Possible Emergence of Sequence Specific RNA Aminoacylation via Peptide Intermediary to Initiate Darwinian Evolution and Code through Origin of Life
Abstract
:1. Introduction
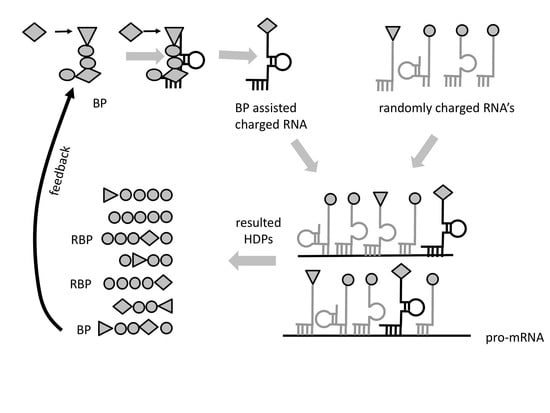
2. Steps of the Proposed RNA-Peptide World Scenario
Steps towards Translation
2.1. Assumptions for Primordial Earth Conditions
2.2. Step 1. Stability Selection of RNA Complexes
2.3. Step 2. Peptide Formation Due to Hybridization of Aminoacylated RNA Oligos
2.4. Step 3. the Amino Acid Content of HDPs
2.5. Step 4. Peptide-Dependent Stability
2.6. Step 5. “Proto-Translation”
2.7. Step 6. Replication
2.8. Step 7. the Evolution of Functions
2.9. Step 8. the Emergence of Standard Genetic Code (SGC)
3. Discussion
3.1. “RNA World” or “RNA-Peptide” World?
3.2. Formation of Standard Genetic Code
Author Contributions
Funding
Conflicts of Interest
References
- Nutman, A.P.; Bennett, V.C.; Friend, C.R.; Van Kranendonk, M.J.; Chivas, A.R. Rapid emergence of life shown by discovery of 3,700-million-year-old microbial structures. Nature 2016, 537, 535–538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tashiro, T.; Ishida, A.; Hori, M.; Igisu, M.; Koike, M.; Mejean, P.; Takahata, N.; Sano, Y.; Komiya, T. Early trace of life from 3.95 Ga sedimentary rocks in Labrador, Canada. Nature 2017, 549, 516–518. [Google Scholar] [CrossRef] [PubMed]
- Schopf, J.W.; Kitajima, K.; Spicuzza, M.J.; Kudryavtsev, A.B.; Valley, J.W. SIMS analyses of the oldest known assemblage of microfossils document their taxon-correlated carbon isotope compositions. Proc. Natl. Acad. Sci. USA 2018, 115, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Pearce, B.K.D.; Tupper, A.S.; Pudritz, R.E.; Higgs, P.G. Constraining the Time Interval for the Origin of Life on Earth. Astrobiology 2018, 18, 343–364. [Google Scholar] [CrossRef] [PubMed]
- Lanier, K.A.; Petrov, A.S.; Williams, L.D. The Central Symbiosis of Molecular Biology: Molecules in Mutualism. J. Mol. Evol. 2017, 85, 8–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gilbert, W. Origin of life: The RNA world. Nature 1986, 319, 618. [Google Scholar] [CrossRef]
- Poole, A.M.; Jeffares, D.C.; Penny, D. The path from the RNA world. J. Mol. Evol. 1998, 46, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Orgel, L.E. Prebiotic chemistry and the origin of the RNA world. Crit. Rev. Biochem. Mol. Biol. 2004, 39, 99–123. [Google Scholar] [CrossRef] [PubMed]
- Wolf, Y.I.; Koonin, E.V. On the origin of the translation system and the genetic code in the RNA world by means of natural selection, exaptation, and subfunctionalization. Biol. Direct. 2007, 2, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bernhardt, H.S.; Tate, W.P. Primordial soup or vinaigrette: Did the RNA world evolve at acidic pH? Biol. Direct. 2012, 7, 4. [Google Scholar] [CrossRef] [PubMed]
- Higgs, P.G.; Lehman, N. The RNA World: molecular cooperation at the origins of life. Nat. Rev. Genet. 2015, 16, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Altman, S.; Kirsebom, L.; Talbot, S. Recent studies of ribonuclease P. FASEB J. 1993, 7, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Cech, T.R. Ribozymes, the first 20 years. Biochem. Soc. Trans. 2002, 30, 1162–1166. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P.; Szostak, J.W. Isolation of new ribozymes from a large pool of random sequences [see comment]. Science 1993, 261, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Robertson, M.P.; Ellington, A.D. In vitro selection of an allosteric ribozyme that transduces analytes to amplicons. Nat. Biotechnol. 1999, 17, 62–66. [Google Scholar] [CrossRef] [PubMed]
- Jaeger, L.; Wright, M.C.; Joyce, G.F. A complex ligase ribozyme evolved in vitro from a group I ribozyme domain. Proc. Natl. Acad. Sci. USA 1999, 96, 14712–14717. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rogers, J.; Joyce, G.F. The effect of cytidine on the structure and function of an RNA ligase ribozyme. RNA 2001, 7, 395–404. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paul, N.; Joyce, G.F. A self-replicating ligase ribozyme. Proc. Natl. Acad. Sci. USA 2002, 99, 12733–12740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, D.E.; Joyce, G.F. Cross-catalytic replication of an RNA ligase ribozyme. Chem. Biol. 2004, 11, 1505–1512. [Google Scholar] [CrossRef] [PubMed]
- Horning, D.P.; Joyce, G.F. Amplification of RNA by an RNA polymerase ribozyme. Proc. Natl. Acad. Sci. USA 2016, 113, 9786–9791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeffares, D.C.; Poole, A.M.; Penny, D. Relics from the RNA world. J. Mol. Evol. 1998, 46, 18–36. [Google Scholar] [CrossRef] [PubMed]
- Khaitovich, P.; Mankin, A.S.; Green, R.; Lancaster, L.; Noller, H.F. Characterization of functionally active subribosomal particles from Thermus aquaticus. Proc. Natl. Acad. Sci. USA 1999, 96, 85–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ban, N.; Nissen, P.; Hansen, J.; Moore, P.B.; Steitz, T.A. The complete atomic structure of the large ribosomal subunit at 2.4 A resolution. Science 2000, 289, 905–920. [Google Scholar] [CrossRef] [PubMed]
- Noller, H.F. Evolution of protein synthesis from an RNA world. Cold Spring Harb. Perspect. Biol. 2012, 4, a003681. [Google Scholar] [CrossRef] [PubMed]
- Sauerwald, A.; Zhu, W.; Major, T.A.; Roy, H.; Palioura, S.; Jahn, D.; Whitman, W.B.; Yates, J.R., 3rd; Ibba, M.; Soll, D. RNA-dependent cysteine biosynthesis in archaea. Science 2005, 307, 1969–1972. [Google Scholar] [CrossRef] [PubMed]
- Mukai, T.; Crnkovic, A.; Umehara, T.; Ivanova, N.N.; Kyrpides, N.C.; Soll, D. RNA-Dependent Cysteine Biosynthesis in Bacteria and Archaea. MBio 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Hohn, M.J.; Park, H.S.; O’Donoghue, P.; Schnitzbauer, M.; Soll, D. Emergence of the universal genetic code imprinted in an RNA record. Proc. Natl. Acad. Sci. USA 2006, 103, 18095–18100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bowman, J.C.; Hud, N.V.; Williams, L.D. The ribosome challenge to the RNA world. J. Mol. Evol. 2015, 80, 143–161. [Google Scholar] [CrossRef] [PubMed]
- Carter, C.W. What RNA World? Why a Peptide/RNA Partnership Merits Renewed Experimental Attention. Life 2015, 5, 294–320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carter C.W., Jr. An Alternative to the RNA World. Nat. Hist. 2016, 125, 28–33. [Google Scholar] [PubMed]
- Shechner, D.M.; Bartel, D.P. The structural basis of RNA-catalyzed RNA polymerization. Nat. Struct. Mol. Biol. 2011, 18, 1036–1042. [Google Scholar] [CrossRef] [PubMed]
- Attwater, J.; Wochner, A.; Pinheiro, V.B.; Coulson, A.; Holliger, P. Ice as a protocellular medium for RNA replication. Nat. Commun. 2010, 1, 76. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, N.; Manapat, M.L.; Chen, I.A.; Xulvi-Brunet, R.; Hayden, E.J.; Lehman, N. Spontaneous network formation among cooperative RNA replicators. Nature 2012, 491, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Attwater, J.; Wochner, A.; Holliger, P. In-ice evolution of RNA polymerase ribozyme activity. Nat. Chem. 2013, 5, 1011–1018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sczepanski, J.T.; Joyce, G.F. A cross-chiral RNA polymerase ribozyme. Nature 2014, 515, 440–442. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, L.K.; Unrau, P.J. Closing the circle: Replicating RNA with RNA. Cold Spring Harb. Perspect. Biol. 2010, 2, a002204. [Google Scholar] [CrossRef] [PubMed]
- Taylor, W.R. Transcription and translation in an RNA world. Philos. Trans. R Soc. Lond. B. Biol. Sci. 2006, 361, 1751–1760. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woese, C.R.; Dugre, D.H.; Saxinger, W.C.; Dugre, S.A. The molecular basis for the genetic code. Proc. Natl. Acad. Sci. USA 1966, 55, 966–974. [Google Scholar] [CrossRef] [PubMed]
- Woese, C.R. The fundamental nature of the genetic code: Prebiotic interactions between polynucleotides and polyamino acids or their derivatives. Proc. Natl. Acad. Sci. USA 1968, 59, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Woese, C.R. Codon recognition: The allosteric ribosome hypothesis. J. Theor. Biol. 1970, 26, 83–88. [Google Scholar] [CrossRef]
- Woese, C.R. Evolution of the genetic code. Naturwissenschaften 1973, 60, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Woese, C.R.; Olsen, G.J.; Ibba, M.; Soll, D. Aminoacyl-tRNA synthetases, the genetic code, and the evolutionary process. Microbiol. Mol. Biol. Rev. 2000, 64, 202–236. [Google Scholar] [CrossRef] [PubMed]
- Yarus, M.; Widmann, J.J.; Knight, R. RNA-amino acid binding: A stereochemical era for the genetic code. J. Mol. Evol. 2009, 69, 406–429. [Google Scholar] [CrossRef] [PubMed]
- Ma, W. The scenario on the origin of translation in the RNA world: In principle of replication parsimony. Biol. Direct. 2010, 5, 65. [Google Scholar] [CrossRef] [PubMed]
- Koonin, E.V.; Novozhilov, A.S. Origin and Evolution of the Universal Genetic Code. Annu. Rev. Genet. 2017, 51, 45–62. [Google Scholar] [CrossRef] [PubMed]
- Fox, G.E. Origin and evolution of the ribosome. Cold Spring Harb. Perspect. Biol. 2010, 2, a003483. [Google Scholar] [CrossRef] [PubMed]
- De Farias, S.T.; Rego, T.G.; Jose, M.V. tRNA Core Hypothesis for the Transition from the RNA World to the Ribonucleoprotein World. Life 2016, 6, 15. [Google Scholar] [CrossRef] [PubMed]
- Altstein, A.D. The progene hypothesis: The nucleoprotein world and how life began. Biol. Direct. 2015, 10, 67. [Google Scholar] [CrossRef] [PubMed]
- Caetano-Anolles, D.; Caetano-Anolles, G. Piecemeal Buildup of the Genetic Code, Ribosomes, and Genomes from Primordial tRNA Building Blocks. Life 2016, 6, 43. [Google Scholar] [CrossRef] [PubMed]
- Wills, P.R.; Carter, C.W., Jr. Insuperable problems of the genetic code initially emerging in an RNA world. Biosystems 2018, 164, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Mirkin, B.G.; Fenner, T.I.; Galperin, M.Y.; Koonin, E.V. Algorithms for computing parsimonious evolutionary scenarios for genome evolution, the last universal common ancestor and dominance of horizontal gene transfer in the evolution of prokaryotes. BMC Evol. Biol. 2003, 3, 2. [Google Scholar] [CrossRef]
- Lanier, K.A.; Williams, L.D. The Origin of Life: Models and Data. J. Mol. Evol. 2017, 84, 85–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poole, A.M.; Jeffares, D.C.; Hoeppner, M.P.; Penny, D. Does the Ribosome Challenge our Understanding of the RNA World? J. Mol. Evol. 2016, 82, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Aravind, L.; Anantharaman, V.; Koonin, E.V. Monophyly of class I aminoacyl tRNA synthetase, USPA, ETFP, photolyase, and PP-ATPase nucleotide-binding domains: implications for protein evolution in the RNA. Proteins 2002, 48, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Theobald, D.L.; Wuttke, D.S. Divergent evolution within protein superfolds inferred from profile-based phylogenetics. J. Mol. Biol. 2005, 354, 722–737. [Google Scholar] [CrossRef] [PubMed]
- Schimmel, P. Classes of aminoacyl-tRNA synthetases and the establishment of the genetic code. Trends Biochem. Sci. 1991, 16, 1–3. [Google Scholar] [CrossRef]
- Burbaum, J.J.; Schimmel, P. Structural relationships and the classification of aminoacyl-tRNA synthetases. J. Biol. Chem. 1991, 266, 16965–16968. [Google Scholar] [PubMed]
- Carter, C.W., Jr.; Wills, P.R. Interdependence, Reflexivity, Fidelity, Impedance Matching, and the Evolution of Genetic Coding. Mol. Biol. Evol. 2018, 35, 269–286. [Google Scholar] [CrossRef] [PubMed]
- Carter C.W., Jr. Coding of Class I and II Aminoacyl-tRNA Synthetases. Adv. Exp. Med. Biol. 2017, 966, 103–148. [Google Scholar] [CrossRef] [PubMed]
- Powner, M.W.; Gerland, B.; Sutherland, J.D. Synthesis of activated pyrimidine ribonucleotides in prebiotically plausible conditions. Nature 2009, 459, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Benner, S.A. Prebiotic stereoselective synthesis of purine and noncanonical pyrimidine nucleotide from nucleobases and phosphorylated carbohydrates. Proc. Natl. Acad. Sci. USA 2017, 114, 11315–11320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sutherland, J.D. The Origin of Life—Out of the Blue. Angew. Chem. Int. Ed. Engl. 2016, 55, 104–121. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, K.; Maurel, M.C. Walking over 4 Gya: Chemical Evolution from Photochemistry to Mineral and Organic Chemistries Leading to an RNA World. Orig. Life Evol. Biosph. 2017, 47, 281–296. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, M.; Yanagawa, H. Prebiotic synthesis of amino acids from formaldehyde and hydroxylamine in a modified sea medium. Orig. Life 1984, 14, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Zaia, D.A.; Zaia, C.T.; De Santana, H. Which amino acids should be used in prebiotic chemistry studies? Orig. Life Evol. Biosph. 2008, 38, 469–488. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Dziedzic, P.; Spacil, Z.; Zhao, G.L.; Nilsson, L.; Ilag, L.L.; Cordova, A. Abiotic synthesis of amino acids and self-crystallization under prebiotic conditions. Sci. Rep. 2014, 4, 6769. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonfio, C.; Valer, L.; Scintilla, S.; Shah, S.; Evans, D.J.; Jin, L.; Szostak, J.W.; Sasselov, D.D.; Sutherland, J.D.; Mansy, S.S. UV-light-driven prebiotic synthesis of iron-sulfur clusters. Nat. Chem. 2017, 9, 1229–1234. [Google Scholar] [CrossRef] [PubMed]
- Monnard, P.A. Taming Prebiotic Chemistry: The Role of Heterogeneous and Interfacial Catalysis in the Emergence of a Prebiotic Catalytic/Information Polymer System. Life 2016, 6, 40. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, S.; Sasselov, D.D. Constraints on the Early Terrestrial Surface UV Environment Relevant to Prebiotic Chemistry. Astrobiology 2017, 17, 169–204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaddour, H.; Sahai, N. Synergism and mutualism in non-enzymatic RNA polymerization. Life 2014, 4, 598–620. [Google Scholar] [CrossRef] [PubMed]
- Mungi, C.V.; Rajamani, S. Characterization of RNA-Like Oligomers from Lipid-Assisted Nonenzymatic Synthesis: Implications for Origin of Informational Molecules on Early Earth. Life 2015, 5, 65–84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pino, S.; Sponer, J.E.; Costanzo, G.; Saladino, R.; Mauro, E.D. From formamide to RNA, the path is tenuous but continuous. Life 2015, 5, 372–384. [Google Scholar] [CrossRef] [PubMed]
- Jackson, J.B. Natural pH Gradients in Hydrothermal Alkali Vents Were Unlikely to Have Played a Role in the Origin of Life. J. Mol. Evol. 2016, 83, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engelhart, A.E.; Powner, M.W.; Szostak, J.W. Functional RNAs exhibit tolerance for non-heriTable 2′-5′ versus 3′-5′ backbone heterogeneity. Nat. Chem. 2013, 5, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Prywes, N.; Blain, J.C.; Del Frate, F.; Szostak, J.W. Nonenzymatic copying of RNA templates containing all four letters is catalyzed by activated oligonucleotides. Elife 2016, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spitzer, J. Emergence of Life on Earth: A Physicochemical Jigsaw Puzzle. J. Mol. Evol. 2017, 84, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Higgs, P.G. The Effect of Limited Diffusion and Wet-Dry Cycling on Reversible Polymerization Reactions: Implications for Prebiotic Synthesis of Nucleic Acids. Life 2016, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Damer, B.; Deamer, D. Coupled phases and combinatorial selection in fluctuating hydrothermal pools: A scenario to guide experimental approaches to the origin of cellular life. Life 2015, 5, 872–887. [Google Scholar] [CrossRef] [PubMed]
- Kee, T.P.; Pierre-Alain, M. On the emergence of a proto-metabolism and the assembly of early protocells. Elements 2016, 12, 5. [Google Scholar] [CrossRef]
- Hanczyc, M.M.; Fujikawa, S.M.; Szostak, J.W. Experimental models of primitive cellular compartments: Encapsulation, growth, and division. Science 2003, 302, 618–622. [Google Scholar] [CrossRef] [PubMed]
- Illangasekare, M.; Sanchez, G.; Nickles, T.; Yarus, M. Aminoacyl-RNA synthesis catalyzed by an RNA. Science 1995, 267, 643–647. [Google Scholar] [CrossRef] [PubMed]
- Illangasekare, M.; Yarus, M. A tiny RNA that catalyzes both aminoacyl-RNA and peptidyl-RNA synthesis. RNA 1999, 5, 1482–1489. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bowler, F.R.; Chan, C.K.; Duffy, C.D.; Gerland, B.; Islam, S.; Powner, M.W.; Sutherland, J.D.; Xu, J. Prebiotically plausible oligoribonucleotide ligation facilitated by chemoselective acetylation. Nat. Chem. 2013, 5, 383–389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suga, H.; Hayashi, G.; Terasaka, N. The RNA origin of transfer RNA aminoacylation and beyond. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2011, 366, 2959–2964. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muller, S. Engineering of ribozymes with useful activities in the ancient RNA world. Ann. N. Y. Acad. Sci 2015, 1341, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.; Bessho, Y.; Wei, K.; Szostak, J.W.; Suga, H. Ribozyme-catalyzed tRNA aminoacylation. Nat. Struct. Biol. 2000, 7, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Higgs, P.G. A four-column theory for the origin of the genetic code: Tracing the evolutionary pathways that gave rise to an optimized code. Biol. Direct. 2009, 4, 16. [Google Scholar] [CrossRef] [PubMed]
- Higgs, P.G.; Pudritz, R.E. A thermodynamic basis for prebiotic amino acid synthesis and the nature of the first genetic code. Astrobiology 2009, 9, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Higgs, P.G. Chemical Evolution and the Evolutionary Definition of Life. J. Mol. Evol. 2017, 84, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Robertson, M.P.; Knudsen, S.M.; Ellington, A.D. In vitro selection of ribozymes dependent on peptides for activity. RNA 2004, 10, 114–127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blanco, C.; Bayas, M.; Yan, F.; Chen, I.A. Analysis of Evolutionarily Independent Protein-RNA Complexes Yields a Criterion to Evaluate the Relevance of Prebiotic Scenarios. Curr. Biol. 2018, 28, 526–537 e525. [Google Scholar] [CrossRef] [PubMed]
- Bayer, T.S.; Booth, L.N.; Knudsen, S.M.; Ellington, A.D. Arginine-rich motifs present multiple interfaces for specific binding by RNA. RNA 2005, 11, 1848–1857. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steitz, T.A. A mechanism for all polymerases. Nature 1998, 391, 231–232. [Google Scholar] [CrossRef] [PubMed]
- Betat, H.; Rammelt, C.; Morl, M. tRNA nucleotidyltransferases: Ancient catalysts with an unusual mechanism of polymerization. Cell Mol. Life Sci. 2010, 67, 1447–1463. [Google Scholar] [CrossRef] [PubMed]
- Timinskas, K.; Balvociute, M.; Timinskas, A.; Venclovas, C. Comprehensive analysis of DNA polymerase III alpha subunits and their homologs in bacterial genomes. Nucleic Acids Res. 2014, 42, 1393–1413. [Google Scholar] [CrossRef] [PubMed]
- Lenstra, R. Evolution of the genetic code through progressive symmetry breaking. J. Theor. Biol. 2014, 347, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Yarus, M. Getting past the RNA world: The initial Darwinian ancestor. Cold Spring Harb. Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Orgel, L.E. Nonenzymatic template-directed synthesis on hairpin oligonucleotides. 3. Incorporation of adenosine and uridine residues. J. Am. Chem. Soc. 1992, 114, 7963–7969. [Google Scholar] [CrossRef] [PubMed]
- Machinek, R.R.; Ouldridge, T.E.; Haley, N.E.; Bath, J.; Turberfield, A.J. Programmable energy landscapes for kinetic control of DNA strand displacement. Nat. Commun. 2014, 5, 5324. [Google Scholar] [CrossRef] [PubMed]
- Frank, D.N.; Pace, N.R. Ribonuclease P: Unity and diversity in a tRNA processing ribozyme. Annu. Rev. Biochem. 1998, 67, 153–180. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.C.; Engelke, D.R. Ribonuclease P: The evolution of an ancient RNA enzyme. Crit. Rev. Biochem. Mol. Biol. 2006, 41, 77–102. [Google Scholar] [CrossRef] [PubMed]
- Petrov, A.S.; Gulen, B.; Norris, A.M.; Kovacs, N.A.; Bernier, C.R.; Lanier, K.A.; Fox, G.E.; Harvey, S.C.; Wartell, R.M.; Hud, N.V.; et al. History of the ribosome and the origin of translation. Proc. Natl. Acad. Sci. USA 2015, 112, 15396–15401. [Google Scholar] [CrossRef] [PubMed]
- Harish, A.; Caetano-Anolles, G. Ribosomal history reveals origins of modern protein synthesis. PLoS ONE 2012, 7, e32776. [Google Scholar] [CrossRef] [PubMed]
- Caetano-Anolles, G. Ancestral Insertions and Expansions of rRNA do not Support an Origin of the Ribosome in Its Peptidyl Transferase Center. J. Mol. Evol. 2015, 80, 162–165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laursen, B.S.; Sorensen, H.P.; Mortensen, K.K.; Sperling-Petersen, H.U. Initiation of protein synthesis in bacteria. Microbiol. Mol. Biol. Rev. 2005, 69, 101–123. [Google Scholar] [CrossRef] [PubMed]
- Root-Bernstein, M.; Root-Bernstein, R. The ribosome as a missing link in the evolution of life. J. Theor. Biol. 2015, 367, 130–158. [Google Scholar] [CrossRef] [PubMed]
- Szathmary, E. Coding coenzyme handles: A hypothesis for the origin of the genetic code. Proc. Natl. Acad. Sci. USA 1993, 90, 9916–9920. [Google Scholar] [CrossRef] [PubMed]
- Rodin, A.S.; Szathmary, E.; Rodin, S.N. On origin of genetic code and tRNA before translation. Biol. Direct 2011, 6, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]




© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kunnev, D.; Gospodinov, A. Possible Emergence of Sequence Specific RNA Aminoacylation via Peptide Intermediary to Initiate Darwinian Evolution and Code through Origin of Life. Life 2018, 8, 44. https://doi.org/10.3390/life8040044
Kunnev D, Gospodinov A. Possible Emergence of Sequence Specific RNA Aminoacylation via Peptide Intermediary to Initiate Darwinian Evolution and Code through Origin of Life. Life. 2018; 8(4):44. https://doi.org/10.3390/life8040044
Chicago/Turabian StyleKunnev, Dimiter, and Anastas Gospodinov. 2018. "Possible Emergence of Sequence Specific RNA Aminoacylation via Peptide Intermediary to Initiate Darwinian Evolution and Code through Origin of Life" Life 8, no. 4: 44. https://doi.org/10.3390/life8040044