1. Introduction
Hemoptysis may be a symptom of diverse respiratory conditions, and is due to lung cancer in about 20% of cases [
1]. The severity of hemoptysis can also vary, ranging from minimal blood-streaked sputum to immediate life-threatening hemorrhage. The effective management of significant hemoptysis includes several interventional procedures and bronchial artery embolization is now the first-line treatment to control bleeding [
2]. In less than 10% of patients, hemoptysis originates from the pulmonary artery [
3] and is associated with increased mortality [
4]. Hemoptysis arising from the pulmonary artery may be traumatic (Swan–Ganz catheter), inflammatory (Behcet’s disease), infectious (tuberculosis with Rasmussen aneurysm, aspergillosis, etc.) or neoplastic [
5]. Several reports have underlined the efficacy of pulmonary artery embolization in cases of infectious [
6,
7,
8,
9] or inflammatory [
10] disease, as well as in cases of iatrogenic pulmonary artery pseudoaneurysms [
11]. Data regarding the use of pulmonary artery embolization in the management of hemoptysis related to lung tumors are still very limited [
12].
The goal of the present study was to evaluate the safety and efficacy of pulmonary artery embolization in patients presenting with intractable hemoptysis related to lung tumors.
2. Materials and Methods
2.1. Patients
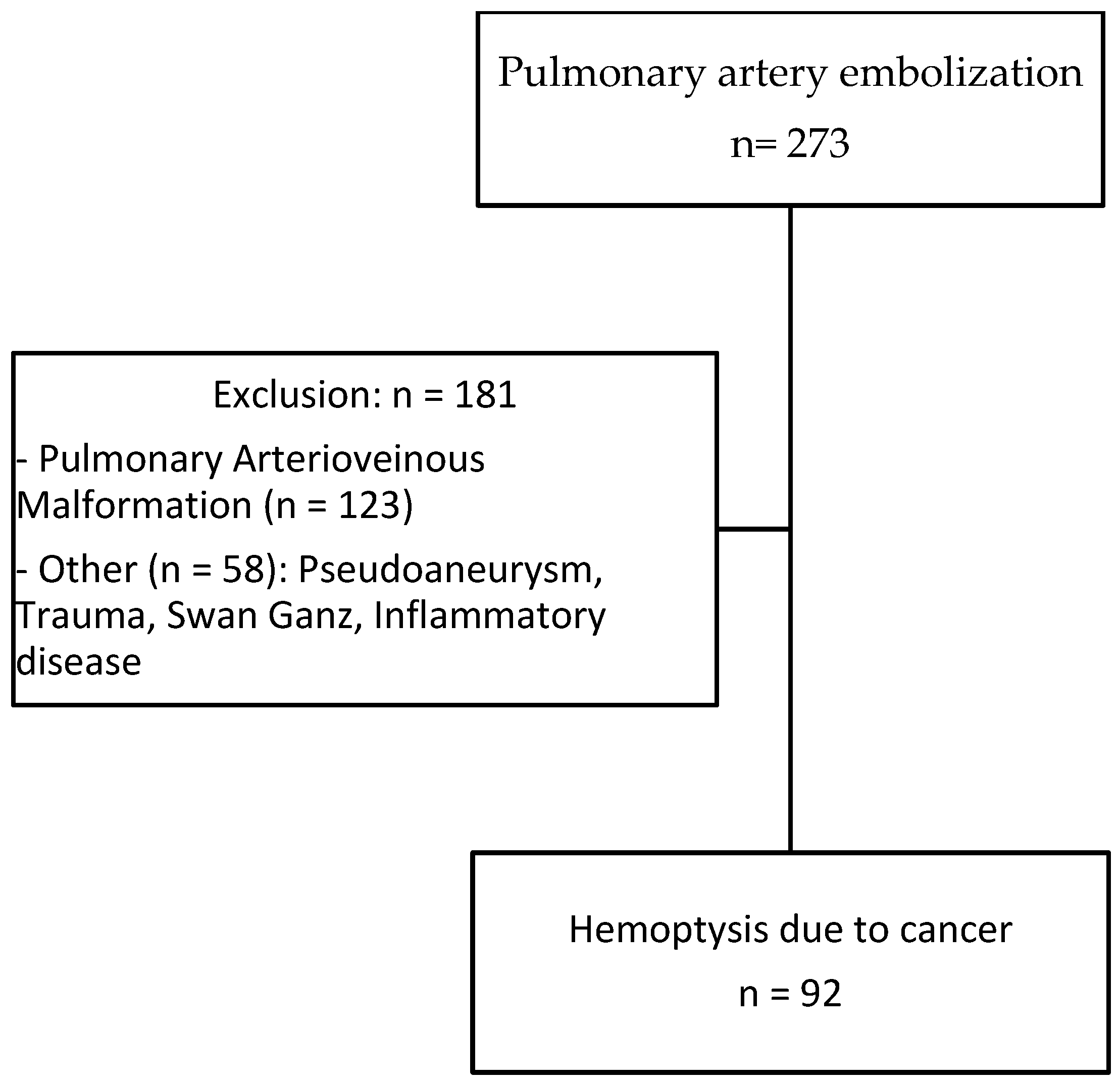
From December 2008 to December 2020, all lung-cancer-related patients presenting with severe hemoptysis treated by pulmonary artery embolization in conjunction with or after a failed bronchial artery embolization at the five participating centers were retrospectively included. The study was promoted by the Interbreizh Research Foundation. Ethical committee approval was obtained (registration number 2317). Patients treated with pulmonary embolization for other causes (traumatic or infectious pseudoaneurysms, pulmonary arterio-venous malformations, etc.) during the study period were excluded.
Each patient was categorized by age, sex, previous medical history, histologic type of cancer, disease stage, previous therapies (chemotherapy, immunotherapy and radiation therapy), hemostasis abnormality, hemoptysis volume and hemodynamic status. Hemoptysis severity was graded on the basis of the quantity of expectorated blood: <50 mL of hemoptysis was considered as minimal, 50 to 200 mL as medium, and >200 mL in 24 h as massive. All patients were examined with 1 mm collimation computed tomography after the administration of iodinated contrast material. Acquisitions were usually started 45 s after intravenous injection, to obtain maximum opacification of the pulmonary and bronchial arteries at the same time. Characteristics of the tumor (long diameter, location proximal vs. distal, presence of necrosis or cavitation) were recorded. All included patients presented with pulmonary artery lesions considered to represent hemoptysis: proximal pulmonary artery invasion, irregularity of arterial wall or vessel narrowing, pseudoaneurysm and tumoral occlusion. The presence of enlarged bronchial arteries was analyzed. The presence of ground-glass attenuation or alveolar consolidations was also recorded.
2.2. Endovascular Management
The initial approach to massive hemoptysis should always begin with airway management and hemodynamic stabilization. Anticoagulant medications should be held for the appropriate period of time and reversal agents employed if necessary. Airway isolation with bronchial blockers and endobronchial use of iced saline and vasoactive agents are among the conservative methods of hemoptysis management. Procedures were performed by 14 interventional radiologists with 2 to 15 years of experience performing embolization (MD, PhD). All interventional procedures were performed under conscious sedation or general anesthesia depending on the hemodynamic status of the patient with continuous monitoring by the intensive care and pulmonology physicians. After the percutaneous introduction of a 6 to 9 Fr vascular sheath in the femoral vein, the pulmonary artery was selectively catheterized using a guiding catheter and different shapes of 5 Fr catheters. In cases of distal vascular involvement, a 2.4–2.8 Fr microcatheter was used to superselectively catheterize the segmental artery causing bleeding. The choice of embolic agents was left at the operators’ discretion according to the location of the anomalies and operator’s habits. The following embolization parameters were recorded: level of occlusion (distal/segmental, lobar or proximal), type of embolization agents, fluoroscopy time and total radiation dose. Particles, gelatin sponge pledgets, acrylic glue, metallic coils, vascular plugs or stent grafts were selected by the interventional radiologist. The major difference between these types of materials is the end result. The stent grafts correct arteries’ abnormalities while preserving their flow. However, stents can only be used for proximal arterial anomalies due to their caliber and for fairly linear arteries because they are more rigid and find it more difficult to take curves. The other different types of materials allow embolization of the target artery. For patients undergoing bronchial embolization, after percutaneous introduction of a 5 Fr sheath in the femoral artery, selective catheterization of the different bronchial arteries was performed with 4 or 5 Fr catheters of different shapes. Target arteries were then superselectively catheterized with a 2.4–2.8 Fr microcatheter. The embolic agent used was microsphere-sized, between 400 to 900 μm, from different vendors. After embolization, all patients were admitted to the intensive care unit.
2.3. Analysis of the Outcome
Technical success was defined as the ability to perform super selective catheterization and embolization of target pulmonary artery branches. Clinical success was defined as cessation of bleeding after embolization. Procedure-associated recurrence of hemoptysis and deaths were evaluated as the main outcomes in two separate analyses. Complications were classified according to the Society of Interventional Radiology grading system (minor vs. major, graded from A to F) [
13]. Patient survival for both outcomes was assessed at 30 days, 6 months and at the end of the study, using the Kaplan–Meier estimator.
p values less than 0.05 were considered statistically significant. Follow-up was defined as the time elapsed between inclusion and December 2020 or the loss to follow-up date.
The Cox proportional hazards model for left-truncated and right-censored data was used in the modeling of the time to the recurrence of hemoptysis and death, in both univariate and multivariate analyses. Potential confounding variables, chosen for their clinical relevance, are indicated in the footnotes of the tables. Analyses were performed using R, version 4.1.1 (R Foundation for Statistical Computing, Vienna, Austria), and RStudio, version 1.4.1717 (Integrated Development Environment for R. RStudio, PBC, Boston, MA, USA).
4. Discussion
Life-threatening or recurrent hemoptysis remains an important clinical concern for chest physicians. CT angiography permits the noninvasive, rapid and accurate assessment of the cause and guides subsequent management [
14]. In the large majority of cases, the source of hemorrhage is of systemic origin, with bronchial artery embolization used as a first-line effective treatment. Control of hemoptysis can be achieved in 65 to 92% of cases depending on the cause [
15]. The main causes of treatment failure are technical challenges and bleeding originating from the pulmonary artery. However, lesions of pulmonary artery branches are not routinely described in radiological reports. Indeed, one study showed that only 46% of pulmonary arteries abnormalities were identified on the initial CT studies [
16]. Patients presenting with large tumors and associated pulmonary artery pseudoaneurysms may benefit from an initial pulmonary artery embolization.
It has been estimated that up to 30% of patients with lung cancer will present hemoptysis and, of these, 10% will experience massive hemoptysis [
17]. Hemoptysis can also reveal cancer [
1]. In patients with tumor-related hemoptysis, overall survival remains low and recurrence is reported in 20 to 30% of cases [
17,
18]. There is no difference in terms of survival between patients who had previously undergone radiation therapy or not [
19].
There are only few studies reporting the use of pulmonary artery embolization and there is no consensus opinion on indications and technique. In previous reports, some authors recommended pulmonary artery embolization only after the failure or recurrence of bronchial artery embolization [
2], whereas others recommended pulmonary artery embolization when an abnormality was detected on pulmonary artery at CT [
3,
9]. Marcelin et al. reported that 7 out of 12 patients treated with pulmonary artery embolization first underwent bronchial artery embolization. They have not reported any case of the simultaneous embolization of both circulations [
12]. However, our data suggest that pulmonary and bronchial embolization performed at the same time may be associated with increased survival. Simultaneous catheterization of both circulations was performed in a few patients when the operator judged that there were pulmonary and bronchial arterial abnormalities. Indeed, hilar tumor lesions can cause pulmonary arterial anomalies but are also associated with systemic tumor hypervascularization. In these cases, it seems interesting to perform simultaneous embolization. This situation is not very common in clinical practice. The difficulty lies in carrying out a double arterial and venous femoral approach for this simultaneous treatment, which extends the procedure time. In cases of severe hemoptysis, the most significant abnormality must be treated first.
In addition, for proximal involvement, there is no consensus opinion regarding whether the pathological branches should be occluded with glue or coils, or if the parent pulmonary artery should be spared using a stent graft. Previous reports recommended stent graft placement for proximal pulmonary abnormalities to maintain vessel patency and minimize lung functional loss [
12,
15]. In our practice, when feasible, ventilation–perfusion lung scintigraphy is carried out prior to proximal embolization to estimate the respective contribution of each lung to the overall function. Nuclear medicine studies are difficult to obtain on an emergency basis and are often not feasible in patients with unstable hemodynamic condition. A higher recurrence and mortality were found in patients embolized with stent grafts. Treatment of proximal lesions may explain these results.
Despite a high technical success rate and initial clinical efficacy associated with embolization, the overall prognosis remains poor. Not surprisingly, in patients with stage III or IV large lung tumors, the 1-year survival rate was 19% in our study. It is well known that pulmonary artery involvement in lung cancer is associated with increased mortality in patients with hemoptysis [
4]. The presence of a large or excavated lesion or a pseudoaneurysm seems to be associated with a poor prognosis. In our series, compared to the experience of Marcelin et al. [
12], the survival rate was lower (i.e., 38% versus 67% at 3 months) and the recurrence rate was also much higher (i.e., 49% versus 0%). The differences we found may be due to the small number of highly selected patients (19 patients) in the former study, which may have led to an underestimation of overall mortality and recurrence. In addition, there were little descriptive data about the tumor in their study, especially regarding the size of the tumor. Furthermore, the higher number of treatments using stent graft for proximal lesions in our series, 32% versus 16% for Marcelin [
12], may have played an important role. The main treatment failures are due to catheterization failures or ruptured arteries during the procedure. Deaths due to massive hemoptysis during the procedure have been reported in our series and also in several studies [
12,
20]. The rupture of the pulmonary artery might have been caused by increased pressure during superselective injection and also by the repeated friction of the catheter tip on the fragile pulmonary artery. These pathological arteries are very fragile, which requires great caution during treatment. Stent treatment requires greater operator experience to properly calibrate it. Complications associated with pulmonary artery embolization are mainly infectious complications, as reported in 16% of cases in our study. No complication was noted by Marcelin et al. [
12].
Our study has some limitations. It was designed as retrospective, one-sample cohort with no control group. The different types of interventions, small population size, large number of years and interventional radiologists for these procedures could be considered other limitations.