Carbon Biogeochemistry of the Estuaries Adjoining the Indian Sundarbans Mangrove Ecosystem: A Review
Abstract
1. Introduction
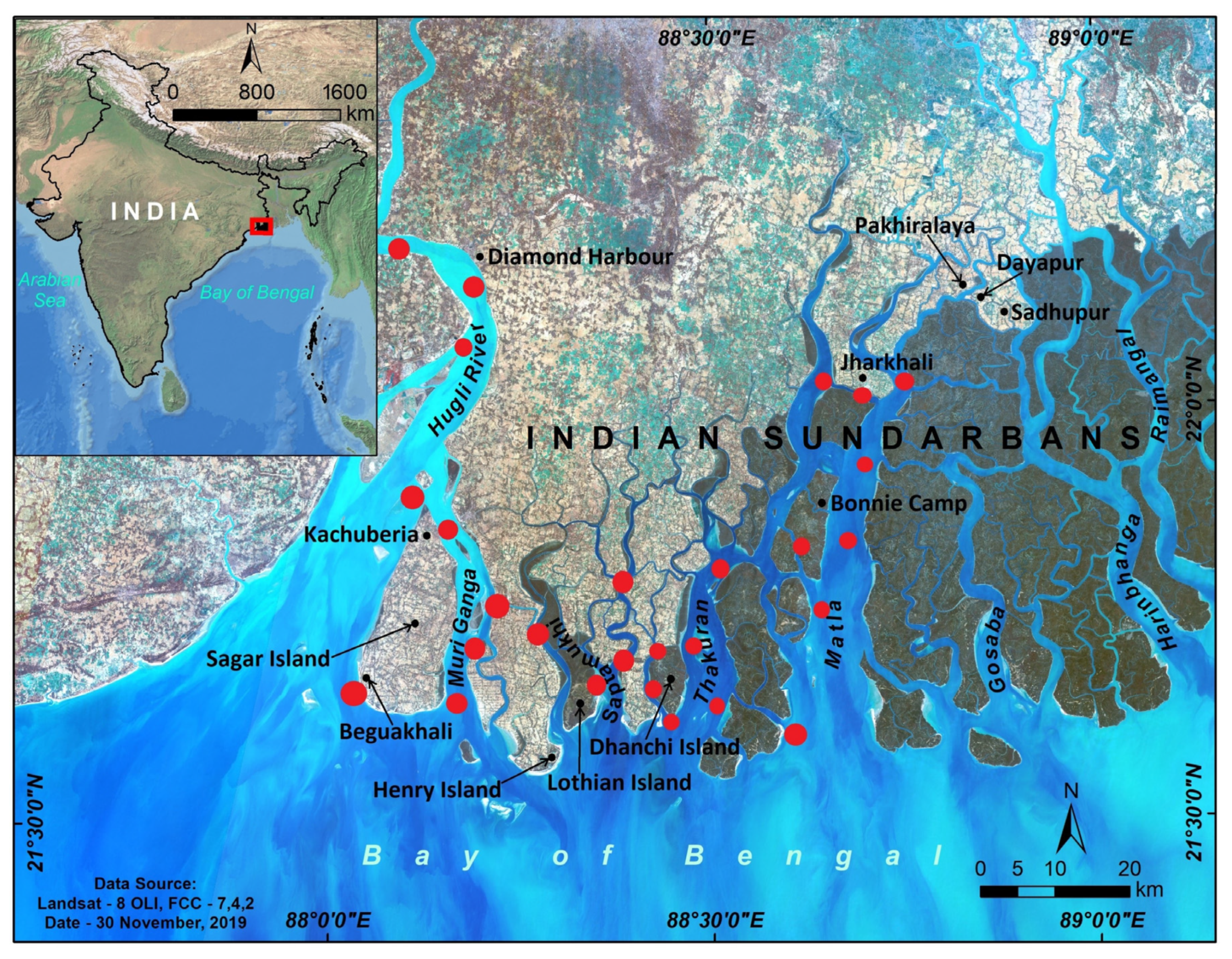
2. Indian Sundarbans Mangrove Forest: A Brief Overview
3. Overview of the Carbon Biogeochemistry Research in Indian Sundarbans
4. Carbon Parameters and Total Alkalinity in Estuarine Water
4.1. Spatial Variability
4.2. Temporal Variability
5. Factors Regulating Air-Water CO2 and CH4 Flux
5.1. Role of Physical Factors
5.2. Role of Biogeochemical Factors
5.3. Role of Hydrological Factors (Pore Water, Groundwater, and Freshwater Discharge)
5.4. Role of the Microbiomes in Carbon Dynamics of Sundarban
6. The Lateral Flux of Carbon to the Bay of Bengal
7. Summary and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Broecker, W.S. Climatic change: Are we on the brink of a pronounced global warming? Science 1975, 189, 460–463. [Google Scholar] [CrossRef] [PubMed]
- de Menocal, P. Wallace Smith Broecker. Nature 2019, 568, 34. [Google Scholar] [CrossRef]
- Chen, C.T.A.; Tsunogai, S. Carbon and nutrients in the ocean. In Asian Change in the Context of Global Change; Galloway, J.N., Melillio, J.M., Eds.; Cambridge University Press: Cambridge, UK, 1998; pp. 271–307. [Google Scholar]
- Chen, C.T.; Huang, T.H.; Chen, Y.C.; Bai, Y.; He, X.; Kang, Y. Air–sea exchanges of CO2 in the world’s coastal seas. Biogeosciences 2013, 10, 6509–6544. [Google Scholar] [CrossRef]
- Dyer, K.R. Response of estuaries to climate change. In Climate Change: Impact on Coastal Habitation; Eisma, D., Ed.; CRC Press: Boca Raton, FL, USA, 1995; pp. 85–110. [Google Scholar]
- Borges, A.V.; Delille, B.; Frankignoulle, M. Budgeting sinks and sources of CO2 in the coastal ocean: Diversity of ecosystems counts. Geophys. Res. Lett. 2005, 32, L14601. [Google Scholar] [CrossRef]
- Borges, A.; Abril, G. Carbon dioxide and methane dynamics in estuaries. In Treatise on Estuarine and Coastal Science; Wolanski, E., McLusky, D.S., Eds.; Academic Press: Waltham, MA, USA, 2010; Volume 5, pp. 119–161. [Google Scholar] [CrossRef]
- Cai, W.J. Estuarine and coastal ocean carbon paradox: CO2 sinks or sites of terrestrial carbon incineration? Annu. Rev. Mar. Sci. 2011, 3, 123–145. [Google Scholar] [CrossRef]
- Chen, X.; Santos, I.R.; Call, M.; Reithmaier, G.M.; Maher, D.; Holloway, C.; Wadnerkar, P.D.; Gómez-Álvarez, P.; Sanders, C.J.; Li, L. The mangrove CO2 pump: Tidally driven pore-water exchange. Limnol. Oceanogr. 2021, 66, 1563–1577. [Google Scholar] [CrossRef]
- Najjar, R.G.; Herrmann, M.; Alexander, R.; Boyer, E.W.; Burdige, D.J.; Butman, D.; Cai, W.J.; Canuel, E.A.; Chen, R.F.; Friedrichs, M.A.; et al. Carbon budget of tidal wetlands, estuaries, and shelf waters of Eastern North America. Glob. Biogeochem. Cycles 2018, 32, 389–416. [Google Scholar] [CrossRef]
- Diaz, R.J.; Rosenberg, R. Spreading dead zones and consequences for marine ecosystems. Science 2008, 321, 926–929. [Google Scholar] [CrossRef]
- Cai, W.J.; Hu, X.; Huang, W.J.; Murrell, M.C.; Lehrter, J.C.; Lohrenz, S.E.; Chou, W.C.; Zhai, W.; Hollibaugh, J.T.; Wang, Y.; et al. Acidification of subsurface coastal waters enhanced by eutrophication. Nat. Geosci. 2011, 4, 766–770. [Google Scholar] [CrossRef]
- Pendleton, L.; Donato, D.C.; Murray, B.C.; Crooks, S.; Jenkins, W.A.; Sifleet, S.; Craft, C.; Fourqurean, J.W.; Kauffman, J.B.; Marbà, N.; et al. Estimating global “blue carbon” emissions from conversion and degradation of vegetated coastal ecosystems. PLoS ONE 2012, 7, e43542. [Google Scholar] [CrossRef]
- Olivier, J.G.J.; Peters, J.A.H.W. Trends in Global CO2 and Total Greenhouse Gas Emissions: 2020 Report; PBL Netherlands Environmental Assessment Agency: The Hague, The Netherlands, 2020. [Google Scholar]
- Kirschke, S.; Bousquet, P.; Ciais, P.; Saunois, M.; Canadell, J.G.; Dlugokencky, E.J.; Bergamaschi, P.; Bergmann, D.; Blake, D.R.; Bruhwiler, L.; et al. Three decades of global methane sources and sinks. Nat. Geosci. 2013, 6, 813–823. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2014: Mitigation of Climate Change; Cambridge University Press: Cambrige, UK, 2014. [Google Scholar]
- IUCN. The management of natural coastal carbon sink. In IUCN World Commission on Protected Areas; Laffoley, D., Drimsditch, G., Eds.; Natural England, UNEP: York, UK, 2009. [Google Scholar]
- Curry, C.L. Modeling the soil consumption of atmospheric methane at the global scale. Glob. Biogeochem. Cycles 2007, 21, GB4012. [Google Scholar] [CrossRef]
- Zhuang, Q.; Melillo, J.M.; Kicklighter, D.W.; Prinn, R.G.; McGuire, A.D.; Steudler, P.A.; Felzer, B.S.; Hu, S. Methane fluxes between terrestrial ecosystems and the atmosphere at northern high latitudes during the past century: A retrospective analysis with a process-based biogeochemistry model. Glob. Biogeochem. Cycles 2004, 18. [Google Scholar] [CrossRef]
- Cicerone, R.J.; Oremland, R.S. Biogeochemical aspects of atmospheric methane. Glob. Biogeochem. Cycles 1988, 2, 299–327. [Google Scholar] [CrossRef]
- Allan, W.; Struthers, H.; Lowe, D.C. Methane carbon isotope effects caused by atomic chlorine in the marine boundary layer: Global model results compared with Southern Hemisphere measurements. J. Geophys. Res. Atmos. 2007, 112. [Google Scholar] [CrossRef]
- Duarte, C.M.; Middelburg, J.J.; Caraco, N. Major role of marine vegetation on the oceanic carbon cycle. Biogeosciences 2005, 2, 1–8. [Google Scholar] [CrossRef]
- Mcleod, E.; Chmura, G.L.; Bouillon, S.; Salm, R.; Björk, M.; Duarte, C.M.; Lovelock, C.E.; Schlesinger, W.H.; Silliman, B.R. A blueprint for blue carbon: Toward an improved understanding of the role of vegetated coastal habitats in sequestering CO2. Front. Ecol. Environ. 2011, 9, 552–560. [Google Scholar] [CrossRef]
- Perera, K.A.R.S.; Amarasinghe, M.D. Carbon sequestration capacity of mangrove soils in micro tidal estuaries and lagoons: A case study from Sri Lanka. Geoderma 2019, 347, 80–89. [Google Scholar] [CrossRef]
- Nagarajan, B.; Pandiarajan, C.; Krishnamoorthy, M.; Sophia, P. Reproductive fitness and success in mangroves: Implication on conservation. In Proceedings of the Taal the 12th World Lake Conference, Jaipur, India, 28 October–2 November 2007. [Google Scholar]
- Donato, D.C.; Kauffman, J.B.; Murdiyarso, D.; Kurnianto, S.; Stidham, M.; Kanninen, M. Mangroves among the most carbon-rich forests in the tropics. Nat. Geosci. 2011, 4, 293–297. [Google Scholar] [CrossRef]
- Kauffman, J.B.; Giovanonni, L.; Kelly, J.; Dunstan, N.; Borde, A.; Diefenderfer, H.; Cornu, C.; Janousek, C.; Apple, J.; Brophy, L. Total ecosystem carbon stocks at the marine-terrestrial interface: Blue carbon of the Pacific Northwest Coast, United States. Glob. Change Biol. 2020, 26, 5679–5692. [Google Scholar] [CrossRef]
- Bouillon, S.; Borges, A.V.; Castañeda-Moya, E.; Diele, K.; Dittmar, T.; Duke, N.C.; Kristensen, E.; Lee, S.Y.; Marchand, C.; Middelburg, J.J.; et al. Mangrove production and carbon sinks: A revision of global budget estimates. Glob. Biogeochem. Cycles 2008, 22. [Google Scholar] [CrossRef]
- Cabezas, A.; Mitsch, W.J.; MacDonnell, C.; Zhang, L.; Bydałek, F.; Lasso, A. Methane emissions from mangrove soils in hydrologically disturbed and reference mangrove tidal creeks in southwest Florida. Ecol. Eng. 2018, 114, 57–65. [Google Scholar] [CrossRef]
- Alongi, D.M. Carbon sequestration in mangrove forests. Carbon Manag. 2012, 3, 313–322. [Google Scholar] [CrossRef]
- Komiyama, A.; Havanond, S.; Srisawatt, W.; Mochida, Y.; Fujimoto, K.; Ohnishi, T.; Ishihara, S.; Miyagi, T. Top/root biomass ratio of a secondary mangrove (Ceriops tagal (Perr.) CB Rob.) forest. For. Ecol. Manag. 2000, 139, 127–134. [Google Scholar] [CrossRef]
- Khan, M.; Islam, N.; Suwa, R.; Hagihara, A. Biomass and aboveground net primary production in a subtropical mangrove stand of Kandelia obovata (S.; L.) Yong at Manko Wetland, Okinawa, Japan. Wetl. Ecol. Manag. 2009, 17, 585–599. [Google Scholar] [CrossRef]
- Eong, O.J. Mangroves-a carbon source and sink. Chemosphere 1993, 27, 1097–1107. [Google Scholar] [CrossRef]
- Gurney, A.; Ahammad, H.; Ford, M. The economics of greenhouse gas mitigation: Insights from illustrative global abatement scenarios modelling. Energy Econ. 2009, 31, S174–S186. [Google Scholar] [CrossRef]
- Gattuso, J.P.; Frankignoulle, M.; Wollast, R. Carbon and carbonate metabolism in coastal aquatic ecosystems. Annu. Rev. Ecol. Syst. 1998, 29, 405–434. [Google Scholar] [CrossRef]
- Bianchi, T.S. Biogeochemistry of Estuaries; Oxford University Press Inc.: Oxford, UK, 2007. [Google Scholar]
- Testa, J.M.; Kemp, W.M.; Hopkinson, C.S.; Smith, S.V. Ecosystem Metabolism. In Estuarine Ecology, 1st ed.; Day, J.W., Crump, B.C., Kemp, W.M., Yáñez-Arancibia, A., Eds.; Wiley, Inc.: Hoboken, NJ, USA, 2012; pp. 381–416. [Google Scholar] [CrossRef]
- Laruelle, G.G.; Dürr, H.H.; Slomp, C.P.; Borges, A.V. Evaluation of sinks and sources of CO2 in the global coastal ocean using a spatially-explicit typology of estuaries and continental shelves. Geophys. Res. Lett. 2010, 37, L15607. [Google Scholar] [CrossRef]
- Laruelle, G.G.; Dürr, H.H.; Lauerwald, R.; Hartmann, J.; Slomp, C.P.; Goossens, N.; Regnier, P.A.G. Global multi-scale segmentation of continental and coastal waters from the watersheds to the continental margins. Hydrol. Earth Syst. Sci. 2013, 17, 2029–2051. [Google Scholar] [CrossRef]
- Crosswell, J.R.; Anderson, I.C.; Stanhope, J.W.; Van Dam, B.; Brush, M.J.; Ensign, S.; Piehler, M.F.; McKee, B.; Bost, M.; Paerl, H.W. Carbon budget of a shallow, lagoonal estuary: Transformations and source-sink dynamics along the river-estuary-ocean continuum. Limnol. Oceanogr. 2017, 62, S29–S45. [Google Scholar] [CrossRef]
- Maher, D.T.; Eyre, B.D. Carbon budgets for three autotrophic Australian estuaries: Implications for global estimates of the coastal air-water CO2 flux. Glob. Biogeochem. Cycles 2012, 26. [Google Scholar] [CrossRef]
- Cotovicz, L.C., Jr.; Knoppers, B.A.; Brandini, N.; Costa Santos, S.J.; Abril, G. A strong CO2 sink enhanced by eutrophication in a tropical coastal embayment (Guanabara Bay, Rio de Janeiro, Brazil). Biogeosciences 2015, 12, 6125–6146. [Google Scholar] [CrossRef]
- Jiang, Z.P.; Cai, W.J.; Lehrter, J.; Chen, B.; Ouyang, Z.; Le, C.; Roberts, B.J.; Hussain, N.; Scaboo, M.K.; Zhang, J.; et al. Spring net community production and its coupling with the CO2 dynamics in the surface water of the northern Gulf of Mexico. Biogeosciences 2019, 16, 3507–3525. [Google Scholar] [CrossRef]
- Joesoef, A.; Huang, W.J.; Gao, Y.; Cai, W.J. Air–water fluxes and sources of carbon dioxide in the Delaware Estuary: Spatial and seasonal variability. Biogeosciences 2015, 12, 6085–6101. [Google Scholar] [CrossRef]
- Crosswell, J.R.; Wetz, M.S.; Hales, B.; Paerl, H.W. Air-water CO2 fluxes in the microtidal Neuse River estuary, North Carolina. J. Geophys. Res. Oceans 2012, 117, C08017. [Google Scholar] [CrossRef]
- Yao, H.; McCutcheon, M.R.; Staryk, C.J.; Hu, X. Hydrologic controls on CO2 chemistry and flux in subtropical lagoonal estuaries of the northwestern Gulf of Mexico. Limnol. Oceanogr. 2020, 65, 1380–1398. [Google Scholar] [CrossRef]
- Li, X.; Han, G.; Liu, M.; Song, C.; Zhang, Q.; Yang, K.; Liu, J. Hydrochemistry and dissolved inorganic carbon (DIC) cycling in a tropical agricultural river, Mun River Basin, Northeast Thailand. Int. J. Environ. Res. Public Health 2019, 16, 3410. [Google Scholar] [CrossRef]
- Joesoef, A.; Kirchman, D.L.; Sommerfield, C.K.; Cai, W.J. Seasonal variability of the inorganic carbon system in a large coastal plain estuary. Biogeosciences 2017, 14, 4949–4963. [Google Scholar] [CrossRef]
- Koné, Y.M.; Borges, A.V. Dissolved inorganic carbon dynamics in the waters surrounding forested mangroves of the Ca Mau Province (Vietnam). Estuar. Coast. Shelf Sci. 2008, 77, 409–421. [Google Scholar] [CrossRef]
- Bridgham, S.D.; Cadillo-Quiroz, H.; Keller, J.K.; Zhuang, Q. Methane emissions from wetlands: Biogeochemical, microbial, and modeling perspectives from local to global scales. Glob. Change Biol. 2013, 19, 1325–1346. [Google Scholar] [CrossRef] [PubMed]
- Turetsky, M.R.; Kotowska, A.; Bubier, J.; Dise, N.B.; Crill, P.; Hornibrook, E.R.; Minkkinen, K.; Moore, T.R.; Myers-Smith, I.H.; Nykänen, H.; et al. A synthesis of methane emissions from 71 northern, temperate, and subtropical wetlands. Glob. Chang. Biol. 2014, 20, 2183–2197. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zimmermann, N.E.; Stenke, A.; Li, X.; Hodson, E.L.; Zhu, G.; Huang, C.; Poulter, B. Emerging role of wetland methane emissions in driving 21st century climate change. Proc. Natl. Acad. Sci. USA 2017, 114, 9647–9652. [Google Scholar] [CrossRef]
- Arai, H.; Yoshioka, R.; Hanazawa, S.; Minh, V.Q.; Tuan, V.Q.; Tinh, T.K.; Phu, T.Q.; Jha, C.S.; Rodda, S.R.; Dadhwal, V.K.; et al. Function of the methanogenic community in mangrove soils as influenced by the chemical properties of the hydrosphere. Soil Sci. Plant Nutr. 2016, 62, 150–163. [Google Scholar] [CrossRef]
- Zheng, X.; Guo, J.; Song, W.; Feng, J.; Lin, G. Methane emission from mangrove wetland soils is marginal but can be stimulated significantly by anthropogenic activities. Forests 2018, 9, 738. [Google Scholar] [CrossRef]
- He, Y.; Guan, W.; Xue, D.; Liu, L.; Peng, C.; Liao, B.; Hu, J.; Yang, Y.; Wang, X.; Zhou, G.; et al. Comparison of methane emissions among invasive and native mangrove species in Dongzhaigang, Hainan Island. Sci. Total Environ. 2019, 697, 133945. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, A.B.; Choudhury, A.; Hussain, Z.; Acharya, G. Mangroves of the Sundarbans: India; IUCN: Bangkok, Thailand, 1994; Volume 1, p. 247. ISBN 13 9782831702094. [Google Scholar]
- Mandal, D.B. Man in Biosphere: A Case Study of Sundarbans Biosphere Reserve, 1st ed.; Gyan Publishing House: New Delhi, India, 2007. [Google Scholar]
- Samanta, S.; Hazra, S.; Mondal, P.P.; Chanda, A.; Giri, S.; French, J.R.; Nicholls, R.J. Assessment and attribution of mangrove Forest changes in the Indian Sundarbanss from 2000 to 2020. Remote Sens. 2021, 13, 4957. [Google Scholar] [CrossRef]
- Bhadra, T.; Mukhopadhyay, A.; Hazra, S. Identification of River Discontinuity Using Geo-Informatics to Improve Freshwater Flow and Ecosystem Services in Indian Sundarbans Delta. In Environment and Earth Observation; Hazra, S., Mukhopadhyay, A., Ghosh, A., Mitra, D., Dadhwal, V., Eds.; Springer Remote Sensing/Photogrammetry; Springer: Cham, Switzerland, 2017. [Google Scholar] [CrossRef]
- Roshith, C.M.; Meena, D.K.; Manna, R.K.; Sahoo, A.K.; Swain, H.S.; Raman, R.K.; Sengupta, A.; Das, B.K. Phytoplankton community structure of the Gangetic (Hooghly-Matla) estuary: Status and ecological implications in relation to eco-climatic variability. Flora 2018, 240, 133–143. [Google Scholar] [CrossRef]
- Chatterjee, M.; Shankar, D.; Sen, G.K.; Sanyal, P.; Sundar, D.; Michael, G.S.; Chatterjee, A.; Amol, P.; Mukherjee, D.; Suprit, K.; et al. Tidal variations in the Sundarbans estuarine system, India. J. Earth Syst. Sci. 2013, 122, 899–933. [Google Scholar] [CrossRef]
- De, T.K.; De, M.; Das, S.; Chowdhury, C.; Ray, R.; Jana, T.K. Phytoplankton abundance in relation to cultural eutrophication at the land-ocean boundary of Sunderbans, NE Coast of Bay of Bengal, India. J. Environ. Sci. Stud. 2011, 1, 169–180. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.K.; Biswas, H.; De, T.K.; Sen, S.; Jana, T.K. Seasonal effects on the air–water carbon dioxide exchange in the Hooghly estuary, NE coast of Bay of Bengal, India. Environ. Monit. Assess. 2002, 4, 549–552. [Google Scholar] [CrossRef] [PubMed]
- Padhy, P.C.; Nayak, R.K.; Dadhwal, V.K.; Salim, M.; Mitra, D.; Chaudhury, S.B.; Rao, P.R.; Rao, K.H.; Dutt, C.B.S. Estimation of partial pressure of carbon dioxide and air-sea fluxes in Hooghly estuary based on in situ and satellite observations. J. Indian Soc. Remote Sens. 2016, 44, 135–143. [Google Scholar] [CrossRef]
- Akhand, A.; Chanda, A.; Watanabe, K.; Das, S.; Tokoro, T.; Chakraborty, K.; Hazra, S.; Kuwae, T. Low CO2 evasion rate from the mangrove-surrounding waters of the Sundarbans. Biogeochemistry 2021, 153, 95–114. [Google Scholar] [CrossRef]
- Akhand, A.; Chanda, A.; Watanabe, K.; Das, S.; Tokoro, T.; Hazra, S.; Kuwae, T. Reduction in Riverine Freshwater Supply Changes Inorganic and Organic Carbon Dynamics and Air-Water CO2 Fluxes in a Tropical Mangrove Dominated Estuary. J. Geophys. Res. Biogeosci. 2021, 126, e2020JG006144. [Google Scholar] [CrossRef]
- Dutta, M.K.; Kumar, S.; Mukherjee, R.; Sanyal, P.; Mukhopadhyay, S.K. The post-monsoon carbon biogeochemistry of the Hooghly–Sundarbans estuarine system under different levels of anthropogenic impacts. Biogeosciences 2019, 16, 289–307. [Google Scholar] [CrossRef]
- Akhand, A.; Chanda, A.; Manna, S.; Das, S.; Hazra, S.; Roy, R.; Choudhury, S.B.; Rao, K.H.; Dadhwal, V.K.; Chakraborty, K.; et al. A comparison of CO2 dynamics and air-water fluxes in a river-dominated estuary and a mangrove-dominated marine estuary. Geophys. Res. Lett. 2016, 43, 11–726. [Google Scholar] [CrossRef]
- Dutta, M.K.; Kumar, S.; Mukherjee, R.; Sharma, N.; Bhushan, R.; Sanyal, P.; Paul, M.; Mukhopadhyay, S.K. Carbon Biogeochemistry of Two Contrasting Tropical Estuarine Ecosystems During Pre-monsoon. Estuaries Coast. 2021, 44, 1916–1930. [Google Scholar] [CrossRef]
- Akhand, A.; Chanda, A.; Watanabe, K.; Das, S.; Tokoro, T.; Hazra, S.; Kuwae, T. Drivers of inorganic carbon dynamics and air–water CO2 fluxes in two large tropical estuaries: Insights from coupled radon (222Rn) and pCO2 surveys. Limnol. Oceanogr. 2022, 67, S118–S132. [Google Scholar] [CrossRef]
- Acharya, A.; Sanyal, P.; Paul, M.; Gupta, V.K.; Bakshi, S.; Bhattacharyya, P.; Mukhopadhyay, S.K. Seasonal quantification of carbonate dissolution and CO2 emission dynamics in the Indian Sundarbans estuaries. Reg. Stud. Mar. Sci. 2022, 53, 102413. [Google Scholar] [CrossRef]
- Dutta, M.K.; Kumar, S.; Mukherjee, R.; Sharma, N.; Acharya, A.; Sanyal, P.; Bhusan, R.; Mukhopadhyay, S.K. Diurnal carbon dynamics in a mangrove-dominated tropical estuary (Sundarbans, India). Estuar. Coast. Shelf Sci. 2019, 229, 106426. [Google Scholar] [CrossRef]
- Ray, R.; Rixen, T.; Baum, A.; Malik, A.; Gleixner, G.; Jana, T.K. Distribution, sources and biogeochemistry of organic matter in a mangrove dominated estuarine system (Indian Sundarbans) during the pre-monsoon. Estuar. Coast. Shelf Sci. 2015, 167, 404–413. [Google Scholar] [CrossRef]
- Biswas, H.; Mukhopadhyay, S.K.; De, T.K.; Sen, S.; Jana, T.K. Biogenic controls on the air—Water carbon dioxide exchange in the Sundarban mangrove environment, northeast coast of Bay of Bengal, India. Limnol. Oceanogr. 2004, 49, 95–101. [Google Scholar] [CrossRef]
- Ghosh, S.; Jana, T.K.; Singh, B.N.; Choudhury, A. Comparative study of carbon dioxide system in virgin and reclaimed mangrove waters of Sundarbans. Mahasagar 1987, 20, 155–161. [Google Scholar]
- Akhand, A.; Chanda, A.; Dutta, S.; Manna, S.; Sanyal, P.; Hazra, S.; Rao, K.H.; Dadhwal, V.K. Dual character of Sundarban estuary as a source and sink of CO2 during summer: An investigation of spatial dynamics. Environ. Monit. Assess. 2013, 185, 6505–6515. [Google Scholar] [CrossRef] [PubMed]
- Biswas, H.; Mukhopadhyay, S.K.; Sen, S.; Jana, T.K. Spatial and temporal patterns of methane dynamics in the tropical mangrove dominated estuary, NE coast of Bay of Bengal, India. J. Mar. Syst. 2007, 68, 55–64. [Google Scholar] [CrossRef]
- Neetha, V. Dissolved Carbon Dioxide and Methane in Estuaries and Waters Surrounding Mangroves on the East Coast of India and the Andaman Islands. Ph.D Thesis, Anna University, Chennai, India, October 2008. Available online: http://hdl.handle.net/10603/29692 (accessed on 17 October 2022).
- Dutta, M.K.; Bianchi, T.S.; Mukhopadhyay, S.K. Mangrove methane biogeochemistry in the Indian Sundarbans: A proposed budget. Front. Mar. Sci. 2017, 4, 187. [Google Scholar] [CrossRef]
- Dutta, M.K.; Mukherjee, R.; Jana, T.K.; Mukhopadhyay, S.K. Biogeochemical dynamics of exogenous methane in an estuary associated to a mangrove biosphere; the Sundarbans, NE coast of India. Mar. Chem. 2015, 170, 1–10. [Google Scholar] [CrossRef]
- Dutta, M.K.; Ray, R.; Mukherjee, R.; Jana, T.K.; Mukhopadhyay, S.K. Atmospheric fluxes and photo-oxidation of methane in the mangrove environment of the Sundarbans, NE coast of India; A case study from Lothian Island. Agric. For. Meteorol. 2015, 213, 33–41. [Google Scholar] [CrossRef]
- Dutta, M.K.; Chowdhury, C.; Jana, T.K.; Mukhopadhyay, S.K. Dynamics and exchange fluxes of methane in the estuarine mangrove environment of the Sundarbans, NE coast of India. Atmos. Environ. 2013, 77, 631–639. [Google Scholar] [CrossRef]
- Ghosh, J.; Chakraborty, K.; Chanda, A.; Akhand, A.; Bhattacharya, T.; Das, S.; Das, I.; Hazra, S.; Choudhury, S.B.; Wells, M. Outwelling of total alkalinity and dissolved inorganic carbon from the Hooghly River to the adjacent coastal Bay of Bengal. Environ. Monit. Assess. 2021, 193, 415. [Google Scholar] [CrossRef]
- Ray, R.; Baum, A.; Rixen, T.; Gleixner, G.; Jana, T.K. Exportation of dissolved (inorganic and organic) and particulate carbon from mangroves and its implication to the carbon budget in the Indian Sundarbans. Sci. Total Environ. 2018, 621, 535–547. [Google Scholar] [CrossRef] [PubMed]
- Dyrssen, D.; Sillén, L.G. Alkalinity and total carbonate in sea water. A plea for p-T-independent data. Tellus 1967, 19, 113–121. [Google Scholar] [CrossRef]
- Smith, S.V.; Hollibaugh, J.T. Coastal metabolism and the oceanic organic carbon balance. Rev. Geophys. 1993, 31, 75–89. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.K.; Biswas, H.D.T.K.; De, T.K.; Jana, T.K. Fluxes of nutrients from the tropical River Hooghly at the land–ocean boundary of Sundarbans, NE Coast of Bay of Bengal, India. J. Mar. Syst. 2006, 62, 9–21. [Google Scholar] [CrossRef]
- Hopkinson, C.S.; Vallino, J.J. The relationships among man’s activities in watersheds and estuaries: A model of runoff effects on patterns of estuarine community metabolism. Estuaries 1995, 18, 598–621. [Google Scholar] [CrossRef]
- Kemp, W.M.; Smith, E.M.; Marvin-DiPasquale, M.; Boynton, W.R. Organic carbon balance and net ecosystem metabolism in Chesapeake Bay. Mar. Ecol. Prog. Ser. 1997, 150, 229–248. [Google Scholar] [CrossRef]
- Cai, W.J.; Wang, Y. The chemistry, fluxes, and sources of carbon dioxide in the estuarine waters of the Satilla and Altamaha Rivers, Georgia. Limnol. Oceanogr. 1998, 43, 657–668. [Google Scholar] [CrossRef]
- Neubauer, S.C.; Anderson, I.C. Transport of dissolved inorganic carbon from a tidal freshwater marsh to the York River estuary. Limnol. Oceanogr. 2003, 48, 299–307. [Google Scholar] [CrossRef]
- Riley, G.A. Particulate organic matter in sea water. In Advances In Marine Biology; Academic Press: Cambridge, MA, USA, 1971; Volume 8, pp. 1–118. [Google Scholar] [CrossRef]
- Happ, G.; Gosselink, J.G.; Day, J.W., Jr. The seasonal distribution of organic carbon in a Louisiana estuary. Estuar. Coast. Mar. Sci. 1977, 5, 695–705. [Google Scholar] [CrossRef]
- Meybeck, M.; Helmer, R. The quality of rivers: From pristine stage to global pollution. Palaeogeogr. Palaeoclimatol. Palaeoecol. 1989, 75, 283–309. [Google Scholar] [CrossRef]
- Peterson, B.; Fry, B.; Hullar, M.; Saupe, S.; Wright, R. The distribution and stable carbon isotopic composition of dissolved organic carbon in estuaries. Estuaries 1994, 17, 111–121. [Google Scholar] [CrossRef]
- Hedges, J.I.; Keil, R.G.; Benner, R. What happens to terrestrial organic matter in the ocean? Org. Geochem. 1997, 27, 195–212. [Google Scholar] [CrossRef]
- Benner, R.; Opsahl, S.; Chin-Leo, G.; Richey, J.E.; Forsberg, B.R. Bacterial carbon metabolism in the Amazon River system. Limnol. Oceanogr. 1995, 40, 1262–1270. [Google Scholar] [CrossRef]
- Moran, M.A.; Sheldon, W.M.; Sheldon, J.E. Biodegradation of riverine dissolved organic carbon in five estuaries of the southeastern United States. Estuaries 1999, 22, 55–64. [Google Scholar] [CrossRef]
- Raymond, P.A.; Bauer, J.E. DOC cycling in a temperate estuary: A mass balance approach using natural 14C and 13C isotopes. Limnol. Oceanogr. 2001, 46, 655–667. [Google Scholar] [CrossRef]
- Gazeau, F.; Gattuso, J.P.; Middelburg, J.J.; Brion, N.; Schiettecatte, L.S.; Frankignoulle, M.; Borges, A.V. Planktonic and whole system metabolism in a nutrient-rich estuary (the Scheldt estuary). Estuaries 2005, 28, 868–883. [Google Scholar] [CrossRef]
- Raymond, P.A.; Bauer, J.E. Bacterial consumption of DOC during transport through a temperate estuary. Aquat. Microb. Ecol. 2000, 22, 1–12. [Google Scholar] [CrossRef]
- Raymond, P.A.; Bauer, J.E.; Cole, J.J. Atmospheric CO2 evasion, dissolved inorganic carbon production, and net heterotrophy in the York River estuary. Limnol. Oceanogr. 2000, 45, 1707–1717. [Google Scholar] [CrossRef]
- Harrison, J.A.; Caraco, N.; Seitzinger, S.P. Global patterns and sources of dissolved organic matter export to the coastal zone: Results from a spatially explicit, global model. Glob. Biogeochem. Cycles 2005, 19. [Google Scholar] [CrossRef]
- Alongi, D.M. Carbon cycling and storage in mangrove forests. Annu. Rev. Mar. Sci. 2014, 6, 195–219. [Google Scholar] [CrossRef]
- Koné, Y.J.M.; Abril, G.; Kouadio, K.N.; Delille, B.; Borges, A.V. Seasonal variability of carbon dioxide in the rivers and lagoons of Ivory Coast (West Africa). Estuaries Coast. 2009, 32, 246–260. [Google Scholar] [CrossRef]
- Van Dam, B.; Fourqurean, J.; Smyth, A. Alkalinity and CO2 fluxes in a tropical seagrass meadow. In Proceedings of the EGU General Assembly Conference Abstracts, Vienna, Austria, 3–8 May 2020. [Google Scholar] [CrossRef]
- Raymond, P.A.; Cole, J.J. Increase in the export of alkalinity from North America’s largest river. Science 2003, 301, 88–91. [Google Scholar] [CrossRef] [PubMed]
- White, A.F.; Blum, A.E. Effects of climate on chemical weathering in watersheds. Geochim. Cosmochim. Acta 1995, 59, 1729–1747. [Google Scholar] [CrossRef]
- Guo, X.; Cai, W.J.; Zhai, W.; Dai, M.; Wang, Y.; Chen, B. Seasonal variations in the inorganic carbon system in the Pearl River (Zhujiang) estuary. Cont. Shelf Res. 2008, 28, 1424–1434. [Google Scholar] [CrossRef]
- White, A.F.; Buss, H.L. Natural weathering rates of silicate minerals. In Treatise on Geochemistry; Drever, J.I., Holland, H.D., Turekian, K.K., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 133–168. [Google Scholar] [CrossRef]
- Cai, W.J.; Xu, Y.Y.; Feely, R.A.; Wanninkhof, R.; Jönsson, B.; Alin, S.R.; Barbero, L.; Cross, J.N.; Azetsu-Scott, K.; Fassbender, A.J.; et al. Controls on surface water carbonate chemistry along North American ocean margins. Nat. Commun. 2020, 11, 2691. [Google Scholar] [CrossRef]
- Su, J.; Cai, W.J.; Brodeur, J.; Chen, B.; Hussain, N.; Yao, Y.; Ni, C.; Testa, J.M.; Li, M.; Xie, X.; et al. Chesapeake Bay acidification buffered by spatially decoupled carbonate mineral cycling. Nat. Geosci. 2020, 13, 441–447. [Google Scholar] [CrossRef]
- Brake, S.; Connors, K.; Romberger, S. A river runs through it: Impact of acid mine drainage on the geochemistry of West Little Sugar Creek pre-and post-reclamation at the Green Valley coal mine, Indiana, USA. Environ. Geol. 2001, 40, 1471–1481. [Google Scholar] [CrossRef]
- Oh, N.H.; Raymond, P.A. Contribution of agricultural liming to riverine bicarbonate export and CO2 sequestration in the Ohio River basin. Glob. Biogeochem. Cycles 2006, 20. [Google Scholar] [CrossRef]
- Zhai, W.; Dai, M.; Cai, W.J.; Wang, Y.; Hong, H. The partial pressure of carbon dioxide and air–sea fluxes in the northern South China Sea in spring, summer and autumn. Mar. Chem. 2005, 96, 87–97. [Google Scholar] [CrossRef]
- Bouillon, S.; Middelburg, J.J.; Dehairs, F.; Borges, A.V.; Abril, G.; Flindt, M.R.; Ulomi, S.; Kristensen, E. Importance of intertidal sediment processes and porewater exchange on the water column biogeochemistry in a pristine mangrove creek (Ras Dege, Tanzania). Biogeosciences 2007, 4, 311–322. [Google Scholar] [CrossRef]
- Borges, A.V.; Djenidi, S.; Lacroix, G.; Théate, J.; Delille, B.; Frankignoulle, M. Atmospheric CO2 flux from mangrove surrounding waters. Geophys. Res. Lett. 2003, 30, 1558. [Google Scholar] [CrossRef]
- Mitra, S.; Ghosh, S.; Satpathy, K.K.; Bhattacharya, B.D.; Sarkar, S.K.; Mishra, P.; Raja, P. Water quality assessment of the ecologically stressed Hooghly River Estuary, India: A multivariate approach. Mar. Pollut. Bull. 2018, 126, 592–599. [Google Scholar] [CrossRef] [PubMed]
- Jeffrey, L.C.; Maher, D.T.; Santos, I.R.; Call, M.; Reading, M.J.; Holloway, C.; Tait, D.R. The spatial and temporal drivers of pCO2, pCH4 and gas transfer velocity within a subtropical estuary. Estuar. Coast. Shelf Sci. 2018, 208, 83–95. [Google Scholar] [CrossRef]
- Tanner, E.L.; Mulhearn, P.J.; Eyre, B.D. CO2 emissions from a temperate drowned river valley estuary adjacent to an emerging megacity (Sydney Harbour). Estuar. Coast. Shelf Sci. 2017, 192, 42–56. [Google Scholar] [CrossRef]
- Lee, K.; Wanninkhof, R.; Feely, R.A.; Millero, F.J.; Peng, T.H. Global relationships of total inorganic carbon with temperature and nitrate in surface seawater. Glob. Biogeochem. Cycles 2000, 14, 979–994. [Google Scholar] [CrossRef]
- Chen, C.T.A.; Huang, T.H.; Fu, Y.H.; Bai, Y.; He, X. Strong sources of CO2 in upper estuaries become sinks of CO2 in large river plumes. Curr. Opin. Environ. Sustain. 2012, 4, 179–185. [Google Scholar] [CrossRef]
- Jayakumar, D.A.; Naqvi, S.W.A.; Narvekar, P.V.; George, M.D. Methane in coastal and offshore waters of the Arabian Sea. Mar. Chem. 2001, 74, 1–13. [Google Scholar] [CrossRef]
- Middelburg, J.J.; Nieuwenhuize, J.; Iversen, N.; Høgh, N.; De Wilde, H.; Helder, W.; Seifert, R.; Christof, O. Methane distribution in European tidal estuaries. Biogeochemistry 2002, 59, 95–119. [Google Scholar] [CrossRef]
- Abril, G.; Iversen, N. Methane dynamics in a shallow non-tidal estuary (Randers Fjord, Denmark). Mar. Ecol. Prog. Ser. 2002, 230, 171–181. [Google Scholar] [CrossRef]
- Padhy, S.R.; Bhattacharyya, P.; Dash, P.K.; Reddy, C.S.; Chakraborty, A.; Pathak, H. Seasonal fluctuation in three mode of greenhouse gases emission in relation to soil labile carbon pools in degraded mangrove, Sundarban, India. Sci. Total Environ. 2020, 705, 135909. [Google Scholar] [CrossRef]
- Weiss, R. Carbon dioxide in water and seawater: The solubility of a non-ideal gas. Mar. Chem. 1974, 2, 203–215. [Google Scholar] [CrossRef]
- Takahashi, T. Carbon dioxide in the atmosphere and in Atlantic Ocean water. J. Geophys. Res. 1961, 66, 477–494. [Google Scholar] [CrossRef]
- Banaszak, A.T.; Neale, P.J. Ultraviolet radiation sensitivity of photosynthesis in phytoplankton from an estuarine environment. Limnol. Oceanogr. 2001, 46, 592–603. [Google Scholar] [CrossRef]
- Ganju, N.K.; Testa, J.M.; Suttles, S.E.; Aretxabaleta, A.L. Spatiotemporal variability of light attenuation and net ecosystem metabolism in a back-barrier estuary. Ocean Sci. 2020, 16, 593–614. [Google Scholar] [CrossRef]
- Domingues, R.B.; Anselmo, T.P.; Barbosa, A.B.; Sommer, U.; Galvão, H.M. Light as a driver of phytoplankton growth and production in the freshwater tidal zone of a turbid estuary. Estuar. Coast. Shelf Sci. 2011, 91, 526–535. [Google Scholar] [CrossRef]
- Sadhuram, Y.; Sarma, V.V.; Murthy, T.V.; Rao, B.P. Seasonal variability of physico-chemical characteristics of the Haldia channel of Hooghly estuary, India. J. Earth Syst. Sci. 2005, 114, 37–49. [Google Scholar] [CrossRef]
- Kumar, M.D.; Naqvi, S.W.A.; George, M.D.; Jayakumar, D.A. A sink for atmospheric carbon dioxide in the northeast Indian Ocean. J. Geophys. Res. Oceans 1996, 101, 18121–18125. [Google Scholar] [CrossRef]
- Frankignoulle, M.; Abril, G.; Borges, A.; Bourge, I.; Canon, C.; Delille, B.; Libert, E.; Théate, J.M. Carbon dioxide emission from European estuaries. Science 1998, 282, 434–436. [Google Scholar] [CrossRef]
- Liu, Q.; Charette, M.A.; Breier, C.F.; Henderson, P.B.; McCorkle, D.C.; Martin, W.; Dai, M. Carbonate system biogeochemistry in a subterranean estuary–Waquoit Bay, USA. Geochim. Cosmochim. Acta 2017, 203, 422–439. [Google Scholar] [CrossRef]
- Chen, C.T.A.; Wang, S.L.; Lu, X.X.; Zhang, S.R.; Lui, H.K.; Tseng, H.C.; Wang, B.J.; Huang, H.I. Hydrogeochemistry and greenhouse gases of the Pearl River, its estuary and beyond. Quat. Int. 2008, 186, 79–90. [Google Scholar] [CrossRef]
- Upstill-Goddard, R.C.; Salter, M.E.; Mann, P.J.; Barnes, J.; Poulsen, J.; Dinga, B.; Fiske, G.J.; Holmes, R.M. The riverine source of CH4 and N2O from the Republic of Congo, western Congo Basin. Biogeosciences 2017, 14, 2267–2281. [Google Scholar] [CrossRef]
- Krupadam, R.J.; Ahuja, R.; Wate, S.R.; Anjaneyulu, Y. Forest bound estuaries are higher methane emitters than paddy fields: A case of Godavari estuary, East Coast of India. Atmos. Environ. 2007, 41, 4819–4827. [Google Scholar] [CrossRef]
- Kristensen, E.; Flindt, M.R.; Ulomi, S.; Borges, A.V.; Abril, G.; Bouillon, S. Emission of CO2 and CH4 to the atmosphere by sediments and open waters in two Tanzanian mangrove forests. Mar. Ecol. Prog. Ser. 2008, 370, 53–67. [Google Scholar] [CrossRef]
- Das, N.; Mondal, A.; Mandal, S. Dynamics of methane and carbon dioxide emissions in the reclaimed islands of Sundarban mangrove ecosystem, India. Austral Ecol. 2022, 47, 412–427. [Google Scholar] [CrossRef]
- Moore, W.S. The effect of submarine groundwater discharge on the ocean. Annu. Rev. Mar. Sci. 2010, 2, 59–88. [Google Scholar] [CrossRef]
- Keating, E.H.; Fessenden, J.; Kanjorski, N.; Koning, D.J.; Pawar, R. The impact of CO2 on shallow groundwater chemistry: Observations at a natural analog site and implications for carbon sequestration. Environ. Earth Sci. 2010, 60, 521–536. [Google Scholar] [CrossRef]
- Jiang, L.Q.; Cai, W.J.; Wang, Y. A comparative study of carbon dioxide degassing in river-and marine-dominated estuaries. Limnol. Oceanogr. 2008, 53, 2603–2615. [Google Scholar] [CrossRef]
- Rudra, K. Changing River courses in the western part of the Ganga-Brahmaputra delta. Geomorphology 2014, 227, 87–100. [Google Scholar] [CrossRef]
- Ghosh, A.; Bhadury, P. Investigating monsoon and post-monsoon variabilities of bacterioplankton communities in a mangrove ecosystem. Environ. Sci. Pollut. Res. 2018, 25, 5722–5739. [Google Scholar] [CrossRef]
- Bhadury, P.; Singh, T. Analysis of marine planktonic cyanobacterial assemblages from Mooriganga Estuary, Indian Sundarbans using molecular approaches. Front. Mar. Sci. 2020, 7, 222. [Google Scholar] [CrossRef]
- Das, S.; De, M.; Ganguly, D.; Maiti, T.K.; Mukherjee, A.; Jana, T.K.; De, T.K. Depth integrated microbial community and physico-chemical properties in mangrove soil of Sundarban, India. Adv. Microbiol. 2012, 2, 234. [Google Scholar] [CrossRef]
- Das, S.; De, M.; Jana, T.K.; De, T.K. Environmental influence on cultivable microbial community in the sediment of Sundarban mangrove forest, India. Afr. J. Microbiol. Res. 2013, 7, 4655–4665. [Google Scholar]
- Mukherjee, R.; Dutta, M.K.; Sanyal, P.; Bhadury, P.; Mukhopadhyay, S.K. Bacterioplankton abundance and community structure during post-monsoon in mangrove dominated estuaries of the Indian Sundarbans; An insight to biogeochemical processes. Estuar. Coast. Shelf Sci. 2020, 243, 106895. [Google Scholar] [CrossRef]
- Dhal, P.K.; Kopprio, G.A.; Gärdes, A. Insights on aquatic microbiome of the Indian Sundarbans mangrove areas. PLoS ONE 2020, 15, 0221543. [Google Scholar] [CrossRef]
- Ciais, P.; Borges, A.V.; Abril, G.; Meybeck, M.; Folberth, G.; Hauglustaine, D.; Janssens, I.A. The impact of lateral carbon fluxes on the European carbon balance. Biogeosciences 2008, 5, 1259–1271. [Google Scholar] [CrossRef]
- Regnier, P.; Friedlingstein, P.; Ciais, P.; Mackenzie, F.T.; Gruber, N.; Janssens, I.A.; Laruelle, G.G.; Lauerwald, R.; Luyssaert, S.; Andersson, A.J.; et al. Anthropogenic perturbation of the carbon fluxes from land to ocean. Nat. Geosci. 2013, 6, 597–607. [Google Scholar] [CrossRef]
- Santos, I.R.; Maher, D.T.; Larkin, R.; Webb, J.R.; Sanders, C.J. Carbon outwelling and outgassing vs. burial in an estuarine tidal creek surrounded by mangrove and saltmarsh wetlands. Limnol. Oceanogr. 2019, 64, 996–1013. [Google Scholar] [CrossRef]
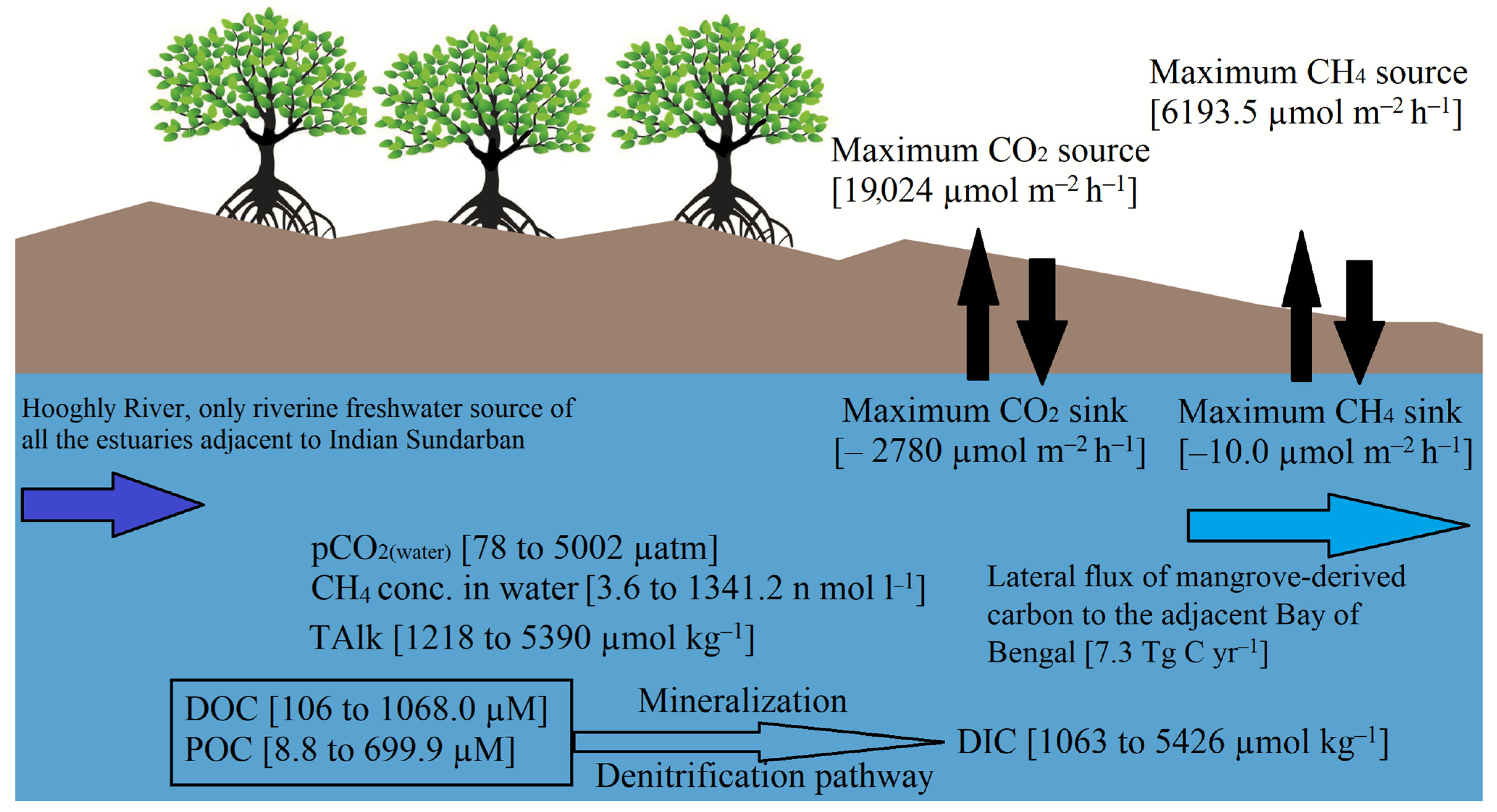
| Parameters | Inner Estuary | Middle Estuary | Outer Estuary |
|---|---|---|---|
| TAlk (µmol kg−1) | 1219–5391 | 1369–4549 | 1218–3189 |
| DIC (µmol kg−1) | 1063–5126 | 1240–4111 | 990–2882 |
| DOC (µmol L−1) | 226.9–1068 | 249–263.6 | 10.6–328.2 |
| POC (µmol L−1) | 8.8–62.3 | 43.2–129.7 | 27–699.9 |
| pCO2(water) (µatm) | 204–5851 | 279–2786 | 98–2015 |
| Air–water CO2 flux (µmol m−2 h−1) | −301–16944 | −249–9743 | −860–7230 |
| CH4 conc. in water (nmol L−1) | 19.67–445.7 | 17.25–53.5 | 3.6–90.91 |
| Air–water CH4 flux (µmol m−2 h−1) | −10.0–2801.2 | 61.6–3658.4 | 0.3–6193.5 |
| Parameters | Pre-Monsoon | Monsoon | Post-Monsoon |
|---|---|---|---|
| TAlk (µmol kg−1) | 1356–5391 | 1473–3300 | 1218–4419 |
| DIC (µmol kg−1) | 990–5126 | 1084–3431 | 1063–4469 |
| DOC (µmol L−1) | 82.7–1068 | 91.8–249.2 | 10.6–662 |
| POC (µmol L−1) | 11–699.9 | 43–87.3 | 8.8–457 |
| pCO2(water) (µatm) | 143–3295 | 123–5851 | 98–4803 |
| Air–water CO2 flux (µmol m−2 h−1) | −860–16,944 | −350–19,024 | −290–9448 |
| CH4 conc. in water (nmol L−1) | 15.4–445.7 | 13.65–327.2 | 3.6–117.2 |
| Air–water CH4 flux (µmol m−2 h−1) | 0.3–6193.5 | 14.2–515.06 | −10.0–1230.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, I.; Chanda, A.; Akhand, A.; Hazra, S. Carbon Biogeochemistry of the Estuaries Adjoining the Indian Sundarbans Mangrove Ecosystem: A Review. Life 2023, 13, 863. https://doi.org/10.3390/life13040863
Das I, Chanda A, Akhand A, Hazra S. Carbon Biogeochemistry of the Estuaries Adjoining the Indian Sundarbans Mangrove Ecosystem: A Review. Life. 2023; 13(4):863. https://doi.org/10.3390/life13040863
Chicago/Turabian StyleDas, Isha, Abhra Chanda, Anirban Akhand, and Sugata Hazra. 2023. "Carbon Biogeochemistry of the Estuaries Adjoining the Indian Sundarbans Mangrove Ecosystem: A Review" Life 13, no. 4: 863. https://doi.org/10.3390/life13040863
APA StyleDas, I., Chanda, A., Akhand, A., & Hazra, S. (2023). Carbon Biogeochemistry of the Estuaries Adjoining the Indian Sundarbans Mangrove Ecosystem: A Review. Life, 13(4), 863. https://doi.org/10.3390/life13040863








