Functional Outcomes and Safety Profile of Trans-Foveal Subthreshold Micropulse Laser in Persistent Central Serous Chorioretinopathy
Abstract
:1. Introduction
2. Materials and Methods
Statistical Analysis
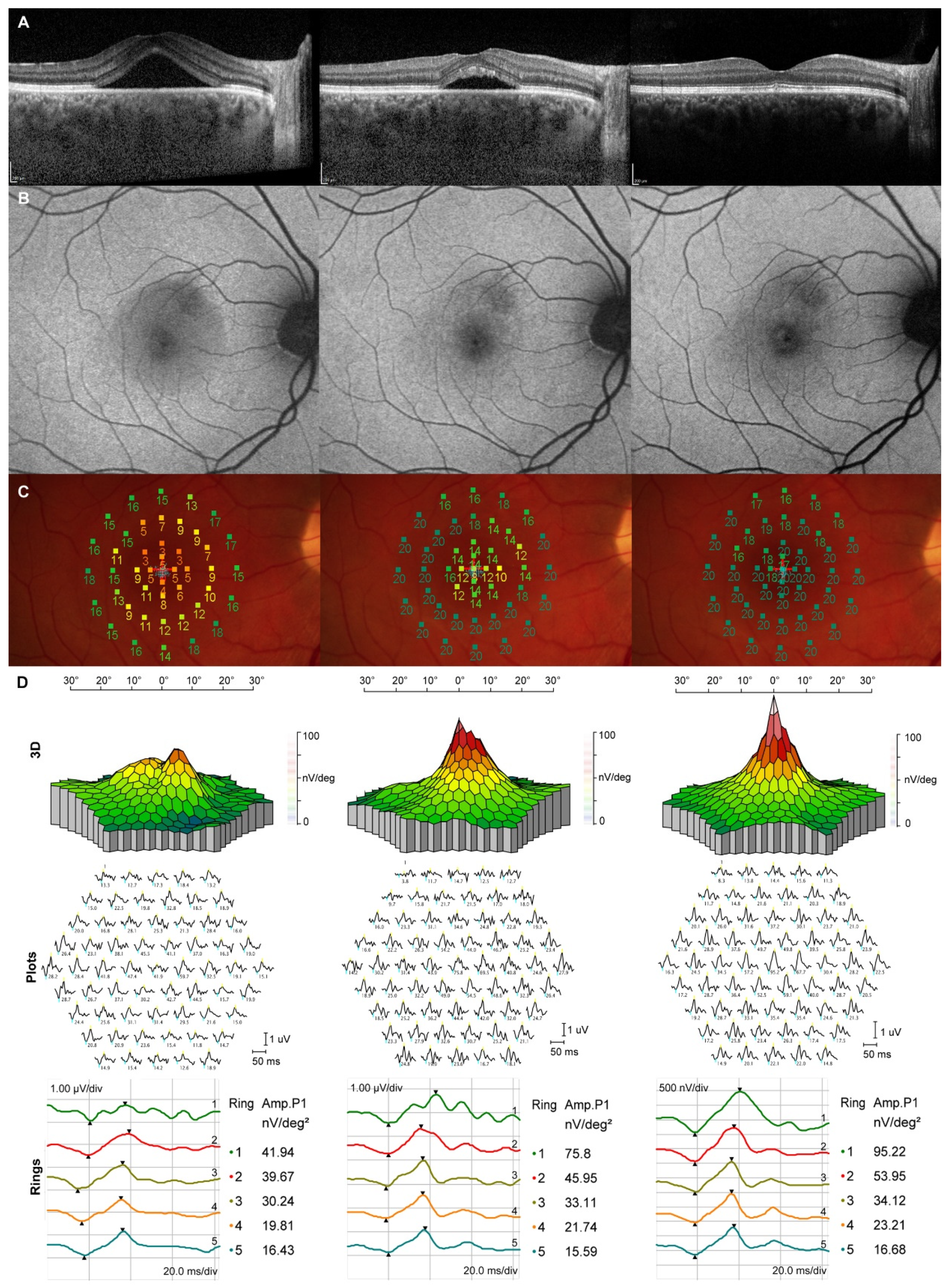
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| gSML | sCSC | p | |
|---|---|---|---|
| BCVA, logmar, ± SD | 0.02 ± 0.04 | 0.03 ± 0.06 | 0.520 |
| CS-1.5, logmar, ± SD | 1.64 ± 0.18 | 1.71 ± 0.19 | 0.210 |
| CS-3.0, logmar, ± SD | 1.8 ± 0.13 | 1.84 ± 0.22 | 0.802 |
| CS-6.0, logmar, ± SD | 1.74 ± 0.22 | 1.77 ± 0.27 | 0.358 |
| CS-12.0, logmar, ± SD | 1.22 ± 0.51 | 1.25 ± 0.62 | 0.433 |
| CS-18.0, logmar, ± SD | 0.81 ± 0.53 | 0.83 ± 0.59 | 0.923 |
| CS-A, logmar, ± SD | 1.44 ± 0.25 | 1.48 ± 0.34 | 0.520 |
| MP-A, db, ± SD | 18.1 ± 1.88 | 17.74 ± 2.38 | 0.880 |
| MP-C, db, ± SD | 17.6 ± 2.72 | 17.22 ± 3.16 | 1.000 |
| MP-P, db, ± SD | 18.29 ± 1.66 | 17.96 ± 2.11 | 1.000 |
| mfERG-A1, nV/deg2, ± SD | 88.66 ± 27.13 | 99.69 ± 27.63 | 0.317 |
| mfERG-A2, nV/deg2, ± SD | 46.5 ± 10.72 | 49.2 ± 10.38 | 0.623 |
| mfERG-A3, nV/deg2, ± SD | 26.39 ± 6.86 | 27.91 ± 5.45 | 0.558 |
| mfERG-A4, nV/deg2, ± SD | 18.25 ± 4.29 | 18.62 ± 3.29 | 0.985 |
| mfERG-A5, nV/deg2, ± SD | 14.12 ± 3.61 | 14.16 ± 2.95 | 0.940 |
| mfERG-IT1, ms, ± SD | 42.09 ± 2.73 | 42.19 ± 2.38 | 0.834 |
| mfERG-IT2, ms, ± SD | 39.25 ± 1.14 | 38.3 ± 1.33 | 0.031 |
| mfERG-IT3, ms, ± SD | 37.11 ± 1.03 | 36.67 ± 1.36 | 0.240 |
| mfERG-IT4, ms, ± SD | 36.99 ± 1.05 | 36.25 ± 1.26 | 0.210 |
| mfERG-IT5, ms, ± SD | 37.37 ± 1.27 | 36.88 ± 1.45 | 0.559 |
References
- Daruich, A.; Matet, A.; Marchionno, L.; De Azevedo, J.D.; Ambresin, A.; Mantel, I.; Behar-Cohen, F. Acute Central Serous Chorioretinopathy: Factors Influencing Episode Duration. Retina 2017, 37, 1905–1915. [Google Scholar] [CrossRef] [PubMed]
- Baran, N.V.; Gürlü, V.P.; Esgin, H. Long-term macular function in eyes with central serous chorioretinopathy. Clin. Exp. Ophthalmol. 2005, 33, 369–372. [Google Scholar] [CrossRef] [PubMed]
- Lourthai, P.; Bhurayanontachai, P. Pattern of Contrast Sensitivity Changes in Acute Central Serous Chorioretinopathy. J. Ophthalmol. 2017, 2017, 9053932. [Google Scholar] [CrossRef] [PubMed]
- Maaranen, T.; Mäntyjärvi, M. Contrast sensitivity in patients recovered from central serous chorioretinopathy. Int. Ophthalmol. 1999, 23, 31–35. [Google Scholar] [CrossRef]
- Ozdemir, H.; Karacorlu, S.A.; Senturk, F.; Karacorlu, M.; Uysal, O. Assessment of macular function by microperimetry in unilateral resolved central serous chorioretinopathy. Eye 2008, 22, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Chappelow, A.V.; Marmor, M.F. Multifocal electroretinogram abnormalities persist following resolution of central serous chorioretinopathy. Arch. Ophthalmol. 2000, 118, 1211–1215. [Google Scholar] [CrossRef]
- Suzuki, K.; Hasegawa, S.; Usui, T.; Ichibe, M.; Takada, R.; Takagi, M.; Abe, H. Multifocal electroretinogram in patients with central serous chorioretinopathy. Jpn. J. Ophthalmol. 2002, 46, 308–314. [Google Scholar] [CrossRef]
- Yu, J.; Xu, G.; Chang, Q.; Ye, X.; Li, L.; Jiang, C.; Zhao, Q. Risk Factors for Persistent or Recurrent Central Serous Chorioretinopathy. J. Ophthalmol. 2019, 2019, 5970659. [Google Scholar] [CrossRef]
- Mrejen, S.; Balaratnasingam, C.; Kaden, T.R.; Bottini, A.; Dansingani, K.; Bhavsar, K.V.; Yannuzzi, N.A.; Patel, S.; Chen, K.C.; Yu, S.; et al. Long-term Visual Outcomes and Causes of Vision Loss in Chronic Central Serous Chorioretinopathy. Ophthalmology 2019, 126, 576–588. [Google Scholar] [CrossRef]
- van Dijk, E.H.C.; Fauser, S.; Breukink, M.B.; Blanco-Garavito, R.; Groenewoud, J.M.M.; Keunen, J.E.E.; Peters, P.J.H.; Dijkman, G.; Souied, E.H.; MacLaren, R.E.; et al. Half-Dose Photodynamic Therapy versus High-Density Subthreshold Micropulse Laser Treatment in Patients with Chronic Central Serous Chorioretinopathy: The PLACE Trial. Ophthalmology 2018, 125, 1547–1555. [Google Scholar] [CrossRef]
- Robertson, D.M.; Ilstrup, D. Direct, indirect, and sham laser photocoagulation in the management of central serous chorioretinopathy. Am. J. Ophthalmol. 1983, 95, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Bousquet, E.; Zhao, M.; Daruich, A.; Behar-Cohen, F. Mineralocorticoid antagonists in the treatment of central serous chorioetinopathy: Review of the pre-clinical and clinical evidence. Exp. Eye Res. 2019, 187, 107754. [Google Scholar] [CrossRef] [PubMed]
- Scholz, P.; Altay, L.; Fauser, S. A Review of Subthreshold Micropulse Laser for Treatment of Macular Disorders. Adv. Ther. 2017, 34, 1528–1555. [Google Scholar] [CrossRef] [PubMed]
- Miura, Y.; Inagaki, K.; Hutfilz, A.; Seifert, E.; Schmarbeck, B.; Murakami, A.; Ohkoshi, K.; Brinkmann, R. Temperature Increase and Damage Extent at Retinal Pigment Epithelium Compared between Continuous Wave and Micropulse Laser Application. Life 2022, 12, 1313. [Google Scholar] [CrossRef]
- Gawęcki, M. Micropulse Laser Treatment of Retinal Diseases. J. Clin. Med. 2019, 8, 242. [Google Scholar] [CrossRef]
- Gawęcki, M.; Jaszczuk-Maciejewska, A.; Jurska-Jaśko, A.; Kneba, M.; Grzybowski, A. Transfoveal Micropulse Laser Treatment of Central Serous Chorioretinopathy within Six Months of Disease Onset. J. Clin. Med. 2019, 8, 1398. [Google Scholar] [CrossRef]
- van Rijssen, T.J.; van Dijk, E.H.C.; Yzer, S.; Ohno-Matsui, K.; Keunen, J.E.E.; Schlingemann, R.O.; Sivaprasad, S.; Querques, G.; Downes, S.M.; Fauser, S.; et al. Central serous chorioretinopathy: Towards an evidence-based treatment guideline. Prog. Retin. Eye Res. 2019, 73, 100770. [Google Scholar] [CrossRef]
- Behnia, M.; Khabazkhoob, M.; Aliakbari, S.; Abadi, A.E.; Hashemi, H.; Pourvahidi, P. Improvement in visual acuity and contrast sensitivity in patients with central serous chorioretinopathy after macular subthreshold laser therapy. Retina 2013, 33, 324–328. [Google Scholar] [CrossRef]
- Arora, S.; Sridharan, P.; Arora, T.; Chhabra, M.; Ghosh, B. Subthreshold diode micropulse laser versus observation in acute central serous chorioretinopathy. Clin. Exp. Optom. 2019, 102, 79–85. [Google Scholar] [CrossRef]
- Goel, N.; Mehta, A.; Gupta, A.K. Multifocal electroretinography-assisted anatomical and functional evaluation of subthreshold green laser in acute central serous chorioretinopathy. Indian J. Ophthalmol. 2021, 69, 2341–2346. [Google Scholar] [CrossRef]
- Long, H.; Liu, M.; Hu, Q.; Li, X. 577 nm subthreshold micropulse laser treatment for acute central serous chorioretinopathy: A comparative study. BMC Ophthalmol. 2022, 22, 105. [Google Scholar] [CrossRef] [PubMed]
- Kiraly, P.; Smrekar, J.; Jaki Mekjavić, P. Morphological parameters predicting subthreshold micropulse laser effectiveness in central serous chorioretinopathy. Lasers Med. Sci. 2022, 37, 3129–3136. [Google Scholar] [CrossRef] [PubMed]
- Lainscak, M.; Pelliccia, F.; Rosano, G.; Vitale, C.; Schiariti, M.; Greco, C.; Speziale, G.; Gaudio, C. Safety profile of mineralocorticoid receptor antagonists: Spironolactone and eplerenone. Int. J. Cardiol. 2015, 200, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Iwase, T.; Yokouchi, H.; Kitahashi, M.; Kubota-Taniai, M.; Baba, T.; Yamamoto, S. Long-Term Effects of Half-Time Photodynamic Therapy on Retinal Sensitivity in Eyes with Chronic Central Serous Chorioretinopathy. BioMed Res. Int. 2020, 2020, 3190136. [Google Scholar] [CrossRef]
- Sun, Z.; Huang, Y.; Nie, C.; Wang, Z.; Pei, J.; Lin, B.; Zhou, R.; Zhang, J.; Chong, V.; Liu, X. Efficacy and safety of subthreshold micropulse laser compared with threshold conventional laser in central serous chorioretinopathy. Eye 2020, 34, 1592–1599. [Google Scholar] [CrossRef]
- Chhablani, J.; Kalra, G.; Alkwatli, L.; Fassbender, B.; Amoroso, F.; Chandra, K.; Ankireddy, S.; Maltsev, D.; Striebe, N.A.; Souied, E. Safety of various parameter sets with navigated microsecond pulsing laser in central serous chorioretinopathy. Int. J. Retin. Vitr. 2021, 7, 62. [Google Scholar] [CrossRef]
- Takahashi, A.; Ooto, S.; Yamashiro, K.; Oishi, A.; Tamura, H.; Nakanishi, H.; Ueda-Arakawa, N.; Tsujikawa, A.; Yoshimura, N. Photoreceptor Damage and Reduction of Retinal Sensitivity Surrounding Geographic Atrophy in Age-Related Macular Degeneration. Am. J. Ophthalmol. 2016, 168, 260–268. [Google Scholar] [CrossRef]
- Gerth, C.; Hauser, D.; Delahunt, P.B.; Morse, L.S.; Werner, J.S. Assessment of multifocal electroretinogram abnormalities and their relation to morphologic characteristics in patients with large drusen. Arch. Ophthalmol. 2003, 121, 1404–1414. [Google Scholar] [CrossRef]
- Kaiser, P.K. Prospective evaluation of visual acuity assessment: A comparison of snellen versus ETDRS charts in clinical practice (An AOS Thesis). Trans. Am. Ophthalmol. Soc. 2009, 107, 311–324. [Google Scholar]
- Hitchcock, E.M.; Dick, R.B.; Krieg, E.F. Visual contrast sensitivity testing: A comparison of two F.A.C.T. test types. Neurotoxicol. Teratol. 2004, 26, 271–277. [Google Scholar] [CrossRef]
- Vujosevic, S.; Midena, E.; Pilotto, E.; Radin, P.P.; Chiesa, L.; Cavarzeran, F. Diabetic macular edema: Correlation between microperimetry and optical coherence tomography findings. Investig. Ophthalmol. Vis. Sci. 2006, 47, 3044–3051. [Google Scholar] [CrossRef] [PubMed]
- Midena, E.; Vujosevic, S.; Cavarzeran, F. Normal values for fundus perimetry with the microperimeter MP1. Ophthalmology 2010, 117, 1571–1576.e1. [Google Scholar] [CrossRef] [PubMed]
- Hood, D.C.; Bach, M.; Brigell, M.; Keating, D.; Kondo, M.; Lyons, J.S.; Marmor, M.F.; McCulloch, D.L.; Palmowski-Wolfe, A.M. ISCEV standard for clinical multifocal electroretinography (mfERG) (2011 edition). Doc. Ophthalmologica. Adv. Ophthalmol. 2012, 124, 1–13. [Google Scholar] [CrossRef]
- Hawlina, M.; Konec, B. New noncorneal HK-loop electrode for clinical electroretinography. Doc. Ophthalmol. Adv. Ophthalmol. 1992, 81, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Midena, E.; Bini, S.; Martini, F.; Enrica, C.; Pilotto, E.; Micera, A.; Esposito, G.; Vujosevic, S. Changes of aqueous humor müller cells’ biomarkers in human patients affected by diabetic macular edema after subthreshold micropulse laser treatment. Retina 2020, 40, 126–134. [Google Scholar] [CrossRef]
- Kiraly, P.; Smrekar, J.; Mekjavić, P.J. Visual function during and after an acute central serous chorioretinopathy episode. Doc. Ophthalmol. Adv. Ophthalmol. 2022, 145, 27–35. [Google Scholar] [CrossRef]
- Moschos, M.; Brouzas, D.; Koutsandrea, C.; Stefanos, B.; Loukianou, H.; Papantonis, F.; Moschos, M. Assessment of central serous chorioretinopathy by optical coherence tomography and multifocal electroretinography. Ophthalmologica 2007, 221, 292–298. [Google Scholar] [CrossRef]
- Luttrull, J.K. Improved retinal and visual function following panmacular subthreshold diode micropulse laser for retinitis pigmentosa. Eye 2018, 32, 1099–1110. [Google Scholar] [CrossRef]
- Luttrull, J.K.; Margolis, B.W. Functionally Guided Retinal Protective Therapy for Dry Age-Related Macular and Inherited Retinal Degenerations: A Pilot Study. Investig. Ophthalmol. Vis. Sci. 2016, 57, 265–275. [Google Scholar] [CrossRef]
- Luttrull, J.K.; Dorin, G. Subthreshold diode micropulse laser photocoagulation (SDM) as invisible retinal phototherapy for diabetic macular edema: A review. Curr. Diabetes Rev. 2012, 8, 274–284. [Google Scholar] [CrossRef]
- Frizziero, L.; Calciati, A.; Midena, G.; Torresin, T.; Parrozzani, R.; Pilotto, E.; Midena, E. Subthreshold Micropulse Laser Modulates Retinal Neuroinflammatory Biomarkers in Diabetic Macular Edema. J. Clin. Med. 2021, 10, 3134. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Kim, S.Y.; Ha, S.; Moon, D.; Seong, S.; Kwon, O.W.; Park, H.S. Short-duration multiple-session subthreshold micropulse yellow laser (577 nm) for chronic central serous chorioretinopathy: Results at 3 years. Eye 2019, 33, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Maltsev, D.S.; Kulikov, A.N.; Chhablani, J. Clinical Application of Fluorescein Angiography-Free Navigated Focal Laser Photocoagulation in Central Serous Chorioretinopathy. Ophthalmic Surg. Lasers Imaging Retin. 2019, 50, e118–e124. [Google Scholar] [CrossRef] [PubMed]
- Donati, M.C.; Murro, V.; Mucciolo, D.P.; Giorgio, D.; Cinotti, G.; Virgili, G.; Rizzo, S. Subthreshold yellow micropulse laser for treatment of diabetic macular edema: Comparison between fixed and variable treatment regimen. Eur. J. Ophthalmol. 2021, 31, 1254–1260. [Google Scholar] [CrossRef]
- Ludwig, K.; Lasser, T.; Sakowski, H.; Abramowski, H.; Wörz, G. Photocoagulation in the edematous and non-edematous retina with the cw-laser of different wavelengths. Der Ophthalmol. Z. Der Dtsch. Ophthalmol. Ges. 1994, 91, 783–788. [Google Scholar]
- van Rijssen, T.J.; van Dijk, E.H.C.; Tsonaka, R.; Feenstra, H.M.A.; Dijkman, G.; Peters, P.J.H.; Diederen, R.M.H.; Hoyng, C.B.; Schlingemann, R.O.; Boon, C.J.F. Half-Dose Photodynamic Therapy versus Eplerenone in Chronic Central Serous Chorioretinopathy (SPECTRA): A Randomized Controlled Trial. Am. J. Ophthalmol. 2022, 233, 101–110. [Google Scholar] [CrossRef]
- Zeng, M.; Chen, X.; Song, Y.; Cai, C. Subthreshold micropulse laser photocoagulation versus half-dose photodynamic therapy for acute central serous chorioretinopathy. BMC Ophthalmol. 2022, 22, 110. [Google Scholar] [CrossRef]
- Raizada, K.; Naik, M. Photodynamic Therapy for the Eye. In StatPearls; Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Sirks, M.J.; van Dijk, E.H.C.; Rosenberg, N.; Hollak, C.E.M.; Aslanis, S.; Cheung, C.M.G.; Chowers, I.; Eandi, C.M.; Freund, K.B.; Holz, F.G.; et al. Clinical impact of the worldwide shortage of verteporfin (Visudyne®) on ophthalmic care. Acta Ophthalmol. 2022, 100, e1522–e1532. [Google Scholar] [CrossRef]
- Inagaki, K.; Shuo, T.; Katakura, K.; Ebihara, N.; Murakami, A.; Ohkoshi, K. Sublethal Photothermal Stimulation with a Micropulse Laser Induces Heat Shock Protein Expression in ARPE-19 Cells. J. Ophthalmol. 2015, 2015, 729792. [Google Scholar] [CrossRef]
- Li, Z.; Song, Y.; Chen, X.; Chen, Z.; Ding, Q. Biological Modulation of Mouse RPE Cells in Response to Subthreshold Diode Micropulse Laser Treatment. Cell Biochem. Biophys. 2015, 73, 545–552. [Google Scholar] [CrossRef]
| Baseline | 3 Months (SML) | 6 Months | p1 | p2 | |
|---|---|---|---|---|---|
| MV, mm3, ± SD | 2.86 ± 0.66 | 2.55 ± 0.25 | 2.49 ± 0.33 | 0.018 | 0.247 |
| CRT, μm, ± SD | 423 ± 135 | 358 ± 82 | 319 ± 94 | 0.008 | 0.044 |
| BCVA, logmar, ± SD | 0.19 ± 0.16 | 0.16 ± 0.15 | 0.09 ± 0.14 | 0.090 | 0.007 |
| CS-1.5, logmar, ± SD | 1.46 ± 0.13 | 1.51 ± 0.14 | 1.59 ± 0.18 | 0.019 | 0.020 |
| CS-3.0, logmar, ± SD | 1.51 ± 0.21 | 1.61 ± 0.19 | 1.69 ± 0.21 | 0.012 | 0.050 |
| CS-6.0, logmar, ± SD | 1.18 ± 0.56 | 1.43 ± 0.43 | 1.47 ± 0.61 | 0.005 | 0.121 |
| CS-12.0, logmar, ± SD | 0.5 ± 0.61 | 0.67 ± 0.63 | 1.02 ± 0.58 | 0.180 | <0.001 |
| CS-18.0, logmar, ± SD | 0.16 ± 0.35 | 0.3 ± 0.42 | 0.62 ± 0.51 | 0.084 | 0.002 |
| CS-A, logmar, ± SD | 0.97 ± 0.28 | 1.1 ± 0.29 | 1.28 ± 0.36 | 0.006 | <0.001 |
| MP-C, db, ± SD | 9.9 ± 5 | 11.5 ± 4.8 | 14.4 ± 5.3 | 0.035 | 0.002 |
| MP-P, db, ± SD | 13.7 ± 4.2 | 14.8 ± 4 | 16.3 ± 3.2 | 0.044 | 0.042 |
| MP-A, db, ± SD | 12.6 ± 4.3 | 13.9 ± 4.2 | 15.8 ± 3.8 | 0.033 | 0.010 |
| mfERG-A1, nV/deg2, ± SD | 58.2 ± 22.5 | 68.1 ± 24 | 77.4 ± 28.1 | 0.004 | 0.065 |
| mfERG-A2, nV/deg2, ± SD | 37.7 ± 10.4 | 40.5 ± 9.4 | 43.4 ± 9.9 | 0.011 | 0.244 |
| mfERG-A3, nV/deg2, ± SD | 25.1 ± 5.4 | 25.9 ± 6.3 | 25.7 ± 5.3 | 0.247 | 0.769 |
| mfERG-A4, nV/deg2, ± SD | 17 ± 3.5 | 16.9 ± 3.7 | 17.6 ± 3.6 | 0.794 | 0.272 |
| mfERG-A5, nV/deg2, ± SD | 13 ± 2.8 | 12.7 ± 2.8 | 13.4 ± 3.1 | 0.350 | 0.267 |
| mfERG-IT1, ms, ± SD | 41.6 ± 4.5 | 42.6 ± 4.7 | 43.3 ± 3 | 0.111 | 0.764 |
| mfERG-IT2, ms, ± SD | 39.2 ± 1.7 | 38.7 ± 1.7 | 39.3 ± 1.3 | 0.243 | 0.206 |
| mfERG-IT3, ms, ± SD | 37.5 ± 1.1 | 37.4 ± 1.1 | 37.2 ± 1.1 | 0.410 | 0.157 |
| mfERG-IT4, ms, ± SD | 37.1 ± 1.1 | 37 ± 1 | 37 ± 1 | 0.320 | 0.878 |
| mfERG-IT5, ms, ± SD | 37.6 ± 1 | 37.6 ± 1.1 | 37.6 ± 1.3 | 0.547 | 0.903 |
| Baseline | 3 Months (SML) | 6 Months | p1 | p2 | |
|---|---|---|---|---|---|
| MV, mm3, ± SD | 2.84 ± 0.59 | 2.52 ± 0.15 | 2.3 ± 0.13 | 0.014 | 0.001 |
| CRT, μm, ± SD | 409 ± 130 | 333 ± 52 | 256 ± 21 | 0.014 | <0.001 |
| BCVA, logmar, ± SD | 0.18 ± 0.16 | 0.12 ± 0.15 | 0.02 ± 0.04 | 0.014 | 0.004 |
| CS-1.5, logmar, ± SD | 1.51 ± 0.14 | 1.54 ± 0.13 | 1.64 ± 0.18 | 0.174 | 0.074 |
| CS-3.0, logmar, ± SD | 1.56 ± 0.21 | 1.68 ± 0.16 | 1.8 ± 0.13 | 0.115 | 0.021 |
| CS-6.0, logmar, ± SD | 1.28 ± 0.56 | 1.51 ± 0.46 | 1.74 ± 0.22 | 0.074 | 0.006 |
| CS-12.0, logmar, ± SD | 0.57 ± 0.65 | 0.87 ± 0.65 | 1.22 ± 0.51 | 0.149 | 0.006 |
| CS-18.0, logmar, ± SD | 0.18 ± 0.4 | 0.33 ± 0.44 | 0.81 ± 0.53 | 0.201 | 0.004 |
| CS-A, logmar, ± SD | 1.02 ± 0.29 | 1.19 ± 0.28 | 1.44 ± 0.25 | 0.108 | |
| MP-C, db, ± SD | 10.5 ± 5.3 | 13.1 ± 5.2 | 17.6 ± 2.7 | 0.022 | 0.001 |
| MP-P, db, ± SD | 14.2 ± 4.1 | 16 ± 3.7 | 18.3 ± 1.7 | 0.080 | 0.013 |
| MP-A, db, ± SD | 13.2 ± 4.3 | 15.2 ± 4 | 18.1 ± 1.9 | 0.080 | 0.004 |
| mfERG-A1, nV/deg2, ± SD | 59.2 ± 20.4 | 69.6 ± 25.5 | 88.7 ± 27.1 | 0.042 | 0.010 |
| mfERG-A2, nV/deg2, ± SD | 39.6 ± 11 | 41 ± 10.2 | 46.5 ± 10.7 | 0.234 | 0.041 |
| mfERG-A3, nV/deg2, ± SD | 25.5 ± 6.5 | 26.3 ± 7.5 | 26.4 ± 6.9 | 0.293 | 0.955 |
| mfERG-A4, nV/deg2, ± SD | 17.7 ± 4.1 | 17.5 ± 4.2 | 18.2 ± 4.3 | 0.529 | 0.691 |
| mfERG-A5, nV/deg2, ± SD | 13.7 ± 3.3 | 13.2 ± 3.2 | 14.1 ± 3.6 | 0.262 | 0.330 |
| mfERG-IT1, ms, ± SD | 41.8 ± 3.9 | 42.9 ± 4.2 | 42.1 ± 2.7 | 0.205 | 0.379 |
| mfERG-IT2, ms, ± SD | 38.7 ± 1.1 | 39.2 ± 1.7 | 39.2 ± 1.1 | 0.270 | 0.915 |
| mfERG-IT3, ms, ± SD | 37.7 ± 1.1 | 37.7 ± 0.9 | 37.1 ± 1 | 0.572 | 0.016 |
| mfERG-IT4, ms, ± SD | 37.3 ± 1 | 37.3 ± 0.8 | 37 ± 1.1 | 1.000 | 0.439 |
| mfERG-IT5, ms, ± SD | 37.6 ± 1 | 37.8 ± 0.8 | 37.4 ± 1.3 | 0.236 | 0.140 |
| Baseline | 3 Months (SML) | 6 Months | p1 | p2 | |
|---|---|---|---|---|---|
| MV, mm3, ± SD | 2.87 ± 0.74 | 2.58 ± 0.32 | 2.68 ± 0.35 | 0.410 | 0.348 |
| CRT, μm, ± SD | 436 ± 142 | 382 ± 99 | 378 ± 98 | 0.224 | 0.940 |
| BCVA, logmar, ± SD | 0.2 ± 0.16 | 0.19 ± 0.15 | 0.17 ± 0.17 | 1.000 | 0.397 |
| CS-1.5, logmar, ± SD | 1.42 ± 0.11 | 1.47 ± 0.13 | 1.55 ± 0.17 | 0.071 | 0.165 |
| CS-3.0, logmar, ± SD | 1.47 ± 0.21 | 1.54 ± 0.2 | 1.58 ± 0.22 | 0.020 | 0.550 |
| CS-6.0, logmar, ± SD | 1.09 ± 0.57 | 1.36 ± 0.41 | 1.21 ± 0.74 | 0.044 | 0.726 |
| CS-12.0, logmar, ± SD | 0.43 ± 0.58 | 0.48 ± 0.57 | 0.83 ± 0.6 | 0.855 | 0.015 |
| CS-18.0, logmar, ± SD | 0.15 ± 0.31 | 0.27 ± 0.42 | 0.45 ± 0.44 | 0.343 | 0.201 |
| CS-A, logmar, ± SD | 0.91 ± 0.28 | 1.02 ± 0.27 | 1.12 ± 0.39 | 0.056 | 0.148 |
| MP-C, db, ± SD | 9.3 ± 4.8 | 10 ± 4.1 | 11.2 ± 5.4 | 0.289 | 0.489 |
| MP-P, db, ± SD | 13.2 ± 4.3 | 13.7 ± 4.1 | 14.4 ± 3.3 | 0.230 | 0.826 |
| MP-A, db, ± SD | 12 ± 4.3 | 12.6 ± 4 | 13.5 ± 3.8 | 0.255 | 0.733 |
| mfERG-A1, nV/deg2, ± SD | 57.2 ± 25 | 66.7 ± 23.1 | 66.8 ± 25.4 | 0.045 | 0.980 |
| mfERG-A2, nV/deg2, ± SD | 35.9 ± 9.8 | 40.1 ± 9 | 40.5 ± 8.4 | 0.031 | 0.782 |
| mfERG-A3, nV/deg2, ± SD | 24.7 ± 4.3 | 25.5 ± 5.2 | 25 ± 3.2 | 0.610 | 0.782 |
| mfERG-A4, nV/deg2, ± SD | 16.3 ± 2.7 | 16.4 ± 3.2 | 17 ± 2.9 | 0.784 | 0.205 |
| mfERG-A5, nV/deg2, ± SD | 12.4 ± 2.1 | 12.3 ± 2.3 | 12.7 ± 2.5 | 0.814 | 0.642 |
| mfERG-IT1, ms, ± SD | 41.5 ± 5.1 | 42.3 ± 5.2 | 44.4 ± 2.9 | 0.284 | 0.208 |
| mfERG-IT2, ms, ± SD | 39.7 ± 2 | 38.2 ± 1.7 | 39.4 ± 1.5 | 0.017 | 0.081 |
| mfERG-IT3, ms, ± SD | 37.4 ± 1 | 37.1 ± 1.1 | 37.2 ± 1.1 | 0.071 | 0.592 |
| mfERG-IT4, ms, ± SD | 37 ± 1.1 | 36.8 ± 1.2 | 37 ± 1.1 | 0.098 | 0.342 |
| mfERG-IT5, ms, ± SD | 37.6 ± 1.1 | 37.5 ± 1.2 | 37.7 ± 1.4 | 0.595 | 0.125 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kiraly, P.; Habjan, M.Š.; Smrekar, J.; Mekjavić, P.J. Functional Outcomes and Safety Profile of Trans-Foveal Subthreshold Micropulse Laser in Persistent Central Serous Chorioretinopathy. Life 2023, 13, 1194. https://doi.org/10.3390/life13051194
Kiraly P, Habjan MŠ, Smrekar J, Mekjavić PJ. Functional Outcomes and Safety Profile of Trans-Foveal Subthreshold Micropulse Laser in Persistent Central Serous Chorioretinopathy. Life. 2023; 13(5):1194. https://doi.org/10.3390/life13051194
Chicago/Turabian StyleKiraly, Peter, Maja Šuštar Habjan, Jaka Smrekar, and Polona Jaki Mekjavić. 2023. "Functional Outcomes and Safety Profile of Trans-Foveal Subthreshold Micropulse Laser in Persistent Central Serous Chorioretinopathy" Life 13, no. 5: 1194. https://doi.org/10.3390/life13051194
APA StyleKiraly, P., Habjan, M. Š., Smrekar, J., & Mekjavić, P. J. (2023). Functional Outcomes and Safety Profile of Trans-Foveal Subthreshold Micropulse Laser in Persistent Central Serous Chorioretinopathy. Life, 13(5), 1194. https://doi.org/10.3390/life13051194






