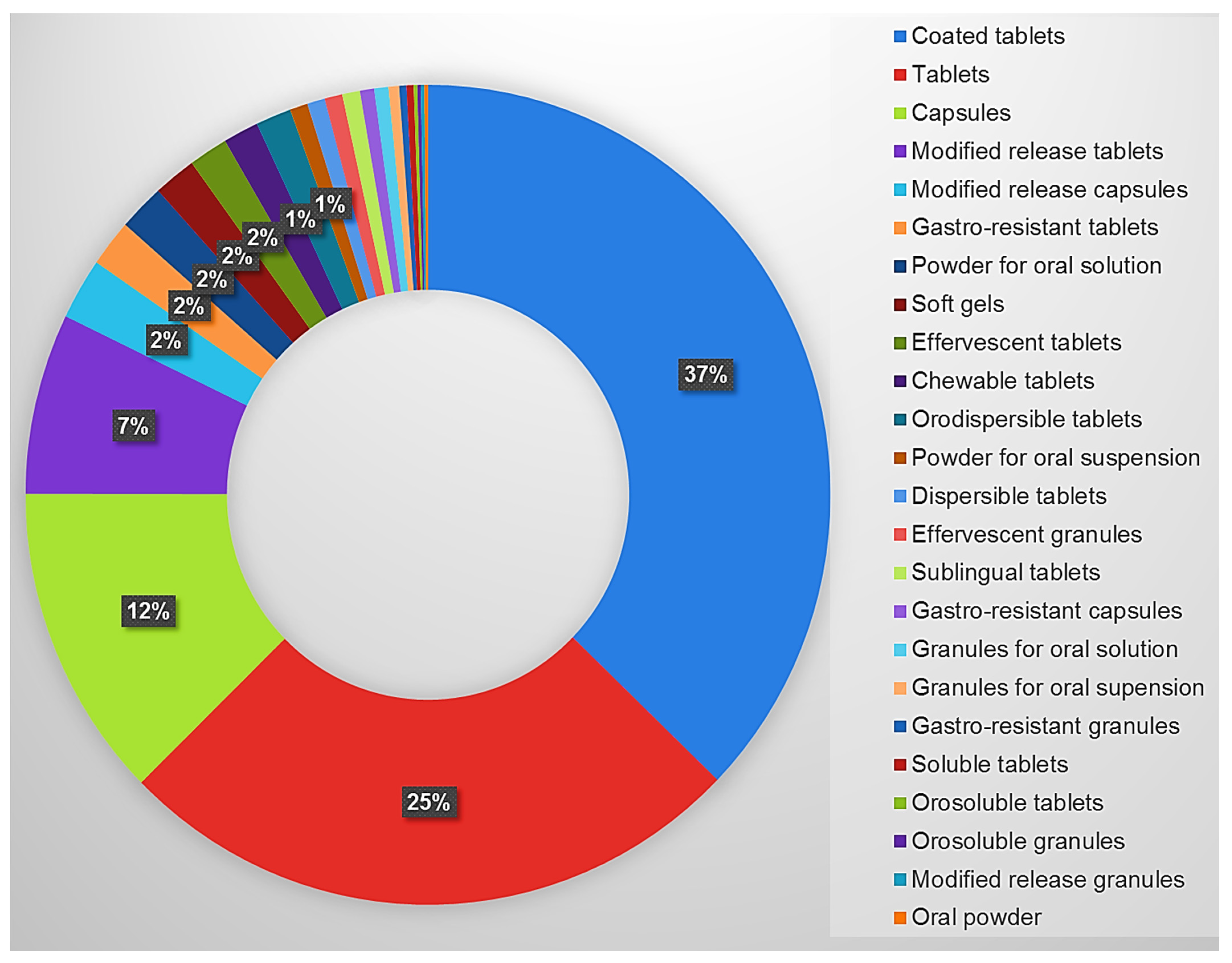
| A02AD01 | Aluminum hydroxide
Magnesium hydroxide
(800 mg) | MAALOX
(SANOFI) | Chewable
tablets | No | Interaction with dietary proteins
Tube obstruction
Use alternative medications | |
| A02AH | Sodium bicarbonate
(500 mg) | SOD BIC NAR
(NOVA ARGENTIA) | Tablets | No | Use other routes of administration | |
| A02BA02 | Ranitidine hydrochloride
(150 mg) | RANIDIL
(A.MENARINI) | Effervescent
tablets | Yes | The tablet should be diluted with at least 75 mL of water (to be avoided in patients on a low sodium diet or with phenylketonuria)
Alternatives: liquid formulation to be diluted with 60 mL of water (contains ethyl alcohol), or sugar-free galenic preparation | |
| A02BA02 | Ranitidine hydrochloride
(150 mg) | ZANTAC
(GLAXOSMITHKLINE) | Coated tablets | No | The effervescent tablets are the first choice (to be avoided in patients on a low sodium diet or with phenylketonuria)
Alternatives: liquid formulation to be diluted with 60 mL of water (contains ethyl alcohol), or Ranitidine 15 mg/mL galenic preparation | [8,9] |
ZANTAC SOLUB
(GLAXOSMITHKLINE) | Effervescent
tablets |
RANITIDINA MYL
(MYLAN) | Coated tablets |
RANITIDINA RATIO
(RATIOPHARM) | Coated tablets |
RANITIDINA TEV
(TEVA) | Coated tablets |
| A02BB01 | Misoprostol
(200 µg) | CYTOTEC 200
(PFIZER) | Tablets | Yes | Crush the tablet in a mortar, add 15 mL water, draw the suspension into the syringe and administer immediately
Rinse the mortar with 15 mL water, draw this water into the syringe and administer
Due to the poor stability of the API better an alternative therapy (Ranitidine or Lansoprazole) | [10] |
| A02BC01 | Omeprazole
(20 mg) | ANTRA
(ASTRAZENECA) | Gastro-resistant capsules | Yes | Open the capsule, suspend the granules contained therein with 20 mL water and then administer after 15–20 min in a tube >8 Fr
The granules must not be crushed
For children the galenic preparation of omeprazole suspension 2 mg/mL is required | [26] |
Omeprazole
(10 mg) | OMEPRAZOLO TEV
(TEVA) |
| A02BC02 | Pantoprazole sodium
sesquihydrate
(20, 40 mg) | PANTORC
(TAKEDA) | Gastro-resistant
tablets | No | Change route of administration or substitute API with Omeprazole or Lansoprazole | [8,9] |
| A02BC03 | Lansoprazole
(15, 30 mg) | LANSOX
(TAKEDA) | Orodispersible
tablets | Yes | Disperse in a small amount of water and administer through a nasogastric tube or oral syringe | SmPC |
| A02BC03 | Lansoprazole
(15, 30 mg) | LANSOX
(TAKEDA) | Capsules | Yes | Open the capsule, suspend the granules with water and administer after 20 min
Prefer orodispersible tablets | [26] |
| A02BC04 | Rabrepazole sodium
(10, 20 mg) | PARIET
(JANSSEN CILAG) | Gastro-resistant
tablets | No | Do not grind gastro-resistant tablets
Alternatives: esomeprazole, lansoprazole or omeprazole | [8,9] |
| A02BC05 | Esomeprazole magnesium
trihydrate
(10 mg) | NEXIUM
(ASTRAZENECA) | Gastro-resistant granules | Yes | To a dose of 10 mg add 15 mL water, mix and leave to thicken for a few minutes
Do not crush the granules, withdraw the suspension with a syringe and inject through the tube of 6 Fr caliber or greater for 30 min
Rinse the tube with 15 mL water | SmPC |
| A02BC05 | Esomeprazole magnesium trihydrate
(10 mg) | LUCEN
(MALESCI) | Gastro-resistant granules | Yes | Mix the granules with 15 mL water, allow to thicken
Pick up the suspension with a syringe and inject it into a tube of 6 Fr caliber or greater | SmPC |
| A02BX02 | Sucralfate
(2 g) | SUCRALFIN
(SANOFI) | Granules for oral suspension | No | Risk of tube occlusion, bezoar formations | [10,21] |
SUCRAMAL
(SCHARPER) |
| A02BX13 | Sodium alginate
Sodium bicarbonate
(250 + 133.5 mg) | GAVISCON
(RECKITT BENCKISER) | Chewable
tablets | No | | [8,9] |
| A03AA05 | Trimebutine maleate
(150 mg) | DEBRIDAT
(SIGMA TAU) | Soft gels | No | Alternatives: solution for injection or suppositories | [8,9] |
| A03AA06 | Rociverin
(10 mg) | RILATEN
(LABORATORI GUIDOTTI) | Coated tablets | Yes | Tablet can be triturated and administered with water | |
| A03AX13 | Simethicone
(40 mg) | MYLICONGAS
(JOHNSON & JOHNSON) | Chewable tablets | No | Alternative: drops to be diluted with 50 mL water and given immediately | [8,9] |
| A03BB01 | Scopolamine methyl bromide
(10 mg) | BUSCOPAN
(BOEHRINGER INGELHEIM) | Coated tablets | Yes | Tablet can be triturated
Alternative: solution for injection | |
| A03FA01 | Metoclopramide hydrochloride monohydrate
(10 mg) | PLASIL
(SANOFI) | Tablets | No | Alternative: syrup which must be diluted and given away from EN | [8,9] |
| A03FA03 | Domperidone maleate
(10 mg) | DOMPERIDONE AGE
(ANGENERICO) | Tablets | No | Alternatives: suppositories or oral suspension (to be diluted with water and administered at least 15 min before the start of EN) | [8,9] |
| A03FA03 | Domperidone
(10 mg) | PERIDON
(ITALCHIMICI) | Coated tablets | No | Alternatives: suppositories or oral suspension (to be diluted with water and administered at least 15 min before the start of EN) | [8,9] |
| A04AA01 | Ondansetron hydrochloride dihydrate
(4 mg) | ZOFRAN
(GLAXOSMITHKLINE) | Coated tablets | No | Prefer orodispersible tablets, syrup or solution for injection | [8,9] |
| A04AA01 | Ondansetron
(8 mg) | ZOFRAN
(GLAXOSMITHKLINE) | Orodispersible
tablets | Yes | | |
| A04AA01 | Ondansetron hydrochloride
(4, 8 mg) | ONDANSETRONE TEV (TEVA) | Coated tablets | No | Alternative: Zofran syrup | [8,9] |
| A04AA02 | Granisetron hydrochloride
(2 mg) | KYTRIL
(ROCHE) | Coated tablets | No | Alternative: solution for injection | [8,9] |
| A04AD12 | Aprepitant
(125, 80 mg) | EMEND
(MSD) | Capsules | No | Open the capsule and administer the granules with water without crushing
Preferred alternative: Ivemend solution for injection | |
| A05AA02 | Ursodeoxycholic acid
(450, 150 mg) | DEURSIL
(CHEPLAPHARM ARZNEIMITTEL) | Modified
release capsules | No | Alternatives: granules for oral suspension, capsules | [8,9] |
| A05AA02 | Ursodeoxycholic acid
(450, 150 mg) | URSILON
(I.B.I.GIOVANNI LORENZINI) | Capsules | Yes | Open the capsule and suspend the granules in 20 mL water then administer immediately | [10] |
| A05BA03 | Sylimarin
(70 mg) | LEGALON
(MEDA PHARMA) | Coated tablets | Yes | | |
| A06AB06 | Senna leaves
(12 mg) | PURSENNID (GLAXOSMITHKLINE) | Coated tablets | No | Prefer other laxatives in syrups | [10] |
| A06AB58 | Sodium picosulfate
Magnesium oxide
Anhydrous citric acid | PICOPREP
(FERRING) | Powder for oral
solution | Yes | Dissolve the powder in 150 mL of water | |
| A06AD11 | Lactulose
(10 g) | LAEVOLAC EPS
(ROCHE) | Granules for oral
solution | Yes | Dilute with at least 60 mL water
Avoid high doses, risk of diarrhea and malabsorption of dietary nutrients | |
| A06AD15 | Macrogol 4000
(4, 9.7, 10 g) | PAXABEL (IPSEN) | Powder for oral
solution | Yes | Dissolve the powder in 125 mL water and administer immediately
Seek dietary advice in case of chronic constipation | |
REGOLINT
(LABORATORI BALDACCI) | |
| LAXIPEG (ZAMBON) | |
| A06AD65 | Macrogol 4000
Sodium sulfate
Sodium bicarbonate
Sodium chloride
Potassium chloride
(17.4, 34.8 g) | ISOCOLAN
(GIULIANI) | Powder for oral
solution | Yes | Dissolve the powder in 250 mL water then
shake and administer immediately | SmPC |
| A06AD65 | Macrogol 3350 Sodium bicarbonate Sodium chloride Potassium chloride
(13.8 g) | MOVICOL
(NORGINE) | Powder for oral
solution | Yes | Dissolve the powder in 125 mL water, shake and administer immediately
Seek dietary advice in case of chronic constipation | |
| A06AD65 | Macrogol 3350
Sodium sulfate
Sodium bicarbonate
Sodium chloride
Potassium chloride
Ascorbic Acid
Sodium Ascorbate
(112 g) | MOVIPREP
(NORGINE) | |
| A07AA06 | Paromomycin sulfate
(250 mg) | HUMATIN
(PFIZER) | Capsules | No | Alternative: syrup to be diluted in at least 60 mL water and then given immediately | [8,9] |
| A07AA09 | Vancomycin hydrochloride
(250 mg) | MAXIVANIL
(GENETIC) | Capsules | No | Alternatives: powder for oral solution or for infusion | [10] |
| A07AA11 | Rifaximin
(200 mg) | NORMIX
(ALFA WASSERMANN) | Coated tablets | No | Alternative: oral suspension | [8,9] |
| A07AA12 | Fidaxomicin
(200 mg) | DIFICLIR
(ASTELLAS) | Coated tablets | Yes | Grind the tablet, suspend the powder in water and administer immediately | [27] |
| A07BC05 | Diosmectite
(3 g) | DIOSMECTAL
(MALESCI) | Powder for oral
solution | Yes | Dissolve the powder in half a glass of water and then administer | |
| A07DA03 | Loperamide hydrochloride
(2 mg) | LOPERAMIDE HEX
(SANDOZ) | Capsules | No | Use other dosage form | |
| A07DA03 | Loperamide hydrochloride
(2 mg) | DISSENTEN
(SOC.PRO.ANTIBIOTICI) | Tablets | Yes | | |
| A07EA06 | Budesonide
(3 mg) | ENTOCIR
(ASTRAZENECA) | Modified
release capsules | No | Alternative: suppositories | [8,9] |
| A07EA07 | Beclomethasone dipropionate
(5 mg) | CLIPPER
(CHIESI FARMACEUTIC)I | Gastro-resistant
tablets | No | Alternative: rectal preparations for distal ulcerative colitis | [8,9] |
| A07EC01 | Sulfasalazine
(500 mg) | SALAZOPYRIN EN
(PFIZER) | Gastro-resistant
tablets | No | | [8,9] |
| A07EC02 | Mesalazine
(400, 500, 800, 1200 mg) | PENTASA
(FERRING) | Modified
release tablets | Yes | Break the tablet and let it disintegrate in water. Alternatives: suppositories or rectal suspension | SmPC |
| A07EC02 | Mesalazine
(400, 500, 800, 1200 mg) | MESAVANCOL
(GIULIANI) | Modified
release tablets | No | Do not crush
Alternatives: rectal suspension or suppositories | [8,9] |
PENTACOL
(SOFAR) |
| A07FA02 | Saccharomyces boulardii
(5 bn) | CODEX
(ZAMBON) | Capsules | No | | |
CODEX
(ZAMBON) | Powder for oral suspension | | |
| A09AA02 | Pancrelipase
(150, 300 mg) | CREON 25000
(BGP PRODUCTS) | Modified
release capsules | Yes | Open the capsule without grinding the granules, suspend them in 20 mL water or in liquids, with a pH <5.5 such as apple, pineapple juice or yogurt, then administer immediately | |
| A10BA02 | Metformin hydrochloride
(500, 850, 1000 mg) | GLUCOPHAGE
(BRUNO FARMACEUTICI) | Coated tablets | Yes | Grind the tablet, suspend the powder in 20 mL water, and administer immediately | [10] |
| A10BA02 | Metformin hydrochloride
(500, 850, 1000 mg) | ZUGLIMET
(ZENTIVA) | Coated tablets | Yes | Grind the tablet, suspend the powder in 20 mL water, then administer immediately | |
| A10BB01 | Glibenclamide
(5 mg) | DAONIL
(SANOFI) | Tablets | Yes | Grind the tablet, suspend the powder in 10 mL water, then administer immediately
Stop EN at least 30 min before drug administration | |
| A10BB09 | Gliclazide
(30, 60 mg) | DIAMICRON
(SERVIER) | Modified
release tablets | No | | [8,9] |
| A10BB09 | Gliclazide
(80 mg) | GLICLAZIDE
(ZENTIVA) | Tablets | Yes | Grind the tablet and suspend the powder in 10 mL water, then administer immediately
Stop EN at least 30 min before drug administration
Divisible tablet. | [10] |
| A10BB12 | Glimepiride
(2 mg) | GLIMEPIRIDE ACC
(ACCORD HEALTHCARE) | Tablets | Yes | Disperse the tablet in 10 mL water until a fine dispersion is created
Administer before the main meal | [10] |
Glimepiride
(3 mg) | GLIMEPIRIDE SAN
(SANDOZ) |
| Glimepiride(1, 2, 3, 4 mg) | AMARYL(SANOFI) |
| A10BD02 | Metformin hydrochloride Glibenclamide
(500 + 5 mg) | GLICONORM
(ABIOGEN PHARMA) | Coated tablets | No | Administer the two drugs separately
Flush with 10 mL water between each one | [8,9] |
Metformin hydrochloride Glibenclamide
(400 + 2.5/500 + 5 mg) | GLIBOMET
(LABORATORI GUIDOTTI) |
Metformin hydrochloride Glibenclamide
(400 + 2.5 mg) | SUGUAN
(SANOFI) |
| A10BD05 | Pioglitazone
Metformin hydrochloride
(15 + 850 mg) | COMPETACT
(TAKEDA) | Coated tablets | No | | [8,9] |
| A10BD07 | Sitagliptin phosphate monohydrate
Metformin hydrochloride
(50 + 850 mg) | JANUMET
(MSD) | Coated tablets | Yes | | |
| A10BD08 | Vildagliptin
Metformin
(50 + 1000 mg) | EUCREAS
(NOVARTIS) | Tablets | No | | [8,9] |
| A10BD10 | Saxagliptin hydrochloride Metformin hydrochloride
(2.5 + 850 mg) | KOMBOGLYZE
(ASTRAZENECA) | Coated tablets | No | | [8,9] |
| A10BF01 | Acarbose
(50, 100 mg) | GLUCOBAY
(BAYER) | Tablets | Yes | The tablets do not disperse easily in water but require gentle stirring for 5 min. A fine suspension is obtained which must be administered immediately, then continue with EN | [10] |
| A10BG03 | Pioglitazone hydrochloride
(15, 30 mg) | PIOGLITAZONE ACV
(ACTAVIS) | Tablets | Yes | | |
ACTOS
(TAKEDA) | | |
| A10BH01 | Sitagliptin phosphate monohydrate
(100 mg) | TESAVEL
(ADDENDA) | Coated tablets | Yes | | |
| A10BH01 | Sitagliptin Phosphate Monohydrate
(50, 100 mg) | JANUVIA
(MSD) | Coated tablets | Yes | Check the glycemia | |
| A10BH02 | Vildagliptin
(50 mg) | GALVUS
(NOVARTIS) | Tablets | No | | |
| A10BH03 | Saxagliptin hydrochloride
(5 mg) | ONGLYZA
(ASTRAZENECA) | Coated tablets | No | | |
| A10BX02 | Repaglinide
(0.5, 1 mg) | REPAGLINIDE EG
(EG) | Tablets | Yes | | |
Repaglinide
(1, 2 mg) | REPAGLINIDE SAN
(SANDOZ) | | |
| A11AA03 | Vitamins Mineral salts | SUPRADYN
(BAYER) | Effervescent
tablets | Yes | Dissolve in water and administer at the end of the effervescence | |
| A11CC04 | Calcitriol
(0.25 µg) | DIFIX
(PROMEDICA) | Soft gels | No | Drug adheres to the walls of the tube, risk of loss | [8,9] |
ROCALTROL
(ROCHE) |
CALCITRIOLO TEV
(TEVA) |
| A11DA01 | Thiamine hydrochloride
(300 mg) | BENERVA
(TEOFARMA) | Gastro-resistant tablets | No | | [8,9] |
| A11DB | Thiamine hydrochloride Cyanocobalamin
Pyridoxine hydrochloride | BENEXOL B12
(BAYER) | Coated tablets | No | Alternative: solution for injection | [8,9] |
| A11DB | Thiamine hydrochloride Cyanocobalamin
Pyridoxine hydrochloride | TRINEVRINA B6
(LABORATORI GUIDOTTI) | Coated tablets | No | Alternative: solution for injection | [8,9] |
| A11HA02 | Pyridoxine hydrochloride
(300 mg) | BENADON
(BAYER) | Gastro-resistant tablets | No | Alternative: solution for injection | [8,9] |
| A11HA03 | Alpha-Tocopherol
(400 IU) | RIGENTEX
(BRACCO) | Soft gels | No | | [8,9] |
| A11JB | Sodium Citrate
Potassium Citrate
Thiamine diphosphate
Riblofavin-5 monophosphate monosodium
Pyridoxine hydrochloride
Citric acid | BIOCHETASI
(SIGMS TAU) | Effervescent granules | Yes | | |
| A12AA04 | Calcium Carbonate
(1000 mg) | CALCIODIE
(SPA) | Effervescent
tablets | Yes | Dissolve in 20 mL water and administer at the end of the effervescence at least 1 h before or 2 h after the NE | |
| A12AX | Calcium Carbonate
Cholecalciferol | IDEOS
(MEDA PHARMA) | Chewable
tablets | Yes | | |
METOCAL VIT.D3
(MEDA PHARMA) | | |
| A12BA01 | Potassium Chloride
(600 mg) | KCL-RETARD
(ASTELLAS) | Modified
release tablets | No | Change route of administration | [8,9] |
| A12BA30 | Potassium Citrate
Potassium Succinate
Potassium Malate
Potassium Tartrate
Potassium Bicarbonate | POTASSION
(ACARPIA) | Effervescent granules | Yes | Dissolve in water, and administer at the end of the effervescence | |
| A16AA02 | Ademetionine Butanedisulfonate
(400 mg) | SAMYR 400
(BGP PRODUCTS) | Gastro-resistant tablets | No | Alternative: solution for injection | [8,9] |
| B01AA03 | Warfarin sodium
(5 mg) | COUMADIN
(BRISTOL-MYERS SQUIBB) | Tablets | Yes | Grind the tablet and suspend the powder in 10 mL water, then administer immediately
Stop feeding at least 1 h before and 2 h after drug administration
Avoid supplementation with vitamin K
Warning: drug-EN interaction, continuous monitoring and dosage adjustment.
If possible administer low molecular weight heparins | [10] |
| B01AA07 | Acenocumarol
(1, 4 mg) | SINTROM
(MERUS LABS LUXCO II SARL) | Tablets | Yes | Grind the tablet, dissolve in 10 mL water and administer immediately
Trituration can alter bioavailability
Check for clotting and prothrombin time | [10] |
| B01AB11 | Sulodexide
(250 LSU) | VESSEL
(ALFA WASSERMANN) | Soft gels | No | Oily excipients do not guarantee correct administration
Alternative: solution for injection | [8,9] |
| B01AC04 | Clopidogrel besylate
(75 mg) | CLOPIDOGREL AURO
(ACTAVIS) | Coated tablets | No | Alternative: Plavix | |
| B01AC04 | Clopidogrel besylate
(75, 300 mg) | PLAVIX
(SANOFI) | Coated tablets | Yes | Crush the tablet and disperse the powder in 10 mL water
Administer by an 8 Fr tube | [10] |
| B01AC05 | Ticlopidine hydrochloride
(250 mg) | TIKLID
(SANOFI) | Coated tablets | Yes | Grind the tablet, dissolve the powder in 20 mL water, and administer immediately
preferably concurrently with EN to avoid gastrointestinal adverse effects | |
| B01AC06 | Acetylsalicylic Acid
(100 mg) | ACIDO ACETILSALICILICO SAN
(SANDOZ) | Gastro-resistant tablets | No | The tablet must not be crushed to avoid irritating effects on the intestine
Alternatives: effervescent tablets, chewable tablets, granules for oral solution | [8,9] |
| B01AC06 | Acetylsalicylic Acid
Magnesium Hydroxide
Algedrate | ASCRIPTIN
(SANOFI) | Tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water, and administer immediately concurrently with EN | |
| B01AC06 | Lysine Acetylsalicylate
(75, 100, 160 mg) | CARDIRENE
(SANOFI) | Powder for oral solution | Yes | Dilute with at least 60 mL of water and administer immediately in conjunction with EN | |
| B01AC07 | Dipyridamole
(75, 200 mg) | PERSANTIN
(BOEHRINGER INGELHEIM) | Coated tablets | Yes | | [10] |
| B01AC07 | Dipyridamole
(75 mg) | PERSANTIN R
(BOEHRINGER INGELHEIM) | Modified
release capsules | No | | [8,9] |
| B01AC10 | Indobufen
(200 mg) | IBUSTRIN
(PFIZER) | Tablets | Yes | | |
| B01AC22 | Prasugrel hydrochloride
(100 mg) | EFIENT
(DAIICHI SANKYO) | Coated tablets | Yes | Grind the tablet and administer immediately
Highly photosensitive hygroscopic drug | |
| B01AC24 | Ticagrerol
(90 mg) | BRILIQUE
(ASTRAZENECA) | Coated tablets | Yes | Crush the tablet to a fine powder, transfer it in half a glass of water and administer
Rinse the tube with water | SmPC |
| B01AC30 | Dipyridamole
Acetylsalicylic Acid
(200 + 25 mg) | AGGRENOX (BOEHRINGER INGELHEIM) | Modified
release capsules | No | The granules can block the tube
Alternatives: change dosage form or route of administration | |
| B01AE07 | Dabigatran etexilate mesylate
(75, 110, 150 mg) | PRADAXA
(BOEHRINGER INGELHEIM) | Capsules | No | The opening of the capsule increases drug bioavailability by 75%
Bleeding risk
Switch to another anticoagulant | |
| B01AF01 | Rivaroxaban
(10, 15, 10 mg) | XARELTO
(BAYER) | Coated tablets | Yes | Crush the tablet, dissolve the powder in water, then administer immediately
Rinse the tube with water
The drug administration should be followed immediately by EN | SmPC |
| B01AF02 | Apixaban
(2.5, 5 mg) | ELIQUIS
(BRISTOL-MYERS SQUIBB) | Coated tablets | Yes | Crush the tablet and suspend the powder in 60 mL water or 5% dextrose water solution, then administer immediately
The crushed tablets are stable up to 4 h when stored at 30 °C | SmPC |
| B02BX05 | Eltrombopag olamine
(25, 50 mg) | REVOLADE
(NOVARTIS) | Coated tablets | No | | |
| B03AA01 | Ferrous (II) glycine sulphate
(100 mg) | NIFEREX
(UCB PHARMA) | Capsules | No | Risk of tube occlusion | |
| B03AA03 | Ferrous (II) gluconate
(80 mg) | PRONTOFERRO
(IBSA FARMACEUTICI) | Effervescent
tablets | Yes | Dissolve the tablet in water, administer before EN
Alternatives: ampoules or syrup | SmPC |
| B03AA07 | Ferrous (II) sulfate
(105 mg) | FERROGRAD
(TEOFARMA) | Modified
release tablets | No | Alternative: ampoules | [8,9] |
| B03BB01 | Folic Acid
(5 mg) | FOLINA
(TEOFARMA) | Capsules | No | Alternative: liquid formulation | [8,9] |
| B06AA | Promelase
(30 mg) | FLAMINASE (GRUNENTHAL) | Gastro-resistant tablets | No | | [8,9] |
| C01AA05 | Digoxin
(0.0625, 0.125 mg) | LANOXIN
(ASPEN PHARMA) | Tablets | Yes | Grind the tablet, dissolve it in 10 mL of water, administer immediately. Stop NE 2 h before and 2 h after the drug administration. Inaccurate dosage, change in bioavailability: monitor the patient
Preferably give liquid formulation | [10] |
| C01AA05 | Digoxin
(0.1, 0.2 mg) | EUDIGOX
(TEOFARMA) | Soft gels | No | Alternative: liquid formulation | [8,9] |
| C01AA08 | Methyl Digoxin
(0.1 mg) | LANITOP
(RIEMSER PHARMA) | Tablets | Yes | Grind the tablet, dissolve it in 10 mL water and administer immediately
Preferably give drops | |
| C01BA | Dihydroquinidine hydrochloride
(250 mg) | IDROCHINIDINA R
(TEOFARMA) | Capsules | Yes | Open the capsule, suspend the powder in 10 mL water, then administer immediately | |
| C01BC03 | Propafenone hydrochloride
(325 mg) | RYTMONORM
(BGP PRODUCTS) | Modified
release capsules | No | Alternatives: Rytmonorm coated tablets or solution for injection | [8,9] |
| C01BC03 | Propafenone hydrochloride
(150, 300 mg) | RYTMONORM
(BGP PRODUCTS) | Coated tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately
Interaction with EN: the effect can be enhanced by the presence of food
Alternative: solution for injection | |
| C01BC04 | Flecanide Acetate
(100 mg) | ALMARYTM
(MEDA PHARMA) | Tablets | Yes | Disperse the tablet in 10 mL water for 2 min
The suspension must be administered immediately | [10] |
| C01BC04 | Flecainide Acetate
(100 mg) | FLECAINIDE TEV
(TEVA) | Tablets | Yes | Divisible tablets | |
| C01BD01 | Amiodarone hydrochloride
(200 mg) | CORDARONE
(SANOFI) | Tablets | Yes | Divisible tablets | |
| C01BD07 | Dronedarone hydrochloride
(400 mg) | MULTAQ
(SANOFI) | Coated tablets | Yes | | |
| C01DA08 | Isosorbide dinitrate
(5 mg) | CARVASIN
(TEOFARMA) | Sublingual
tablets | No | Do not grind
Reduction of the absorption of the drug due to the hepatic first pass effect
Administer sublingually only if the patient is conscious | [8,9] |
| C01DA14 | Isosorbide mononitrate
(60 mg) | DURONITRIN (ASTRAZENECA) | Modified
release tablets | No | Alternative: Nitroglycerine transdermal formulation | [8,9] |
Isosorbide mononitrate
(60 mg) | MONOKET MULTITAB (CHIESI FARMACEUTICI) | Modified
release tablets |
Isosorbide mononitrate
(20 mg) | MONOKET
(CHIESI FARMACEUTICI) | Modified
release tablets |
Isosorbide mononitrate
(50 mg) | MONOKET RETARD
(CHIESI FARMACEUTICI) | Modified
release capsules |
Isosorbide mononitrate
(20, 40, 50, 80 mg) | MONOCINQUE R
(IST.LUSOFARMACOI) | Modified
release capsules |
| C01EB17 | Ivabradine hydrochloride | PROCORALAN
(SERVIER) | Coated tablets | Yes | Crush the tablet, suspend in water and administer immediately | |
| Ivabradine hydrochloride | CORLENTOR
(STRODER) | |
| C01EB18 | Ranolazine
(375, 500, 750 mg) | RANEXA
(A.MENARINI) | Modified
release tablets | No | | [8,9] |
| C02AB01 | Methyldopa
(250 mg) | ALDOMET
(IROKO PRODUCTS) | Coated tablets | No | Change antihypertensive drug | |
| C02AC01 | Clonidine hydrochloride | CATAPRESAN
(BOEHRINGER INGELHEIM) | Tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water and administer immediately | [10] |
| C02CA04 | Doxazosin mesylate
(2, 4 mg) | CARDURA
(PFIZER) | Tablets | Yes | Grind the tablet, dissolve the powder with 10 mL sterile water and administer immediately
Monitor the patient and adjust the therapy | [10] |
| C02DC01 | Loniten
(5 mg) | MINOXIDIL
(PFIZER) | Tablets | Yes | The tablet is dispersed in water to give a fine suspension which must be administered immediately | [10] |
| C02KX01 | Bosentan monohydrate
(62.5, 125 mg) | TRACLEER
(ACTELION PHARMA) | Coated tablets | Yes | Crush the tablet and dissolve it in at least 50 mL water, it is not very soluble
Use individual protections for handling
Dispersible tablets (Tracleer 32 mg) for pediatrics | |
| C02KX02 | Ambriesentan
(5 mg) | VOLIBRIS
(GLAXOSMITHKLINE) | Coated tablets | No | | |
| C02KX04 | Macitentan
(10 mg) | OPSUMIT
(ACTELION PHARMA) | Coated tablets | Yes | Crush the tablet into a fine powder and add water, to have a suspension (drug not soluble)
Rinse the tube several times after administration
Use individual protections for handling
Teratogen drug | |
| C02KX05 | Riociguat
(2.5 mg) | ADEMPAS
(MSD) | Coated tablets | Yes | The tablet can be crushed and mixed with water | SmPC |
| C03AA03 | Hydrochlorothiazide
(25 mg) | ESIDREX
(NOVARTIS) | Tablets | Yes | | |
| C03BA04 | Chlorthalidone
(25 mg) | IGROTON
(AMDIPHARM) | Tablets | Yes | The tablet disperses in water within 2 min | [10] |
| C03BA08 | Metolazone
(5, 10 mg) | ZAROXOLYN
(TEOFARMA) | Tablets | Yes | Grind the tablet, dissolve it with 10 mL water and administer immediately
Monitor the patient, drug bioavailability variable | [10] |
| C03CA01 | Furosemide
(25 mg) | FUROSEMIDE
(LAB.FARMACOLOGICO MILANESE) | Tablets | Yes | Grind the tablet, dissolve it in 10 mL water and administer immediately preferably concomitantly with EN to minimize gastrointestinal effects
It is recommended to use the liquid form | |
Furosemide
(25, 500 mg) | LASIX
(SANOFI) |
Furosemide
(500 mg) | FUROSEMIDE TEV
(TEVA) |
| C03CA04 | Torasemide
(10 mg) | TORASEMIDE TEV
(TEVA) | Tablets | Yes | Divisible tablet | |
| C03DA01 | Spironolactone
(100 mg) | ALDACTONE
(SANOFI) | Coated tablets | Yes | Grind, dissolve the powder in 10 mL water and administer immediately
Pay attention to concomitant administration with potassium. | [10] |
Spironolactone
(25 mg) | ALDACTONE
(SANOFI) | Capsules | Yes | Open the capsule, dissolve in 10 mL of water and administer immediately
Pay attention to concomitant administration with potassium. |
| C03DA01 | Spironolactone
(25 mg) | URACTONE
SPA (SOC.PRO.ANTIBIOTICI) | Tablets | Yes | | |
| C03DA03 | Canrenone
(50, 100 mg) | LUVION
(THERABEL GIENNE PHARMA) | Tablets | No | Alternative: solution for injection | |
| C03EA01 | Amiloride hydrochloride Hydrochlorothiazide
(5 + 50 mg) | MODURETIC
(MSD) | Tablets | Yes | Grind, dissolve in 10 mL of water and administer immediately. | [10] |
| C03EA14 | Potassium Canrenoate
Butizide
(5 + 50 mg) | KADIUR
(THERABEL GIENNE PHARMA) | Tablets | No | | [8,9] |
| C03EB01 | Furosemide
Spironolactone
(25 + 37 mg) | LASITONE
(SANOFI) | Capsules | No | Administer the two drugs separately
Flush with 10 mL water between each one | [8,9] |
| C04AD03 | Pentoxifylline
(400 mg) | TRENTAL
(SANOFI) | Modified
release tablets | No | | [8,9] |
| C05CA53 | Diosmin
Hesperidin
(500 mg) | DAFLON
(SERVIER) | Coated tablets | Yes | Grind the tablet, dissolve the powder in 10 mL of water and administer it immediately | |
| C05CX | Escin
(40 mg) | EDEVEXIN
(I.B.I.GIOVANNI LORENZINI) | Coated tablets | No | Alternatives: solution for injection or cutaneous gel | |
| C07AA05 | Propranolol hydrochloride
(40 mg) | INDERAL
(ASTRAZENECA) | Tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water and administer immediately
Alternative: galenic preparation (Propranolol 2 or 5 mg/mL oral suspension) | |
| C07AA07 | Sotalol hydrochloride
(80 mg) | RYTMOBETA
(BGP PRODUCTS) | Tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water by shaking for 5 min, then administer immediately
Stop EN 2 h before and 2 h after drug administration
Follow the same administration schedule | [10] |
| C07AA12 | Nadolol
(80 mg) | NADOLOLO SFV
(SANOFI) | Tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water and administer immediately
If possible, use another beta blocker available in liquid form | |
| C07AB02 | Metoprolol tartrate
(100 mg) | LOPRESOR
(DAIICHI SANKYO) | Coated tablets | Yes | It is recommended to use a beta blocker liquid preparation
If Metoprolol is non-replaceable, prepare an extemporaneous suspension of Metoprolol 10 mg/mL with simple syrup, or grind the tablet and dissolve it in 10 mL of water. | [10] |
| METOPROLOLO HEX (SANDOZ) | Tablets |
| C07AB03 | Atenolol
(100 mg) | ATENOLOLO RAT (RATIOPHARM) | Coated tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water under shaking for 5 min, then administer immediately Stop EN 30 min before and 30 min after drug administration
Divisible tablets | [10] |
| ATENOLOLO HEX (SANDOZ) | Grind the tablet, dissolve the powder in 10 mL water, then administer immediately
Stop EN 30 min before and 30 min after drug administration | [10] |
| C07AB07 | Bisoprolol fumarate
(10 mg) | CONCOR
(BRACCO) | Tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water under shaking for 5 min, then administer immediately preferably in the morning after EN | [10] |
Bisoprolol hemifumarate
(2.5 mg) | CONGESCOR
(DAIICHI SANKYO) | Coated tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water under shaking for 5 min, then administer immediately preferably in the morning after EN | [10] |
Bisoprolol hemifumarate
(1.25, 2.5, 3.75, 5, 10 mg) | CARDICOR
(RECORDATI) | Coated tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water under shaking for 5 min, then administer immediately preferably in the morning after EN
Divisible tablet | [10] |
| C07AB12 | Nebivolol hydrochloride
(5 mg) | NEBIVOLOLO SAN (SANDOZ) | Tablets | No | Alternative: Atenolol | |
| C07AG01 | Labetalol hydrochloride
(100 mg) | TRANDATE
(TEOFARMA) | Tablets | No | | |
| C07AG02 | Carvedilol
(6.25 mg) | OMERIA
(MEDIOLANUM FARMACEUTICI) | Tablets | Yes | Grind and dissolve the tablet in 10 mL water
Administer with EN to reduce the risk of orthostatic hypotension | [10] |
| C07AG02 | Carvedilol
(25 mg) | Carvedilol ZTV
(ZENTIVA) | Tablets | Yes | Grind and dissolve the tablet in 10 mL water
Administer with EN to reduce the risk of orthostatic hypotension | [10] |
| C08CA01 | Amlodipine besylate
(5, 10 mg) | NORVASC
(PFIZER) | Tablets | Yes | Dissolve the tablet in 10 mL water and administer immediately concurrently with EN to minimize the gastrointestinal side effects | [10] |
Amlodipine maleate
(10 mg) | AMLODIPINA WIN
(ZENTIVA) |
| C08CA02 | Felodipine
(5 mg) | FELODIPINA WPI
(ZENTIVA) | Modified
release tablets | No | | [8,9] |
| C08CA05 | Nifedipine
(20,30,60 mg) | ADALAT CRONO
(BAYER) | Modified
release tablets | No | Do not grind, use the liquid formulation (Nifedicor 20 mg/mL drops) to be diluted with 60 mL water and administer immediately | [8,9] |
Nifedipine
(10 mg) | ADALAT
(BAYER) | Soft gels | No | Alternative: Nifedicor 20 mg/mL drop solution to be diluted with 60 mL of water |
Nifedipine
(30 mg) | CORAL
(SO.SE.PHARM) | Modified
release tablets | No | Alternative: Nifedicor 20 mg/mL drop solution to be diluted with 60 mL of water |
| C08CA06 | Nimodipine
(30 mg) | NIMOTOP
(BAYER) | Coated tablets | Yes | Photosensitive drug, use amber syringe
Not compatible with PVC tube | |
| C08CA09 | Lacipidine
(4 mg) | LACIPIL
(GLAXOSMITHKLINE) | Coated tablets | No | Drug poorly soluble in water and photosensitive
Divisible tablets
Alternative: Amlodipine formulations | |
| C08CA11 | Manidipine hydrochloride
(20 mg) | IPERTEN
(CHIESI FARMACEUTICI) | Tablets | No | | |
MANIDIPINA MYL
(MYLAN) | Yes | |
MANIDIPINA TEV
(TEVA) | Yes | Divisible tablets |
| C08CA13 | Lecardipine hydrochloride
(10 mg) | ZANEDIP
(RECORDATI) | Coated tablets | Yes | Administer away from EN
Alternative: Amlodipine formulations | |
| C08DA01 | Verapamil hydrochloride
(40, 80, 180 mg) | ISOPTIN
(BGP PRODUCTS) | Coated tablets | Yes | | [10] |
| C08DA01 | Verapamil hydrochloride
(120 mg) | ISOPTIN R
(BGP PRODUCTS) | Modified
release tablets | No | Do not grind
Alternative: solution for injection | [8,9] |
VERAPAMIL HEX
(SANDOZ) |
| C08DB01 | Diltiazem hydrochloride
(120 mg) | ALTIAZEM (IST.LUSOFARMACO) | Modified
release tablets | No | Alternative: 60 mg Diltiazem divisible tablets dosage form | [8,9] |
Diltiazem hydrochloride
(300 mg) | ALTIAZEM (IST.LUSOFARMACO) | Modified
release capsules |
Diltiazem hydrochloride
(200, 300 mg) | TILDIEM
(SANOFI) | Modified
release tablets |
Diltiazem Hydrochloride
(60, 120 mg) | TILDIEM
(SANOFI) | Modified
release tablets |
| C09AA01 | Captopril
(25, 50 mg) | CAPTOPRIL RAT (RATIOPHARM) | Tablets | Yes | Grind, dissolve the tablet in 10 mL water and administer immediately
Stop EN at least two h before drug administration. Divisible tablets | [10] |
| C09AA02 | Enalapril maleate
(5, 20 mg) | ENAPREN
(MSD) | Tablets | Yes | Grind the tablet, dissolve the powder in 10 mL sterile water and administer immediately | [10] |
| C09AA03 | Lisinopril dehydrate
(5, 20 mg) | ZESTRIL
(ASTRAZENECA) | Tablets | Yes | Divisible tablets
It is possible to prepare an extemporaneous galenic solution/suspension: stable for 30 days at 2–8 °C | [10] |
| C09AA03 | Lisinopril dehydrate
(20 mg) | LISINOPRIL WPI
(ZENTIVA) | Tablets | Yes | | |
| C09AA04 | Perindopril arginine
(5 mg) | COVERSYL
(SERVIER) | Coated tablets | Yes | | |
| C09AA04 | Perindopril tosilate
(5 mg) | PERINDOPRIL TEV
(TEVA) | Coated tablets | No | Switch to another ACE inhibitor | |
| C09AA05 | Ramipril
(10 mg) | QUARK
(POLIFARMA) | Tablets | Yes | Dissolve the tablet in 20 mL water and administer immediately
Divisible tablets | [10] |
Ramipril
(2.5, 5, 10 mg) | TRIATEC
(SANOFI) |
| C09AA13 | Moexipril hydrochloride
(15 mg) | FEMIPRES
(UCB PHARMA) | Coated tablets | No | | [10] |
| C09AA15 | Zofenopril calcium
(30 mg) | BIFRIL
(IST.LUSOFARMACO) | Coated tablets | Yes | | |
| C09BA04 | Perindopril arginine
Indapamide
(2.5 + 0.625/5 + 1.25 mg) | PRETERAX
(SERVIER) | Coated tablets | Yes | Grind, dissolve the tablet in 10 mL water and administer immediately | |
Perindopril arginine
Indapamide
(5 + 1.25 mg) | PRELECTAL
(STRODER) | |
| C09BA05 | Ramipril
Piretanide
(5 + 6 mg) | PRILACE
(SANOFI) | Coated tablets | No | Divisible tablets | [8,9] |
| C09BA06 | Quinapril hydrochloride
hydrochlorothiazide
(20 + 12.5 mg) | ACEQUIDE
(RECORDATI) | Coated tablets | Yes | Crushable and divisible tablets | |
| C09BA09 | Fosinopril sodium
Hydrochlorothiazide
(20 + 12.5 mg) | FOSICOMBI
(A.MENARINI) | Tablets | No | Switch to another ACE inhibitor | [8,9] |
| C09CA01 | Losartan Potassium
(50 mg) | LORTAAN
(MSD) | Coated tablets | Yes | Grind, dissolve the tablet in 10 mL water and administer immediately | [10] |
| C09CA03 | Valsatrtan
(40 mg) | TAREG
(NOVARTIS) | Coated tablets | Yes | | |
Valsatrtan
(80, 160 mg) | NOVARTIS | Capsules | | |
| C09CA04 | Ibersartan
(150, 300 mg) | APROVEL
(SANOFI) | Tablets | Yes | The tablet is dispersed in 10 mL water under stirring for 5–10 min
No blockage risk with an 8 Fr tube | |
Ibersartan
(150 mg) | KARVEA
(SANOFI) | Stirring to disperse the tablet | |
| C09CA06 | Candesartan Cilexetil
(8 mg) | RATACAND
(ASTRAZENECA) | Tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water and administer immediately | [10] |
Candesartan Cilexetil
(16 mg) | BLOPRESS
(TAKEDA) | Grind the tablet, dissolve the powder in 10 mL water and administer immediately. Alternative: Ibersartan |
Candesartan Cilexetil
(8, 16 mg) | CANDESARTAN TEV
(TEVA) | Grind the tablet, dissolve the powder in 10 mL water and administer immediately
Divisible tablets. |
| C09CA07 | Telmisartan
(20, 80 mg) | PRITOR
(BAYER) | Tablets | Yes | Dissolve the tablet in 5 mL water under stirring and administer immediately
Hygroscopic drug | [10] |
Telmisartan
(20, 40, 80 mg) | MICARDIS
(BOEHRINGER INGELHEIM) |
| C09CA08 | Olmesartan medoxomil
(10, 20 mg) | OLPRESS
(MENARINI) | Coated tablets | No | Alternative: Ibersartan | |
Olmesartan medoxomil
(10, 20, 40 mg) | PLAUNAC
(MENARINI) |
| C09DA08 | Olmesartan medoxomil
Hydrochlorothiazide
(20 + 12.5 mg) | PLAUNAZIDE
(MENARINI) | Coated tablets | No | Administer the two drugs separately
Flush with 10 mL water between each one | [8,9] |
Olmesartan medoxomil
Hydrochlorothiazide
(20 + 25/40 + 25 mg) | OLPREZIDE
(MENARINI) |
| C09DB02 | Olmesartan medoxomil
Amlodipine besylate
(40 + 5 mg) | SEVIKAR
(IST.LUSOFARMACO) | Coated tablets | No | Alternative: administer separately another angiotensin II antagonist drug (eg Irbesartan) and the calcium channel blocker | [8,9] |
| C09XA02 | Aliskiren hemifumarate
(150, 300 mg) | RASILEZ
(NOVARTIS) | Coated tablets | Yes | | |
| C10AA01 | Simvastatin
(10, 20 mg) | SINVACOR
(MSD) | Coated tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water and administer immediately, preferably together with the last course of EN | [10] |
Simvastatin
(10, 20 mg) | SIMVASTAT.RAT (RATIOPHARM) | Grind the tablet, dissolve the powder in 10 mL water and administer immediately, preferably together with the last course of EN
Divisible tablets |
Simvastatin
(10 mg) | SIMVASTAT.TEV
(TEVA) | Grind the tablet, dissolve the powder in 10 mL water and administer immediately, preferably together with the last course of EN
Divisible tablets |
Simvastatin
(40 mg) | SIMVASTAT.ZTV
(ZENTIVA) | Grind the tablet, dissolve the powder in 10 mL water and administer immediately, preferably together with the last course of EN. |
| C10AA03 | Pravastatin sodium
(20 mg) | PRAVASTAT.RAT
(TEVA) | Tablets | Yes | Divisible tablets | [10] |
| C10AA04 | Fluvastatin sodium
(80 mg) | LESCOL
(NOVARTIS) | Modified
release tablets | No | Alternative: Fluvastatin 20 or 40 mg capsules, or Atorvastatin or Pravastatin | [8,9] |
| C10AA05 | Atorvastatin calcium trihydrate
(10, 20, 40 mg) | TORVAST
(PFIZER) | Coated tablets | Yes | The tablet is dispersed within 2–5 min in 10 mL water
Photosensitive drug
Absorption not affected by the presence of food | |
| C10AA07 | Rosuvastatin calcium
(5, 10, 20 mg) | CRESTOR
(ASTRAZENECA) | Coated tablets | Yes | Disperse the tablet in water, after 5 min a milky pink dispersion is obtained, then administer immediately
Better to use Atorvastatin | [10] |
| C10AB04 | Gemfibrozil
(600, 900 mg) | LOPID
(PFIZER) | Coated tablets | Yes | Crushable tablets | |
| C10AB05 | Fenofibrate
(145 mg) | FULCROSUPRA
(BGP PRODUCTS) | Coated tablets | No | | |
Fenofibrate
(200 mg) | FENOFIBRATO SAN
(SANDOZ) | Capsules | No | | |
| C10AC01 | Cholestyramine hydrochloride
(4 g) | QUESTRAN
(BRISTOL-MYERS SQUIBB) | Powder for oral suspension | No | Risk of occlusion of the tube due to the formation of a semi-solid mass
Possible interference with the absorption of other drugs | |
| C10AX06 | Polyenoic Omega (ethyl esters of polyunsaturated fatty acids)
(1000 mg) | SEACOR
(SOC.PRO.ANTIBIOTICI) | Soft gels | No | | [8,9] |
| C10AX09 | Ezetimibe
(10 mg) | EZETROL
(MERCK SHARP & DOHME) | Tablets | Yes | Disperse the tablet in 10 mL water under stirring Administer the suspension immediately | |
| C10BA02 | Ezetimibe
Simvastatin
(10 + 10 mg) | INEGY
(MERCK SHARP & DOHME) | Tablets | No | Administer the two drugs separately
Flush with 10 mL water between each one | [8,9] |
Ezetimibe
Simvastatin
(10 + 20 mg) | VYTORIN
(NEOPHARMED GENTILI) |
| D01BA02 | Terbinafine hydrochloride
(250 mg) | TERBINAFINA SAN
(SANDOZ) | Tablets | Yes | Disperse the tablet under stirring in 10 mL water for 5 min, to give a fine dispersion that must be immediately administered
Divisible tablets
Be careful when handling | [10] |
| D05BB02 | Acitretin
(10, 25 mg) | NEOTIGASON
(AUROBINDO) | Capsules | Yes | Open the capsule, disperse the content in water and administer immediately together with EN
If possible change the drug | |
| D10BA01 | Isotretinoin
(10 mg) | ISOTRETINOINA DIF
(DIFA COOPER) | Soft gels | No | | [8,9] |
| D11AH04 | Alitretinoin
(30 mg) | TOCTINO
GLAXOSMITHKLINE | Soft gels | No | | [8,9] |
| G02AB01 | Methylergometrine maleate
(0.125 mg) | METHERGIN
(NOVARTIS) | Coated tablets | No | Alternative: solution for injection | |
| G02CB01 | Bromocriptine mesylate
(2.5 mg) | PARLODEL
(MEDA PHARMA) | Tablets | Yes | Administer together with EN | [10] |
| G02CB03 | Cabergoline
(0.5 mg) | DOSTINEX
(PFIZER) | Tablets | No | | |
| G03AA10 | Ethinylestradiol
Gestodene | GINODEN
(BAYER) | Coated tablets | No | Alternative: transdermal patches | [8,9] |
ESTINETTE
(EFFIK) |
| G03AD01 | Levonorgestrel
(1.5 mg) | NORLEVO
(LABORATOIRE HRA PHARMA) | Tablets | No | Alternative: transdermal patches | [8,9] |
| G03DA04 | Prgesterone
(100, 200 mg) | PROGEFFIK
(EFFIK) | Capslues | No | Alternatives: insert the capsule vaginally or use the solution for injection | [8,9] |
| G03DB04 | Nomegestrol acetate
(5 mg) | LUTENYL
(RATIOPHARM) | Tablets | No | | |
| G03DC02 | Norethisterone acetate
(10 mg) | PRIMOLUT NOR
(BAYER) | Tablets | Yes | Disperse the tablet in 10 mL water under stirring for 5 min, to give a fine dispersion that must be immediately administered
Be careful when handling | [10] |
| G03HA01 | Cyproterone acetate
(50 mg) | ANDROCUR
(BAYER) | Tablets | Yes | Grind, disperse the tablet in 10 mL water and administer immediately. Where possible switch to intramuscular administration | |
| G03XB01 | Mifepristone
(200 mg) | MIFEGYNE
(EXELGYN) | Tablets | Yes | | |
| G03XB02 | Ulipristal acetate
(5 mg) | ESMYA
(GEDEON RICHTER) | Tablets | Yes | Crushable tablets | |
| G04BD | Flavoxate
Propyphenazone
(200 + 30 mg) | CISTALGAN
(MEDA PHARMA) | Coated tablets | No | | [8,9] |
| G04BD04 | Oxybutynin hydrochloride
(5 mg) | DITROPAN
(SANOFI) | Tablets | Yes | | |
| G04BD08 | Solifenacin succinate
(5 mg) | VESIKER
(ASTELLAS) | Coated tablets | No | | |
| G04BD12 | Mirabregon
(50 mg) | BETMIGA
(ASTELLAS) | Modified
release tablets | No | | [8,9] |
| G04BE03 | Sildenafil citrate
(20 mg) | REVATIO
(PFIZER) | Coated tablets | No | Alternative: oral suspension | |
Sildenafil citrate
(50, 100 mg) | VIAGRA
(PFIZER) | Tablets | Yes | Grind, disperse the tablet in 10 mL water and administer immediately | |
| G04BE08 | Tadalafil
(5, 10, 20 mg) | CIALIS
(ELI LILLY) | Coated tablets | No | | |
Tadalafil
(20 mg) | ADCIRCA
(ELI LILLY) | | |
| G04CA01 | Alfuzosin hydrochloride
(2.5 mg) | MITTOVAL
(SANOFI) | Modified
release tablets | No | Alternative: Doxasozin | [8,9] |
XATRAL
(SANOFI) |
| G04CA01 | Alfuzosin hydrochloride
(2.5 mg) | XATRAL
(SANOFI | Coated tablets | Yes | Alternative: Doxasozin | |
| G04CA02 | Tamsulisin hydrochloride
(0.4 mg) | PRADIF
(BOEHRINGER INGELHEIM) | Capsules | No | Risk of blockage of the tube
Alternative: Doxasozin | |
| G04CA03 | Terazosin hydrochloride
(5 mg) | TERAPROST
(MALESCI) | Tablets | Yes | | |
Terazosin hydrochloride
(2, 5 mg) | TERAZOSINA TEV
(TEVA) | Tablets | No | | |
| G04CA04 | Silodosin
(4, 8 mg) | UROREC
(RECORDATI) | Capsules | Yes | Open the capsule and suspend the content in water | |
| G04CB01 | Finasteride
(5 mg) | FINASTERIDE (NEOPHARMED GENTILI) | Coated tablets | Yes | Crushable tablets
Teratogen drug | [10] |
| G04CB02 | Dutasteride
(0.5 mg) | AVODART
(GLAXOSMITHKLINE) | Capsules | No | Content of the capsule liquid, irritant and teratogen | |
| H01BA02 | Desmopressin acetate
(60, 120 µg) | MINIRIN/DDAVP
(FERRING) | Sublingual
tablets | No | Do not crush the tablet
Reduction of the absorption of the drug due to the hepatic first pass effect | [8,9] |
| H02AB01 | Betamethasone sodium phosphate | BENTELAN
(SIGMATAU) | Effervescent
tablets | Yes | Dissolve the tablet in 10 mL water and administer immediately | SmPC |
| H02AB04 | Methylprednisolone
(4, 16 mg) | MEDROL
(PFIZER) | Tablets | Yes | Grind the tablet, dissolve it in 10 mL water and administer immediately | [10] |
| H02AB07 | Prednisone
(5, 25 mg) | DELTACORTENE
DELTACROTENE FTE
(BRUNO FARMACEUTICI) | Tablets | Yes | Grind the tablet, dissolve it the tablet in 10 mL water and administer immediately | [10] |
| H02AB07 | Prednisone
(5 mg) | LODOTRA
(MUNDIPHARMA) | Modified
release tablets | No | Alternative: Deltacortene 5 mg | [8,9] |
| H02AB09 | Hydrocortisone
(5, 20 mg) | PLENADREN
(SHIRE) | Modified release tablets | No | | [8,9] |
| H02AB10 | Cortisone acetate
(25 mg) | CORTONE ACETATO (TEOFARMA) | Tablets | Yes | | |
| H03AA01 | Levothyroxine sodium
(50, 100 µg) | TIROSINT
(IBSA FARMACEUTICI) | Tablets | Yes | Grind the tablet, dissolve it in 10 mL water.
Interaction with EN: suspend it 1 h before and resume it 2 h later drug administration
Alternative: Tirosint oral solution which is not affected by simultaneous food intake | SmPC |
| H03AA01 | Levothyroxine sodium
(25, 50, 75, 100 µg) | EUTIROX
(MERCK SERONO) | Tablets | Yes | Grind the tablet and dissolve it in 10 mL water, then administer immediately
Suspend EN 1 h before and resume it 2 h later drug administration
Monitor TSH levels
Divisible tablets | [10] |
Levothyroxine sodium
(25 µg) | LEVOTIROXINA TEV
(TEVA) |
| H03BB02 | Thiamazole (Methimazole)
(5 mg) | TAPAZOLE
(TEOFARMA) | Tablets | Yes | Grind the tablet and disperse it in 10 mL water, then administer immediately
Divisible tablets | |
| H05BX01 | Cinacalcet hydrochloride
(30, 60, 90 mg) | MIMPARA
(AMGEN) | Coated tablets | No | | |
| H05BX02 | Paricalcitol
(1 µg, 2 µg) | ZEMPLAR
(ABBVIE) | Capsules | No | Alternative: Zemplar solution for injection | |
| J01AA02 | Doxycycline hyclate
(100 mg) | BASSADO
(PFIZER) | Tablets | Yes | Grind the tablet and dissolve it in 10 mL water. Administer immediately with plenty of water to prevent irritation | [10] |
| J01AA08 | Minocycline hydrochloride
(100 mg) | MINOCIN
(TEOFARMA) | Capsules | No | | |
| J01CA01 | Ampicillin
(500 mg) | AMPLITAL
(PFIZER) | Capsules | No | Alternative: powder for solution for injection | |
| J01CA04 | Amoxicillin trihydrate
(1 g) | ZIMOX
(PFIZER) | Tablets | No | Alternatives: switch to other formulations (drops, oral suspension, powder for oral suspension) | |
| J01CA04 | Amoxicillin trihydrate
(1 g) | ZIMOX
(PFIZER) | Chewable
tablets | Yes | Dissolve the tablets in a half glass of water | SmPC |
| J01CR02 | Amoxicillin trihydrate
Clavulanate potassium
(875 + 125 g) | ABBA
(FIDIA FARMACEUTICI) | Powder for oral suspension | Yes | Dissolve the powder in 50 mL water
Administer together with EN to minimize the gastrointestinal side effects | SmPC |
| AUGMENTIN (GLAXOSMITHKLINE) |
| J01CR02 | Amoxicillin trihydrate
Clavulanate potassium
(875 + 125 g) | NEODUPLAMOX
(VALEAS) | Coated tablets | No | Alternatives: switch to other formulations (suspension, powder for oral suspension) | [8,9] |
| J01CR04 | Sultamicillin tosylate
(750 mg) | UNASYN
(PFIZER) | Coated tablets | No | Alternatives: powder for oral solution, or solution for injection | [8,9] |
| J01DB01 | Cephalexin monohydrate
(1 g) | KEFORAL
(CRINOS) | Tablets | No | Switch to another cephalosporin or use the oral suspension available on the market | [8,9] |
Cephalexin
(1 g) | CEPOREX
(TEOFARMA) | Coated tablets |
| J01DC02 | Cefuroxime acetoxyethyl
(250 mg) | ZINNAT (GLAXOSMITHKLINE) | Coated tablets | No | Use the syrup which must be diluted with 60 mL water and administered immediately | [8,9] |
| J01DD08 | Cefixime
(400 mg) | UNIXIME
(F.I.R.M.A.) | Coated tablets | No | Alternatives: dispersible tablets or oral suspension. Stop the EN at least 1 h before and resume it 2 h after drug administration | [8,9] |
| J01DD13 | Cefpodoxime proxetil
(100, 200 mg) | CEFODOX
(SCHARPER) | Coated tablets | Yes | Better to switch to the oral suspension | [8,9] |
| J01EE01 | Trimethoprim
Sulfamethoxazole
(160 + 800 mg) | BACTRIM
(ROCHE) | Tablets | No | Alternative: oral suspension | [8,9] |
| J01FA09 | Clarithromycin
(250, 500 mg) | KLACID
(BGP PRODUCTS) | Coated tablets | No | Alternatives: oral suspension, solution for injection
Photosensitive drug | [8,9] |
Clarithromycin
(500 mg) | CLARITROMICINA TEV
(TEVA) | Alternative: oral suspension which must be diluted with 60 mL water and administered immediately |
| J01FA10 | Azithromycin monohydrate
(500 mg) | AZITROMICINA EG
(EG) | Coated tablets | No | Alternative: oral suspension which must be diluted with 60 mL water and administered immediately
It does not interact with EN | [8,9] |
Azithromycin monohydrate
(500 mg) | AZITROMICINA MYL
(MYLAN) | Alternative: oral suspension which must be diluted
Administer 1 h before or 2 h after EN |
Azithromycin dihydrate
(500 mg) | ZITROMAX
(PFIZER) | Alternative: oral suspension which must be diluted
Administer 1 h before or 2 h after EN |
Azithromycin dihydrate
(500 mg) | AZITROMICINA PRG
(PROGE FARM) | Alternative: oral suspension which must be diluted
Administer 1 h before or 2 h after EN |
Azithromycin monohydrate
(500 mg) | AZITROMICINA SAN
(SANDOZ) | Alternative: oral suspension which must be diluted
Administer 1 h before or 2 h after EN |
| J01FA10 | Azithromycin dihydrate
(500 mg) | TROZAMIL
(SO.SE.PHARM) | Coated tablets | Yes | Alternative: oral suspension which must be diluted
Administer 1 h before or 2 h after EN | |
| J01FF01 | Clindamycin hydrochloride
(150 mg) | DALACIN-C
(PFIZER) | Capsules | Yes | Disperse the content in water and then administer Alternatives: solution for injection or other route of administration (cutaneous gel) | |
| J01MA02 | Ciprofloxacin hydrochloride
(250, 500, 750, 1000 mg) | CIPROXIN
(BAYER) | Coated tablets | No | EN-drug interaction: decreased absorption and therapeutic failure
Alternatives: solution for infusion or granules for oral suspension (EN must be suspended 30 min before and 30 min after drug administration) | |
Ciprofloxacin hydrochloride monohydrate
(250 mg) | CIPROFLOXAC.RAT (RATIOPHARM) |
Ciprofloxacin hydrochloride monohydrate
(750 mg) | CIPROFLOXAC.SAN (SANDOZ) |
| J01MA12 | Levofloxacin hemihydrate
(500 mg) | LEVOFLOXACINA SAN (SANDOZ) | Coated tablets | No | Alternatives: solution for injection or switch to more bioavailable Ofloxacin | |
| J01MA14 | Moxifloxacin hydrochloride
(400 mg) | AVALOX
(BAYER) | Coated tablets | Yes | The coating exerts only taste masking | |
| J01MA17 | Prulifloxacin
(600 mg) | UNIDROX
(ANGELINI) | Coated tablets | yes | Administer immediately after dissolution of the tablet in water | |
| J01XE01 | Nitrofurantoin macrocrystals
(50 mg) | NEOFURADANTIN (GRUNENTHAL) | Capsules | No | Risk of tube blockage | |
| J01XX01 | Fosfomycin trometamol salt
(3 g) | BERNY
(SO.SE.PHARM) | Granules for oral solution | Yes | Administer away from the EN, usually at bedtime | |
| J01XX08 | Linezolid
(600 mg) | ZYVOXID
(PFIZER) | Coated tablets | Yes | Grind the tablet, dissolve the powder in 20 mL water then administer immediately
Prefer oral suspension or solution for infusion
Any interaction with EN | [10] |
| J02AC01 | Fluconazole
(100 g) | FLUCONAZOLO EG
(EG) | Capsules | Yes | Open the capsule, disperse the content in 20 mL water, then administer immediately
Prefer oral suspension or solution for injection
If dietary product is rich in fiber, suspend EN 1 h before and 1 h after drug administration | [10] |
DIFLUCAN
(PFIZER) |
FLUCONAZOLO SAN
(SANDOZ) |
FLUCONAZOLO HEX
(SANDOZ) |
FLUCONAZOLO RAT
(TEVA) |
| J02AC02 | Itraconazole
(100 g) | SPORANOX
(JANSSEN CILAG) | Capsules | No | Alternatives: solution for infusion or oral solution to be diluted with water
The absorption occurs at acidic pH
Administer together with EN | |
| J02AC03 | Voriconazole
(200 mg) | VFEND
(PFIZER) | Coated tablets | Yes | Grind the tablet, disperse the powder in water and administer away from EN (1 h before or 2 h after EN)
Alternatives: oral suspension or powder for solution for injection | [10] |
| J04AB02 | Rifampicin
(300, 400 mg) | RIFADIN
(SANOFI) | Capsules | No | Alternative: syrup to be diluted and administered away from EN | [8,9] |
| J04AC01 | Isoniazid
(200 mg) | NICOZID
(PIAM FARMACEUTICI) | Tablets | Yes | Grind the tablet, dissolve the powder in 20 mL water then administer immediately
Stop EN 1 h before and 2 h after drug administration | [10] |
| J04AK01 | Pyrazinamide
(500 mg) | PIRALDINA
(BRACCO) | Tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water then administer immediately | [10] |
| J04AK02 | Ethambutol hydrochloride
(400 mg) | ETAPIAM
(PIAM FARMACEUTICI) | Coated tablets | Yes | Grind the tablet, dissolve the powder in 10 mL water then administer immediately 2 h after EN | [10] |
| J04AM02 | Rifampicin
Isoniazid
(300 + 150 mg) | RIFINAH 300
(SANOFI) | Coated tablets | No | Administer the two drugs separately
Flush with 10 mL water between each one | [8,9] |
| J04AM05 | Isoniazid
Pyrazinamide
Rifampicin
(50 + 300 +120 mg) | RIFATER
(SANOFI) | Coated tablets | Yes | Suspend EN 1 h before drug administration | |
| J05AB01 | Acyclovir
(400 mg) | ACICLOVIR EG
(EG) | Tablets | Yes | Grind the tablet, dissolve it in 30 mL water then administer immediately
Alternatives: oral suspension or solution for injection | [10] |
Acyclovir
(800 mg) | ACICLIN
(FIDIA FARMACEUTICI) |
Acyclovir
(400 mg) | ACICLOVIR DRM
(TEVA) |
| J05AB04 | Ribavirin
(200 mg) | REBETOL
(MSD) | Capsules | No | Alternative: Ribavirin (Rebetol) oral solution | [8,9] |
COPEGUS
(ROCHE) | Coated tablets |
RIBAVIRINA SAN
(SANDOZ) | Capsules |
RIBAVIRINA TEV
(TEVA) | Capsules |
| J05AB11 | Valacyclovir hydrochloride
(1 g) | ZELITREX (GLAXOSMITHKLINE) | Coated tablets | Yes | Better to switch to Acyclovir
The tablets are difficult to crush and the powder does not suspend well
Alternative: galenic suspension of Valaciclovir 50 mg/mL, by triturating the tablets and using simple syrup (good stability up to 21 days at 2–8 °C) | |
| VALACICLOVIR MYL (MYLAN) |
| J05AB14 | Valgancyclovir hydrochloride
(450 mg) | DARILIN
(ROCHE) | Coated tablets | No | Alternative: Valgancyclovir oral suspension (Novir) | [8,9] |
| J05AE03 | Ritonavir
(100 mg) | NORVIR
(ABBVIE) | Coated tablets | No | Alternative: Ritonavir oral suspension
Tablets not crushable | [8,9] |
| J05AE07 | Fosamprenavir calcium
(700 mg) | TELZIR
(VIIV HEALTHCARE) | Coated tablets | No | Alternative: Fosamprenavir oral suspension (Telzir) | [8,9] |
| J05AE08 | Atazanavir sulfate
(200, 300 mg) | REYATAZ
(BRISTOL-MYERS SQUIBB) | Capsules | Yes | Open the capsule, dissolve the content in water, administer it via tube, then continue with EN | |
| J05AE10 | Darunavir ethanolate
(600, 800 mg) | PREZISTA
(JANSSEN CILAG) | Coated tablets | No | Alternative: Darunavir ethanolate oral suspension | |
| J05AE11 | Telaprevir
(375 mg) | INCIVO
(JANSSEN CILAG) | Coated tablets | No | | |
| J05AE12 | Boceprevir
(200 mg) | VICTRELIS
(MSD) | Capsules | No | | |
| J05AE14 | Simeprevir
(150 mg) | OLYSIO
(JANSSEN CILAG) | Capsules | No | | |
| J05AF02 | Didanosine
(250 mg) | VIDEX 250
(BRISTOL-MYERS SQUIBB) | Capsules | No | | |
| J05AF05 | Lamivudine
(100 mg) | ZEFFIX (GLAXOSMITHKLINE) | Coated tablets | Yes | Disperse the tablet in 10 mL water, a pale orange solution is obtained, then administer immediately
Prefer: Lamivudine oral solution | [10] |
| J05AF05 | Lamivudine
(100 mg) | LAMIVUDINA MYL
(MYLAN) | Coated tablets | No | Prefer liquid formulation | [8,9] |
| J05AF07 | Tenofovir Disoproxil Fumarate
(245 mg) | VIREAD
(GILEAD SCIENCES) | Coated tablets | Yes | Dissolve the tablet in 100 mL water or orange juice and administer | SmPC |
| J05AF08 | Adefovir dipivoxil
(10 mg) | HESPERA
(GILEAD SCIENCES) | Coated tablets | No | Switch to Tenofovir | |
| J05AF09 | Emtricitabine
(200 mg) | EMTRIVA
(GILEAD SCIENCES) | Capsules | No | Alternative: Emtricitabine oral solution
Increase the dose by 20% with respect to the tablets, due to reduced bioavailability of the oral solution | |
| J05AF10 | Entecavir
(0.5, 1 mg) | BARACLUDE
(BRISTOL-MYERS SQUIBB) | Coated tablets | No | Switch to oral solution | [8,9] |
| J05AG01 | Nevirapine
(400 mg) | VIRAMUNE
(BOEHRINGER INGELHEIM) | Tablets | No | Switch to oral suspension | [8,9] |
Nevirapine
(200 mg) | NEVIRAPINA TEV
(TEVA) | Alternative: liquid formulation |
| J05AG03 | Efavirenz
(200 mg) | SUSTIVA
(BRISTOL-MYERS SQUIBB) | Capsules | Yes | | |
| J05AG03 | Efavirenz
(600 mg) | EFAVIRENZ MYL
(MYLAN) | Coated tablets | No | Alternative: liquid formulation | [8,9] |
| J05AG04 | Etravirine
(200 mg) | INTELENCE
(JANSSEN CILAG) | Tablets | Yes | Dissolve the tablet in 10 mL water and administer | SmPC |
| J05AG05 | Rilpivirine hydrochloride
(25 mg) | EDURANT
(JANSSEN CILAG) | Coated tablets | No | | |
| J05AH02 | Oseltamivir phosphate
(30, 45 mg) | TAMIFLU
(ROCHE) | Capsules | No | Alternative: oral suspension | [8,9] |
| J05AR01 | Lamivudine
Zidovidine
(150 + 300 mg) | LAMIV+ZIDOV MYL
(MYLAN) | Coated tablets | No | Administer the two drugs separately in liquid form | [8,9] |
| J05AR02 | Abacavir sulfate
Lamivudine
(600 + 300 mg) | KIVEXA
(VIIV HEALTHCARE) | Coated tablets | No | Separate preparations of the two drugs are available in the form of oral suspension | [8,9] |
| J05AR03 | Emtricitabine
Tenofovir disoproxil
(200 + 245 mg) | TRUVADA
(GILEAD SCIENCES) | Coated tablets | Yes | The tablets can be dissolved in about 100 mL of water, orange juice or grape juice and taken immediately | SmPC |
| J05AR04 | Abacavir sulfate
Lamivudine
Zidovudine
(300 + 150 + 300 mg) | TRIZIVIR
(VIIV HEALTHCARE) | Coated tablets | No | Separate preparations of the three drugs are available in the form of oral suspension | [8,9] |
| J05AR06 | Efavirenz
Emtricitabine
Tenofovir disoproxil
(600 +200 +245 mg) | ATRIPLA
(GILEAD SCIENCES) | Coated tablets | No | | [8,9] |
| J05AR08 | Emtricitabine
Rilpivirine hydrochloride
Tenofovir disoproxil fumarate
(200 + 25 + 245 mg) | EVIPLERA
(GILEAD SCIENCES) | Coated tablets | No | | [8,9] |
| J05AR09 | Emtricitabine
Tenofovir disoproxil fumarate
Elvitegravir
Cobicistat
(200 + 300 + 150 + 150 mg) | STRIBILD
(GILEAD SCIENCES) | Coated tablets | No | | [8,9] |
| J05AR10 | Lopinavir
Ritonavir
(200 + 50 mg) | KALETRA
(ABBVIE) | Coated tablets | No | Tablet crushing reduces AUC by approximately 40% Preferred alternative: Kaletra oral solution (slightly viscous, contains alcohol and propylene glycol) | [8,9] |
| J05AX08 | Raltegravir potassium
(400 mg) | ISENTRESS
(MSD) | Coated tablets | No | Drug insoluble in water | |
| J05AX09 | Maraviroc
(150, 300 mg) | CELSENTRI
(VIIV HEALTHCARE) | Coated tablets | No | | |
| J05AX12 | Dolutegravir sodium
(50 mg) | TIVICAY
(VIIV HEALTHCARE) | Coated tablets | Yes | Grind the tablet, disperse it in water and administer away from EN (2 h before or 6 h after EN) | |
| J05AX14 | Daclatasvir dihydrochloride
(60 mg) | DAKLINZA
(BRISTOL-MYERS SQUIBB) | Coated tablets | Yes | The coating serves only to cover the drug bitter taste | |
| J05AX15 | Sofosbuvir
(400 mg) | SOVALDI
(GILEAD SCIENCES) | Coated tablets | Yes | The coating serves only to cover the drug bitter taste
The tablets can be triturated and administered via tube concurrently with EN | |
| J05AX16 | Dasabuvir
(250 mg) | EXVIERA
(ABBVIE) | Coated tablets | No | Not crushable tablet | |
| J05AX65 | Ledipasvir
Sofosbuvir
(90 + 400 mg) | HARVONI
(GILEAD SCIENCES) | Coated tablets | No | | [8,9] |
| J05AX67 | Ombitasvir
Paritaprevir
Ritonavir
(12.5 + 75 + 50 mg) | VIEKIRAX
(ABBVIE) | Coated tablets | No | Not crushable tablet | [8,9] |
| L01AA01 | Cyclophosphamide
(50 mg) | ENDOXAN
(BAXTER) | Coated tablets | No | Alternative: solution for injection | [8,9] |
| L01AA02 | Chlorambucil
(2 mg) | LEUKERAN
(GLAXOSMITHKLINE) | Coated tablets | No | | |
| L01AA03 | Melfalan
(25 mg) | ALKERAN
(ASPEN PHARMA) | Coated tablets | No | Alternative: solution for injection | [8,9] |
| L01AB01 | Busulfan
(2 mg) | MYLERAN
(ASPEN PHARMA) | Coated tablets | No | Alternative: galenic suspension of Busulfan 2 mg/mL by triturating the tablet
Shelf life 30 days if stored between 2–8 °C | |
| L01AX03 | Temozolomide
(100 mg) | TEMODAL
(MSD) | Capsules | No | | |
Temozolomide
(100 mg) | TEMOZOLOMIDE SUH (RANBAXY ITALIA) | | |
Temozolomide
(5, 20, 140, 180, 250 mg) | TEMOZOLOMIDE SUH
(SUN PHARMACEUTICALS) | | |
| L01BA01 | Methotrexate
(2.5 mg) | METHOTREXATE
(PFIZER) | Tablets | No | Alternative: Methotrexate solution for injection
Tablets are divisible but not crushable | [8,9] |
| L01BB05 | Fludarabine phosphate
(10 mg) | FLUDARA
(GENZYME) | Coated tablets | No | Alternative: solution for injection | [8,9] |
| L01BC06 | Capecitabine
(150, 500 mg) | CAPECITABINA ACC
(ACCORD HEALTHCARE) | Coated tablets | Yes | Disperse the tablet in 200 mL warm water and then administer immediately
Attention to handling, use individual protection measures | |
CAPECITABINA MYL
(MYLAN) |
XELODA
(ROCHE) |
| L01CA04 | Vinorelbine ditartrate(20, 30 mg) | NAVELBINE
(PIERRE FABRE PHARMA) | Soft gels | No | Alternative: concentrate for solution for infusion | [8,9] |
| L01XE01 | Imatinib mesylate
(100 mg) | GLIVEC
(NOVARTIS) | Capsules | Yes | Open the capsule, dilute the content in water or apple juice
Attention to handling, use individual protection measures | SmPC |
| L01XE02 | Gefitinib
(250 mg) | IRESSA
(ASTRAZENECA) | Coated tablets | Yes | Disperse the tablet in half glass water (it takes 20 min)
Administer the suspension immediately | SmPC |
| L01XE03 | Erlotinib hydrochloride
(100, 150 mg) | TARCEVA
(ROCHE) | Coated tablets | No | | |
| L01XE04 | Sunitinib maleate
(12.5, 25, 50 mg) | SUTENT
(PFIZER) | Capsules | Yes | Attention to handling, use individual protection measures | |
| L01XE05 | Sorafenib tosilate
(200 mg) | NEXAVAR
(BAYER) | Coated tablets | No | No specific data are available | |
| L01XE06 | Dasatinib monohydrate
(50, 100, 140 mg) | BRISTOL-MYERS SQUIBB | Coated tablets | No | No specific data are available
Attention to handling, use individual protection measures | |
| L01XE07 | Lapatinib ditosylate monohydrate
(250 mg) | TYVERB
(NOVARTIS) | Coated tablets | No | | |
| L01XE08 | Nilotinib hydrochloride
(150, 200 mg) | TASIGNA
(NOVARTIS) | Capsules | Yes | | |
| L01XE10 | Everolimus
(5, 10 mg) | AFINITOR
(NOVARTIS) | Tablets | No | | |
| L01XE11 | Pazopanib hydrochloride
(400 mg) | VOTRIENT
(NOVARTIS) | Coated tablets | No | | |
| L01XE13 | Afatinib dimaleate
(40 mg) | GIOTRIF
(BOEHRINGER INGELHEIM) | Coated tablets | Yes | Disperse the tablet in water without breaking it, under stirring for 15 min until a fine dispersion is obtained, then administer immediately away from the EN
Be careful when handling | SmPC |
| L01XE16 | Crizotinib
(250 mg) | XALKORI
(PFIZER) | Capsules | No | | |
| L01XE18 | Ruxolitinib phosphate
(5, 15 mg) | JAKAVI
(NOVARTIS) | Tablets | No | | |
| L01XE21 | Regorafenib
(40 mg) | STIVARGA
(BAYER) | Coated tablets | No | | |
| L01XX05 | Hydroxycarbamide
(500 mg) | ONCO CARBIDE
(TEOFARMA) | Capsules | Yes | Attention to handling, use individual protection measures
Antineoplastic drug | [10] |
| L01XX14 | Tretinoin
(10 mg) | VESANOID
(CHEPLAPHARM ARZNEIMITTEL) | Capsules | No | | |
| L01XX23 | Mitotane
(500 mg) | LYSODREN
(HRA PHARMA) | Tablets | Yes | The company confirms that the tablet can be crushed and the API dissolves in water
Use gloves for handling
Antineoplastic drug | Manufacturer upon request |
| L01XX35 | Anagrelide hydrochloride
(0.5 mg) | XAGRID
(SHIRE) | Capsules | No | | |
| L02AB01 | Megestrol acetate
(160 mg) | GESTROLTEX
(PHARMATEX) | Tablets | No | | |
| L02BA01 | Tamoxifen citrate
(20 mg) | NOMAFEN
(ITALIAN DEVICES) | Coated tablets | No | | |
KESSAR
(ORION) | Tablets | | |
| L02BB01 | Flutamide
(250 mg) | FLUTAMIDE FDI
(FIDIA FARMACEUTICI) | Tablets | Yes | Tablet disaggregates slowly, maintain it under stirring in 10 mL water for 10 min
Attention to handling | [10] |
| L02BB03 | Bicalutamide
(50, 150 mg) | CASODEX
(ASTRAZENECA) | Coated tablets | Yes | If possible use other antiandrogens administered as a subcutaneous implant
Grind the tablet and disperse the powder in water
Work in a closed system (disperse the tablet in the body of a syringe)
Administer immediately after the EN
Attention to handling | [10] |
Bicalutamide
(150 mg) | BICALUTAMIDE HIK
(HIKMA FARMACEUTICA) | Coated tablets | No | If possible use other antiandrogens administered by subcutaneous implant | [10] |
| L02BB03 | Bicalutamide
(50, 150 mg) | BICALUTAMIDE TEV
(TEVA) | Coated tablets | No | If possible use other antiandrogens administered by subcutaneous implant | |
| L02BG03 | Anastrozole
(1 mg) | ANASTROZOLO ACC
(ACCORD HEALTHCARE) | Coated tablets | No | Data not available | |
ARIMIDEX
(ASTRAZENECA) | Yes | Disperse the tablet in 10 mL water in a closed system (disperse the tablet in the body of a syringe)
Drug slightly soluble in water | |
| L02BG04 | Letrozole
(2.5 g) | LETROZOLO ACC
(ACCORD HEALTHCARE) | Coated tablets | No | | |
LETROZOLO SAN
(SANDOZ) | | |
| L02BG06 | Exemestane
(25 mg) | AROMASIN
(PFIZER) | Coated tablets | No | | |
| L02BX03 | Abiraterone acetate
(25 mg) | ZYTIGA
(JANSSEN CILAG) | Coated tablets | No | | |
| L04AA06 | Mycophenolate sodium
(180, 360 mg) | MYFORTIC
(NOVARTIS) | Gastro-resistant tablets | No | Alternative: Mycofenolate mofetil (Cellept) oral solution | [8,9] |
Mycofenolate mofetil
(250, 500 mg) | CELLCEPT
(ROCHE) | Capsules |
Mycofenolate mofetil
(250, 500 mg) | MYFENAX
(TEVA) | Coated tablets |
| L04AA10 | Sirolimus
(0.5, 1 mg) | RAPAMUNE
(PFIZER) | Coated tablets | No | Alternative: Sirolimus (Rapamune) oral solution | [8,9] |
| L04AA13 | Leflunomide
(20 mg) | ARAVA
(SANOFI) | Coated tablets | Yes | Dissolve the tablet in 10 mL water and administer immediately | [10] |
| L04AA18 | Everolimus
(0.25, 0.75 mg) | CERTICAN
(NOVARTIS) | Tablets | No | Alternative: orosoluble tablets to be dissolved in 10 mL water
Stop EN 1 h before and 2 h after drug administration | [8,9] |
| L04AA27 | Fingolimod hydrochloride
(0.5 mg) | GILENYA
(NOVARTIS) | Capsules | No | | |
| L04AA31 | Teriflunomide
(14 mg) | AUBAGIO
(GENZYME) | Coated tablets | No | | |
| L04AD01 | Cyclosporine
(10, 25, 50, 100 mg) | SANDIMMUN NEORAL (NOVARTIS) | Capsules | No | Alternative: Cyclosporine oral solution | [8,9] |
| L04AD02 | Tacrolimus
(1 mg) | TACROLIMUS ACC
(ACCORD HEALTHCARE) | Capsules | No | Alternatives: Porgraf capsules or Tacni capsules | |
Tacrolimus monohydrate
(0, 5, 1, 3, 5 mg) | ADVAGRAF
(ASTELLAS) |
Tacrolimus monohydrate
(0, 5, 1, 5 mg) | PROGRAF
(ASTELLAS) | Yes | Open the capsule and dissolve the content in water
Administer through an 8 Fr caliber tube
Do not use a PVC tube
Beware of handling
Take on an empty stomach or at least 1 h before or 2–3 h after eating | |
Tacrolimus
(0.5 mg) | TACROLIMUS MYL
(MYLAN) | No | Alternative: Porgraf capsules | |
Tacrolimus monohydrate
(1 mg) | ADOPORT
(SANDOZ) | No | Alternative: Porgraf capsules | |
| L04AX01 | Azathioprine
(50 mg) | AZATIOPRINA
(ASPEN PHARMA) | Coated tablets | Yes | Disperse the tablet in 10 mL water in the body of a syringe (closed system)
Cytotoxic drug
Use personal protective equipment | [10] |
| L04AX02 | Thalidomide
(50 mg) | THALIDOMIDE CELGENE (CELGENE) | Capsules | No | The capsules must not be opened
Teratogen drug | |
| L04AX04 | Lenalomide
(5, 10, 15, 25 mg) | REVLIMID
(CELGENE) | Capsules | No | The capsules must not be opened
Teratogen drug | |
| L04AX05 | Pirfenidone
(267 mg) | ESBRIET
(ROCHE) | Capsules | No | | |
| L04AX06 | Pomalidomide
(4 mg) | IMNOVID
(CELGENE) | Capsules | No | Not available data on drug manipulation | |
| M01AB01 | Indomethacin
(25, 50 mg) | INDOXEN
(SIGMATAU) | Capsules | No | Alternative: suppositories | [8,9] |
| M01AB05 | Diclofenac sodium
(50, 150 mg) | FLOGOFENAC (A.MENARINI) | Modified
release capsules | No | Alternatives: dispersible tablets, solution for injection, cutaneous formulations | [8,9] |
| M01AB05 | Diclofenac sodium
(50, 150 mg) | DICLOREUM
(ALFA WASSERMANN) | Gastro-resistant tablets | No | Alternatives: dispersible tablets, solution for injection, cutaneous formulations | [8,9] |
| M01AB15 | Ketorolac tromethamine
(10 mg) | TORADOL
(RECORDATI) | Coated tablets | No | Alternatives: drops, solution for injection | [8,9] |
| M01AC01 | Piroxicam
(20 mg) | FELDENE SOL
(PFIZER) | Soluble tablets | Yes | | [8,9] |
| M01AE01 | Ibuprofen
(600 mg) | BRUFEN
(BGP PRODUCTS) | Coated tablets | No | Alternatives: effervescent granules, oral suspension to be diluted with the same water amount | [8,9] |
Ibuprofen
(600 mg) | BRUFEN
(BGP PRODUCTS) | Effervescent granules | Yes | Dissolve the granules in plenty of water |
Ibuprofen
(200 mg) | NUROFEN
(RECKITT BENCKISER) | Coated tablets | No | Alternative: oral suspension |
Ibuprofen arginine salt
(400 mg) | SPIDIDOL
(ZAMBON) | Coated tablets | No | Alternative: oral suspension |
| M01AE03 | Ketoprofen lysine salt
(80 mg) | OKI
(DOMPE’ FARMACEUTICI) | Granules for oral solution | Yes | Alternatives: solution for injection, suppositories, cutaneous gel | [8,9] |
Ketoprofen
(200 mg) | KETOPROFENE EG
(EG) | Capsules | No |
Ketoprofen lysine salt
(80 mg) | KETOPROFENE MYL (MYLAN) | Powder for oral solution | Yes |
Ketoprofen
(200 mg) | ORUDIS RETARD
(SANOFI) | Capsules | No |
| M01AH01 | Celecoxib
(200 mg) | CELEBREX
(PFIZER) | Capsules | Yes | Open the capsule and disperse the content in 10 mL water | [10] |
| M01AH05 | Etoricoxib
(60, 90 mg) | TAUXIB
(ADDENDA) | Coated tablets | Yes | The tablet immersed in 10 mL water, swells and then releases fine granules that must be administered in an 8 Fr caliber tube | [10] |
Etoricoxib
(60 mg) | ARCOXIA
(MSD) |
Etoricoxib
(60, 90 mg) | ALGIX
(NEOPHARMED GENTILI) |
| M01AX17 | Nimesulide
(100 mg) | AULIN
(HELSINN BIREX PHARMAC.) | Granules for oral solution | Yes | Disperse the tablet in 30 mL water and then administer immediately
Administer together with EN to reduce gastrointestinal side effects | |
| M03BX01 | Baclofen
(10, 25 mg) | LIORESAL
(NOVARTIS) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately | [10] |
| M03BX03 | Pridinol mesilate
(4 mg) | LYSEEN (GLAXOSMITHKLINE) | Tablets | Yes | Alternative: solution for injection | |
| M03BX05 | Thiocolchicoside
(4 mg) | MUSCORIL
(SANOFI) | Capsules | No | Alternative: solution for injection | [8,9] |
| M04AA01 | Allopurinol
(100, 300 mg) | ALLOPURINOLO TEV
(TEVA) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately | [10] |
| M04AA03 | Febuxostat
(80 mg) | ADENURIC
(A.MENARINI) | Coated tablets | No | Alternative: Allopurinol tablets | |
| M04AC01 | Colchicine
(1 mg) | COLCHICINA LIRCA (PHARMAFAR) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately | [10] |
| M05BA04 | Alendronate sodium
(10 mg) | ALENDROS
(ABIOGEN PHARMA) | Tablets | No | Interaction with EN
Irritant for mucosal membranes
If necessary grind the tablet and dissolve the powder in 50 mL water, then administer 30 min before the first daily administration of EN keeping the patient in a semi-sitting position
Rinse the tube with 100 mL water after administration | |
Alendronate sodium trihydrate
(70 mg) | ADRONAT
(NEOPHARMED GENTILI) |
Alendronate sodium
(10 mg) | DRONAL
(SIGMATAU) |
Alendronate sodium monohydrate
(70 mg) | ALENDRONATO TEV
(TEVA) |
| M05BA06 | Ibandronate sodium monohydrate
(150 mg) | BONVIVA
(ROCHE) | Coated tablets | No | | |
AC.IBANDR.TEV
(TEVA) | | |
| M05BB03 | Alendronate sodium trihydrate
Cholecalciferol
(70 mg + 5600 IU) | FOSAVANCE
(MSD) | Tablets | No | | |
| M05BX03 | Strontium ranelate
(2 g) | PROTELOS
(SERVIER) | Granules for oral
suspension | Yes | | |
| N02AA01 | Morphine sulfate
(10, 30 mg) | MS CONTIN
(MUNDIPHARMA) | Modified
release tablets | No | Alternative: drops to be diluted with 50 mL water and administered immediately | [8,9] |
| N02AA03 | Hydromorphone hydrochloride
(4, 8, 16 mg) | JURNISTA
(JANSSEN CILAG) | Modified
release tablets | No | | [8,9] |
| N02AA05 | Oxycodone hydrochloride
(5, 10, 20 mg) | OXYCONTIN
(MUNDIPHARMA) | Modified
release tablets | No | | [8,9] |
Oxycodone hydrochloride
(40 mg) | OXICODONE SAN
(SANDOZ) | |
| N02AA55 | Oxycodone hydrochloride
Paracetamol
(10 + 325/20 + 325 mg) | DEPALGOS
(MOLTENI & C. F.LLI ALITTI) | Coated tablets | No | | [8,9] |
| N02AA55 | Oxycodone hydrochloride
Naloxone hydrochloride dihydrate
(10 + 325/20 + 325 mg) | TARGIN
(MUNDIPHARMA) | Modified
release tablets | No | Alternative: switch to another analgesic opioid transdermal formulation | [8,9] |
| N02AA59 | Paracetamol
Codeine phosphate
(500 + 30 mg) | ANGELINI | Effervescent granules | Yes | Administer only after the end of the effervescence | |
| Coated tablets | The tablet can be triturated and dispersed in water | |
| N02AB03 | Fentanyl citrate
(100, 200, 300, 400, 600 µg) | ABSTRAL
(PROSTRAKAN) | Sublingual
tablets | No | | [8,9] |
Fentanyl citrate
(100, 200, 300, 400 µg) | EFFENTORA
(TEVA) | Orosoluble tablets | Yes | |
| N02AX02 | Tramadol hydrochloride
(100 mg) | CONTRAMAL (GRUNENTHAL) | Modified
release tablets | No | Alternatives: suppositories, orosoluble tablets, suppositories, drops to be diluted with 50 mL water | [8,9] |
| N02AX06 | Tapentadol hydrochloride
(50, 100 mg) | PALEXIA
(GRUNENTHAL) | Modified
release tablets | No | | [8,9] |
| N02AX52 | Tramadol hydrochloride
Paracetamol
(37.5 + 325 mg) | PATROL
(ALFA WASSERMANN) | Coated tablets | No | Alternative: effervescent tablets to be dissolved in water and administered after the end of the effervescence | [8,9] |
KOLIBRI
(ALFA WASSERMANN) |
| N02BA01 | Acetylsalicylic acid
(100 mg) | ASPIRINETTA
(BAYER) | Tablets | Yes | Immerse directly the tablet in water
Administer together with EN to reduce gastrointestinal side effects | SmPC |
| N02BE01 | Paracetamol
(500 mg) | TACHIPIRINA
(ANGELINI) | Tablets | Yes | Grind the tablet and disperse the powder in 10 mL water, then administer immediately
Alternatives: orosoluble tablets, granules, suppositories, syrup or solution for injection | |
Paracetamol
(1000 mg) | Effervescent tablets | Dissolve the tablet in 30 mL water and administer only at the end of the effervescence
Alternatives: orosoluble tablets, granules, suppositories or solution for injection | |
| N02BE01 | Paracetamol
(250, 500 mg) | TACHIPIRINA FLASHTAB
(ANGELINI) | Orodispersible tablets | Yes | | [8,9] |
Paracetamol
(250, 1000 mg) | TACHIPIRINA OROS (ANGELINI) | Orosoluble granules | |
| N02CC01 | Sumatriptan succinate
(50 mg) | SUMATRIPTAN DOC
(DOC GENERICI) | Coated tablets | Yes | Alternatives: solution for injection or nasal spray | [8,9] |
| N03AA02 | Phenobarbital
(100 mg) | LUMINALE BRACCO
(BRACCO) | Tablets | Yes | Grind the tablet and disperse the powder in 10 mL water, then administer immediately
Administer together with EN to reduce gastrointestinal side effects
Do not administer solution for injection via tube due to the presence of glycol | SmPC |
| N03AA02 | Phenobarbital
(100 mg) | GARDENALE
(SANOFI) | Tablets | Yes | Grind the tablet and disperse the powder in 10 mL water, then administer immediately
Administer together with EN to reduce gastrointestinal side effects
Do not administer solution for injection via tube due to the presence of glycol | |
| N03AA03 | Primidone
(250 mg) | MYSOLINE
(SIT LABORATORIO FARMACEUTICO) | Tablets | Yes | Grind the tablet and disperse the powder in 10 mL water, then administer immediately | |
| N03AB02 | Phenytoin sodium
(100 mg) | DINTOINA
(RECORDATI) | Coated tablets | Yes | Grind the tablet and disperse the powder in 10 mL water, then administer immediately
Stop EN 2 h before and 2 h after drug administration
Wash the tube with 50 mL water
Alternative: solution for injection | [10] |
| N03AB52 | Phenytoin
Methylphenobarbital
(100 + 40 mg) | DINTOINALE
(RECORDATI) | Tablets | Yes | | |
| N03AE01 | Clonazepam
(2 mg) | RIVOTRIL
(ROCHE) | Tablets | Yes | Alternative: Clonazepam drops to be diluted with 50 mL water and administered immediately | |
| N03AF01 | Carbamazepine
(200, 400 mg) | CARBAMAZEPINA
(EG) | Tablets | Yes | Grind the tablet and disperse the powder in 10 mL sterile water, then administer immediately
Stop EN 1 h before and 2 h after drug administration
Requires close monitoring, it interacts with the EN Can adhere to the walls of the tube and have a reduced absorption
Better to use syrup | [10] |
| N03AF01 | Carbamazepine
(400 mg) | TEGRETOL
(NOVARTIS) | Modified
release tablets | No | Alternatives: syrup or tablets | [8,9] |
| N03AF01 | Carbamazepine
(400 mg) | TEGRETOL
(NOVARTIS) | Tablets | Yes | Grind the tablet and disperse the powder in 10 mL sterile water, then administer immediately
Stop EN at least 1 h before and 2 h after the dose
Requires close monitoring, it interacts with the EN Can adhere to the walls of the tube and have a reduced absorption
Better to use syrup | [10] |
| N03AF02 | Oxcarbazepine
(300, 600 mg) | TOLEP
(NOVARTIS) | Tablets | Yes | Grind the tablet and disperse the powder in 10 mL water, then administer immediately | |
| N03AG01 | Valproic acid
Valproate sodium
(300, 500 mg) | AC VAL/S.VALP.RAT
(RATIOPHARM) | Modified
release tablets | No | Alternatives: oral solution or drops to be diluted with 100 mL water | [8,9] |
Valproic acid
Valproate sodium
(500 mg) | AC VALPROICO/SOD VALPR SAN
(SANDOZ) | Modified
release tablets |
Valproate sodium
(200, 500 mg) | DEPAKIN
(SANOFI) | Gastro-resistant tablets |
Valproic acid
Valproate sodium
(100, 250, 500, 750, 1000 mg) | DEPAKIN
(SANOFI) | Modified
release granules |
Valproate magnesium
(500 mg) | DEPAMAG
(SIGMATAU) | Gastro-resistant tablets |
| N03AG02 | Valpromide
(300 mg) | DEPAMIDE
(SANOFI) | Gastro-resistant tablets | No | | [8,9] |
| N03AG04 | Vugabatrin
(500 mg) | SABRIL
(SANOFI) | Coated tablets | No | Alternative: granules | |
| N03AX09 | Lamotrigine
(25, 200 mg) | LAMOTRIGINA EG
(EG) | Dispersible
tablets | Yes | Dissolve the tablet in 10 mL water and administer the solution immediately | |
| N03AX09 | Lamotrigine
(25, 50, 100, 200 mg) | LAMICTAL (GLAXOSMITHKLINE) | Dispersible
tablets | Yes | | SmPC |
| N03AX11 | Topiramate
(25 mg) | TOPIRAMATO DOC
(DOC GENERICI) | Coated tablets | Yes | The tablet is not easily dispersed in water, due to the coating, but requires gentle stirring for 5 min
A fine suspension is obtained which should be administered immediately
Better to use capsules | [10] |
Topiramate
(25, 50, 100 mg) | TOPIRAMATO EG
(EG) |
Topiramate
(25, 50, 100 mg) | TOPAMAX
(JANSSEN CILAG) |
Topiramate
(25 mg) | TOPIRAMATO SAN (SANDOZ) |
| N03AX11 | Topiramate
(25 mg) | TOPAMAX
(JANSSEN CILAG) | Capsules | Yes | Open the capsule, disperse the content in 20 mL water, then administer immediately | SmPC |
| N03AX12 | Gabapentin
(400 mg) | DOC GENERICI | Capsules | Yes | Open the capsule, disperse the content in 20 mL water, then administer immediately | [10] |
Gabapentin
(100, 300, 400 mg) | GABAPENTIN TEV
(TEVA) |
| N03AX14 | Levetiracetam
(500 mg) | LEVETIRACETAM DOC
(DOC GENERICI) | Coated tablets | Yes | Grind the tablet and dissolve the powder in 20 mL water, then administer immediately
Better to use oral solution | [10] |
Levetiracetam
(500, 1000 mg) | LEVETIRACETAM SAN (SANDOZ) |
| N03AX15 | Zonisamide
(50, 100 mg) | ZONEGRAN
(EISAI) | Capsules | Yes | | |
| N03AX16 | Pregabalin
(25, 75, 150 mg) | LYRICA
(PFIZER) | Capsules | Yes | Open the capsules and dissolve the content in water
Drug water soluble | |
| N03AX18 | Lacosamide
(50, 100 mg) | VIMPAT
(UCB PHARMA) | Coated tablets | Yes | Alternative: solution for injection | |
| N04AA02 | Biperiden hydrochloride
(4 mg) | AKINETON
(SIT LABORATORIO FARMACEUTICO) | Modified
release tablets | No | Alternative: Biperiden hydrochloride
2 mg | [8,9] |
| N04AA02 | Biperiden hydrochloride
(2 mg) | AKINETON
(SIT LABORATORIO FARMACEUTICO) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately | |
| N04AA11 | Bornaprine hydrochloride
(4 mg) | SORMODREN
(TEOFARMA) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately
Wash the tube with 50 mL water | |
| N04BA02 | Levodopa
Carbidopa
(100 + 25/200 + 50 mg) | SINEMET
(MSD) | Modified
release tablets | No | Alternative: dispersible tablets | [8,9] |
| N04BA02 | Levodopa
Carbidopa
(100 + 25/250 + 25 mg) | SINEMET
(MSD) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately
Stop EN 2 h before and 2 h after drug administration
Do not administer together with high-protein diets
Photosensitive drug | [10] |
| N04BA02 | Levodopa
Benserazide hydrochloride
(100 + 25 mg) | MADOPAR
(ROCHE) | Modified
release capsules | No | Alternative: dispersible tablets | [8,9] |
Levodopa
Benserazide hydrochloride
(100 + 25 mg) | MADOPAR
(ROCHE) | Capsules |
Levodopa
Benserazide hydrochloride
(100 + 25 mg) | MADOPAR
(ROCHE) | Tablets |
| N04BA02 | Levodopa
Benserazide hydrochloride
(100 + 25 mg) | MADOPAR
(ROCHE) | Dispersible
tablets | Yes | Disperse the tablet in 25–50 mL water until a fine opalescent suspension is obtained
Stop EN 1 h before drug administration | SmPC |
| N04BA02 | Levodopa
Carbidopa
(200 + 50 mg) | LEVOD/CARB HEXAL
(SANDOZ) | Modified
release tablets | No | Alternative: dispersible tablets | [8,9] |
| N04BA02 | Levodopa
Benserazide hydrochloride
(200 + 50 mg) | LEVODOPA/BENSERAZIDE TEV
(TEVA) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately
Stop EN 2 h before and 2 h after drug administration
Do not administer together with high-protein diets Splittable tablet | [10] |
| N04BA03 | Levodopa
Carbidopa
Entecapone
(all dosages) | STALEVO
(NOVARTIS) | Coated tablets | No | | [8,9] |
| N04BA05 | Melevodopa hydrochloride
Carbidopa hydrate
(12.5 + 125/25 + 100 mg) | SIRIO
(CHIESI FARMACEUTICI) | Effervescent
tablets | Yes | Dissolve in 150 mL water
and wait until the end of the effervescence | |
| N04BB01 | Amantadine hydrochloride
(100 mg) | MANTADAN
(BOEHRINGER INGELHEIM) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately | [10] |
| N04BC04 | Ropirinole hydrochloride
(4, 8 mg) | REQUIP
(GLAXOSMITHKLINE) | Modified
release tablets | No | Alternative: Requip tablets | [8,9] |
| N04BC04 | Ropirinole hydrochloride
(0.25, 0.5, 1, 2 mg) | REQUIP
(GLAXOSMITHKLINE) | Coated tablets | Yes | Dissolve quickly the tablet in 10 mL water to obtain a fine dispersion to be administered immediately | [10] |
| N04BC04 | Ropirinole hydrochloride
(4 mg) | ROPINIROLO SAN
(SANDOZ) | Modified
release tablets | No | Alternative: Requip tablets | [8,9] |
| N04BC05 | Pramipexole dihydrochloride monohydrate
(0.26, 0.52, 1.05, 2.1, 3.15 mg) | MIRAPEXIN
(BOEHRINGER INGELHEIM) | Modified
release tablets | No | Alternative: Pramipexol tablets | [8,9] |
| N04BC05 | Pramipexole dihydrochloride monohydrate
(0.7 mg) | PRAMIPEXOLO EG
(EG) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately
Photosensitive drug | |
Pramipexole dihydrochloride monohydrate
(0.18, 0.7 mg) | PRAMIPEXOLO
(TEVA) |
| N04BC06 | Cabergoline
(2 mg) | CABASER
(PFIZER) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately | [10] |
| N04BD01 | Selegiline hydrochloride
(5, 10 mg) | JUMEX
(CHIESI FARMACEUTICI) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately
Administer in the morning before starting EN | |
| N04BD02 | Rasagiline mesylate
(1 mg) | AZILECT
(TEVA) | Tablets | Yes | | |
| N04BX01 | Tolcapone
(100 mg) | TASMAR
(MEDA PHARMA) | Coated tablets | Yes | | |
| N04BX02 | Entacapone
(200 mg) | COMTAN
(NOVARTIS) | Coated tablets | Yes | Grind the tablet and dissolve the powder in 20 mL water, then administer immediately | [10] |
| N05AA01 | Chlorpromazine hydrochloride
(100 mg) | PROZIN
(IST.LUSOFARMACO) | Coated tablets | No | Alternatives: drops or oral solution | [8,9] |
| N05AA02 | Levomepromazine maleate
(25, 100 mg) | NOZINAN
(SANOFI) | Coated tablets | No | | |
| N05AB03 | Perfenazine
(2, 4, 8 mg) | TRILAFON
(NEOPHARMED GENTILI) | Coated tablets | Yes | | |
| N05AB06 | Trifluoperazine dihydrochloride
(1 mg) | MODALINA
(IST LABORATORIO FARMACEUTICO) | Coated tablets | No | | |
| N05AD01 | Haloperidol
(1, 5 mg) | HALDOL
(JANSSEN CILAG) | Tablets | Yes | Better to use drops diluted in 50 mL water and administered 1 h before or 2 h after EN | |
| N05AF05 | Zuclopenthixol dihydrochloride
(10 mg) | CLOPIXOL
(LUNDBECK) | Coated tablets | Yes | Immerse the tablet in water, after 10 min a fine suspension is obtained, then administer immediately
Alternative: solution for injection | |
| N05AH02 | Clozapine
(25 mg) | CLOZAPINA CHS
(CHIESI FARMACEUTICI) | Coated tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately | |
Clozapine
(25, 100 mg) | LEPONEX
(NOVARTIS) | Tablets |
Clozapine
(100 mg) | CLOZAPINA
(ORION) | Tablets |
| N05AH03 | Olanzapine
(5, 10 mg) | ZYPREXA VELOTAB
(ELI LILLY) | Orodispersible
tablets | Yes | Dissolve in 30 mL water, apple juice, orange juice, or milk
Irritant drug
Use personal protective equipment | SmPC |
| N05AH03 | Olanzapine
(2.5 mg) | ZYPREXA
(ELI LILLY) | Coated tablets | Yes | Better to use orodispersible tablets
Irritant to mucous membranes and eyes | |
Olanzapine
(5, 10 mg) | OLANZAPINA SUN
(SUN PHARMACEUTICALS) | Orodispersible tablets | Yes | Dissolve the tablet in water and administer immediately | |
Olanzapine
(2.5, 5, 10 mg) | OLANZAPINA TEV
(TEVA) | Coated tablets | No | Better to use orodispersible tablets | |
| N05AH03 | Olanzapine
(5, 10 mg) | OLANZAPINA TEV
(TEVA) | Orodispersible
tablets | Yes | Dissolve the tablet in water and administer immediately | SmPC |
| N05AH04 | Quetiapine fumarate
(50, 150, 200, 300, 400 mg) | SEROQUEL
(ASTRAZENECA) | Modified
release tablets | No | Alternative: Seroquel coated tablets | [8,9] |
| N05AH04 | Quetiapine fumarate
(100, 200, 300 mg) | SEROQUEL
(ASTRAZENECA) | Coated tablets | Yes | Grind the tablet and dissolve the powder in 25 mL water, then administer immediately | |
Quetiapine fumarate
(50, 200, 300, 400 mg) | QUETIAPINA TEV
(TEVA) | No | | |
| N05AH04 | Quetiapine fumarate
(50, 200, 300, 400 mg) | QUETIAPINA TEV
(TEVA) | Modified
release tablets | No | | [8,9] |
| N05AH05 | Asenapine maleate
(5, 10 mg) | SYCREST
(LUNDBECK) | Sublingual
tablets | No | | [8,9] |
| N05AH06 | Clotiapine
(40 mg) | ENTUMIN
(LAB.JUVISE’ PHARMACEUTICALS) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately
Alternative: Entumin drops to be diluted with 50 mL water | |
| N05AL03 | Tiapride hydrochloride
(100 mg) | SEREPRILE
(SANOFI) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately
Administer at a fixed time | |
| N05AL05 | Amilsupride
(50 mg) | AMISULPRIDE EG
(EG) | Tablets | Yes | Alternative: oral solution | [10] |
Amilsupride
(50, 200, 400 mg) | AMISULPRIDE MYL
(MYLAN) | Tablets |
Amilsupride
(400 mg) | AMISULPRIDE SAN
(SANDOZ) | Coated tablets |
Amilsupride
(400 mg) | SOLIAN
(SANOFI) | Coated tablets |
| N05AL07 | Levosulpride
(50, 100 mg) | LEVOPRAID
(TEOFARMA) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately | [10] |
| N05AN01 | Lithium carbonate
(300 mg) | LITIO CARBONATO
(NOVA ARGENTIA) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately
Attention drug with a narrow therapeutic index | |
CARBOLITHIUM
(TEVA) | Capsules |
| N05AX08 | Risperidone
(1, 2 mg) | RISPERIDONE SAN
(SANDOZ) | Coated tablets | No | Alternative: liquid formulation | [8,9] |
Risperidone
(2, 3, 4 mg) | RISPERIDONE TEV
(TEVA) |
| N05AX12 | Aripripazole
(5, 10, 15 mg) | ABILIFY
(OTSUKA) | Tablets | Yes | Alternatives: orodispersible tablets or oral solution | [8,9] |
Aripripazole
(10, 15 mg) | ABILIFY
(OTSUKA) | Orodispersible tablets | | [8,9] |
| N05AX13 | Paliperidone
(3, 6, 9 mg) | INVEGA
(JANSSEN CILAG) | Modified
release tablets | No | Alternative: Xeplion modified release suspension for injection | [8,9] |
| N05BA | Delorazepam
(0.5, 1, 2 mg) | DELORAZEPAM
(AUROBINDO) | Tablets | No | Alternative: drops | [8,9] |
Delorazepam
(0.5, 1, 2 mg) | EN
(BGP PRODUCTS) |
Delorazepam
(2 mg) | DELORAZEPAM RAT (RATIOPHARM) |
Delorazepam
(0.5, 1 mg) | DELORAZEPAM WPI (ZENTIVA) |
| N05BA01 | Diazepam
(2 mg) | VALIUM
(ROCHE) | Capsules | Yes | Open the capsule, disperse the content in 10 mL water, then administer immediately | [10] |
Diazepam
(5 mg) | VATRAN
(VALEAS) | Tablets | Disperse the tablet in 10 mL water, then administer immediately |
| N05BA04 | Oxazepam
(15 mg) | SERPAX
(MEDA PHARMA) | Tablets | Yes | Grind the tablet and disperse it in 10 mL water, then administer immediately
Divisible tablet
Better to use Diazepam or Lorazepam | [10] |
| N05BA06 | Lorazepam
(1, 2.5 mg) | TAVOR
(PFIZER) | Tablets | Yes | Disperse the tablet in 10 mL water, then administer immediately
Prefer liquid or the orosoluble formulation | [10] |
| N05BA08 | Bromazepam
(1.5 mg) | COMPENDIUM
(POLIFARMA) | Capsules | No | Alternative: drops | [8,9] |
Bromazepam
(3 mg) | BROMAZEPAM RAT
(RATIOPHARM) | Tablets |
| N05BA09 | Clobazam
(10 mg) | FRISIUM
(SANOFI) | Capsules | Yes | Open the capsule, disperse the content in 10 mL water, then administer immediately | [10] |
| N05BA11 | Prazepam
(10 mg) | PRAZENE
(PFIZER) | Tablets | No | Alternative: drops that must be diluted | [8,9] |
| N05BA12 | Alprazolam
(0.25, 0.5 mg) | XANAX
(PFIZER) | Tablets | Yes | Disperse the tablet in 10 mL water, then administer immediately
Better to use the drops | [10] |
| N05BB01 | Hydroxyzine hydrochloride
(25 mg) | ATARAX
(UCB PHARMA) | Coated tablets | Yes | Prefer Atarax syrup | [10] |
| N05CD01 | Flurazepam monohydrochloride
(15, 30 mg) | VALDORM
(VALEAS) | Capsules | Yes | Open the capsule, disperse the content in 20 mL water, then administer immediately
Administer before the last EN | [10] |
| N05CD02 | Nitrazepam
(5 mg) | MOGADON
(MEDA PHARMA) | Tablets | Yes | Dissolve the tablet in a water glass and then administer immediately | SmPC |
| N05CD05 | Triazolam
(250 µg) | HALCION
(PFIZER) | Tablets | Yes | | |
| N05CD09 | Brotizolam
(0.25 mg) | LENDORMIN
(BOEHRINGER INGELHEIM) | Tablets | Yes | Divisible tablets | |
| N05CF02 | Zolpidem tartrate
(10 mg) | ZOLPIDEM RAT
(RATIOPHARM) | Coated tablets | Yes | Grind the tablet and disperse it in 10 mL water, then administer immediately
Better to administer at bedtime to have a hypnotic effect
Divisible tablets | |
| N06AA04 | Clomipramine hydrochloride
(75 mg) | ANAFRANIL
(SIGMATAU) | Modified
release tablets | No | | [8,9] |
| N06AA04 | Clomipramine hydrochloride
(10, 25 mg) | ANAFRANIL
(SIGMATAU) | Coated tablets | Yes | Grind the tablet and disperse it in 10 mL water, then administer immediately | [10] |
| N06AA09 | Amitriptyline hydrochloride
(10, 25 mg) | LAROXYL
(TEOFARMA) | Coated tablets | No | Alternative: drops that must be diluted with 50 mL water | [8,9] |
| N06AA10 | Nortriptyline hydrochloride
(10, 25 mg) | NORITREN
(LUNDBECK) | Coated tablets | Yes | | |
| N06AB03 | Fluoxetine hydrochloride
(20 mg) | FLUOXETINA EG
(EG) | Capsules | Yes | Open the capsule, disperse the content in 20 mL water, then administer immediately
Prefer liquid formulation or orosoluble tablets
The solution should not be administered in the fast due to the acidic pH | [10] |
| N06AB03 | Fluoxetine hydrochloride
(20 mg) | FLUOXETINA FDI
(FIDIA FARMACEUTICI) | Capsules | Yes | Preferably use the liquid form or the dispersible tablets | [8,9] |
| N06AB03 | Fluoxetine hydrochloride
(20 mg) | FLUOXETINA RAT (RATIOPHARM) | Soluble tablets | Yes | Dissolve the tablet in 50 mL water and administer immediately | SmPC |
| N06AB03 | Fluoxetine hydrochloride
(20 mg) | XEREDIEN
(VALEAS) | Dispersible tablets | Yes | Dissolve the tablet in water and administer immediately | [8,9] |
| N06AB04 | Citalopram hydrobromide
(20 mg) | CITALOPRAM RAT (RATIOPHARM) | Coated tablets | Yes | Grind the tablet and disperse it in 10 mL water, then administer immediately
Preferably use drops
Incompatible with PVC tubes | [10] |
CITALOPRAM HEX
(SANDOZ) | [10] |
| N06AB05 | Paroxetine hydrochloride
(20 mg) | SEREUPIN (GLAXOSMITHKLINE) | Coated tablets | Yes | Grind the tablet and disperse the powder in 10 mL water, then administer immediately
Alternatives: drops or oral suspension | [10] |
| N06AB06 | Sertraline hydrochloride
(50 mg) | SERTRALINA MYL
(MYLAN) | Coated tablets | Yes | Grind the tablet and disperse the powder in 10 mL water, then administer immediately
Rinse the tube well after administration
Preferably switch to another selective serotonin reuptake inhibitor | |
ZOLOFT
(PFIZER) |
| N06AB08 | Fluvoxamine maleate
(50 mg) | FEVARIN
(BGP PRODUCTS) | Coated tablets | No | Preferably switch to another selective serotonin reuptake inhibitor in liquid formulation | [8,9] |
MAVERAL
(BGP PRODUCTS) |
| N06AB10 | Escitalopram oxalate
(10, 20 mg) | CIPRALEX
(LUNDBECK) | Coated tablets | Yes | Alternative: Cipralex drops
Divisible tablets | [10] |
Escitalopram oxalate
(20 mg) | ENTACT
(LUNDBECK) |
| N06AX03 | Mianserin hydrochloride
(30 mg) | LANTANON
(MSD) | Coated tablets | Yes | Grind the tablet and disperse the powder in 10 mL water, then administer immediately | |
| N06AX05 | Tradozone hydrochloride
(75, 150 mg) | TRITTICO
(ANGELINI) | Modified
release tablets | No | Alternatives: coated tablets or drops | [8,9] |
| N06AX05 | Tradozone hydrochloride
(75, 150 mg) | TRITTICO
(ANGELINI) | Coated tablets | Yes | Grind the tablet and disperse the powder in 10 mL water, then administer immediately
Divisible tablets | |
| N06AX11 | Mirtazapine
(30 mg) | MIRTAZAPINA SAN
(SANDOZ) | Coated tablets | Yes | | |
| N06AX12 | Bupropion hydrochloride
(150 mg) | WELLBUTRIN (GLAXOSMITHKLINE) | Modified
release tablets | No | | [8,9] |
| N06AX16 | Venlafaxine hydrochloride
(75, 150 mg) | ZARELIS
(ITALFARMACO) | Modified
release tablets | No | Alternative: oral solution | [8,9] |
Venlafaxine hydrochloride
(37.5, 75, 150 mg) | EFEXOR
(PFIZER) | Modified
release capsules |
Venlafaxine hydrochloride
(37.5 mg) | VENLAFAX TEV
(TEVA) | Modified
release capsules |
| N06AX21 | Duloxetine hydrochloride
(30, 60 mg) | CYMBALTA
(ELI LILLY) | Modified
release capsules | No | Risk of probe occlusion | [8,9] |
Duloxetine hydrochloride
(60 mg) | XERISTAR
(QUINTILES) | Gastro-resistant capsules | No | Do not grind the granules | [8,9] |
| N06BA04 | Methylphenidate hydrochloride
(10 mg) | RITALIN
(NOVARTIS) | Tablets | Yes | | |
| N06BA04 | Methylphenidate hydrochloride
(10, 20 mg) | EQUASYM
(SHIRE) | Modified
release capsules | No | | [8,9] |
| N06BA07 | Modafinil
(100 mg) | PROVIGIL
(TEVA) | Tablets | Yes | Grind the tablet and disperse the powder in water, then administer immediately | [10] |
| N06BA09 | Atomoxetine hydrochloride
(40, 60 mg) | STRATTERA
(ELI LILLY) | Capsules | No | | |
| N06BX12 | Acetyl-L-carnitine
(500 mg) | NICETILE
(SIGMATAU) | Gastro-resistant tablets | No | | [8,9] |
| N06BX12 | Acetyl-L-carnitine
(500 mg) | NICETILE
(SIGMATAU) | Powder for oral solution | Yes | | |
| N06CA01 | Amitriptyline
Perphenazine
(10 + 4 mg) | MUTABON MITE
(NEOPHARMED GENTILI) | Coated tablets | No | | [8,9] |
| N06DA02 | Donepezil hydrochloride
(10 mg) | ARICEPT 10
(PFIZER) | Coated tablets | Yes | Grind the tablet and disperse the powder in 20 mL water, then administer within 15 min
Preferably use Donezepil orodispersible tablets | [10] |
Donepezil hydrochloride
(5, 10 mg) | DONEPEZIL ACV
(ACTAVIS) |
MEMAC
(BRACCO) |
DONEPEZIL EG
(EG) |
| N06DA02 | Donepezil hydrochloride monohydrate
(5 mg) | DONEPEZIL MYL
(MYLAN) | Orodispersible tablets | Yes | Dissolve the tablet in 10 mL water and administer immediately | [8,9] |
Donepezil hydrochloride
(5, 10 mg) | DONEPEZIL TEV
(TEVA) |
| N06DA03 | Rivastigmine hydrogen tartrate
(1.5, 3, 4.5 mg) | EXELON
(NOVARTIS) | Capsules | No | Alternatives: transdermal patches or oral solution | [8,9] |
Rivastigmine hydrogen tartrate
(1.5, 3, 4.5, 6 mg) | RIVASTIGMINA SAN
(SANDOZ) |
| N06DA04 | Galantamine hydrobromide
(8, 16, 24 mg) | REMINYL
(JANSSEN CILAG) | Modified
release capsules | No | Alternative: oral solution | [8,9] |
| N06DA04 | Galantamine hydrobromide
(8, 16, 24 mg) | REMINYL
(JANSSEN CILAG) | Coated tablets | Yes | Grind the tablet and disperse the powder in 10 mL water, then administer
Preferably use Galantamine oral solution to be diluted before administration | [10] |
| N06DX01 | Memantine hydrochloride
(10 mg) | EBIXA
(LUNDBECK) | Coated tablets | No | Alternative: Ebixa oral solution | [8,9] |
Memantine hydrochloride
(10, 20 mg) | MEMANTINA MYL
(MYLAN) |
| N07AA02 | Pyridostigmine hydrobromide
(180 mg) | MESTINON
(MEDA PHARMA) | Modified
release tablets | No | Alternative: Mestinton 60 mg | [8,9] |
| N07AA02 | Pyridostigmine hydrobromide
(60 mg) | MESTINON
(MEDA PHARMA) | Tablets | Yes | Divisible tablets | |
| N07AX01 | Pilocarpine hydrochloride
(5 mg) | SALAGEN
(MERUS LABS LUXCO II SARL) | Coated tablets | No | | |
| N07BB01 | Disulfiram
(400 mg) | ANTABUSE DISPERGETTES (AUROBINDO) | Effervescent
tablets | Yes | Dissolve the tablet in 10 mL water and administer at the end of the effervescence | |
| N07BB03 | Acamprosate calcium
(333 mg) | CAMPRAL
(BRUNO FARMACEUTICI) | Coated tablets | No | | |
| N07BC01 | Buprenorphine hydrochloride
(2, 8 mg) | BUPRENORFINA MOL (MOLTENI & C. F.LLI ALITTI) | Sublingual
tablets | No | Administer sublingually only if the patient is conscious | [8,9] |
| N07CA01 | Betahistine dihydrochloride
(8 mg) | MICROSER
(GRUNENTHAL) | Tablets | Yes | Grind the tablet and disperse the powder in 20 mL water, then administer
Preferably use drops | [10] |
| N07CA02 | Cinnarizine
(75 mg) | STUGERON FTE
(JANSSEN CILAG) | Capsules | No | Alternative: drops | [8,9] |
STUGERON FTE
(JANSSEN CILAG) | Tablets |
| N07XX02 | Riluzole
(50 mg) | RILUZOLO SAN
(SANDOZ) | Coated tablets | Yes | Grind the tablet and disperse the powder in water, then administer
Preferably change therapy | [10] |
| N07XX06 | Tetrabenazine
(25 mg) | XENAZINA
(CHIESI FARMACEUTICI) | Tablets | Yes | Divisible and crushable tablet | |
| N07XX09 | Dimethyl fumarate
(120, 240 mg) | TECFIDERA
(BIOGEN ITALIA) | Gastro-resistant capsules | No | Do not grind the granules | [8,9] |
| P01AB01 | Metronidazole
(250 mg) | VAGILEN
(ALFA WASSERMANN) | Capsules | Yes | Open the capsule, disperse the content in 20 mL water, then administer immediately 1 h before EN
Alternatives: solution for injection or vaginal ovules | |
FLAGYL
(ZAMBON) | Tablets | No | Alternatives: solution for injection or vaginal ovules | |
| P01BA01 | Chloroquine diphosphate
(250 mg) | CLOROCHINA
(BAYER) | Coated tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately preferably after EN | [10] |
| P01BA02 | Hydroxychloroquine sulfate
(200 mg) | PLAQUENIL
(SANOFI) | Coated tablets | Yes | | [10] |
| P01BF05 | Piperaquine tetraphosphate
Dihydroartemisinin
(320 + 40 mg) | EURARTESIM
(SIGMATAU) | Coated tablets | Yes | | |
| P02CA03 | Albendazole
(400 mg) | ZENTEL
(GLAXOSMITHKLINE) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer immediately | SmPC |
| P02DA01 | Niclosamide
(500 mg) | YOMESAN
(BAYER) | Chewable
tablets | Yes | Dissolve the tablet in water and administer | SmPC |
| R03DA04 | Theophylline anhydrous
(200, 300 mg) | THEO-DUR
(RECORDATI) | Modified
release tablets | No | Alternative: liquid formulation to be diluted with 20 mL water and administered 1 h after EN
Monitor blood levels and adjust dosage | [8,9] |
RESPICUR
(TAKEDA) | Modified
release capsules |
| R03DA08 | Bamifylline hydrochloride
(600 mg) | BAMIFIX
(CHIESI FARMACEUTICI) | Coated tablets | No | Alternative: suppositories but only for pediatrics | |
| R03DA11 | Doxofylline
(400 mg) | ANSIMAR
(ABC FARMACEUTICI) | Tablets | Yes | Alternatives: Ansimar syrup or solution for injection | |
| R03DC03 | Montelukast sodium
(5 mg) | SINGULAIR
(MSD) | Chewable
tablets | Yes | | |
Montelukast sodium
(10 mg) | SINGULAIR
(MSD) | Coated tablets | Disperse the tablet in 10 mL water under gentle stirring, then administer immediately | |
Montelukast sodium
(5 mg) | MONTELUKAST SAN
(SANDOZ) | Chewable tablets | | |
Montelukast sodium
(5 mg) | MONTELUKAST TEV
(TEVA) | Coated tablets | Grind the tablet and disperse the powder in water, then administer away from EN
Preferably use oral granules | |
| R05CB01 | Acetylcysteine
(600 mg) | ACETILCISTEINA RAT (RATIOPHARM) | Effervescent
tablets | Yes | Administer at the end of the effervescence | |
FLUIMUCIL
(ZAMBON) | Granules for oral solution | Mix with water then administer | |
FLUIMUCIL
(ZAMBON) | Effervescent
tablets | Administer at the end of the effervescence | |
| R06AE06 | Oxatomide
(30 mg) | TINSET
(GRUNENTHAL) | Tablets | No | Alternative: drops | [8,9] |
| R06AE07 | Cetirizine hydrochloride
(10 mg) | CERCHIO
(MEDIOLANUM FARMACEUTICI) | Tablets | Yes | Alternative: drops | |
CETIRIZINA SAN
(SANDOZ) | Coated tablets | |
| R06AX25 | Mizolastine
(10 mg) | ZOLISTAM
(SANOFI) | Modified
release tablets | No | | [8,9] |
| R06AX26 | Fexofenadine hydrochloride
(180 mg) | TELFAST
(SANOFI) | Coated tablets | No | Alternatives: Cetirizine or Loratadine | |
| R06AX27 | Desloratadine
(2.5 mg) | AERIUS
(MSD) | Orodispersible tablets | Yes | | |
| S01EC01 | Acetazolamide
(250 mg) | DIAMOX
(TEOFARMA) | Tablets | Yes | | |
| V03AC03 | Deferasirox
(125, 500 mg) | EXJADE
(NOVARTIS) | Dispersible tablets | Yes | Disperse in 200 mL water until a suspension is obtained
Take on an empty stomach before EN | SmPC |
| V03AE02 | Sevelamer hydrochloride
(800 mg) | RENAGEL
(GENZYME) | Coated tablets | No | Alternative: Renvela powder for oral suspension | [8,9] |
RENVELA
(GENZYME) |
| V03AE02 | Sevelamer carbonate
(2.4 g mg) | RENVELA
(GENZYME | Powder for oral suspension | Yes | Disperse the powder in 60 mL water and administer immediately
Rinse well the tube | |
| V03AE03 | Lanthanum(III) carbonate hydrate
(1 g) | FOZNOL
(SHIRE) | Oral powder | No | | |
Lanthanum(III) carbonate hydrate
(1 g) | FOZNOL
(SHIRE) | Chewable
tablets | Yes | | |
| V03AE04 | Calcium acetate
Magnesium carbonate
(435 + 235 mg) | OSVAREN
(VIFOR FRESENIUS) | Coated tablets | No | | |
| V03AF | Calcium mefolinate
(15 mg) | PREFOLIC
(ZAMBON) | Gastro-resistant tablets | No | | [8,9] |
| V03AF04 | Calcium levofolinate
(7.5 mg) | LEDERFOLIN
(PFIZER) | Tablets | Yes | Grind the tablet and dissolve the powder in 10 mL water, then administer
Alternative: powder for reconstitution for injection | [10] |
| V03AF04 | Calcium levofolinate
(2.5 mg) | LEDERFOLIN
(PFIZER) | Granules for oral solution | Yes | | |