Eutherian-Specific Functions of BetaM Acquired through Atp1b4 Gene Co-Option in the Regulation of MyoD Expression
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Animals
2.2. Cell Culture and Transfections
2.3. Protein Isolation and Western Blotting
2.4. RNA Extraction and Reverse-Transcription-PCR (RT-PCR)
- MyoD:
- Forward: GCA GGC TCT GCT GCG CGA CC
- Reverse: TGT AAT CCA TCA TGC CAT
- BetaM:
- Forward: GAG CTT GGA GAT CCT GTG AAG G
- Reverse: GGA GGT CAA AAG AAG CCG ACT
- SKIP
- Forward: TGA CCA AAG GCT CTT CAA CCA
- Reverse: GCC ATA TCT TTC CCA CCT CTC C
- GAPDH:
- Forward: TGC ACC ACC AAC TGC TTA G
- Reverse: GAG GCA GGG ATG ATG TTC
2.5. Luciferase Assays
2.6. MyoD DRR Luciferase Deletion Constructs
- Ebox-4 Deletion:
- Forward: GGG CAG GGT GCG TGA AGG GTT TCC AGA GGC TAT ATA TAT A
- Reverse: TAT ATA TAT AGC CTC TGG AAA CCC TTC ACG CAC CCT GCC C
- CArG Deletion:
- Forward: CAC ATT CCT TTC CAG AGG GCA GCC AAG GGA GCT GAG AGG G
- Reverse: CCC TCT CAG CTC CCT TGG CTG CCC TCT GGA AAG GAA TGT G
2.7. Chromatin Immunoprecipitation Assays (ChIP)
- Rat PRR
- Forward TAG GCA CTG GAG AGA CTT GG
- Reverse GCC TCA AGC CAA TAG GAG TGT AG
- Rat DRR2
- Forward TAG ACA CAA GCC AGC AAT GC
- Reverse TAT AAA TGG AGA GCT GGC TT
- Rat CE
- Forward ACA TGA GCC CCA CAG CAT TTG
- Reverse GAG CTA GAG AAA CCG GAG AAG A
- Rat GAPDH
- Forward CAT TAA CGT CAA CTA CAT GG
- Reverse TGA TGA CCA GCT TCC CAT TCT CAG C
- Mouse DRR
- Forward GGGCTGGTCCTGTTCCACC
- Reverse GCTATAAATGGAGAGCTGGCTTTT
- Mouse IgH enhancer
- Forward GCCGATCAGAACCAGAACACC
- Reverse TGGTGGGGCTGGACAGAGTGTTTC
2.8. Electrophoretic Mobility Shift Assays (EMSA)
2.9. Statistical Analysis
3. Results
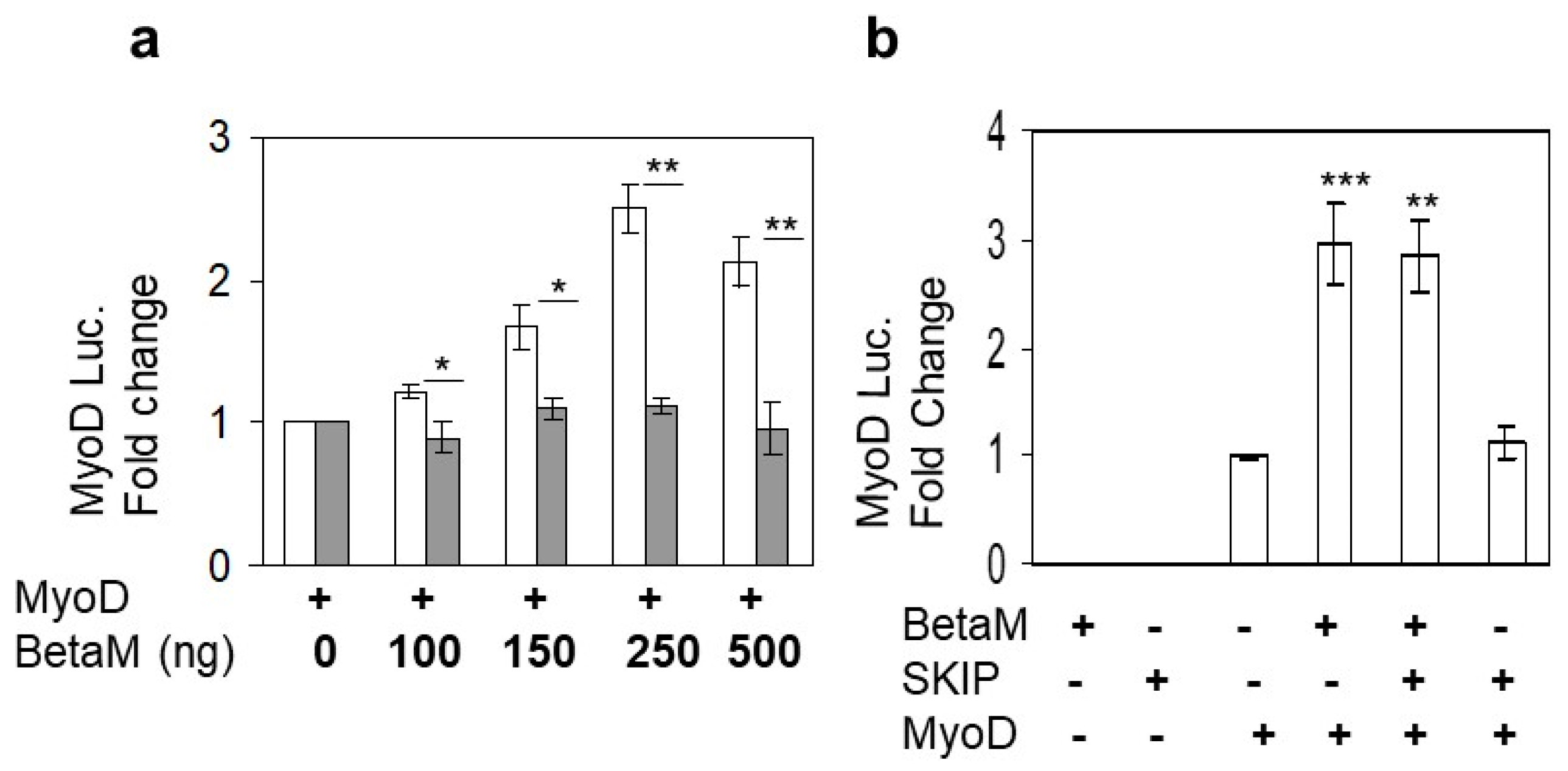
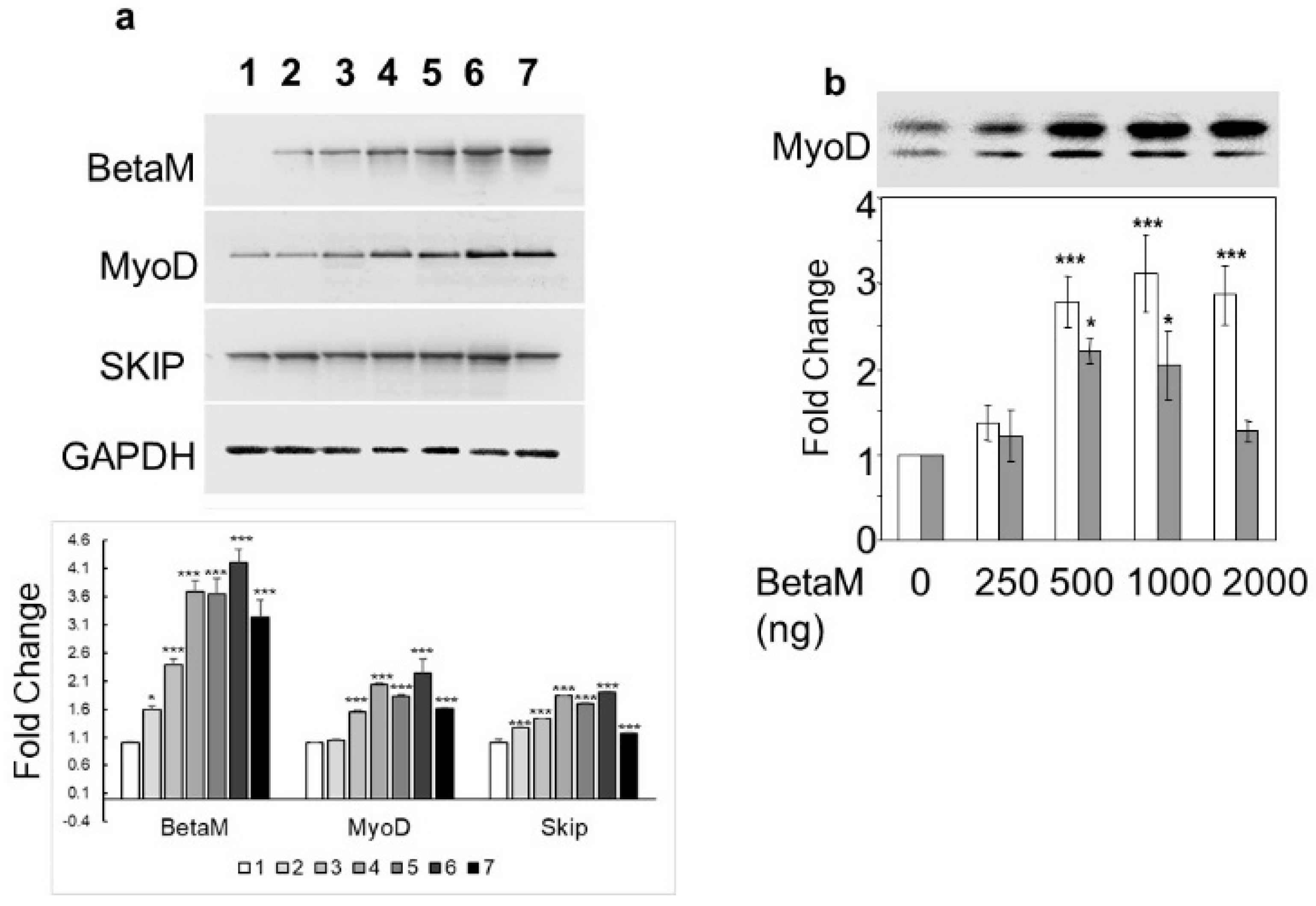
3.1. Beta M Promotes Expression of the Muscle Determining Factor, MyoD in C2C12 Myoblasts
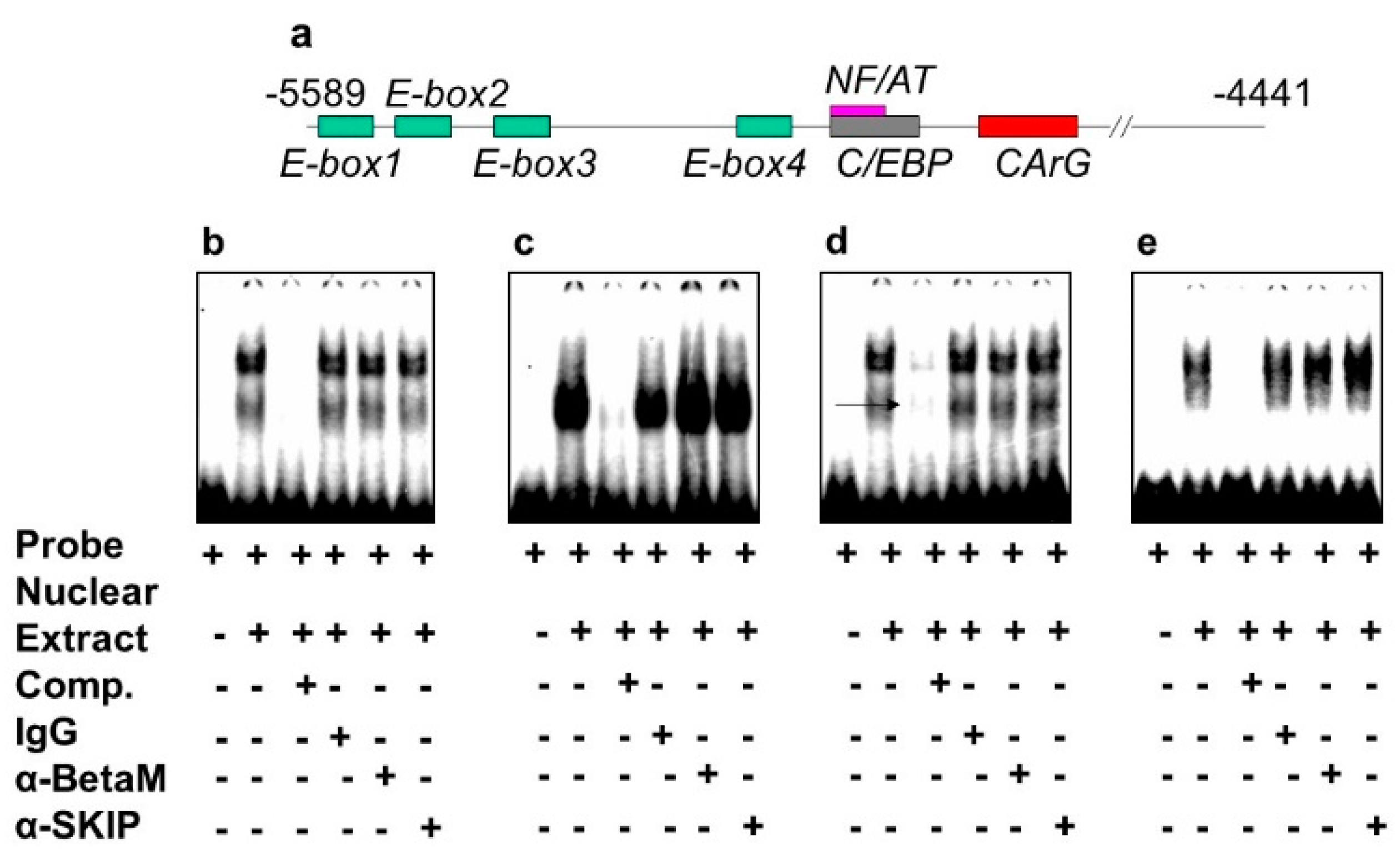
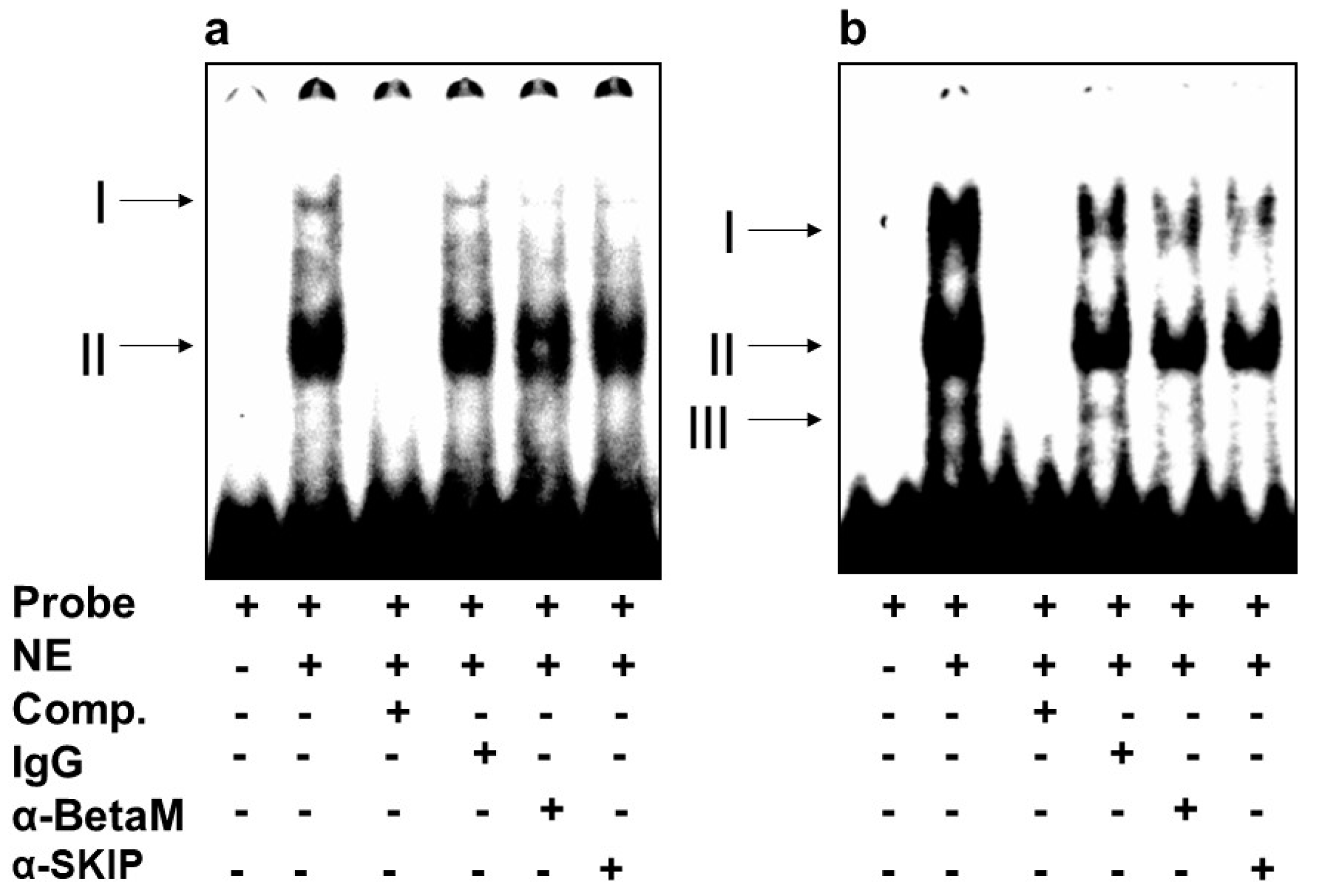
3.2. BetaM Binds to an E-Box Element and the CArG Box in the DRR Region of MyoD in Rat Skeletal Muscle
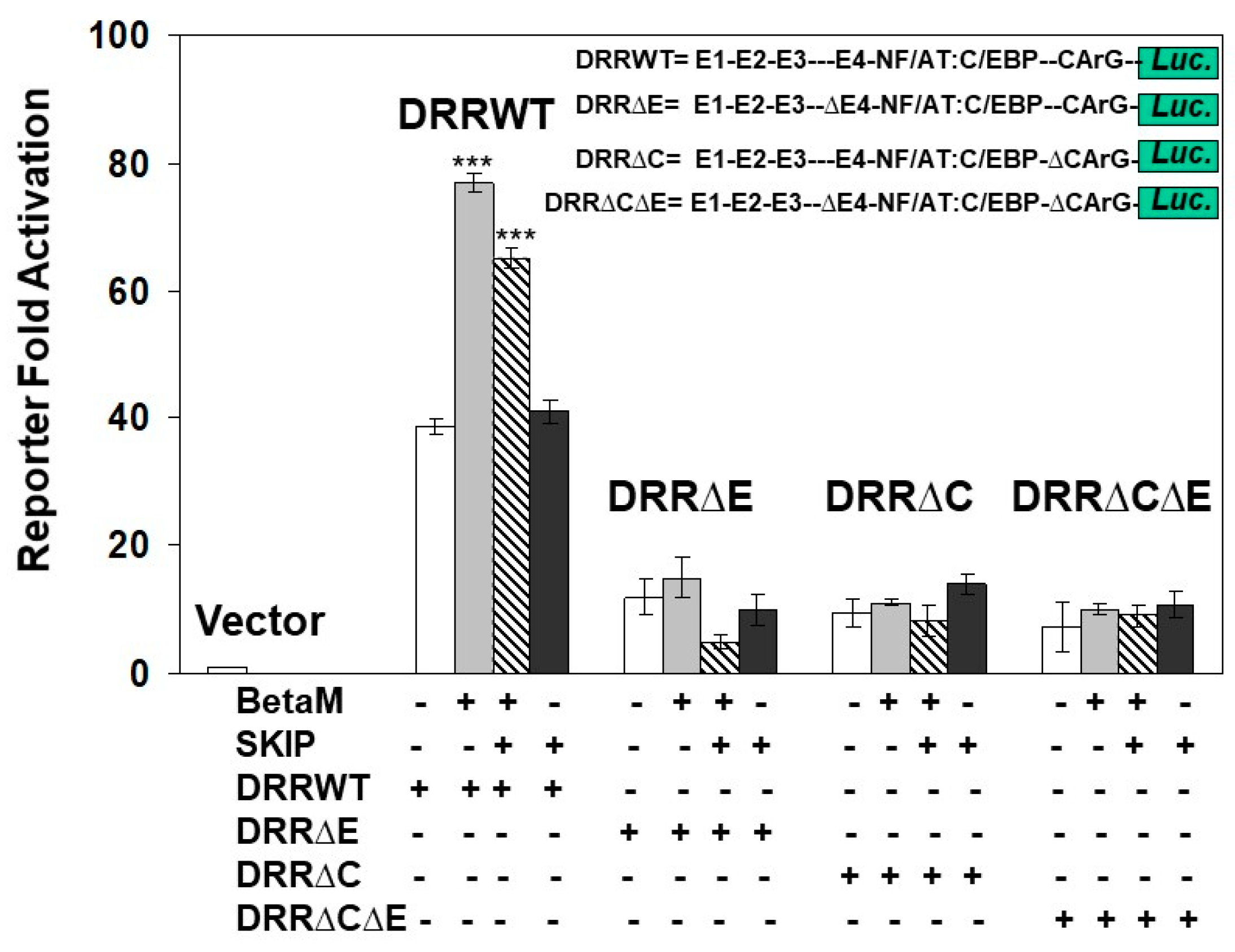
3.3. Transactivation of the MyoD DRR by BetaM Requires E-Box4 and CArG Elements
3.4. BetaM Promotes BRG1 Recruitment and Histone Modifications on the DRR of the MyoD Locus
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- True, J.R.; Carroll, S.B. Gene Co-Option in Physiological and Morphological Evolution. Annu. Rev. Cell Dev. Biol. 2002, 18, 53–80. [Google Scholar] [CrossRef] [PubMed]
- Pestov, N.B.; Ahmad, N.; Korneenko, T.V.; Zhao, H.; Radkov, R.; Schaer, D.; Roy, S.; Bibert, S.; Geering, K.; Modyanov, N.N. Evolution of Na,K-ATPase beta m-subunit into a coregulator of transcription in placental mammals. Proc. Natl. Acad. Sci. USA 2007, 104, 11215–11220. [Google Scholar] [CrossRef] [PubMed]
- Geering, K. The functional role of beta subunits in oligomeric P-type ATPases. J. Bioenerg. Biomembr. 2001, 33, 425–438. [Google Scholar] [CrossRef] [PubMed]
- Pestov, N.; Korneenko, T.V.; Zhao, H.; Adams, G.; Kostina, M.B.; Shakhparonov, M.I.; Modyanov, N.N. The betam protein, a member of the X,K-ATPase beta-subunits family, is located intracellularly in pig skeletal muscle. Arch. Biochem. Biophys. 2001, 396, 80–88. [Google Scholar] [CrossRef]
- Pestov, N.B.; Adams, G.; Shakhparonov, M.I.; Modyanov, N.N. Identification of a novel gene of the X,K-ATPase beta-subunit family that is predominantly expressed in skeletal and heart muscles. FEBS Lett. 1999, 456, 243–248. [Google Scholar] [CrossRef]
- Zhao, H.; Pestov, N.B.; Korneenko, T.V.; Shakhparonov, M.I.; Modyanov, N.N. Accumulation of beta (m), a structural member of X,K-ATPase beta-subunit family, in nuclear envelopes of perinatal myocytes. Am. J. Physiol. Physiol. 2004, 286, C757–C767. [Google Scholar] [CrossRef]
- Boulikas, T. Nuclear import of protein kinases and cyclins. J. Cell. Biochem. 1996, 60, 61–82. [Google Scholar] [CrossRef]
- Garza, A.S.; Ahmad, N.; Kumar, R. Role of intrinsically disordered protein regions/domains in transcriptional regulation. Life Sci. 2009, 84, 189–193. [Google Scholar] [CrossRef]
- Faux, N.G.; Bottomley, S.P.; Lesk, A.M.; Irving, J.A.; Morrison, J.R.; de la Banda, M.G.; Whisstock, J.C. Functional insights from the distribution and role of homopeptide repeat-containing proteins. Genome Res. 2005, 15, 537–551. [Google Scholar] [CrossRef]
- Pestov, N.B.; Crambert, G.; Zhao, H.; Korneenko, T.V.; Shakhparonov, M.I.; Geering, K.; Modyanov, N.N. The muscle-specific beta m protein is functionally different from other members of the X,K-ATPase beta-subunit family. Ann. N. Y. Acad. Sci. 2003, 986, 304–305. [Google Scholar] [CrossRef]
- Pestov, N.B.; Korneenko, T.V.; Zhao, H.; Adams, G.; Shakhparonov, M.I.; Modyanov, N.N. Immunochemical demonstration of a novel beta-subunit isoform of X, K-ATPase in human skeletal muscle. Biochem. Biophys. Res. Commun. 2000, 277, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, H.; Abdollah, S.; Qiu, Y.; Cai, J.; Xu, Y.Y.; Grinnell, B.W.; Richardson, M.A.; Topper, J.N.; Gimbrone, M.A.; Wrana, J.L.; et al. The MAD-related protein Smad7 associates with the TGFbeta receptor and functions as an antagonist of TGFbeta signaling. Cell 1997, 89, 1165–1173. [Google Scholar] [CrossRef] [PubMed]
- Massagué, J.; Seoane, J.; Wotton, D. Smad transcription factors. Genes Dev. 2005, 19, 2783–2810. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-J.; Noguchi, S.; Hayashi, Y.K.; Tsukahara, T.; Shimizu, T.; Arahata, K. The product of an oculopharyngeal muscular dystrophy gene, poly(A)-binding protein 2, interacts with SKIP and stimulates muscle-specific gene expression. Hum. Mol. Genet. 2001, 10, 1129–1139. [Google Scholar] [CrossRef] [PubMed]
- Vicente-García, C.; Hernández-Camacho, J.D.; Carvajal, J.J. Regulation of myogenic gene expression. Exp. Cell Res. 2022, 419, 113299. [Google Scholar] [CrossRef] [PubMed]
- Wardle, F.C. Master control: Transcriptional regulation of mammalian Myod. J. Muscle Res. Cell Motil. 2019, 40, 211–226. [Google Scholar] [CrossRef]
- Carvajal, J.J.; Rigby, P.W. Regulation of gene expression in vertebrate skeletal muscle. Exp. Cell Res. 2010, 316, 3014–3018. [Google Scholar] [CrossRef] [PubMed]
- Reuveni, Z.Y.; Rivera, A.J. Temporal expression of regulatory and structural muscle proteins during myogenesis of satellite cells on isolated adult rat fibers. Dev. Biol. 1994, 164, 588–603. [Google Scholar] [CrossRef]
- Tajbakhsh, S.; Rocancourt, D.; Cossu, G.; Buckingham, M. Redefining the genetic hierarchies controlling skeletal myogenesis: Pax-3 and Myf-5 act upstream of MyoD. Cell 1997, 89, 127–138. [Google Scholar] [CrossRef]
- Kablar, B.; Krastel, K.; Ying, C.; Asakura, A.; Tapscott, S.J.; A Rudnicki, M. MyoD and Myf-5 differentially regulate the development of limb versus trunk skeletal muscle. Development 1997, 124, 4729–4738. [Google Scholar] [CrossRef]
- Grifone, R.; Demignon, J.; Giordani, J.; Niro, C.; Souil, E.; Bertin, F.; Laclef, C.; Xu, P.-X.; Maire, P. Eya1 and Eya2 proteins are required for hypaxial somitic myogenesis in the mouse embryo. Dev. Biol. 2007, 302, 602–616. [Google Scholar] [CrossRef] [PubMed]
- Coletti, D.; Daou, N.; Hassani, M.; Li, Z.; Parlakian, A. Serum Response Factor in Muscle Tissues: From Development to Ageing. Eur. J. Transl. Myol. 2016, 26, 6008. [Google Scholar] [CrossRef] [PubMed]
- Havis, E.; Coumailleau, P.; Bonnet, A.; Bismuth, K.; Bonnin, M.-A.; Johnson, R.; Fan, C.-M.; Relaix, F.; Shi, D.-L.; Duprez, D. Sim2 prevents entry into the myogenic program by repressing MyoD transcription during limb embryonic myogenesis. Development 2012, 139, 1910–1920. [Google Scholar] [CrossRef] [PubMed]
- Alter, J.; Bengal, E. Stress-induced C/EBP homology protein (CHOP) represses MyoD transcription to delay myoblast differentiation. PLoS ONE 2011, 6, e29498. [Google Scholar] [CrossRef]
- Brunk, B.P.; Goldhamer, D.J.; Emerson, C.P., Jr. Regulated demethylation of the myoD distal enhancer during skeletal myogenesis. Dev. Biol. 1996, 177, 490–503. [Google Scholar] [CrossRef] [PubMed]
- Hamed, M.; Khilji, S.; Chen, J.; Li, Q. Stepwise acetyltransferase association and histone acetylation at the Myod1 locus during myogenic differentiation. Sci. Rep. 2013, 3, 2390. [Google Scholar] [CrossRef]
- Boonsanay, V.; Zhang, T.; Georgieva, A.; Kostin, S.; Qi, H.; Yuan, X.; Zhou, Y.; Braun, T. Regulation of Skeletal Muscle Stem Cell Quiescence by Suv4-20h1-Dependent Facultative Heterochromatin Formation. Cell Stem Cell 2016, 18, 229–242. [Google Scholar] [CrossRef]
- Zhang, C.; Dowd, D.R.; Staal, A.; Gu, C.; Lian, J.B.; Van Wijnen, A.J.; Stein, G.S.; MacDonald, P.N. Nuclear coactivator-62 kDa/Ski-interacting protein is a nuclear matrix-associated coactivator that may couple vitamin D receptor-mediated transcription and RNA splicing. J. Biol. Chem. 2003, 278, 35325–35336. [Google Scholar] [CrossRef]
- Hunter, J.G.; van Delft, M.F.; Rachubinski, R.A.; Capone, J.P. Peroxisome proliferator-activated receptor gamma ligands differentially modulate muscle cell differentiation and MyoD gene expression via peroxisome proliferator-activated receptor gamma -dependent and -independent pathways. J. Biol. Chem. 2001, 276, 38297–38306. [Google Scholar] [CrossRef]
- Ahmad, N.; Lingrel, J.B. Kruppel-like factor 2 transcriptional regulation involves heterogeneous nuclear ribonucleoproteins and acetyltransferases. Biochemistry 2005, 44, 6276–6285. [Google Scholar] [CrossRef]
- Tapscott, S.J.; Davis, R.L.; Thayer, M.J.; Cheng, P.-F.; Weintraub, H.; Lassar, A.B. MyoD1: A nuclear phosphoprotein requiring a Myc homology region to convert fibroblasts to myoblasts. Science 1988, 242, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Kitzmann, M.; Fernández, A. Crosstalk between cell cycle regulators and the myogenic factor MyoD in skeletal myoblasts. Cell. Mol. Life Sci. 2001, 58, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Asakura, A.; Lyons, G.E.; Tapscott, S.J. The Regulation of Myod Gene-Expression: Conserved Elements Mediate Expression in Embryonic Axial Muscle. Dev. Biol. 1995, 171, 386–398. [Google Scholar] [CrossRef]
- Goldhamer, D.J.; Faerman, A.; Shani, M.; Emerson, C.P. Regulatory elements that control the lineage-specific expression of myoD. Science 1992, 256, 538–542. [Google Scholar] [CrossRef]
- Padilla-Benavides, T.; Nasipak, B.T.; Paskavitz, A.L.; Haokip, D.T.; Schnabl, J.M.; Nickerson, J.A.; Imbalzano, A.N. Casein kinase 2-mediated phosphorylation of Brahma-related gene 1 controls myoblast proliferation and contributes to SWI/SNF complex composition. J. Biol. Chem. 2017, 292, 18592–18607. [Google Scholar] [CrossRef]
- de la Serna, I.L.; Carlson, K.A.; Imbalzano, A.N. Imbalzano, Mammalian SWI/SNF complexes promote MyoD-mediated muscle differentiation. Nat. Genet. 2001, 27, 187–190. [Google Scholar] [CrossRef] [PubMed]
- de la Serna, I.L.; Ohkawa, Y.; Berkes, C.A.; Bergstrom, D.A.; Dacwag, C.S.; Tapscott, S.J.; Imbalzano, A.N. MyoD targets chromatin remodeling complexes to the myogenin locus prior to forming a stable DNA-bound complex. Mol. Cell. Biol. 2005, 25, 3997–4009. [Google Scholar] [CrossRef]
- Simone, C.; Forcales, S.; A Hill, D.; Imbalzano, A.N.; Latella, L.; Puri, P.L. p38 pathway targets SWI-SNF chromatin-remodeling complex to muscle-specific loci. Nat. Genet. 2004, 36, 738–743. [Google Scholar] [CrossRef]
- Folk, P.; Půta, F.; Skružný, M. Transcriptional coregulator SNW/SKIP: The concealed tie of dissimilar pathways. Cell. Mol. Life Sci. 2004, 61, 629–640. [Google Scholar] [CrossRef]
- Miyake, T.; Alli, N.S.; McDermott, J.C. Nuclear Function of Smad7 Promotes Myogenesis. Mol. Cell. Biol. 2010, 30, 722–735. [Google Scholar] [CrossRef]
- Figueroa, J.D.; Hayman, M.J. Differential effects of the Ski-interacting protein (SKIP) on differentiation induced by transforming growth factor-beta1 and bone morphogenetic protein-2 in C2C12 cells. Exp. Cell Res. 2004, 296, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Korneenko, T.V.; Pestov, N.B.; Ahmad, N.; Okkelman, I.A.; Dmitriev, R.I.; Shakhparonov, M.I.; Modyanov, N.N. Evolutionary diversification of the BetaM interactome acquired through co-option of the ATP1B4 gene in placental mammals. Sci. Rep. 2016, 6, 22395. [Google Scholar] [CrossRef]
- Kayman-Kurekci, G.; Talim, B.; Korkusuz, P.; Sayar, N.; Sarioglu, T.; Oncel, I.; Sharafi, P.; Gundesli, H.; Balci-Hayta, B.; Purali, N.; et al. Mutation in TOR1AIP1 encoding LAP1B in a form of muscular dystrophy: A novel gene related to nuclear envelopathies. Neuromuscul. Disord. 2014, 24, 624–633. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.-Y.; Méndez-López, I.; Wang, Y.; Hays, A.P.; Tanji, K.; Lefkowitch, J.H.; Schulze, P.C.; Worman, H.J.; Dauer, W.T. Lamina-associated polypeptide-1 interacts with the muscular dystrophy protein emerin and is essential for skeletal muscle maintenance. Dev. Cell 2013, 26, 591–603. [Google Scholar] [CrossRef] [PubMed]
- Heller, S.A.; Shih, R.; Kalra, R.; Kang, P.B. Emery-Dreifuss muscular dystrophy. Muscle Nerve 2020, 61, 436–448. [Google Scholar] [CrossRef] [PubMed]
- Perovanovic, J.; Dell’Orso, S.; Gnochi, V.F.; Jaiswal, J.K.; Sartorelli, V.; Vigouroux, C.; Mamchaoui, K.; Mouly, V.; Bonne, G.; Hoffman, E.P. Laminopathies disrupt epigenomic developmental programs and cell fate. Sci. Transl. Med. 2016, 8, 335ra58. [Google Scholar] [CrossRef]
- Håkelien, A.M.; Delbarre, E.; Gaustad, K.G.; Buendia, B.; Collas, P. Expression of the myodystrophic R453W mutation of lamin A in C2C12 myoblasts causes promoter-specific and global epigenetic defects. Exp. Cell Res. 2008, 314, 1869–1880. [Google Scholar] [CrossRef]



| Region | Forward | Reverse | |
|---|---|---|---|
| Ebox1 | ACT GCT GTG AGA ACA TCT GAC ATC CAC CAC | GTG GTG GAT GTC AGA TGT TCT CAC AGC AGT | WT |
| Ebox2 | CCA CCA CCT AGT TCA TTT GCC AGA CTC CCA AGG | CCT TGG GAG TCT GGC AAA TGA ACT AGG TGG TGG | WT |
| Ebox3 | GAC CAG GAC CAC ATC TGC GCC CAG CCA CAA | TTG TGG CTG GGC GCA GAT GTG GTC CTG GTC | WT |
| Ebox4 | GAG GCT TGG GGC AGG TGC TGG TTG GAT CCG | CGG ATC CAA CCA GCA CCT GCC CCA AGC CTC | WT |
| Ebox4 M1 | GAG GCT TGG GGt tGG gaC TGG TTG GAT CCG | CGG ATC CAA CCA Gtc CCa aCC CCA AGC CTC | Mutant |
| Ebox4 M2 | GAG GCT TGG GGC Atc TGC TGG TTG GAT CCG | CGG ATC CAA CCA GCA gaT GCC CCA AGC CTC | Mutant |
| Ebox4 M3 | GAG GCT acc aaC AGG TGC TGG TTG GAT CCG | CGG ATC CAA CCA GCA CCT Gtt ggt AGC CTC | Mutant |
| Ebox4 M4 | GAG GCT TGG GGC AGG TGC TGG Tca ctc CCG | CGG gag tgA CCA GCA CCT GCC CCA AGC CTC | Mutant |
| NFAT | TTG GAT CCG GTT TCC AGA GGC TAT ATA TAT AAA | TTT ATA TAT ATA GCC TCT GGA AAC CGG ATC CAA | WT |
| CArG | CCA GCT CTC CAT TTA TAG CCC CTG GGC AGA | TCT GCC CAG GGG CTA TAA ATG GAG AGC TGG | WT |
| CArG M1 | CCA GCT CTa aAT TTA Tcc CCg gTG GGC AGA | TCT GCC CAc cGG ggA TAA ATt tAG AGC TGG | Mutant |
| CArG M2 | ggA cga taC CAT TTA TAG CCC CTG GGC AGA | TCT GCC CAG GGG CTA TAA ATG Gta tcg Tcc | Mutant |
| CArG M3 | CCA GCT CTC Cca ccg acG CCC CTG GGC AGA | TCT GCC CAG GGG Cgt cgg tgG GAG AGC TGG | Mutant |
| CArG M4 | CCA GCT CTC CAT TTA TAG CCt aaG ccC tat | ata Ggg Ctt GGG CTA TAA ATG GAG AGC TGG | Mutant |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, N.; de la Serna, I.L.; Marathe, H.G.; Fan, X.; Dube, P.; Zhang, S.; Haller, S.T.; Kennedy, D.J.; Pestov, N.B.; Modyanov, N.N. Eutherian-Specific Functions of BetaM Acquired through Atp1b4 Gene Co-Option in the Regulation of MyoD Expression. Life 2023, 13, 414. https://doi.org/10.3390/life13020414
Ahmad N, de la Serna IL, Marathe HG, Fan X, Dube P, Zhang S, Haller ST, Kennedy DJ, Pestov NB, Modyanov NN. Eutherian-Specific Functions of BetaM Acquired through Atp1b4 Gene Co-Option in the Regulation of MyoD Expression. Life. 2023; 13(2):414. https://doi.org/10.3390/life13020414
Chicago/Turabian StyleAhmad, Nisar, Ivana L. de la Serna, Himangi G. Marathe, Xiaoming Fan, Prabhatchandra Dube, Shungang Zhang, Steven T. Haller, David J. Kennedy, Nikolay B. Pestov, and Nikolai N. Modyanov. 2023. "Eutherian-Specific Functions of BetaM Acquired through Atp1b4 Gene Co-Option in the Regulation of MyoD Expression" Life 13, no. 2: 414. https://doi.org/10.3390/life13020414
APA StyleAhmad, N., de la Serna, I. L., Marathe, H. G., Fan, X., Dube, P., Zhang, S., Haller, S. T., Kennedy, D. J., Pestov, N. B., & Modyanov, N. N. (2023). Eutherian-Specific Functions of BetaM Acquired through Atp1b4 Gene Co-Option in the Regulation of MyoD Expression. Life, 13(2), 414. https://doi.org/10.3390/life13020414









