New Litter Trap Devices Outperform Pitfall Traps for Studying Arthropod Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Description of the Traps
2.1.1. Pitfall Traps
2.1.2. Cul-de-sac Traps
2.1.3. Basket Traps
2.1.4. Comparison among Pitfall, Cul-de-sac, and Basket Traps
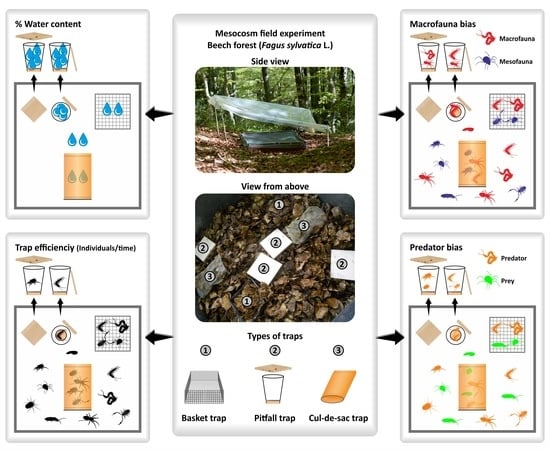
2.2. Experimental Design
2.2.1. Study Sites
2.2.2. General Description of the Experiment
2.2.3. Predrying Period
2.2.4. Trapping Establishment and Plot Watering
2.3. Trap Collection and Counting of Fauna
2.4. Assessment of Fauna Abundances Outside the Traps
2.5. Assessment of Trap Bias
2.6. Statistical Analysis
3. Results
3.1. Water Content
3.2. Mesofauna Activity
3.2.1. Collembola Activity
3.2.2. Mite Activity
3.3. Macrofauna Activity
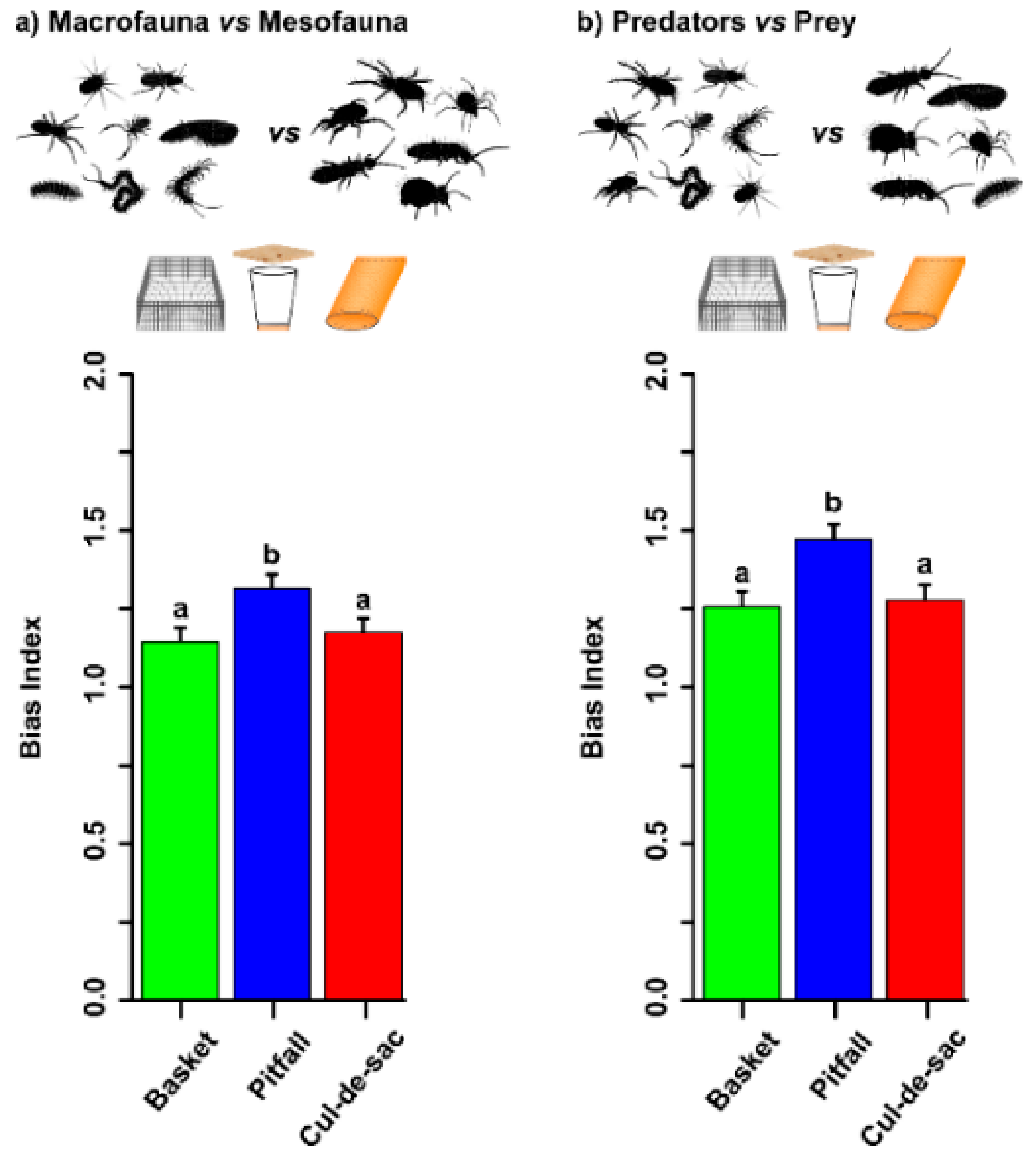
3.4. Trap Bias Index
4. Discussion
- (a)
- Collect litter from the habitat to be studied and carefully defaunate it in the laboratory. Using LED bulbs for longer periods (e.g., 7–14 days) instead of incandescent bulbs will minimize the death of animals in the litter, which could have some unwanted effects in terms of attracting scavengers and fungivores feeding on the fungi growing on the carcasses. In our case, we did check for large animals in the litter visually, and since the bias of a few inadvertently dead animals in the litter was the same for all trap types, we do not believe that it affected our results.
- (b)
- Check the weather forecasts around the study area and get ready to set up the traps in the field right after the first sufficiently abundant rainfall event (e.g., of >10 mm). Set the traps for the experiment before rainfall starts or before too much rain has fallen. This will ensure that the litter in the traps gets as wet as the litter in the habitat. Alternatively, set the trap when it has not been raining for a long time (i.e., when the litter in the field is as dry as the one that will be brought from the laboratory). However, in dry conditions, it is much less likely to be able to collect high amounts of fauna, and it may be necessary to wait until the next rainfall to collect the traps. If the intention is to include the traps in a mesocosm experiment, they must set in the inner parts of the plot and not at the edges. In our case, we set a pair of pitfall traps a few centimeters from the edge of the plot to minimize the bias due to catches from animals that were trying to leave the plot. A second pair of pitfall traps was set in the center of the plot to further dilute potential edge effects. Unfortunately, in our case, we pooled the data from edge and center traps without previously testing for edge effects. If traps of any type are to be included at the edges, it is important to test for differences between edge and inner traps before pooling the data.
- (c)
- Check the traps 2–3 days after rainfall ceases (or a different number of days, which may depend on the particular system). Collect the traps and extract the fauna in the laboratory. Be aware that basket traps need to be collected fast and with care. Always try to prevent animals from escaping down into the litter or out from the sides. Place the trap in a bag or closed container as soon as it is removed from the field.
- (d)
- The weather conditions on the day and timing of trap collection and differences among ecosystems can determine what one catches in the traps. Thus, for each ecosystem type, select a time of the day in which the edaphic fauna is active in the relevant the weather conditions. For instance, in the peak of summer in relatively dry ecosystems, most epigeic fauna are only active at night [40]. In addition, collect enough amounts of litter from the microhabitat’s surrounding area (or inside the mesocosm) to estimate the abundances of all organisms under study.
- (e)
- Run a GLMM or GLM to test for activity (animals caught by unit of time), and include the logarithm of the abundance or density of the group of interest as a covariate to control for animal availability in the litter to ensure that only activity is being tested for (not abundance). In our case, density was significant only in the case of macrofauna. If density is not significant, it means that activity is decoupled from the availability of individuals.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Accessibility Statement
References
- Lavelle, P. Faunal activities and soil processes: Adaptive strategies that determine ecosystem function. Adv. Ecol. Res. 1997, 27, 93–132. [Google Scholar]
- Lavelle, P.; Decaëns, T.; Aubert, M.; Barot, S.; Blouin, M.; Bureau, F.; Margerie, P.; Mora, P.; Rossi, J.P. Soil invertebrates and ecosystem services. Eur. J. Soil Biol. 2006, 42, S3–S5. [Google Scholar] [CrossRef]
- Briones, M.J.I. The serendipitous value of soil fauna in ecosystem functioning: The unexplained explained. Front. Environ. Sci. 2018, 6, 149. [Google Scholar] [CrossRef]
- Lang, A.; Filser, J.; Henschel, J.R. Predation by ground beetles and wolf spiders on herbivorous insects in maize crop. Agric. Ecosyst. Environ. 1999, 72, 189–199. [Google Scholar] [CrossRef]
- Snyder, W.E.; Wise, D.H. Predator interference and the establishment of generalist predator populations for biocontrol. Biol. Control 1999, 15, 283–292. [Google Scholar] [CrossRef]
- McNabb, D.M.; Halaj, J.; Wise, D.H. Inferring trophic positions of generalist predators and their linkage to the detrital food web in agroecosystems: A stable isotope analysis. Pedobiologia 2001, 45, 289–297. [Google Scholar] [CrossRef]
- Thiele-Bruhn, S.; Bloem, J.; de Vries, F.T.; Kalbitz, K.; Wagg, C. Linking soil biodiversity and agricultural soil management. Curr. Opin. Environ. Sustain. 2012, 4, 523–528. [Google Scholar] [CrossRef]
- Birkhofer, K.; Fließbach, A.; Wise, D.H.; Scheu, S. Arthropod food webs in organic and conventional wheat farming systems of an agricultural long-term experiment: A stable isotope approach. Agric. For. Entomol. 2011, 13, 197–204. [Google Scholar] [CrossRef]
- Moya-Laraño, J.; Wise, D.H. Direct and indirect effects of ants on a forest-floor food web. Ecology 2007, 88, 1454–1465. [Google Scholar] [CrossRef]
- Krey, K.L.; Blubaugh, C.K.; Chapman, E.G.; Lynch, C.A.; Snyder, G.B.; Jensen, A.S.; Fu, A.; Prischmann-Voldseth, D.A.; Harwood, J.D.; Snyder, W.E. Generalist predators consume spider mites despite the presence of alternative prey. Biol. Control 2017, 115, 157–164. [Google Scholar] [CrossRef]
- Ibánez, J.J.; Krasilnikow, P.V.; Saldaña, A. Archive and refugia of soil organisms: Applying a pedodiversity framework for the conservation of biological and non-biological heritages. J. Appl. Ecol. 2012, 49, 1267–1277. [Google Scholar] [CrossRef]
- Bardgett, R.D.; van der Putten, W.H. Belowground biodiversity and ecosystem functioning. Nature 2014, 515, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Dent, D.R.; Walton, M.P. Methods in Ecological and Agricultural Entomology; CABI Publishing: Wallingford, Oxon, UK, 1997. [Google Scholar]
- Woodcock, B.A. Pitfall trapping in ecological studies. In Insect Sampling in Forest Ecosystems; Leather, S.R., Ed.; Blackwell Science Ltd.: Oxford, UK, 2005; pp. 37–57. [Google Scholar]
- Sabu, T.K.; Shiju, R.T. Efficacy of pitfall trapping, Winkler and Berlese extraction methods for measuring ground-dwelling arthropods in moist-deciduous forests in the Western Ghats. J. Insect Sci. 2010, 10. [Google Scholar] [CrossRef] [PubMed]
- Thiele, H.U. Carabid Beetles in Their Environment: A Study on Habitat Selection by Adaptation in Physiology and Behavior; Springer-Verlag: New York, NY, USA, 1977. [Google Scholar]
- Frank, J.H. Staphylinidae. In An Introduction to Immature Insects of North America; Stehr, F.W., Ed.; Kendall-Hunt: Dubuque, IA, USA, 1991; pp. 341–352. [Google Scholar]
- Battirola, L.D.; Marques, M.I.; Brescovit, A.D.; Neto, G.H.R.; dos Anjos, K.C. Comunidade edáfica de Araneae (Arthropoda, Arachnida) em uma floresta sazonalmente inundável na região Norte do Pantanal de Mato Grosso, Brasil. Biota Neotrop. 2010, 10, 173–183. [Google Scholar] [CrossRef]
- Lang, A. The pitfalls of pitfalls: A comparison of pitfall trap catches and absolute density estimates of epigeal invertebrate predators in Arable Land. J. Pest Sci. 2000, 73, 99–106. [Google Scholar]
- Wagner, J.D.; Toft, S.; Wise, D.H. Spatial stratification in litter depth by forest-floor spiders. J. Arachnol. 2003, 31, 28–39. [Google Scholar] [CrossRef]
- Curtis, D. Pitfalls in spider community studies (Arachnida, Araneae). J. Arachnol. 1980, 8, 271–280. [Google Scholar]
- Maelfait, J.P.; Baert, L. Contribution to the knowledge of the arachno- and entomofauna of different woodhabitats. Part I. Sampled habitats, theoretical study of the pitfall method, survey of the captured taxa. Biol. Jaarb. Dodonaea 1975, 43, 175–196. [Google Scholar]
- Heydemann, B. Die Biotopstruktur als Raumwiderstand und Raumfulle für die Tierwelt. Verh. Dtsch. Zool. Ges. 1957, 47, 332–347. [Google Scholar]
- Tretzel, E. Reife- und Fortpflanzungszeit bei Spinnen. Z. Morphol. Ökologie Tiere 1954, 42, 634–691. [Google Scholar] [CrossRef]
- Topping, C.J.; Sunderland, K.D. Limitations to the use of pitfall traps in ecological studies exemplified by a study of spiders in a field of winter wheat. J. Appl. Ecol. 1992, 29, 485–491. [Google Scholar] [CrossRef]
- Shultz, B.J.; Lensing, J.R.; Wise, D.H. Effects of altered precipitation and wolf spiders on the density and activity of forest-floor Collembola. Pedobiologia 2006, 50, 43–50. [Google Scholar] [CrossRef]
- De Mas, E.; Riebera, C.; Moya-Laraño, J. Resurrecting the differential mortality model of sexual size dimorphism. J. Evol. Biol. 2009, 22, 1739–1749. [Google Scholar] [CrossRef] [PubMed]
- Melbourne, B.A. Bias in the effect of habitat structure on pitfall traps: An experimental evaluation. Aust. J. Ecol. 1999, 24, 228–239. [Google Scholar] [CrossRef] [Green Version]
- Buddle, C.M.; Hammond, H.E.J. Comparison of ground beetles (Coleoptera: Carabidae) and spiders (Araneae) collected in pan and pitfall traps. Can. Entomol. 2003, 135, 609–611. [Google Scholar] [CrossRef]
- Greenslade, P.; Greenslade, P.J.M. The use of baits and preservatives in pitfall traps. Aust. J. Entomol. 1971, 10, 253–260. [Google Scholar] [CrossRef]
- Skvarla, M.J.; Larson, J.L.; Dowling, P.G. Pitfalls and preservatives: A review. J. Entomol. Soc. Ont. 2014, 145, 15–43. [Google Scholar]
- Ninyerola, M.; Pons, X.; Roure, J.M. Atlas Climático Digital de la Península Ibérica. Metodología y Aplicaciones en Bioclimatología y Geobotánica, 1st ed.; Anman Gràfiques del Vallès, S.L.: Barcelona, Spain, 2005. [Google Scholar]
- Armas, C.; Ordiales, R.; Pugnaire, F.I. Measuring plant interaction: A new comparative index. Ecology 2004, 85, 2682–2686. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.M.; Walker, S.C. Fitting Linear Mixed-Effects Models using lme4. J. Stat. Softw. 2015, arXiv:1406.582365. [Google Scholar]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. LmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 82. [Google Scholar] [CrossRef]
- Zuur, A.F.; Ieno, E.N.; Walker, N.J.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R, 1st ed.; Springer-Verlag: New York, NY, USA, 2009. [Google Scholar]
- Moya-Laraño, J. Can temperature and water availability contribute to the maintenance of latitudinal diversity by increasing the rate of biotic interactions? Open Ecol. J. 2010, 3, 1–13. [Google Scholar] [CrossRef]
- Lima, S.T.; Dill, L.M. Behavioral decisions made under the risk of predation: A review and prospectus. Can. J. Zool. 1990, 68, 619–640. [Google Scholar] [CrossRef]
- Whitford, W.G.; de Soyza, A.G.; van Zee, J.W.; Herrick, J.E.; Havstad, K.M. Vegetation, soil, and animal indicators of rangeland health. Environ. Monit. Assess. 1998, 51, 179–200. [Google Scholar] [CrossRef]
- Cloudsley-Thompson, J.L. Biotic Interaction in Arid Lands (Adaptations of Desert Organisms), 1st ed.; Springer: Berlin, Germany, 1996; p. 208. [Google Scholar]




© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz-Lupión, D.; Pascual, J.; Melguizo-Ruiz, N.; Verdeny-Vilalta, O.; Moya-Laraño, J. New Litter Trap Devices Outperform Pitfall Traps for Studying Arthropod Activity. Insects 2019, 10, 147. https://doi.org/10.3390/insects10050147
Ruiz-Lupión D, Pascual J, Melguizo-Ruiz N, Verdeny-Vilalta O, Moya-Laraño J. New Litter Trap Devices Outperform Pitfall Traps for Studying Arthropod Activity. Insects. 2019; 10(5):147. https://doi.org/10.3390/insects10050147
Chicago/Turabian StyleRuiz-Lupión, Dolores, Jordi Pascual, Nereida Melguizo-Ruiz, Oriol Verdeny-Vilalta, and Jordi Moya-Laraño. 2019. "New Litter Trap Devices Outperform Pitfall Traps for Studying Arthropod Activity" Insects 10, no. 5: 147. https://doi.org/10.3390/insects10050147