Optimizing Photoperiod, Exposure Time, and Host-to-Parasitoid Ratio for Mass-Rearing of Telenomus remus, an Egg Parasitoid of Spodoptera frugiperda, on Spodoptera litura Eggs
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Insects
2.2. Parasitism and Fitness of T. remus under Different Photoperiods
2.3. Effects of Exposure Time and Host Density
2.4. Statistical Analysis
3. Results
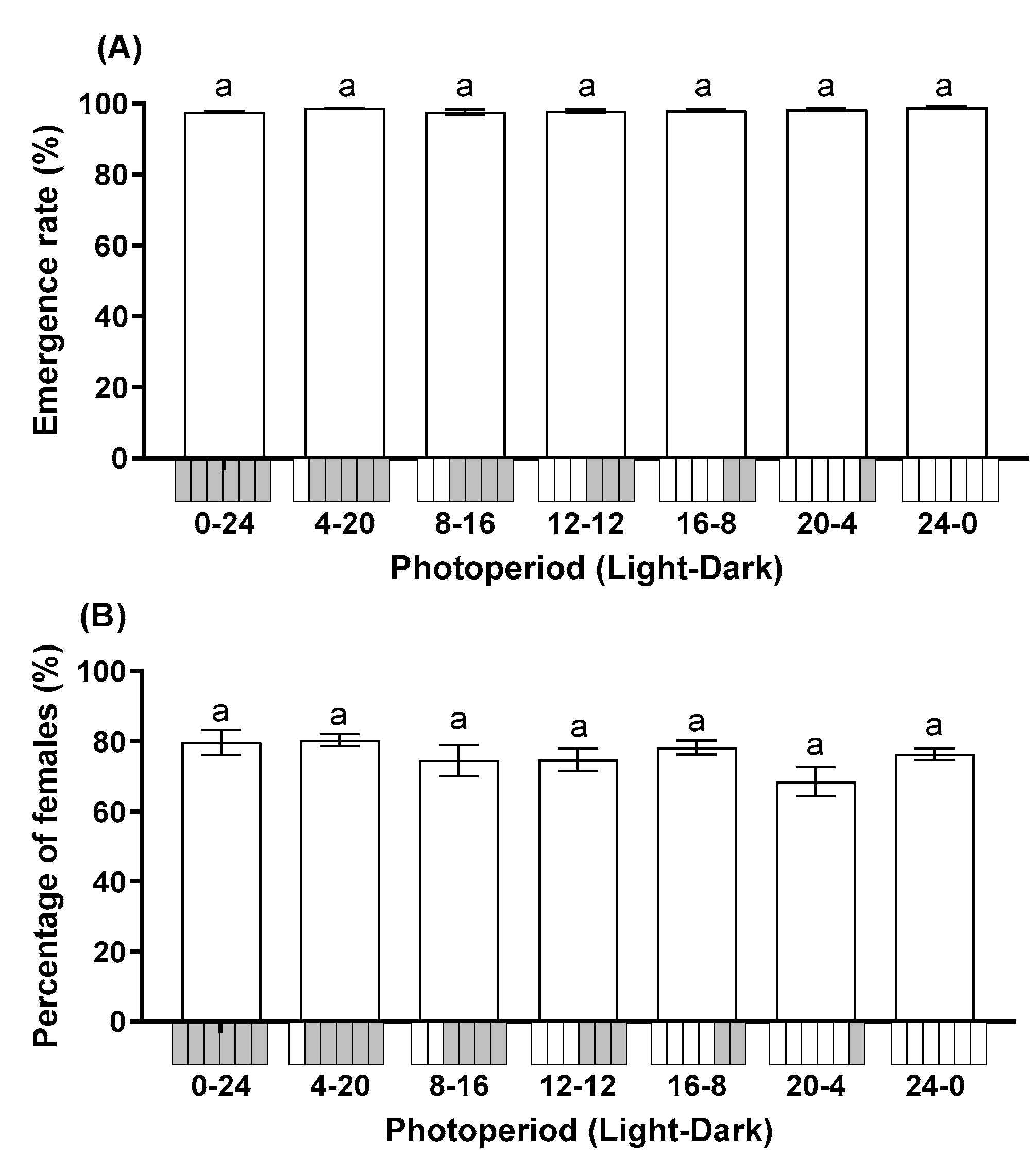
3.1. Effect of Photoperiod on Parasitoid Performance
3.2. Life table of T. remus under Different Photoperiods
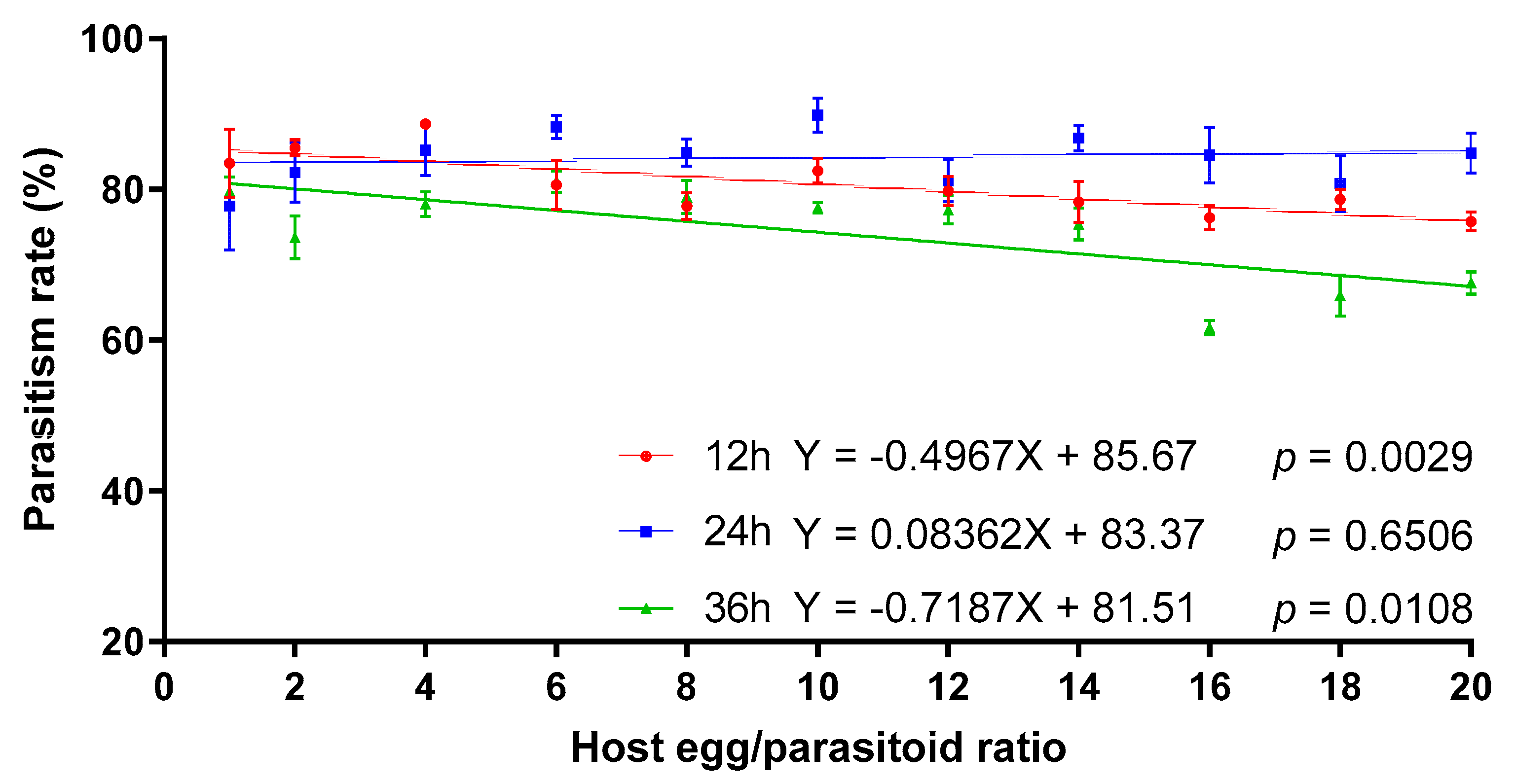
3.3. Parasitism Rate of T. remus under Different Combinations of Exposure Time and Host Egg:Parasitoid Ratio
3.4. Offspring Fitness of T. remus under Different Combinations of Exposure Time and Host Egg:Parasitoid Ratio
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Todd, E.L.; Poole, R.W. Keys and illustrations for the armyworm moths of the noctuid genus Spodoptera Guenee from the Western Hemisphere. Ann. Entomol. Soc. Am. 1980, 73, 722–738. [Google Scholar] [CrossRef]
- Feldmann, F.; Rieckmann, U.; Winter, S. The spread of the fall armyworm Spodoptera frugiperda in Africa—What should be done next? J. Plant. Dis. Protect. 2019, 126, 97–101. [Google Scholar] [CrossRef]
- CABI. Spodoptera frugiperda (Fall Armyworm). Available online: https://www.cabi.org/isc/datasheet/29810#tonaturalEnemies (accessed on 20 September 2021).
- Goergen, G.; Kumar, P.L.; Sankung, S.B.; Togola, A.; Tamò, M. First report of outbreaks of the fall armyworm Spodoptera frugiperda (J E Smith) (Lepidoptera, Noctuidae), a new alien invasive pest in west and central Africa. PLoS ONE 2016, 11, e165632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- China National Agro-Tech Extension and Service Center. Fall Armyworm, the Major Pest has Invaded in Yunnan, the Field Scouting and Monitoring for this Pest to Start Immediately within China. Available online: https://www.natesc.org.cn/news/des?id=c004cc6d-d305-40d7-a360-603f2919813d&Category=%E5%85%A8%E6%96%87%E6%90%9C%E7%B4%A2&CategoryId=d6a35339-e804-4f90-bf93-927382b1fd22 (accessed on 1 September 2021).
- China National Agro-Tech Extension and Service Center. Occurrence Trend of Spodoptera frugiperda in Autumn Maize. Available online: https://www.natesc.org.cn/News/des?id=b4ca3130-c5a2-4f0d-9762-f248e0b94985&kind=HYTX&Category=%E6%A4%8D%E6%A3%80%E6%A4%8D%E4%BF%9D&CategoryId=07e72766-0a38-4dbd-a6a3-c823ce1172bd (accessed on 1 September 2021).
- Wright, M.G. Biological control of invasive insect pests. In Integrated Pest Management; Abrol, D.P., Ed.; Academic Press: London, UK, 2014; pp. 267–281. [Google Scholar]
- Henneberry, T.J. Insect pest management. In Encyclopedia of Pest Management; Pimental, D., Ed.; Taylor and Francis: Boca Raton, FL, USA, 2007; pp. 258–261. [Google Scholar]
- Day, R.; Abrahams, P.; Bateman, M.; Beale, T.; Clottey, V.; Cock, M.; Colmenarez, Y.; Corniani, N.; Early, R.; Godwin, J.; et al. Fall armyworm: Impacts and implications for Africa. Outlooks Pest Manag. 2017, 28, 196–201. [Google Scholar] [CrossRef] [Green Version]
- Wu, K.M. Management strategies of fall armyworm (Spodoptera frugiperda) in China. Plant Prot. 2020, 46, 1–5. [Google Scholar] [CrossRef]
- Montezano, D.G.; Specht, A.; Sosa-Gómez, D.R.; Roque-Specht, V.F.; Sousa-Silva, J.C.; Paula-Moraes, S.V.; Peterson, J.A.; Hunt, T.E. Host plants of Spodoptera frugiperda (Lepidoptera: Noctuidae) in the Americas. Afr. Entomol. 2018, 26, 286–300. [Google Scholar] [CrossRef] [Green Version]
- Guo, J.F.; Wu, S.Y.; Zhang, F.; Huang, C.L.; He, K.L.; Babendreier, D.; Wang, Z.Y. Prospects for microbial control of the fall armyworm Spodoptera frugiperda: A review. BioControl 2020, 65, 647–662. [Google Scholar] [CrossRef]
- Bhusal, S.; Chapagain, E. Threats of fall armyworm (Spodoptera frugiperda) incidence in Nepal and it’s integrated management—A review. J. Agr. Nat. Resour. 2020, 3, 345–359. [Google Scholar] [CrossRef]
- Tambo, J.A.; Day, R.K.; Lamontagne-Godwin, J.; Silvestri, S.; Beseh, P.K.; Oppong-Mensah, B.; Phir, N.A.; Matimelo, M. Tackling fall armyworm (Spodoptera frugiperda) outbreak in Africa: An analysis of farmers’ control actions. Int. J. Pest Manag. 2020, 66, 298–310. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.Z.; Liu, Y.Q.; Shi, M.; Huang, J.H.; Chen, X.X. Parasitoid wasps as effective biological control agents. J. Integr. Agr. 2019, 18, 705–715. [Google Scholar] [CrossRef] [Green Version]
- Okuma, D.M.; Bernardi, D.; Horikoshi, R.J.; Bernardi, O.; Silva, A.P.; Omoto, C. Inheritance and fitness costs of Spodoptera frugiperda (Lepidoptera: Noctuidae) resistance to spinosad in Brazil. Pest Manag. Sci. 2017, 78, 1441–1448. [Google Scholar] [CrossRef] [PubMed]
- Masry, S.H.D.; El-Wakeil, N. Egg parasitoid production and their role in controlling insect pests. In Cottage Industry of Biocontrol Agents and Their Applications; El-Wakeil, N., Saleh, M., Abu-hashim, M., Eds.; Springer: Cham, Switzerland, 2019; pp. 3–47. [Google Scholar]
- Salazar-Mendoza, P.; Rodriguez-Saona, C.; Fernandes, O.A. Release density, dispersal capacity, and optimal rearing conditions for Telenomus remus, an egg parasitoid of Spodoptera frugiperda, in maize. Biocontrol Sci. Technol. 2020, 30, 1040–1059. [Google Scholar] [CrossRef]
- Cave, R.D. Biology, ecology and use in pest management of Telenomus remus. Biocontrol News Inf. 2000, 21, 21–26. [Google Scholar]
- Waddill, H.; Whitcomb, W.H. Release of Telenomus remus (Hym. Scelionidae) against Spodoptera frugiperda (Lep.: Noctuidae) in Florida, U.S.A. Entomophaga 1982, 27, 159–162. [Google Scholar] [CrossRef]
- Pomari, A.F.; Bueno, A.F.; Bueno, R.C.O.F.; Junior, M.; de Oliceriras, A.; Fonseca, A.C.P.F. Releasing number of Telenomus remus (Nixon) (Hymenoptera: Platygastridae) against Spodoptera frugiperda Smith (Lepidoptera: Noctuidae) in corn, cotton and soybean. Cienc. Rural 2013, 43, 377–382. [Google Scholar] [CrossRef] [Green Version]
- Dong, H.; Zhu, K.H.; Zhao, Q.; Bai, X.P.; Zhou, J.C.; Zhang, L.S. Morphological defense of the egg mass of Spodoptera frugiperda (Lepidoptera: Noctuidae) affects parasitic capacity and alters behaviors of egg parasitoid wasps. J. Asia-Pac. Entomol. 2021, 24, 671–678. [Google Scholar] [CrossRef]
- Finney, G.L.; Fisher, T.W. Culture of entomophagous insects and their host. In Biological Control of Insect Pests and Weeds; De Bach, P., Sclinger, E.I., Eds.; Chapman & Hall Ltd.: London, UK, 1964; pp. 328–355. [Google Scholar]
- Parra, J.R.P. Mass rearing of egg parasitoids for biological control programs. In Egg Parasitoids in Agroecosystems with Emphasis on Trichogramma; Consoli, F.L., Parra, J.R.P., Zucchi, R.A., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 267–292. [Google Scholar]
- Pomari-Fernandes, A.; Bueno, A.F.; de Bortoli, S.A.; Favetti, B.M. Dispersal capacity of the egg parasitoid Telenomus remus Nixon (Hymenoptera: Platygastridae) in maize and soybean crops. Biol. Control 2018, 126, 158–168. [Google Scholar] [CrossRef] [Green Version]
- Queiroz, A.P.; Bueno, A.F.; Pomari-Fernandes, A.; Grande, M.L.M.; Bortolotto, O.C.; Silva, D.M. Quality control of Telenomus remus (Hymenoptera: Platygastridae) reared on the factitious host Corcyra cephalonica (Lepidoptera: Pyralidae) for successive generations. Bull. Entomol. Res. 2017, 107, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, A.P.; Bueno, A.F.; Pomari-Fernandes, A.; Grande, M.L.M.; Bortolotto, O.C.; Silva, D.M. Low temperature storage of Telenomus remus (Nixon) (Hymenoptera: Platygastridae) and its factitious host Corcyra cephalonica (Stainton) (Lepidoptera: Pyralidae). Neotrop. Entomol. 2017, 46, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Pomari-Fernandes, A.; Queiroz, A.P.; Bueno, A.F.; Sanzovo, A.W.; De Bortoli, S.A. The importance of relative humidity for Telenomus remus (Hymenoptera: Platygastridae) parasitism and development on Corcyra cephalonica (Lepidoptera: Pyralidae) and Spodoptera frugiperda (Lepidoptera: Noctuidae) eggs. Ann. Entomol. Soc. Am. 2015, 108, 11–17. [Google Scholar] [CrossRef]
- Dai, P.; Sun, J.W.; Chen, Y.M.; Bao, H.P.; Zhang, L.S.; Nkunika, P.O.Y.; Zang, L.S. Discovery of three egg parasitoid species for the control of Spodoptera frugiperda (Smith). J. Jilin. Agric. Univ. 2019, 41, 505–509. [Google Scholar] [CrossRef]
- Huo, L.X.; Zhou, J.C.; Ning, S.F.; Zhao, Q.; Zhang, L.X.; Zhang, Z.T.; Zhang, L.S.; Dong, H. Biological characteristics of Telenomus remus against Spodoptera frugiperda and Spodoptera litura eggs. Plant Prot. 2019, 45, 60–64. [Google Scholar] [CrossRef]
- Chen, W.B.; Li, Y.Y.; Wang, M.Q.; Mao, J.J.; Zhang, L.S. Evaluating the potential of using Spodoptera litura eggs for mass-rearing Telenomus remus, a promising egg parasitoid of Spodoptera frugiperda. Insects 2021, 12, 384. [Google Scholar] [CrossRef] [PubMed]
- Van Nieuwenhove, G.A.; Bezdjian, L.P.; Ovruski, S.M. Effect of exposure time and ratio of hosts to female parasitoids on offspring production of Diachasmimorpha longicaudata (Hymneoptera: Braconidae) reared on Anastrepha fraterculus (Diptera: Tephritidae) larvae. Fla. Entomol. 2012, 95, 99–104. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.G.; Aparicio, E.M.; Duan, J.J.; Gould, J.; Hoelmer, K.A. Optimizing parasitoid and host densities for efficient rearing of Ontsira mellipes (Hymenoptera: Braconidae) on Asian Longhorned Beetle (Coleoptera: Cerambycidae). Environ. Entomol. 2020, 49, 1041–1048. [Google Scholar] [CrossRef]
- Wei, K.; Gao, S.K.; Tang, Y.L.; Wang, X.Y.; Yang, Z.Q. Determination of the optimal parasitoid-to-host ratio for efficient mass-rearing of the parasitoid, Sclerodermus pupariae (Hymenoptera: Bethylidae). J. Appl. Entomol. 2017, 141, 181–188. [Google Scholar] [CrossRef]
- Wang, Z.Y.; He, K.L.; Zhang, F.; Lu, X.; Babendreier, D. Mass rearing and release of Trichogramma for biological control of insect pests of corn in China. Biol. Control 2014, 68, 136–144. [Google Scholar] [CrossRef]
- Zilch, K.C.F.; Jahnke, S.M.; Köhler, A.; Bender, E. Effect of diet, photoperiod and host density on parasitism of Anisopteromalus calandrae on the tobacco beetle and biological parameters of the parasitoid. Am. J. Plant. Sci. 2017, 8, 3218–3232. [Google Scholar] [CrossRef] [Green Version]
- Pomari, A.F.; Bueno, A.F.; Bueno, R.C.O.F.; Menezes, A.O. Telenomus remus Nixon egg parasitization of three species of Spodoptera under different temperatures. Neotrop. Entomol. 2013, 42, 399–406. [Google Scholar] [CrossRef] [Green Version]
- Gautam, R.D. Effect of different temperatures and relative humidities on the efficiency of parasitoid, Telenomus remus Nixon (Scelionidae: Hymenoptera) in the laboratory. J. Entomol. Res. 1986, 10, 34–39. [Google Scholar]
- Greene, G.L.; Leppla, N.C.; Dickerson, W.A. Velvetbean caterpillar: A rearing procedure and artificial medium. J. Econ. Entomol. 1976, 69, 487–488. [Google Scholar] [CrossRef]
- Chen, Q.J.; Li, G.H.; Pang, Y. A simple artificial diet for mass rearing of some noctuid species. Entomol. Knowl. 2000, 37, 325–327. [Google Scholar]
- Huang, S.S.; Dai, Z.Y.; Wu, D.Z. Compilation and application of life table of Trichogramma population. Acta. Phytoph. Sin. 1996, 23, 209–212. [Google Scholar]
- Kehoe, R.; Sanders, D.; Cruse, D.; Silk, M.; Gaston, K.J.; Bridle, J.R.; van Veen, F. Longer photoperiods through range shifts and artificial light lead to a destabilizing increase in host–parasitoid interaction strength. J. Anim. Ecol. 2020, 89, 2508–2516. [Google Scholar] [CrossRef]
- Grande, M.L.M.; Queiroz, A.P.; Gonçalves, J.; Hayashida, R.; Ventura, M.U.; Bueno, A.F. Impact of environmental variables on parasitism and emergence of Trichogramma pretiosum, Telenomus remus and Telenomus podisi. Neotrop. Entomol. 2021, 50, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Scott, S.M.; Barlow, C.A. Effect of prey availability during development on the reproductive output of Metasyrphus corollae (Diptera: Syrphidae). Environ. Entomol. 1984, 13, 669–674. [Google Scholar] [CrossRef]
- Ozkan, C. Effect of food, light and host instar on the egg load of the synovigenic endoparasitoid Venturia canescens (Hymenoptera: Ichneumonidae). J. Pest Sci. 2007, 80, 79–83. [Google Scholar] [CrossRef]
- Mbata, G.N.; Warsi, S.; Payton, M.E. Influence of temperature and photoperiod on the fecundity of Habrobracon hebetor Say (Hymenoptera: Braconidae) and on the paralysis of host larvae, Plodia interpunctella (Hübner) (Lepidoptera: Pyralidae). Insects 2021, 12, 753. [Google Scholar] [CrossRef]
- Hu, S.; Wang, X.Y.; Yang, Z.Q.; Duan, J.J. Effects of photoperiod and light intensity on wing dimorphism and development in the parasitoid Sclerodermus pupariae (Hymenoptera: Bethylidae). Biol. Control 2019, 133, 117–122. [Google Scholar] [CrossRef]
- Gu, H.; Dorn, S. How do wind velocity and light intensity influence host-location success in Cotesia glomerata (Hym., Braconidae)? J. Appl. Entomol. 2001, 125, 115–120. [Google Scholar] [CrossRef]
- Zilahi-balogh, G.M.G.; Shipp, J.L.; Cloutier, C.; Brodeur, J. Influence of light intensity, photoperiod, and temperature on the efficacy of two aphelinid parasitoids of the greenhouse whitefly. Environ. Entomol. 2006, 35, 581–589. [Google Scholar] [CrossRef]
- Mousumi, D.; Ganguly, A.; Haldar, P. Determination of optimum temperature and photoperiod for mass production of Oxya hyla hyla (Serville). Turk. J. Zool. 2012, 36, 329–339. [Google Scholar] [CrossRef]
- Musolin, D.L.; Saulich, A.K. Photoperiodic control of nymphal growth in true bugs (Heteroptera). Entomol. Rev. 1997, 77, 768–780. [Google Scholar]
- Reznik, S.Y.; Vaghina, N.P. Effect of photoperiod on parasitization by Trichogramma principium (Hymenoptera: Trichogrammatidae). Eur. J. Entomol. 2007, 104, 705–713. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Tan, X.L.; Guo, X.J.; Zhang, F. Effect of temperature and photoperiod on the development, reproduction, and predation of the predatory ladybird Cheilomenes sexmaculata (Coleoptera: Coccinellidae). J. Econ. Entomol. 2013, 106, 2621–2629. [Google Scholar] [CrossRef]
- Malaquias, J.B.; Ramalho, F.S.; Fernandes, F.S.; Souza, J.V.S.; Azeredo, T.L. Effects of photoperiod on the development and growth of Podisus nigrispinus, a predator of cotton leafworm. Phytoparasitica 2009, 37, 241–248. [Google Scholar] [CrossRef]
- Bradshaw, W.E.; Holzapfel, C.M. Evolution of animal photoperiodism. Annu. Rev. Ecol. Evol. Syst. 2007, 38, 1–25. [Google Scholar] [CrossRef]
- Qian, H.T.; Cong, B.; Zhang, Z.L.; Dai, Q.H. Effect of some environmental and biological factors on reproductive characters of Trichogramma spp. Afr. J. Agric. Res. 2013, 8, 2195–2203. [Google Scholar] [CrossRef]
- Somjee, U.; Ablard, K.; Crespi, B.; Schaefer, P.W.; Gries, G. Local mate competition in the solitary parasitoid wasp Ooencyrtus kuvanae. Behav. Ecol. Sociobiol. 2011, 65, 1071–1077. [Google Scholar] [CrossRef]
- Chambers, D.L. Quality control in mass rearing. Annu. Rev. Entomol. 1977, 22, 289–308. [Google Scholar] [CrossRef]
- Yazdani, M.; Keller, M. Mutual interference in Dolichogenidea tasmanica (Cameron) (Hymenoptera: Braconidae) when foraging for patchily-distributed light brown apple moth. Biol. Control 2015, 86, 1–6. [Google Scholar] [CrossRef]
- Gobault, M.; Mack, A.F.S.; Hardy, C.W. Encountering competitors reduces clutch size and increases offspring size in a parasitoid with female–female fighting. Proc. R. Soc. B 2007, 274, 2571–2577. [Google Scholar] [CrossRef] [Green Version]
- Smith, S.M. Biological control with Trichogramma: Advances, successes, and potential of their use. Annu. Rev. Entomol. 1996, 41, 375–406. [Google Scholar] [CrossRef]
- Godfray, H.C.J. Parasitoids: Behavioral and Evolutionary Ecology; Princeton University Press: Princeton, NJ, USA, 1994. [Google Scholar]
- Ullah, F.; Farooq, M.; Honey, S.F.; Zada, N. Parasitism potential of Dirhinus giffardii (Silvestri) (Hymenoptera: Chalcididae) on pupae of the fruit fly species, Zeugodacus cucurbitae (Coquillett) and Bactrocera dorsalis (Hendel) (Diptera: Tephritidae), during variable exposure durations. Egypt. J. Biol. Pest Control 2021, 31, 9. [Google Scholar] [CrossRef]
- Li, L.; Wei, L.; Liu, Z.D.; Sun, J.H. Host adaptation of a gregarious parasitoid Sclerodermus harmandi in artificial rearing. BioControl 2010, 55, 465–472. [Google Scholar] [CrossRef]
- Zhou, J.C.; Liu, Q.Q.; Wang, Q.R.; Ning, S.F.; Che, W.N.; Dong, H. Optimal clutch size for quality control of bisexual and Wolbachia-infected thelytokous lines of Trichogramma dendrolimi Matsumura (Hymenoptera: Trichogrammatidae) mass reared on eggs of a substitutive host, Antheraea pernyi Guérin-Méneville (Lepidoptera: Saturniidae). Pest Manag. Sci. 2020, 76, 2635–2644. [Google Scholar] [CrossRef]
- Chen, W.B.; He, K.L.; Wang, Q.Y.; Wang, Z.Y. Effects of yellow peach moth Conogethes punctiferalis egg age on parasitism and oviposition behaviour of four indigenous Trichogramma strains in China. Biocontrol Sci. Technol. 2021, 31, 739–753. [Google Scholar] [CrossRef]
- Godin, C.; Boivin, G. Effects of host age on parasitism and progeny allocation in Trichogrammatidae. Entomol. Exp. Appl. 2000, 97, 149–160. [Google Scholar] [CrossRef]
- Duan, J.J.; Oppel, C. Critical rearing parameters of Tetrastichus planipennisi (Hymenoptera: Eulophidae) as affected by host plant substrate and host-parasitoid group structure. J. Econ. Entomol. 2012, 105, 792–801. [Google Scholar] [CrossRef] [Green Version]
- Montoya, P.; Cancino, J.; Pérez-Lachaud, G.; Liedo, P. Host size, superparasitism and sex ratio in mass-reared Diachasmimorpha longicaudata, a fruit fly parasitoid. BioControl 2011, 56, 11–17. [Google Scholar] [CrossRef]
- González, P.I.; Montoya, P.; Perez-Lachaud, G.; Cancino, J.; Liedo, P. Superparasitism in mass reared Diachasmimorpha longicaudata (Ashmead) (Hymenoptera: Braconidae), a parasitoid of fruit flies (Diptera: Tephritidae). Biol. Control 2007, 40, 320–326. [Google Scholar] [CrossRef]

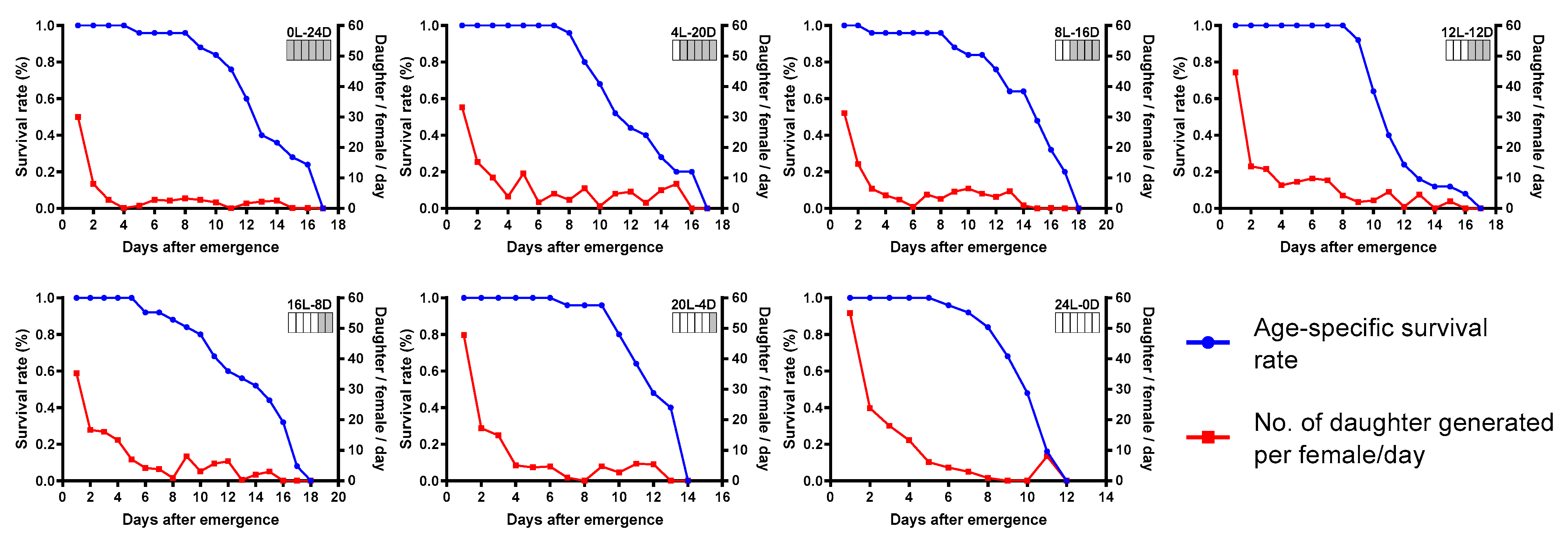

| Photoperiod (Light-Dark) | Life Table Parameters | |||
|---|---|---|---|---|
| Net Reproductive Rate (R0) | Intrinsic Rate of Increase (rm) | Finite Rate of Increase (λ) | Mean Generation Time (T) | |
| 0–24 | 55.16 ± 2.93 d | 0.354 ± 0.021 b | 1.424 ± 0.029 b | 11.5 ± 0.7 ab |
| 4–20 | 97.81 ± 3.26 bc | 0.383 ± 0.016 b | 1.470 ± 0.023 b | 12.0 ± 0.5 ab |
| 8–16 | 88.40 ± 3.86 c | 0.359 ± 0.020 b | 1.432 ± 0.029 b | 12.6 ± 0.6 a |
| 12–12 | 117.96 ± 6.75 a | 0.414 ± 0.013 ab | 1.514 ± 0.019 ab | 11.5 ± 0.3 ab |
| 16–8 | 116.08 ± 3.82 a | 0.395 ± 0.013 ab | 1.484 ± 0.019 ab | 12.1 ± 0.3 ab |
| 20–4 | 107.96 ± 3.33 ab | 0.423 ± 0.019 ab | 1.526 ± 0.029 ab | 11.1 ± 0.5 ab |
| 24–0 | 125.20 ± 1.89 a | 0.463 ± 0.007 a | 1.590 ± 0.010 a | 10.4 ± 0.1 b |
| F | 36.631 | 5.524 | 5.944 | 2.167 |
| df | 6, 28 | 6, 28 | 6, 28 | 6, 28 |
| p | <0.0001 | 0.001 | <0.0001 | 0.077 |
| Parameters | Source | df | F | p |
|---|---|---|---|---|
| Parasitism rate (%) | ET | 2 | 41.753 | <0.0001 |
| HPR | 10 | 4.422 | <0.0001 | |
| ET × HPR | 20 | 2.364 | 0.002 | |
| Error | 132 | |||
| Emergence rate (%) | ET | 2 | 11.101 | <0.0001 |
| HPR | 10 | 0.981 | 0.463 | |
| ET × HPR | 20 | 2.710 | <0.0001 | |
| Error | 132 | |||
| Percentage of female progeny (%) | ET | 2 | 5.117 | 0.007 |
| HPR | 10 | 63.486 | <0.0001 | |
| ET × HPR | 20 | 3.297 | <0.0001 | |
| Error | 132 | |||
| No. of emerged adults/egg | ET | 2 | 4.042 | 0.02 |
| HPR | 10 | 4.48 | <0.0001 | |
| ET × HPR | 20 | 1.458 | 0.107 | |
| Error | 132 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, W.; Weng, Q.; Nie, R.; Zhang, H.; Jing, X.; Wang, M.; Li, Y.; Mao, J.; Zhang, L. Optimizing Photoperiod, Exposure Time, and Host-to-Parasitoid Ratio for Mass-Rearing of Telenomus remus, an Egg Parasitoid of Spodoptera frugiperda, on Spodoptera litura Eggs. Insects 2021, 12, 1050. https://doi.org/10.3390/insects12121050
Chen W, Weng Q, Nie R, Zhang H, Jing X, Wang M, Li Y, Mao J, Zhang L. Optimizing Photoperiod, Exposure Time, and Host-to-Parasitoid Ratio for Mass-Rearing of Telenomus remus, an Egg Parasitoid of Spodoptera frugiperda, on Spodoptera litura Eggs. Insects. 2021; 12(12):1050. https://doi.org/10.3390/insects12121050
Chicago/Turabian StyleChen, Wanbin, Qingfen Weng, Rui Nie, Hongzhi Zhang, Xiaoyu Jing, Mengqing Wang, Yuyan Li, Jianjun Mao, and Lisheng Zhang. 2021. "Optimizing Photoperiod, Exposure Time, and Host-to-Parasitoid Ratio for Mass-Rearing of Telenomus remus, an Egg Parasitoid of Spodoptera frugiperda, on Spodoptera litura Eggs" Insects 12, no. 12: 1050. https://doi.org/10.3390/insects12121050
APA StyleChen, W., Weng, Q., Nie, R., Zhang, H., Jing, X., Wang, M., Li, Y., Mao, J., & Zhang, L. (2021). Optimizing Photoperiod, Exposure Time, and Host-to-Parasitoid Ratio for Mass-Rearing of Telenomus remus, an Egg Parasitoid of Spodoptera frugiperda, on Spodoptera litura Eggs. Insects, 12(12), 1050. https://doi.org/10.3390/insects12121050