LINC00084/miR-204/ZEB1 Axis Mediates Myofibroblastic Differentiation Activity in Fibrotic Buccal Mucosa Fibroblasts: Therapeutic Target for Oral Submucous Fibrosis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Reagents
2.3. Tissue Acquisition
2.4. Quantitative Real-Time PCR and Western Blot Analysis
2.5. Lentiviral-Mediated Knockdown or Overexpression of LINC00084
2.6. Collagen Gel Contraction
2.7. Cell Migration and Invasion Assays
2.8. Dual-Luciferase Reporter Gene Assay
2.9. RIP (RNA Immunoprecipitation) Assay
2.10. Statistical Analysis
3. Results
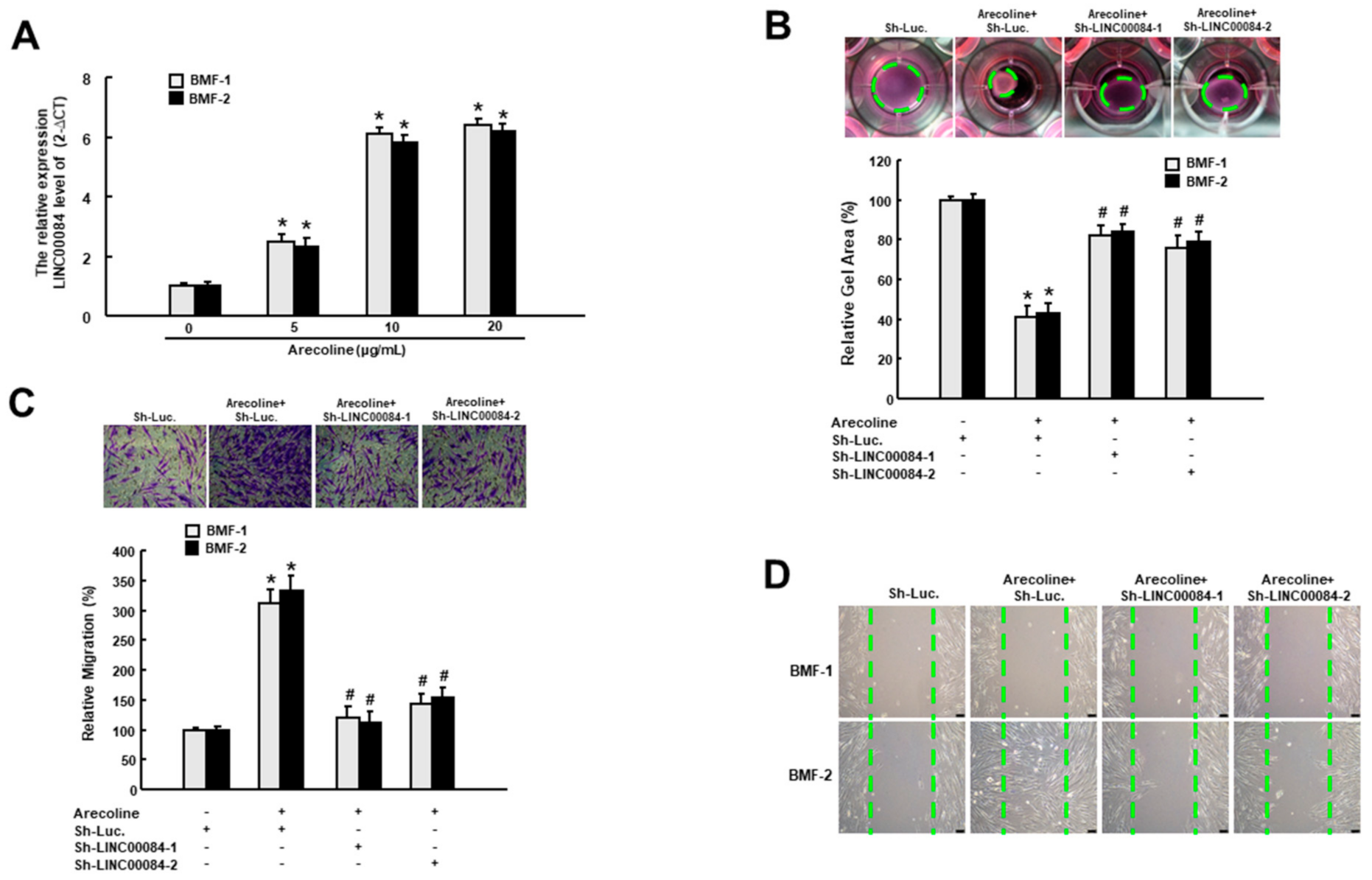
3.1. Arecoline-Induced LINC00084 Promotes Myofibroblast Activation
3.2. The Upregulated LINC00084 in OSF Tissues Is Positively Associated with Several Fibrosis Markers
3.3. Silencing LINC00084 in fBMFs Mitigates the Myofibroblasts Features
3.4. LINC00084 Acts as a Molecular Sponge of miR-204
3.5. MiR-204 Regulates the Myofibroblast Activities by Directly Targeting ZEB1
3.6. LINC00084 Mediates the Myofibroblast Activation via Sponge Regulation of miR-204-ZEB1 Interaction
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Lee, C.H.; Ko, Y.C.; Huang, H.L.; Chao, Y.Y.; Tsai, C.C.; Shieh, T.Y.; Lin, L.M. The precancer risk of betel quid chewing, tobacco use and alcohol consumption in oral leukoplakia and oral submucous fibrosis in southern Taiwan. Br. J. Cancer 2003, 88, 366–372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiang, W.F.; Liu, S.Y.; Lin, J.F.; Chiu, S.F.; Gou, S.B.; Chiou, C.T.; Chang, C.H. Malignant development in patients with oral potentially malignant disorders detected through nationwide screening: Outcomes of 5-year follow-up at a single hospital. Head Neck 2020, 42, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Chuang, S.L.; Wang, C.P.; Chen, M.K.; Su, W.W.Y.; Su, C.W.; Chen, S.L.S.; Chiu, S.Y.H.; Fann, J.C.Y.; Yen, A.M.F. Malignant transformation to oral cancer by subtype of oral potentially malignant disorder: A prospective cohort study of Taiwanese nationwide oral cancer screening program. Oral Oncol. 2018, 87, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Iocca, O.; Sollecito, T.P.; Alawi, F.; Weinstein, G.S.; Newman, J.G.; De Virgilio, A.; Di Maio, P.; Spriano, G.; López, S.P.; Shanti, R.M. Potentially malignant disorders of the oral cavity and oral dysplasia: A systematic review and meta-analysis of malignant transformation rate by subtype. Head Neck 2020, 42, 539–555. [Google Scholar] [CrossRef]
- Angadi, P.V.; Kale, A.D.; Hallikerimath, S. Evaluation of myofibroblasts in oral submucous fibrosis: Correlation with disease severity. J. Oral Pathol. Med. 2010, 40, 208–213. [Google Scholar] [CrossRef]
- Sobral, L.M.; Bufalino, A.; Lopes, M.A.; Graner, E.; Salo, T.; Coletta, R.D. Myofibroblasts in the stroma of oral cancer promote tumorigenesis via secretion of activin A. Oral Oncol. 2011, 47, 840–846. [Google Scholar] [CrossRef] [Green Version]
- Cheng, R.H.; Wang, Y.P.; Chang, J.Y.; Pan, Y.H.; Chang, M.C.; Jeng, J.H. Genetic Susceptibility and Protein Expression of Extracellular Matrix Turnover-Related Genes in Oral Submucous Fibrosis. Int. J. Mol. Sci. 2020, 21, 8104. [Google Scholar] [CrossRef]
- Peng, C.Y.; Liao, Y.W.; Lu, M.Y.; Yang, C.M.; Hsieh, P.L.; Yu, C.C. Positive Feedback Loop of SNAIL-IL-6 Mediates Myofibroblastic Differentiation Activity in Precancerous Oral Submucous Fibrosis. Cancers 2020, 12, 1611. [Google Scholar] [CrossRef] [PubMed]
- Pant, I.; Kumar, N.; Khan, I.; Rao, S.G.; Kondaiah, P. Role of Areca Nut Induced TGF-β and Epithelial-Mesenchymal Interaction in the Pathogenesis of Oral Submucous Fibrosis. PLoS ONE 2015, 10, e0129252. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.C.; Tsai, C.H.; Lai, Y.L.; Yu, C.C.; Chi, W.Y.; Li, J.J.; Chang, W.W. Arecoline-induced myofibroblast transdifferentiation from human buccal mucosal fibroblasts is mediated by ZEB 1. J. Cell. Mol. Med. 2014, 18, 698–708. [Google Scholar] [CrossRef] [PubMed]
- Szymański, M.; Barciszewska, M.Z.; Zywicki, M.; Barciszewski, J. Noncoding RNA transcripts. J. Appl. Genet. 2003, 44, 1–20. [Google Scholar]
- Marchese, F.P.; Raimondi, I.; Huarte, M. The multidimensional mechanisms of long noncoding RNA function. Genome Biol. 2017, 18, 206. [Google Scholar] [CrossRef] [Green Version]
- Yu, C.C.; Liao, Y.W.; Hsieh, P.L.; Chang, Y.C. Targeting lncRNA H19/miR-29b/COL1A1 Axis Impedes Myofibroblast Activities of Precancerous Oral Submucous Fibrosis. Int. J. Mol. Sci. 2021, 22, 2216. [Google Scholar]
- Gabbiani, G. The myofibroblast in wound healing and fibrocontractive diseases. J. Pathol. 2003, 200, 500–503. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.C.; Lin, L.D.; Wu, H.L.; Ho, Y.S.; Hsien, H.C.; Wang, T.M.; Jeng, P.Y.; Cheng, R.H.; Hahn, L.J.; Jeng, J.H. Areca nut-induced buccal mucosa fibroblast contraction and its signaling: A potential role in oral submucous fibrosis—A precancer condition. Carcinogenesis 2013, 34, 1096–1104. [Google Scholar] [CrossRef] [Green Version]
- Thangjam, G.S.; Agarwal, P.; Khan, I.; Verma, U.P.; Balapure, A.K.; Rao, S.G.; Kondaiah, P. Transglutaminase-2 Regulation by Arecoline in Gingival Fibroblasts. J. Dent. Res. 2009, 88, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Hinz, B.; Celetta, G.; Tomasek, J.J.; Gabbiani, G.; Chaponnier, C. Alpha-Smooth Muscle Actin Expression Upregulates Fibroblast Contractile Activity. Mol. Biol. Cell 2001, 12, 2730–2741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hutchinson, J.N.; Ensminger, A.W.; Clemson, C.M.; Lynch, C.R.; Lawrence, J.B.; Chess, A. A screen for nuclear transcripts identifies two linked noncoding RNAs associated with SC35 splicing domains. BMC Genom. 2007, 8, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Zhang, P.; Cao, L.; Zhou, R.; Yang, X.; Wu, M. The lncRNA Neat1 promotes activation of inflammasomes in macrophages. Nat. Commun. 2019, 10, 1–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- An, H.; Tan, J.T.; Shelkovnikova, T.A. Stress granules regulate stress-induced paraspeckle assembly. J. Cell Biol. 2019, 218, 4127–4140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, F.; Jiang, Z.; Chen, B.; Dong, P.; Zheng, J. NEAT1 accelerates the progression of liver fibrosis via regulation of microRNA-122 and Kruppel-like factor 6. J. Mol. Med. 2017, 95, 1191–1202. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Xu, Y.; Ge, X.; Xu, B.; Peng, W.; Jiang, X.; Shen, L.; Xia, L. Long noncoding RNA NEAT1 accelerates the proliferation and fibrosis in diabetic nephropathy through activating Akt/mTOR signaling pathway. J. Cell. Physiol. 2019, 234, 11200–11207. [Google Scholar] [CrossRef] [PubMed]
- Kenneweg, F.; Bang, C.; Xiao, K.; Boulanger, C.M.; Loyer, X.; Mazlan, S.; Schroen, B.; Hermans-Beijnsberger, S.; Foinquinos, A.; Hirt, M.N.; et al. Long Noncoding RNA-Enriched Vesicles Secreted by Hypoxic Cardiomyocytes Drive Cardiac Fibrosis. Mol. Ther.-Nucleic Acids 2019, 18, 363–374. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jia, H.; Wang, X.; Sun, Z. Exploring the molecular pathogenesis and biomarkers of high risk oral premalignant lesions on the basis of long noncoding RNA expression profiling by serial analysis of gene expression. Eur. J. Cancer Prev. 2018, 27, 370–378. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; He, X.; Wei, X.L. lncRNA NEAT1 promotes cell proliferation and invasion by regulating miR-365/RGS20 in oral squamous cell carcinoma. Oncol. Rep. 2018, 39, 1948–1956. [Google Scholar] [CrossRef]
- Chen, S.J.; Wu, P.; Sun, L.J.; Zhou, B.; Niu, W.; Liu, S.; Lin, F.J.; Jiang, G.R. miR-204 regulates epithelial-mesenchymal transition by targeting SP1 in the tubular epithelial cells after acute kidney injury induced by ischemia-reperfusion. Oncol. Rep. 2016, 37, 1148–1158. [Google Scholar] [CrossRef] [Green Version]
- Sui, H.; Fan, S.; Liu, W.; Li, Y.; Zhang, X.; Du, Y.; Bao, H. LINC00028 regulates the development of TGFβ1-treated human tenon capsule fibroblasts by targeting miR-204-5p. Biochem. Biophys. Res. Commun. 2020, 525, 197–203. [Google Scholar] [CrossRef]
- Lu, Y.; Li, T.; Wei, G.; Liu, L.; Chen, Q.; Xu, L.; Zhang, K.; Zeng, D.; Liao, R. The long non-coding RNA NEAT1 regulates epithelial to mesenchymal transition and radioresistance in through miR-204/ZEB1 axis in nasopharyngeal carcinoma. Tumor Biol. 2016, 37, 11733–11741. [Google Scholar] [CrossRef]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, Y.-H.; Liao, Y.-W.; Lu, M.-Y.; Hsieh, P.-L.; Yu, C.-C. LINC00084/miR-204/ZEB1 Axis Mediates Myofibroblastic Differentiation Activity in Fibrotic Buccal Mucosa Fibroblasts: Therapeutic Target for Oral Submucous Fibrosis. J. Pers. Med. 2021, 11, 707. https://doi.org/10.3390/jpm11080707
Lee Y-H, Liao Y-W, Lu M-Y, Hsieh P-L, Yu C-C. LINC00084/miR-204/ZEB1 Axis Mediates Myofibroblastic Differentiation Activity in Fibrotic Buccal Mucosa Fibroblasts: Therapeutic Target for Oral Submucous Fibrosis. Journal of Personalized Medicine. 2021; 11(8):707. https://doi.org/10.3390/jpm11080707
Chicago/Turabian StyleLee, Yu-Hsien, Yi-Wen Liao, Ming-Yi Lu, Pei-Ling Hsieh, and Cheng-Chia Yu. 2021. "LINC00084/miR-204/ZEB1 Axis Mediates Myofibroblastic Differentiation Activity in Fibrotic Buccal Mucosa Fibroblasts: Therapeutic Target for Oral Submucous Fibrosis" Journal of Personalized Medicine 11, no. 8: 707. https://doi.org/10.3390/jpm11080707





