Mountains as Islands: Species Delimitation and Evolutionary History of the Ant-Loving Beetle Genus Panabachia (Coleoptera, Staphylinidae) from the Northern Andes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field Collection
2.2. DNA Extraction, Amplification and Sequencing
2.3. Phylogenetic Analyses
2.4. Species Delimitation Analyses
3. Results
3.1. Sequence Data and Polymorphisms
3.2. Species Delimitation Analyses
3.3. Phylogenetic Analyses and Divergence Time Estimates
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gregory-Wodzicki, K.M. Uplift history of the Central and Northern Andes: A review. Geol. Soc. Am. Bull. 2000, 112, 1091–1105. [Google Scholar] [CrossRef]
- Veblen, T.T.; Young, K.R.; Orme, A. The Physical Geography of South America; Oxford University Press: New York, NY, USA, 2007; pp. 1–60. [Google Scholar]
- Hoorn, C.T.; Wesselingh, F.P.; Ter Steege, H.; Bermudez, M.A.; Mora, A.; Sevink, J.; Sanmartín, I.; Sanchez-Meseguer, A.; Anderson, C.L.; Figueiredo, J.P.; et al. Amazonia Through Time: Andean Uplift, Climate Change, Landscape Evolution, and Biodiversity. Science 2010, 330, 927–931. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antonelli, A. Multiple origins of mountain life. Nature 2015, 524, 300–301. [Google Scholar] [CrossRef] [PubMed]
- Flantua, S.G.A.; O’Dea, A.; Onstein, R.E.; Giraldo, C.; Hooghiemstra, H. The flickering connectivity system of the north Andean páramos. J. Biogeogr. 2019, 46, 1808–1825. [Google Scholar] [CrossRef]
- Orme, A.R. The tectonic framework of South America. In The Physical Geography of South America; Veblen, T.T., Young, K.R., Orme, A.R., Eds.; Oxford University Press: New York, NY, USA, 2007; pp. 3–22. [Google Scholar]
- Schubert, C.; Clapperton, C.M. Quaternary Glaciations in the northern Andes (Venezuela, Colombia and Ecuador). Quat. Sci. Rev. 1990, 9, 123–135. [Google Scholar] [CrossRef]
- Ehlers, J.; Gibbard, P.L.; Hughes, P.D. Quaternary Glaciations–Extent and Chronology: A Closer Look; Elsevier: Oxford, UK, 2011; Volume 15, pp. 151–163. [Google Scholar]
- Madriñán, S.; Cortés, A.J.; Richardson, J.E. Páramo is the world’s fastest evolving and coolest biodiversity hotspot. Front. Genet. 2013, 4, 192. [Google Scholar] [CrossRef] [Green Version]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; da Fonseca, G.A.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef]
- Villota, A.; Behling, H. Late Glacial and Holocene environmental change inferred from the paramo of Cajanuma in the Podocarpus National Park, Southern Ecuador. Caldasia 2014, 36, 345–364. [Google Scholar] [CrossRef] [Green Version]
- Hazzi, N.A.; Moreno, J.S.; Ortiz-Movliav, C.; Palacio, R.D. Biogeographic regions and events of isolation and diversification of the endemic biota of the tropical Andes. Proc. Natl. Acad. Sci. USA 2018, 115, 7985–7990. [Google Scholar] [CrossRef] [Green Version]
- Gómez-Gutiérrez, M.C.; Pennington, R.T.; Neavez, L.E.; Milne, R.I.; Madriñán, S.; Richardson, J.E. Genetic diversity in the Andes: Variation within and between the South American species of Oreobolus. Alp. Bot. 2017, 127, 155–170. [Google Scholar] [CrossRef]
- Mac Vean, C.; Schuster, J.C. Altitudinal Distribution of Passalid Beetles (Coleoptera, Passalidae) and Pleistocene Dispersal on the Volcanic Chain of Northern Central America. Biotropica 1981, 13, 29–38. [Google Scholar] [CrossRef]
- Muñoz-Mendoza, C.; D’Elia, G.; Panzera, A.; Villalobos-Leiva, A.; Sites, J.W., Jr.; Victoriano, P.F. Geography and past climate changes have shaped the evolution of a widespread lizard from the Chilean hotspot. Mol. Phylogenet. Evol. 2017, 116, 157–171. [Google Scholar] [CrossRef] [PubMed]
- Hooghiemstra, H.; Flantua, S.G.A. Colombia in the Quaternary: An overview of environmental and climatic change. In The Geology of Colombia; Gómez-Tapias, J., Pinilla-Pachon, A.O., Eds.; Servicio Geológico Colombiano, Publicaciones Geológicas Especiales: Bogota, Colombia, 2019; Volume 4, pp. 33–57. [Google Scholar]
- Anthelme, F.; Jacobsen, D.; Macek, P.; Meneses, R.I.; Moret, P.; Beck, S.; Dangles, O. Biodiversity Patterns and Continental Insularity in the Tropical High Andes Biodiversity patterns and continental insularity in the tropical High Andes. Arct. Antarct. Alp. Res. 2014, 46, 811–828. [Google Scholar] [CrossRef] [Green Version]
- Neil, D.A. Geología. In Catálogo De Las Plantas Vasculares Del Ecuador; Jorgensen, P.M., Leon-Yanez, S., Eds.; Monographs in Systematic Botany from the Missouri Botanical Garden: St. Louis, MO, USA, 1999; pp. 1–1181. [Google Scholar]
- Hodkinson, I.D. Terrestrial insects along elevation gradients: Species and community responses to altitude. Biol. Rev. 2005, 80, 489–513. [Google Scholar] [CrossRef] [Green Version]
- Dillon, M.E. Into thin air: Physiology and evolution of alpine insects. Integr. Comp. Biol. 2006, 46, 49–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Somme, L.; Block, W. Adaptations to Alpine and Polar Environments in Insects and Other Terrestrial Arthropods. In Insects at Low Temperature; Lee, R.E., Denlinger, D.L., Eds.; Springer Science & Business Media: New York, NY, USA, 1991; pp. 318–359. [Google Scholar]
- Helgen, K.M.; Kays, R.; Helgen, L.E.; Tsuchiya-jerep, M.T.N.; Pinto, C.M.; Koepfli, K.; Eizirik, E.; Maldonado, J.E. Taxonomic boundaries and geographic distributions revealed by an integrative systematic overview of the mountain coatis, Nasuella (Carnivora: Procyonidae). Carnivore 2009, 41, 65–74. [Google Scholar]
- Guayasamin, J.M.; Bonaccorso, E.; Duellman, W.E.; Coloma, L.A. Genetic differentiation in the nearly extinct Atelopus, with emphasis on the A. ignescens and A. bomolochos. Zootaxa 2010, 68, 55–68. [Google Scholar] [CrossRef]
- Hines, H.M. Historical biogeography, divergence times, and diversification patterns of bumble bees (Hymenoptera: Apidae: Bombus). Syst. Biol. 2008, 57, 58–75. [Google Scholar] [CrossRef]
- Elias, M.; Joron, M.; Willmott, K.; Silva-BrandÃo, K.L.; Kaiser, V.; Arias, C.F.; Piñerez, L.M.G.; Uribe, S.; Brower, A.V.Z.; Freitas, A.V.L.; et al. Out of the Andes: Patterns of diversification in clearwing butterflies. Mol. Ecol. 2009, 18, 1716–1729. [Google Scholar] [CrossRef]
- Maddison, D.R. An unexpected clade of South American ground beetles (Coleoptera, Carabidae, Bembidion). Zookeys 2014, 155, 113–155. [Google Scholar] [CrossRef] [Green Version]
- Muñoz–Tobar, S.I. Weak Genetic Differentiation among Populations of the Andean Ground Beetle Pelmatellus columbianus (Reiche, 1843) (Coleoptera: Carabidae). Coleopt. Bull. 2019, 73, 411–427. [Google Scholar] [CrossRef]
- Muñoz–Tobar, S.I.; Caterino, M.S. The role of dispersal for shaping phylogeographical structure of flightless beetles from the Andes. PeerJ 2019, 7, e7226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Graham, C.H.; Carnaval, A.C.; Cadena, C.D.; Zamudio, K.R.; Roberts, T.E.; Parra, J.L.; Mccain, C.M.; Bowie, R.C.K.; Moritz, C.; Baines, S.B.; et al. The origin and maintenance of montane diversity: Integrating evolutionary and ecological processes. Ecography 2014, 37, 711–719. [Google Scholar] [CrossRef]
- Newton, A.F. Beetles (Coleoptera) of Peru: A Survey of the Families. Mycteridae Blanchard, 1845. J. Kansas Entomol. Soc. 2015, 89, 283–304. [Google Scholar]
- Thayer, M.K. Staphylinidae Latreille, 1802. In Coleoptera, Beetles. Volume 1: Morphology and Systematics (Archostemata, Adephaga, Myxophaga, Polyphaga Partim); Kristensen, P., Beutel, R.G., Eds.; Walter de Gruyter: Berlin, Germany; New York, NY, USA, 2005; pp. 296–344. [Google Scholar]
- Asenjo, A.; Irmler, U.; Klimaszewski, J.; Chandler, D.S.; Fierros-López, H.E.; Vieira, J.S. Staphylinidae (Insecta: Coleoptera) in Latin America: Synopsis, annotated catalog, diversity and distribution. Zootaxa 2019, 4621, 1–406. [Google Scholar] [CrossRef]
- Sánchez-Baracaldo, P.; Thomas, G.H. Adaptation and convergent evolution within the Jamesonia–Eriosorus complex in high–elevation biodiverse andean hotspots. PLoS ONE 2014, 9, e110618. [Google Scholar] [CrossRef]
- Hubb, N.; Lllambi, L.D.; Ramirez, L.; Callaway, R.M. Alpine cushion plants have species–specific effects on microhabitat and community structure in the tropical Andes. ARPN J. Eng. Appl. Sci. 2017, 28, 928–938. [Google Scholar]
- Park, O. A Study in Neotropical Pselaphidae; Pantagraph Press: Bloomington, IN, USA, 1942; pp. 125–153. [Google Scholar]
- Navarrete-Heredia, J.L.; Newton, A.F.; Thayer, M.K. Guia Ilustrada Para Los Géneros de Staphylinidae (Coleoptera) de Mexico; Universidad de Guadalajara; Comission Nacional para el Conocimiento y Uso de la Biodiversidad (CONABIO): Guadalajara, Mexico, 2002; pp. 53–89.
- Sharp, D. Insecta, Coleoptera Volume 1, Part 2. In Biologia Centrali-Americana; Godman, F.D., Savin, O., Eds.; Taylor & Francis: London, UK, 1887; pp. 30–31. [Google Scholar]
- Newton, A.F.; Chandler, D.S. World catalog of the genera of Pselaphidae (Coleoptera). Fieldiana Zool. New Ser. 1989, 53, 1–93. [Google Scholar]
- Simon, C.; Frati, F.; Beckenbach, A.; Crespi, B.; Liu, H.; Flook, P. Evolution, weighting, and phylogenetic utility of mitochondrial gene sequences and a compilation of conserved polymerase chain reaction primers. Ann. Entomol. Soc. Am. 1994, 87, 651–701. [Google Scholar] [CrossRef]
- Caterino, M.S.; Tishechkin, A.K. New genera and species of Neotropical Exosternini (Coleoptera, Histeridae). Zookeys 2014, 78, 11–78. [Google Scholar] [CrossRef] [Green Version]
- Wild, A.L.; Maddison, D.R. Evaluating nuclear protein–coding genes for phylogenetic utility in beetles. Mol. Phylogenet. Evol. 2008, 48, 877–891. [Google Scholar] [CrossRef] [PubMed]
- Parker, J.; Grimaldi, D.A. Specialized myrmecophily at the ecological dawn of modern ants. Curr. Biol. 2014, 24, 2428–2434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Darriba, D.; Taboada, G.L.; Doallo, R.; Posada, D. Europe PMC Funders Group jModelTest 2: More models, new heuristics and high–performance computing. Nat. Methods 2012, 9, 6–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clement, M.; Posada, D.; Crandall, K.A. TCS: A computer program to estimate gene genealogies. Mol. Ecol. 2000, 9, 1657–1660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rozas, J.; Sanchez-DelBarrio, J.C.; Messeguer, X.; Rozas, R. DnaSP, DNA polymorphism analyses by the coalescent and other methods. Bioinformatics 2003, 19, 2496–2497. [Google Scholar] [CrossRef]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post–analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef]
- Maddison, D.R.; Maddison, W.P. Zephyr: A Mesquite Package for Interacting with External Phylogeny Inference Programs, Version 3.0. Available online: http://zephyr.mesquiteproject.org (accessed on 30 May 2018).
- Huelsenbeck, J.P.; Ronquist, F. MrBayes: Bayesian inference of phylogenetic trees. Bioinformatics 2001, 17, 754–755. [Google Scholar] [CrossRef] [Green Version]
- Bouckaert, R.; Heled, J.; Kühnert, D.; Vaughan, T.; Wu, C.-H.; Xie, D.; Suchard, M.A.; Rambaut, A.; Drummond, A.J. BEAST 2: A Software Platform for Bayesian Evolutionary Analysis. PLoS Comput. Biol. 2014, 10, e1003537. [Google Scholar] [CrossRef] [Green Version]
- Chandler, D.S. Biology, Morphology, and Systematics of the Ant–Like Litter Beetle Genera of Australia (Coleoptera: Staphylinidae: Pselaphinae), 15th ed.; Memoirs on Entomology International; American Entomological Institute: Gainesville, FL, USA, 2001; pp. 1–560. [Google Scholar]
- Zhang, J.; Kapli, P.; Pavlidis, P.; Stamatakis, A. A general species delimitation method with applications to phylogenetic placements. Bioinformatics 2013, 29, 2869–2876. [Google Scholar] [CrossRef] [Green Version]
- Fujisawa, T.; Barraclough, T.G. Delimiting species using single–locus data and the generalized mixed yule coalescent approach: A revised method and evaluation on simulated data sets. Syst. Biol. 2013, 62, 707–724. [Google Scholar] [CrossRef] [Green Version]
- Jones, G. Algorithmic improvements to species delimitation and phylogeny estimation under the multispecies coalescent. J. Math. Biol. 2017, 74, 447–467. [Google Scholar] [CrossRef] [PubMed]
- Rambaut, A.; Suchard, M.A.; Xie, D.; Drummond, A.J. Tracer v1.6. Available online: http://beast.bio.ed.ac.uk/Tracer (accessed on 11 November 2016).
- Swofford, D.L. PAUP*. Phylogenetic Analysis Using Parsimony (*and Other Methods), Version 4. 2003. Available online: https://paup.phylosolutions.com/ (accessed on 2 March 2018).
- Hoorn, C.; Perrigo, A.; Antonelli, A. Mountains, Climate and Biodiversity: An Introduction. In Mountains, Climate and Biodiversity; Hoorn, C., Perrigo, A., Antonelli, A., Eds.; Wiley: Oxford, UK, 2018; pp. 1–5. [Google Scholar]
- Antonelli, A.; Kissling, W.D.; Flantua, S.G.A.; Bermúdez, M.A.; Mulch, A.; Muellner-Riehl, A.N.; Kreft, H.; Linder, H.P.; Badgley, C.; Fjeldså, J.; et al. Geological and climatic influences on mountain biodiversity. Nat. Geosci. 2018, 11, 718–725. [Google Scholar] [CrossRef]
- Fujita, M.K.; Leaché, A.D.; Burbrink, F.T.; McGuire, J.A.; Moritz, C. Coalescent–based species delimitation in an integrative taxonomy. Trends Ecol. Evol. 2012, 27, 480–488. [Google Scholar] [CrossRef]
- Sklenář, P.; Dušková, E.; Balslev, H. Tropical and Temperate: Evolutionary History of Paramo Flora. Bot. Rev. 2011, 77, 71–108. [Google Scholar] [CrossRef]
- Luteyn, J.L. Paramos: A Checklist of Plan Diversity, Geographical Distribution and Botanical Literature, 84th ed.; Memoirs of the New York Botanical Garden: New York, NY, USA, 1999; pp. 1–38. [Google Scholar]
- Merckx, V.S.F.T.; Hendriks, K.P.; Beentjes, K.K.; Mennes, C.B.; Becking, L.E.; Peijnenburg, K.T.C.A.; Afendy, A.; Arumugam, N.; de Boer, H.; Biun, A.; et al. Evolution of endemism on a young tropical mountain. Nature 2015, 524, 347–350. [Google Scholar] [CrossRef]
- Barraclough, T.G.; Nee, S. Phylogenetics and speciation. Trends Ecol. Evol. 2001, 16, 391–399. [Google Scholar] [CrossRef]
- Robin, C.; Eissen, J.P.; Samaniego, P.; Martin, H.; Hall, M.; Cotten, J. Evolution of the late Pleistocene Mojanda–Fuya Fuya volcanic complex (Ecuador), by progressive adakitic involvement in mantle magma sources. Bull. Volcanol. 2009, 71, 233–258. [Google Scholar] [CrossRef] [Green Version]
- Monsalve, M.L.; Laverde, C.A. Actividad de los volcanes Chiles y Cerro Negro (Frontera Colombo–Ecuatoriana). Bol. Geol. 2016, 38, 61–78. [Google Scholar]
- Jordan, E.; Hastenrath, S.L. Glaciers of South America: Glaciers of Ecuador. In US Geological Survey Professional Paper; Williams, R.S., Ferrigno, J.G., Eds.; United States Government Printing Office: Washington, DC, USA, 1998; pp. 131–150. [Google Scholar]
- Edwards, J.S.; Sugg, P. Arthropod Fallout as a Resource in the Recolonization of Mount St. Helens. Ecol. Soci. 1993, 74, 954–958. [Google Scholar] [CrossRef]
- Elizalde, L. Volcanism and arthropods: A review. Ecol. Austral 2014, 24, 3–16. [Google Scholar]
- Lester, S.; Ruttenberg, B.; Gaines, S.D.; Kinlan, B.P. The relationship between dispersal ability and geographic range size. Ecol. Lett. 2007, 10, 745–758. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, H.; Nishikawa, M.; Sota, T. Loss of flight promotes beetle diversification. Nat. Commun. 2012, 3, 647–648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mcculloch, G.A.; Wallis, G.P.; Waters, J.M. Does wing size shape insect biogeography? Evidence from a diverse regional stonefly assemblage. Glob. Ecol. Biogeogr. 2017, 26, 93–101. [Google Scholar] [CrossRef]
- Sklenář, P.; Hedberg, I.; Cleef, A.M. Island biogeography of tropical alpine floras. J. Biogeogr. 2014, 41, 287–297. [Google Scholar] [CrossRef]
- Moret, P. Los Coleópteros Carabidae Del Páramo En Los Andes Del Ecuador; Museo de Zoología; Centro de Biodiversidad y Ambiente; Escuela de Biología; Pontificia Universidad Católica del Ecuador: Quito, Ecuador, 2005; pp. 11–273. [Google Scholar]
- Moret, P. Altitudinal distribution, diversity and endemicity of Carabidae (Coleoptera) in the páramos of Ecuadorian Andes Pierre. Ann. Société Entomol. Fr. 2009, 45, 500–510. [Google Scholar] [CrossRef] [Green Version]
- Martinez, E.; Duque, P.; Wolff, M. Succession pattern of carrion–feeding insects in Paramo, Colombia. Forensic Sci. Int. 2007, 166, 182–189. [Google Scholar] [CrossRef]
- Cuesta, F.; Peralvo, M.; Merino-Viteri, A.; Bustamante, M.; Baquero, F.; Freile, J.F.; Muriel, P.; Torres-Carvajal, O. Priority areas for biodiversity conservation in mainland Ecuador. Neotrop. Biodivers. 2017, 3, 93–106. [Google Scholar] [CrossRef]
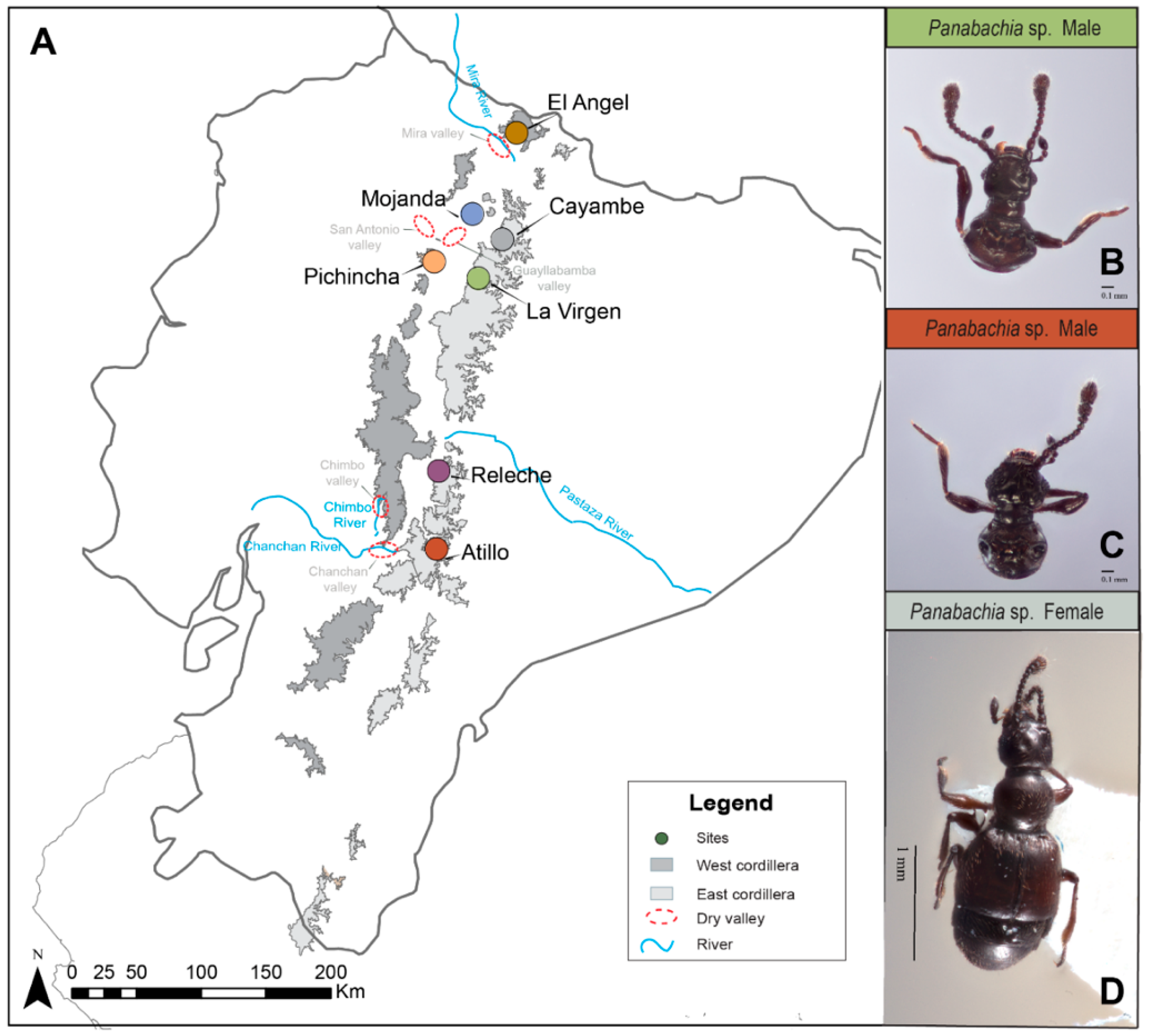






| Mountain Range | No. | Site | N | Latitude | Longitude | Elevation | Collecting Date |
|---|---|---|---|---|---|---|---|
| West | 1 | El Angel | 10 | 00°42.3521′ N | 77°57.985′ W | 3301 m | 26 July 2016 |
| 2 | Mojanda | 10 | 00°08.710′ N | 78°16.753′ W | 3715 m | 12 July 2016 | |
| 3 | Pichincha | 11 | 00°11.259′ S | 78°32.432′ W | 3897 m | 22 June 2016 | |
| East | 4 | Cayambe | 10 | 00°02.101′ S | 78°03.608′ W | 3743 m | 1 June 2016 |
| 5 | La Virgen | 10 | 00°18.477′ S | 78°13.953′ W | 3694 m | 28 June 2016 | |
| 6 | Releche | 7 | 01°38.400′ S | 78°30.426′ W | 3124 m | 8 July 2016 | |
| 7 | Atillo | 10 | 02°11.265′ S | 78°31.2601′ W | 3501 m | 7 July 2016 |
| Voucher ID | Genus | Species | Site | Haplotype | COI | Haplotype | Wingless | Reference |
|---|---|---|---|---|---|---|---|---|
| SIMT248 | Panabachia | sp. | El Angel | – | – | H8/H9 | MK674905 | This study |
| SIMT249 | Panabachia | sp. | El Angel | H15 | MN536380 | H11 | MK674907 | This study |
| SIMT288 | Panabachia | sp. | El Angel | H24 | MN536401 | H30 | MK674927 | This study |
| SIMT289 | Panabachia | sp. | El Angel | H25 | MN536402 | H31 | MK674928 | This study |
| SIMT290 | Panabachia | sp. | El Angel | H24 | MN536403 | H32/H33 | MK674929 | This study |
| SIMT291 | Panabachia | sp. | El Angel | H24 | MN536404 | H34/H35 | MK674930 | This study |
| SIMT292 | Panabachia | sp. | El Angel | H15 | MN536405 | H36 | MK674931 | This study |
| SIMT293 | Panabachia | sp. | El Angel | H15 | MN536406 | H37 | MK674932 | This study |
| SIMT294 | Panabachia | sp. | El Angel | H27 | MN536407 | H38 | MK674933 | This study |
| SIMT295 | Panabachia | sp. | El Angel | H27 | MN536408 | H39 | MK674934 | This study |
| SIMT252 | Panabachia | sp. | Mojanda | H12 | MN536371 | – | – | This study |
| SIMT297 | Panabachia | sp. | Mojanda | H13 | MN536410 | H40/H41 | MK674936 | This study |
| SIMT298 | Panabachia | sp. | Mojanda | H21 | MN536411 | H42 | MK674937 | This study |
| SIMT299 | Panabachia | sp. | Mojanda | H20 | MN536412 | H43 | MK674938 | This study |
| SIMT300 | Panabachia | sp. | Mojanda | H26 | MN536413 | H44/H45 | MK674939 | This study |
| SIMT301 | Panabachia | sp. | Mojanda | H12 | MN536414 | H46 | MK674940 | This study |
| SIMT302 | Panabachia | sp. | Mojanda | H20 | MN536415 | H47 | MK674941 | This study |
| SIMT303 | Panabachia | sp. | Mojanda | H20 | MN536416 | H48 | MK674942 | This study |
| SIMT304 | Panabachia | sp. | Mojanda | H14 | MN536417 | H40 | MK674943 | This study |
| SIMT253 | Panabachia | sp. | Mojanda | H28 | MN536382 | H13 | MK674909 | This study |
| SIMT250 | Panabachia | sp. | Pichincha | H8 | MN536381 | H12 | MK674908 | This study |
| SIMT251 | Panabachia | sp. | Pichincha | H29 | MN536370 | H2 | MK674899 | This study |
| SIMT266 | Panabachia | sp. | Pichincha | H9 | MN536375 | H6 | MK674903 | This study |
| SIMT267 | Panabachia | sp. | Pichincha | H29 | MN536376 | H7 | MK674904 | This study |
| SIMT268 | Panabachia | sp. | Pichincha | H29 | MN536377 | – | – | This study |
| SIMT269 | Panabachia | sp. | Pichincha | H29 | MN536378 | – | – | This study |
| SIMT270 | Panabachia | sp. | Pichincha | H29 | MN536384 | H15/H16 | MK674911 | This study |
| SIMT271 | Panabachia | sp. | Pichincha | H29 | MN536385 | H17 | MK674912 | This study |
| SIMT272 | Panabachia | sp. | Pichincha | H29 | MN536386 | H17 | MK674913 | This study |
| SIMT273 | Panabachia | sp. | Pichincha | H29 | MN536387 | H15/H18 | MK674914 | This study |
| SIMT296 | Panabachia | sp. | Pichincha | H7 | MN536409 | H10 | MK674935 | This study |
| SIMT254 | Panabachia | sp. | Cayambe | H6 | MN536383 | H14 | MK674910 | This study |
| SIMT255 | Panabachia | sp. | Cayambe | H5 | MN536372 | H3 | MK674900 | This study |
| SIMT278 | Panabachia | sp. | Cayambe | H4 | MN536392 | – | – | This study |
| SIMT279 | Panabachia | sp. | Cayambe | H1 | MN536393 | H10/H20 | MK674919 | This study |
| SIMT280 | Panabachia | sp. | Cayambe | H1 | MN536394 | H21/H22 | MK674920 | This study |
| SIMT281 | Panabachia | sp. | Cayambe | H2 | MN536395 | H10 | MK674921 | This study |
| SIMT282 | Panabachia | sp. | Cayambe | H3 | MN536396 | H23 | MK674922 | This study |
| SIMT309 | Panabachia | sp. | Cayambe | H1 | MN536422 | H51 | MK674947 | This study |
| SIMT310 | Panabachia | sp. | Cayambe | H1 | MN536423 | H52/H53 | MK674948 | This study |
| SIMT311 | Panabachia | sp. | Cayambe | H5 | MN536424 | H10/H54 | MK674949 | This study |
| SIMT246 | Panabachia | sp. | La Virgen | H11 | MN536369 | H1 | MK674898 | This study |
| SIMT247 | Panabachia | sp. | La Virgen | H9 | MN536379 | H10 | MK674906 | This study |
| SIMT274 | Panabachia | sp. | La Virgen | H10 | MN536388 | H11 | MK674915 | This study |
| SIMT275 | Panabachia | sp. | La Virgen | H22 | MN536389 | H14 | MK674916 | This study |
| SIMT276 | Panabachia | sp. | La Virgen | H10 | MN536390 | H19 | MK674917 | This study |
| SIMT277 | Panabachia | sp. | La Virgen | H9 | MN536391 | H19 | MK674918 | This study |
| SIMT305 | Panabachia | sp. | La Virgen | H22 | MN536418 | H49 | MK674944 | This study |
| SIMT306 | Panabachia | sp. | La Virgen | H22 | MN536419 | H50 | MK674945 | This study |
| SIMT307 | Panabachia | sp. | La Virgen | H22 | MN536420 | H50 | MK674946 | This study |
| SIMT308 | Panabachia | sp. | La Virgen | H22 | MN536421 | – | – | This study |
| SIMT256 | Panabachia | sp. | Releche | H16 | MN536373 | H4 | MK674901 | This study |
| SIMT257 | Panabachia | sp. | Releche | H16 | MN536374 | H5 | MK674902 | This study |
| SIMT283 | Panabachia | sp. | Releche | H16 | MN536397 | H24/H25 | MK674923 | This study |
| SIMT284 | Panabachia | sp. | Releche | H10 | MN536398 | H26 | MK674924 | This study |
| SIMT340 | Panabachia | sp. | Releche | H16 | MN536425 | – | – | This study |
| SIMT341 | Panabachia | sp. | Releche | H17 | MN536426 | H55/H56 | MK674950 | This study |
| SIMT342 | Panabachia | sp. | Releche | H16 | MN536427 | H4 | MK674951 | This study |
| SIMT285 | Panabachia | sp. | Atillo | H23 | MN536399 | H27 | MK674925 | This study |
| SIMT286 | Panabachia | sp. | Atillo | H23 | MN536400 | H28/H29 | MK674926 | This study |
| SIMT287 | Panabachia | sp. | Atillo | H23 | MN536434 | H65 | MK674959 | This study |
| SIMT343 | Panabachia | sp. | Atillo | H18 | MN536428 | MK674952 | This study | |
| SIMT344 | Panabachia | sp. | Atillo | H18 | MN536429 | H59 | MK674953 | This study |
| SIMT345 | Panabachia | sp. | Atillo | H30 | MN536430 | H60 | MK674954 | This study |
| SIMT346 | Panabachia | sp. | Atillo | H19 | MN536431 | H61 | MK674955 | This study |
| SIMT347 | Panabachia | sp. | Atillo | H18 | MN536432 | H61 | MK674956 | This study |
| SIMT348 | Panabachia | sp. | Atillo | – | – | H62 | MK674957 | This study |
| SIMT349 | Panabachia | sp. | Atillo | H18 | MN536433 | H63/H64 | MK674958 | This study |
| – | Bryaxis | curtisi | Outgroup | – | KM350460 | – | KM350297 | Parker and Grimaldi (2014). |
| – | Tychobythinus | sp. | Outgroup | – | KM350498 | – | KM350290 | Parker and Grimaldi (2014). |
| Gene | N | S | Ps | θ | Π | D |
|---|---|---|---|---|---|---|
| COI | 67 | 240 | 66 | 0.054 | 0.113 | 1.73 (p > 1.78) |
| wingless | 62 | 118 | 105 | 0.056 | 0.047 | −0.51 (p > 0.10) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muñoz-Tobar, S.I.; Caterino, M.S. Mountains as Islands: Species Delimitation and Evolutionary History of the Ant-Loving Beetle Genus Panabachia (Coleoptera, Staphylinidae) from the Northern Andes. Insects 2020, 11, 64. https://doi.org/10.3390/insects11010064
Muñoz-Tobar SI, Caterino MS. Mountains as Islands: Species Delimitation and Evolutionary History of the Ant-Loving Beetle Genus Panabachia (Coleoptera, Staphylinidae) from the Northern Andes. Insects. 2020; 11(1):64. https://doi.org/10.3390/insects11010064
Chicago/Turabian StyleMuñoz-Tobar, Sofía I., and Michael S. Caterino. 2020. "Mountains as Islands: Species Delimitation and Evolutionary History of the Ant-Loving Beetle Genus Panabachia (Coleoptera, Staphylinidae) from the Northern Andes" Insects 11, no. 1: 64. https://doi.org/10.3390/insects11010064
APA StyleMuñoz-Tobar, S. I., & Caterino, M. S. (2020). Mountains as Islands: Species Delimitation and Evolutionary History of the Ant-Loving Beetle Genus Panabachia (Coleoptera, Staphylinidae) from the Northern Andes. Insects, 11(1), 64. https://doi.org/10.3390/insects11010064





