto, A.R.; Simões, C.M.; Zanetti, C.R. Evaluation of antiviral activity of phenolic compounds and derivatives against rabies virus. Vet. Microbiol. 2006, 116, 53–59. [Google Scholar] [CrossRef]
- Behbahani, M.; Sayedipour, S.; Pourazar, A.; Shanehsazzadeh, M. In vitro anti-HIV-1 activities of kaempferol and kaempferol-7-Oglucoside isolated from Securigera securidaca. Res. Pharm. Sci. 2014, 9, 463–469. [Google Scholar] [PubMed]
- Özçelik, B.; Kartal, M.; Orhan, I. Cytotoxicity, antiviral and antimicrobial activities of alkaloids, flavonoids, and phenolic acids. Pharm. Biol. 2011, 49, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-L.; Guo, Y.-S.; Wang, C.-H.; Li, G.-Q.; Xu, J.-J.; Chung, H.Y.; Ye, W.-C.; Li, Y.-L.; Wang, G.-C. Phenolic compounds from Origanum vulgare and their antioxidant and antiviral activities. Food Chem. 2014, 152, 300–306. [Google Scholar] [CrossRef]
- Ishikawa, T.; Watanabe, T.; Tanigawa, H.; Saito, T.; Kotake, K.-I.; Ohashi, Y.; Ishii, H. Nitrosation of phenolic substrates under mildly basic conditions: Selective preparation of p-quinone monooximes and their antiviral activities. J. Org. Chem. 1996, 61, 2774–2779. [Google Scholar] [CrossRef]
- Zhang, Y.-B.; Wu, P.; Zhang, X.-L.; Xia, C.; Li, G.-Q.; Ye, W.-C.; Wang, G.-C.; Li, Y.-L. Phenolic compounds from the flowers of Bombax malabaricum and their antioxidant and antiviral activities. Molecules 2015, 20, 19947–19957. [Google Scholar] [CrossRef] [Green Version]
- Nile, S.H.; Kim, D.H.; Nile, A.; Park, G.S.; Gansukh, E.; Kai, G. Probing the effect of quercetin 3-glucoside from Dianthus superbus L. against influenza virus infection- In vitro and in silico biochemical and toxicological screening. Food Chem. Toxicol. 2020, 135, 110985. [Google Scholar] [CrossRef]
- Takeda, Y.; Okuyama, Y.; Nakano, H.; Yaoita, Y.; Machida, K.; Ogawa, H.; Imai, K. Antiviral Activities of Hibiscus sabdariffa L. tea extract against human influenza a virus rely largely on acidic pH but partially on a low-pH-independent mechanism. Food Environ. Virol. 2019, 12, 9–19. [Google Scholar] [CrossRef] [PubMed]
- You, H.-L.; Huang, C.-C.; Chen, C.-J.; Chang, C.-C.; Liao, P.-L.; Huang, S.-T. Anti-pandemic influenza A (H1N1) virus potential of catechin and gallic acid. J. Chin. Med Assoc. 2018, 81, 458–468. [Google Scholar] [CrossRef] [PubMed]
- Nicolaou, K.C. Organic synthesis: The art and science of replicating the molecules of living nature and creating others like them in the laboratory. Proc. R. Soc. 2014, 479, 20130690. [Google Scholar] [CrossRef] [PubMed]
- Crane, E.A.; Gademann, K. Capturing biological activity in natural products fragments by chemical synthesis. Angew. Chem. Int. Ed. 2016, 55, 3882–3902. [Google Scholar] [CrossRef] [Green Version]
- Newman, D.J.; Cragg, G.M. Natural products as source of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef] [Green Version]
- Yi, L.; Li, Z.; Yuan, K.; Qu, X.; Chen, J.; Wang, G.; Zhang, H.; Luo, H.; Zhu, L.; Jiang, P.; et al. Small molecules blocking the entry of severe acute respiratory syndrome coronavirus into host cells. J. Virol. 2004, 78, 11334–11339. [Google Scholar] [CrossRef] [Green Version]
- Lin, C.-W.; Tsai, F.-J.; Tsai, C.-H.; Lai, C.-C.; Wan, L.; Ho, T.-Y.; Hsieh, C.-C.; Chao, P.-D.L. Anti-SARS coronavirus 3C-like protease effects of Isatis indigotica root and plant-derived phenolic compounds. Antivir. Res. 2005, 68, 36–42. [Google Scholar] [CrossRef]
- Ho, T.-Y.; Wu, S.-L.; Chen, J.-C.; Li, C.-C.; Hsiang, C.-Y. Emodin blocks the SARS coronavirus spike protein and angiotensin-converting enzyme 2 interaction. Antivir. Res. 2007, 74, 92–101. [Google Scholar] [CrossRef]
- Park, J.-Y.; Kim, J.H.; Kwon, J.M.; Kwon, H.-J.; Jeong, H.J.; Kim, Y.M.; Kim, D.; Lee, W.S.; Ryu, Y.B. Dieckol, a SARS-CoV 3CLpro inhibitor, isolated from the edible brown algae Ecklonia cava. Bioorganic Med. Chem. 2013, 21, 3730–3737. [Google Scholar] [CrossRef]
- Park, J.-Y.; Ko, J.A.; Kim, D.W.; Kim, Y.M.; Kwon, H.J.; Jeong, H.J.; Kim, C.Y.; Park, K.H.; Lee, W.S.; Ryu, Y.B. Chalcones isolated from Angelica keiskei inhibit cysteine proteases of SARS-CoV. J. Enzym. Inhib. Med. Chem. 2016, 31, 23–30. [Google Scholar] [CrossRef] [Green Version]
- Park, J.-Y.; Yuk, H.J.; Ryu, H.W.; Lim, S.H.; Kim, K.S.; Park, K.H.; Ryu, Y.B.; Lee, W.S. Evaluation of polyphenols from Broussonetia papyrifera as coronavirus protease inhibitors. J. Enzym. Inhib. Med. Chem. 2017, 32, 504–512. [Google Scholar] [CrossRef] [Green Version]
- Park, J.-Y.; Kim, J.H.; Kim, Y.M.; Jeong, H.J.; Kim, D.W.; Park, K.H.; Kwon, H.; Park, S.; Lee, W.S.; Ryu, Y.B. Tanshinones as selective and slow-binding inhibitors for SARS-CoV cysteine proteases. Bioorganic Med. Chem. 2012, 20, 5928–5935. [Google Scholar] [CrossRef] [PubMed]
- Bahbah, E.I.; Negida, A.; Nabet, M.S. Purposing saikosaponins for the treatment of COVID-19. Med. Hypotheses 2020, 140, 109782. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Su, X.; Gong, S.; Qin, Y.; Liu, W.; Li, J.; Yu, H.; Xu, Q. Anti-SARS coronavirus 3C-like protease effects of Rheum palmatum L. extracts. Biosci. Trends 2009, 3, 124–126. [Google Scholar] [PubMed]
- Kim, D.W.; Seo, K.H.; Curtis-Long, M.J.; Oh, K.Y.; Oh, J.W.; Cho, J.K.; Lee, K.H.; Park, K.H. Phenolic phytochemical displaying SARS-CoV papain-like protease inhibition from the seeds of Psoralea corylifolia. J. Enzym. Med. Chem. 2014, 29, 59–63. [Google Scholar] [CrossRef] [Green Version]
- Schwarz, S.; Sauter, D.; Wang, K.; Zhang, R.; Sun, B.; Karioti, A.; Bilia, A.R.; Efferth, T.; Schwarz, W. Kaempferol derivatives as antiviral drugs against the 3a channel protein of coronavirus. Planta Medica 2014, 80, 177–182. [Google Scholar] [CrossRef] [Green Version]
- Cheng, P.-W.; Ng, L.-T.; Chiang, L.-C.; Lin, C.-C. Antiviral effects of saikosaponins on human coronavirus 229E in vitro. Clin. Exp. Pharmacol. Physiol. 2006, 33, 612–616. [Google Scholar] [CrossRef]
- Zhang, T.; Chen, D. Anticomplementary principles of a Chinese multiherb remedy for the treatment and prevention of SARS. J. Ethnopharmacol. 2008, 117, 351–361. [Google Scholar] [CrossRef]
- Karioti, A.; Bilia, A.R.; Skaltsa, H. Quercus ilex L.: A rich source of polyacylated flavonoid glycosides. Food Chem. 2010, 123, 131–142. [Google Scholar] [CrossRef]
- Orfali, R.; Rateb, M.; Hassan, H.; Alonazi, M.; Gomaa, M.; Mahrous, N.; GabAllah, M.; Kandeil, A.; Perveen, S.; Abdelmohsen, U.; et al. Sinapic Acid Suppresses SARS CoV-2 Replication by Targeting Its Envelope Protein. Antibiotics 2021, 10, 420. [Google Scholar] [CrossRef]
- Zannella, C.; Giugliano, R.; Chianese, A.; Buonocore, C.; Vitale, G.A.; Sanna, G.; Sarno, F.; Manzin, A.; Nebbioso, A.; Termolino, P.; et al. Antiviral Activity of Vitis vinifera Leaf Extract against SARS-CoV-2 and HSV-1. Viruses 2021, 13, 1263. [Google Scholar] [CrossRef] [PubMed]
- Boroduske, A.; Jekabsons, K.; Riekstina, U.; Muceniece, R.; Rostoks, N.; Ilva, N. Wild Sambucus nigra L. from north-east edge of the species range: A valuable germplasm with inhibitory capacity against SARS-CoV2 S-protein RBD and hACE2 binding in vitro. Ind. Crops Prod. 2021, 165, 113438. [Google Scholar] [CrossRef] [PubMed]
- Signer, J.; Jonsdottir, H.R.; Albrich, W.C.; Strasser, M.; Züst, R.; Ryter, S.; Ackerman-Gäumann, R.; Lenz, N.; Siegrist, D.; Suter, A.; et al. In vitro virucidal activity of Echinaforce, an Echinacea purpurea preparation, against coronaviruses, including common cold coronavirus 229E and SARS-CoV-2. Virol. J. 2020, 17, 136. [Google Scholar] [CrossRef] [PubMed]
- Cinatl, J.; Morgenstern, B.; Bauer, G.; Chandra, P.; Rabenau, H.; Doerr, H. Glycyrrhizin, and active component of liquorice roots, and replication of SARS-associated coronavirus. Lancet 2003, 361, 2045–2046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, H.; Ye, F.; Sun, Q.; Liang, H.; Li, C.; Li, S.; Lu, R.; Huang, B.; Tna, W.; Lai, L. Scutellaria baicalensis extract and baicalein inhibit replication of SARS-CoV-2 and its 3C-like protease in vitro. J. Enzym. Inhib. Med. Chem. 2021, 36, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Ohishi, T.; Hishiki, T.; Baig, M.S.; Rajpoot, S.; Saqib, U.; Takasaki, T.; Hara, Y. Epigallocatechin gallate (EGCG) attenuates severe acute respiratory coronavirus disease 2 (SARS-CoV-2) infection by blocking the interaction of SARS-CoV-2 spike protein receptor-binding domain to human angiotensin-converting enzyme 2. PLoS ONE 2022, 17, e0271112. [Google Scholar] [CrossRef]
- Tran, H.T.T.; Gigl, M.; Le, N.P.K.; Dawid, C.; Lamy, E. In vitro effect of Taraxacum officinale lear aqueous extract on the interaction between ACE2 cell surface receptor and SARS-CoV-2 spike protein D614 and four mutants. Pharmaceuticals 2021, 14, 1055. [Google Scholar] [CrossRef]
- Touret, F.; Gilles, M.; Barral, K.; Nougairède, A.; van Helden, J.; Decroly, E.; de Lamballerie, X.; Coutard, B. In vitro screening of a FDA approved chemical library reveals potential inhibitors of SARS-CoV-2 replication. Sci. Rep. 2020, 10, 13093. [Google Scholar]
- Brown, A.S.; Ackerley, D.F.; Calcott, M.J. High-throughput screening for inhibitors of the SARS-CoV-2 protease using a FRET-biosensor. Molecules 2020, 25, 4666. [Google Scholar] [CrossRef]
- Jin, Z.; Du, X.; Xu, Y.; Deng, Y.; Liu, M.; Zhao, Y.; Zhang, B.; Li, X.; Zhang, L.; Peng, C.; et al. Structure of Mpro from SARS-CoV-2 and discovery of its inhibitors. Nature 2020, 582, 289–293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coelho, C.; Gallo, G.; Campos, C.B.; Hardy, L.; Würtele, M. Biochemical screening for SARS-CoV-2 main protease inhibitors. PLoS ONE 2020, 15, e0240079. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Zheng, W.; Huang, R. High-throughput screening assays for SARS-CoV-2 drug development: Current status and future directions. Drug Discov. Today 2021, 26, 2439–2444. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, Y.; Qiao, W.; Zhang, J.; Qi, Z. Baricitinib, a drug with potential effect to prevent SARS-CoV-2 from entering target cells and control cytokine storm induced by COVID-19. Int. Immunopharmacol. 2020, 86, 106749. [Google Scholar] [CrossRef]
- Zhao, J.; Guo, S.; Yi, D.; Li, Q.; Ma, L.; Zhang, Y.; Wang, J.; Li, X.; Guo, F.; Lin, R.; et al. A cell-based assay to discover inhibitors of SARS-CoV-2 RNA dependent RNA polymerase. Antivir. Res. 2021, 190, 105078. [Google Scholar] [CrossRef]
- Nie, J.; Li, Q.; Wu, J.; Zhao, C.; Hao, H.; Liu, H.; Zhang, L.; Nie, L.; Qin, H.; Wang, M.; et al. Establishment and validation of a pseudovirus neutralization assay for SARS-CoV-2. Emerg. Microbes Infect. 2020, 9, 680–686. [Google Scholar] [CrossRef] [Green Version]
- Govinda, K.C.; Giovanni, B.; Srijan, V.; Mahmudulla, H.; Jayme, H.; Jeremy, Y. A machine learning platform to estimate anti-SARS-CoV-2 activities. Nat. Mach. Intell. 2021, 3, 527–535. [Google Scholar]
- Lan, J.; Ge, J.; Yu, J.; Shan, S.; Zhou, H.; Fan, S.; Zhang, Q.; Shi, X.; Wang, Q.; Zhang, L.; et al. Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature 2020, 581, 215–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.-L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef] [Green Version]
- Xiu, S.; Dick, A.; Ju, H.; Mirzaie, S.; Abdi, F.; Cocklin, S.; Zhan, P.; Liu, X. Inhibitors of SARS-CoV-2 entry: Current and future opportunities. J. Med. Chem. 2020, 63, 12256–12274. [Google Scholar] [CrossRef]
- Lundin, A.; Dijkman, R.; Bergström, T.; Kann, N.; Adamiak, B.; Hannoun, C.; Kindler, E.; Jónsdóttir, H.R.; Muth, D.; Kint, J.; et al. Targeting membrane-bound viral RNA synthesis reveals potent inhibition of diverse coronaviruses including the middle east respiratory syndrome virus. PLoS Pathol. 2014, 10, e1004166. [Google Scholar] [CrossRef] [Green Version]
- Gurevich, E.V.; Gurevich, V.V. Therapeutic potential of small molecules and engineered proteins. In Arrestins-Pharmacology and Therapeutic Potential; Springer: Berlin/Heidelberg, Germany, 2014; pp. 1–12. [Google Scholar]
- Ngo, H.X.; Garneau-Tsodikova, S. What are the drugs of the future? MedChemComm 2018, 9, 757–758. [Google Scholar] [CrossRef] [PubMed]
- Milewska, A.; Ciejka, J.; Kaminski, K.; Karewicz, A.; Bielska, D.; Zeglen, S.; Wojciech, K.; Nowakowska, M.; Potempa, J.; Bosch, B.J.; et al. Novel polymeric inhibitors of HCoV-NL63. Antivir. Res. 2013, 97, 112–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitsuki, Y.Y.; Ohnishi, K.; Takagi, H.; Oshima, M.; Yamamoto, T.; Mizukoshi, F.; Terahara, K.; Kobayashi, K.; Yamamoto, N.; Yamaoka, S.; et al. A single amino acid substitution in the S1 and S2 Spike protein domains determines the neutralization escape phenotype of SARS-CoV. Microbes Infect. 2008, 10, 908–915. [Google Scholar] [CrossRef] [PubMed]
- Wahedi, H.M.; Ahmad, S.; Abbasi, S.W. Stilbene-based natural compounds as promising drug candidates against COVID-19. J. Biomol. Struct. Dyn. 2020, 39, 3225–3234. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Rivera, O.A.; Shukla, S.; Shin, M.D.; Chen, A.; Beiss, V.; Moreno-Gonzalez, M.A.; Zheng, Y.; Clark, A.E.; Carlin, A.F.; Pokorski, J.K.; et al. Cowpea mosaic virus nanoparticle vaccine candidate displaying peptide epitopes can neutralize the severe acute respiratory syndrome coronavirus. ACS Infect. Dis. 2021, 7, 3096–3110. [Google Scholar] [CrossRef]
- Patrick, R.; Garad, R.; Snell, T.; Enticott, J.; Meadows, G. Australians report climate change as a bigger concern than COVID-19. J. Clim. Chang. Health 2021, 3, 100032. [Google Scholar] [CrossRef]
- Beyer, R.M.; Manica, A.; Mora, C. Shifts in global bat diversity suggest a possible role in climate change in the emergence of SARS-CoV-1 and SARS-CoV-2. Sci. Total Environ. 2021, 767, 145413. [Google Scholar] [CrossRef]
- Galeotti, C.; Bayry, J. Autoimmune and inflammatory diseases following COVID-19. Nat. Rev. Rheumatol. 2020, 16, 413–414. [Google Scholar] [CrossRef]
- Toscano, G.; Palmerini, F.; Ravaglia, S.; Ruiz, L.; Invernizzi, P.; Cuzzoni, M.G.; Baldanti, F.; Daturi, R.; Postorino, P.; Cavallini, A.; et al. Guillain-Barré syndrome associated with SARS-CoV-2. N. Engl. J. Med. 2020, 382, 2574–2576. [Google Scholar] [CrossRef]
- Zhou, Y.; Han, T.; Chen, J.; Hou, C.; Hua, L.; He, S.; Guo, Y.; Zhang, S.; Wang, Y.; Yuan, J.; et al. Clinical and autoimmune characteristics of severe and critical cases of COVID-19. Clin. Transl. Sci. 2020, 13, 1077–1086. [Google Scholar] [CrossRef]



| Vaccine | Type | Manufacturer | Approval |
|---|---|---|---|
| Approved | |||
| Comirnaty | mRNA | Pfizer-BioNTech | EU and US |
| Spikevax (former Moderna) | mRNA | Moderna | EU and US |
| Vaxzevria (former AstraZeneca) | Modified adenovirus | AstraZeneca | EU |
| Janssen | Modified adenovirus | Janssen | EU and US |
| Novavax | Spike glycoprotein subunit | Novavax | EU and US |
| Under Rolling Review | |||
| Sanofi (Vidprevtyn) | Spike glycoprotein subunit | Sanofi-GSK | Sought in EU |
| HIPRA Human Health | Spike glycoprotein subunit | HIPRA | Sought in the EU |
| Valneva | Inactivated coronavirus | Valneva | Sought in the EU |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heleno, S.A.; Carocho, M.; Reis, F.S.; Pires, T.C.S.P.; Pintado, M.; Ferreira, I.C.F.R.; Barros, L. Plant Extracts and SARS-CoV-2: Research and Applications. Life 2023, 13, 386. https://doi.org/10.3390/life13020386
Heleno SA, Carocho M, Reis FS, Pires TCSP, Pintado M, Ferreira ICFR, Barros L. Plant Extracts and SARS-CoV-2: Research and Applications. Life. 2023; 13(2):386. https://doi.org/10.3390/life13020386
Chicago/Turabian StyleHeleno, Sandrina A., Marcio Carocho, Filipa S. Reis, Tânia C. S. P. Pires, Manuela Pintado, Isabel C. F. R. Ferreira, and Lillian Barros. 2023. "Plant Extracts and SARS-CoV-2: Research and Applications" Life 13, no. 2: 386. https://doi.org/10.3390/life13020386









