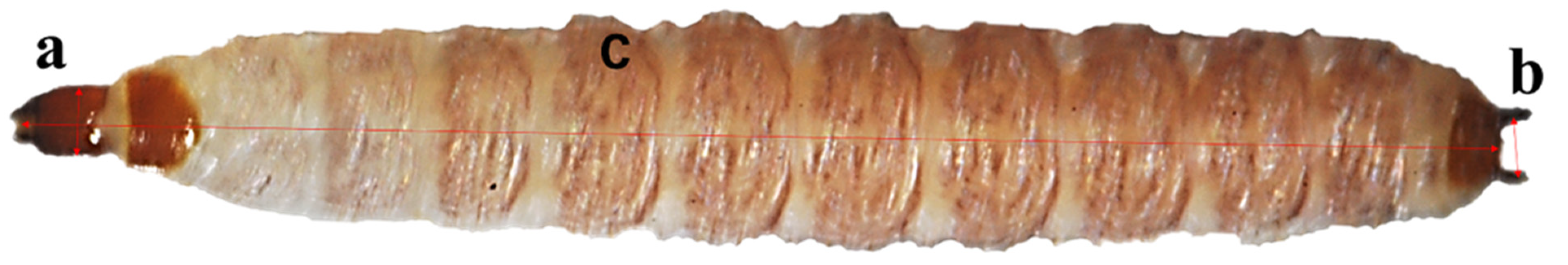
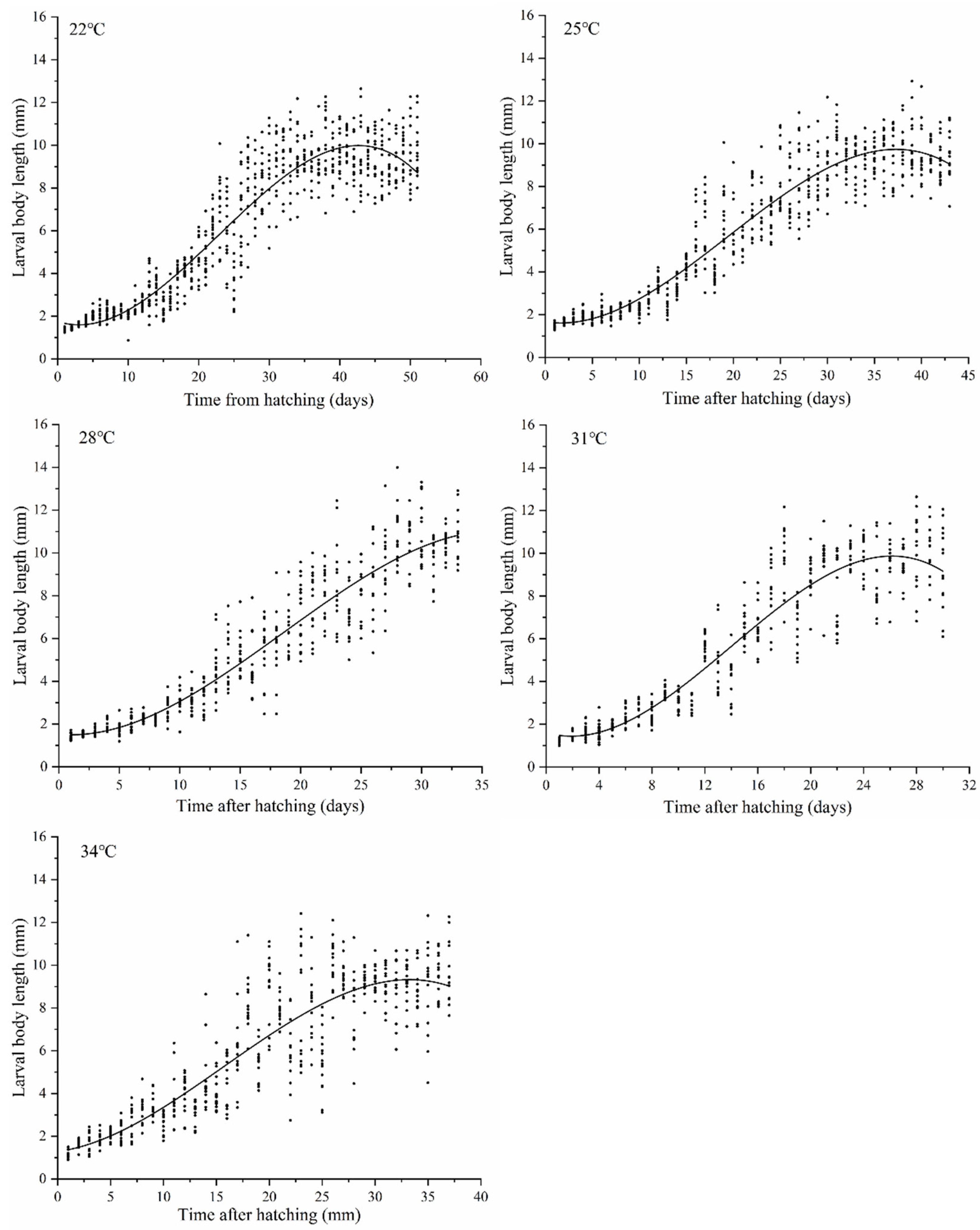
1. Introduction
Forensic entomology provides the scientific basis for postmortem interval (PMI) estimations through the development and succession patterns of sarcosaprophagous insects [
1], and its feasibility has been verified by a large number of practical cases [
2,
3]. At present, the method based on insects is one of the most accurate for estimating the PMI of a corpse in the first 1–2 months of death [
4]. This is because the application of forensic entomology at present is mainly based on fly studies [
5,
6,
7], since the time required for flies to develop one generation is typically within 1–2 months [
8,
9,
10,
11].
Sarcosaprophagous beetles are one of the other important groups of insects that share corpse resources with flies [
12,
13], and they are also key indicator insects for PMI estimation [
5]. Sarcosaprophagous beetles colonize the corpse at a relatively late stage [
5,
14]. Some species belonging to the families Silphidae and Staphylinidae [
12,
15], arrive at a corpse on the 2nd–3rd day (i.e., at an early stage of decomposition). Most species in the families Dermestidae, Cleridae, Nitidulidae, and Scarabaeidae, do not appear on the corpse until a late stage of decomposition or even at the skeletonization stage [
14,
15,
16,
17]. In addition, under the same conditions, the development time of sarcosaprophagous beetles is usually much longer than that of flies [
18]. When all the flies at one scene of death have accomplished one generation of development (with only empty puparia remaining), the sarcosaprophagous beetles on the corpse can still be at the pupal stage, and can therefore be used to estimate PMI [
4,
19]. This suggests that research on sarcosaprophagous beetles could not only expand the PMI time window, which is currently determined by flies, but also improve the accuracy of PMI estimation in the “post-fly stage”, provide a scientific basis for PMI estimations of highly decomposed and skeletonized corpses, and finally, used together with the fly indicators to provide more accurate PMI results for corpses at the early and middle stages of decomposition [
18].
Previous studies on sarcosaprophagous beetles have mainly focused on insect succession [
5,
14,
16,
20,
21], but there are relatively few studies on their development in relation to PMI estimation. However, in recent years, the number of studies on the growth and development of sarcosaprophagous beetles has increased, and dozens of species in the families Silphidae [
22], Staphylinidae [
23,
24,
25], Nitidulidae [
26], Dermestidae [
27,
28], Leiodidae [
29], and Histeridae [
30] have been examined. They provided important basic developmental data, including developmental duration, developmental accumulated temperature, larval body length changes, and instar discriminant parameters for related species used for PMI estimation, while isomorphen diagram, isomegalen diagram, and thermal summation models were established for the convenience of application. Simultaneously, research has also investigated the effects of drugs [
31], foods [
32,
33], in vivo measurement method [
34], and pupation substrate [
33,
35] on the growth and development of different sarcosaprophagous beetles, as well as the developmental rate differences between different sexes [
25]. Although a lot of research has been conducted, basic developmental data are still missing on a large number of important sarcosaprophagous beetles.
Necrobia ruficollis (Fabricius, 1775) (Coleoptera: Cleridae) is mainly characterized by a metallic blue to blue-green color, except for the pronotum, wings base, sternum, and legs which appear reddish brown. It mainly damages dried meat, animal medicinal materials, fur, dried fish and fish meal, cocoons, as well as cotton clothes [
36,
37,
38], and is a globally distributed storage pest. Despite its destructive nature,
N. ruficollis is also an important species in forensic science [
38] and has been recorded in death cases and insect succession studies in many areas around the world [
15,
16,
20,
39,
40]. Previous succession studies have indicated that
N. ruficollis mainly appear on carcasses during the advanced decay stage and skeleton stage [
5,
16,
40]. However, when the temperature is relatively higher, the adult beetles also appear on carcasses at the bloating stage [
15]. In some areas,
N. ruficollis frequently appears together with
Necrobia rufipes (De Greer, 1775) (Coleoptera: Cleridae) [
5]. After arriving at a corpse, the adult
N. ruficollis mainly feeds on corpse tissues, but also preys on other insects on the corpse [
38]. The larvae mainly feed on the decomposed corpse. Mature larvae usually pupate in the pupal chamber [
41]. Since
N. ruficollis is an important storage pest of significant forensic value, research on the developmental pattern of
N. ruficollis has significance for both pest control and estimation of the minimum PMI.
At present, research on
N. ruficollis mainly includes mitochondrial whole genome analyses [
38], cytological detection [
42], descriptions of biological and morphological properties [
41], as well as damage and control in relation to stored products [
36,
37]. However, basic developmental data for pest control and forensic entomological applications are still insufficient. The results of the existing succession studies showed that
N. ruficollis mainly appears on the corpse at the late stage of decomposition or even the skeletonization stage, and is one of the insects that arrive at the corpse last [
5,
16,
40]. Study on
N. rufipes, a closely related species of
N. ruficollis, has suggested that certain morphological indexes, including head capsule width, pronotum width, and mesonotum width could be used for the determination of larval instars [
43]. Thus, we supposed that such indexes may also be used to determine larval instars of
N. ruficollis. In view of this, we conducted a study at five constant temperatures between 22–34 °C to determine (1) the developmental duration and thermal summation constant of the
N. ruficollis; (2) whether the variation of larval body length can be used to estimate the PMI; and (3) whether two indexes, head capsule width and distance between urogomphi, can feasibly be used to estimate the larval instar, in order to better use this species to estimate the PMI.
4. Discussion
As a storage pest,
N. ruficollis has a wide-ranging diet, and many animal products can be used as its food [
36,
38]. In this study, the dried pork slices were fed to
N. ruficollis adults, considering that the larvae would often drill into the gaps of large meat pieces, making them difficult to observe; the dried pork slices were therefore smashed into small pieces before being fed to the larvae. The pork slices were easy to obtain and process, and in this study, it was found that both the adults and larvae raised on this food exhibited good growth. Nevertheless, we were unable to evaluate whether the development data of
N. ruficollis obtained in this study were consistent with the development pattern on a corpse under field conditions; this requires further studies which should evaluate the effects of different food types on the development of
N. ruficollis, in order to obtain the experimental data which would better represent its development in the natural environment.
Previous studies showed that
N. ruficollis will pupate on multiple substrates, such as existing cavities (including empty puparium of fly), sawdust, cork, and even banana peels [
41], but all tended to pupate in a hidden place. In this study, the sandy soil, cotton, wood, and paper tissue were provided as the pupation substrates for larvae at the post-feeding stage, and it was found that they preferred to pupate in cotton. The larvae produced filamentous secretions that formed a slightly hardened pupal chamber and the formed pupal chamber was similar to a silkworm cocoon, but smaller and thinner. The contours of the insect body in the pupal chamber could be observed by the transmission light of the microscope. There existed a pre-pupal stage between pupal chamber formation and pupation during which the larval length gradually shortened to a point similar to the pupal length, which was followed by pupation after a period of time. If the pupal chamber was opened before the larval body length shortened, larvae would leave the pupal chamber. After crawling away, some larvae would choose to repair the damaged pupal chamber and continued to pupate, while other larvae would re-form the pupal chamber. Due to the delay in pupation time caused by the destruction of the pupal chamber, the insect body changes in the pupal chamber were observed through transmission light and the pupation time could be accurately judged by the phenomenon that the pupae would twist their abdomens. In this study, in order to record the exact adult emergence time and facilitate the observation, the pupal chambers were opened after the larvae had developed into pupae. The pupae can continue its development process and almost all pupae were emerged successfully, and we did not find the obvious effects on development velocity and eclosion rate of pupae. However, as there were no pervious data to prove the effect of opening the pupal chamber on pupal development velocity and eclosion rate, and the comparative studies were not conducted in this study, whether this operation will affect the development velocity and eclosion rate of pupae needs to be studied further. The head and thorax of a newly formed pupa is milk white and translucent, and the abdomen is covered with irregular black dark spots. With development, the insect will become colored in the order from eyes, mouthparts, and legs/wings. In the study by Novák et al. [
47], the age of the pupal stage of Silphidae could be estimated through quantifying the changes in compound eye color of the pupa. In future research, observations of external morphological changes in
N. ruficollis pupae could provide a basis for pupal age estimation.
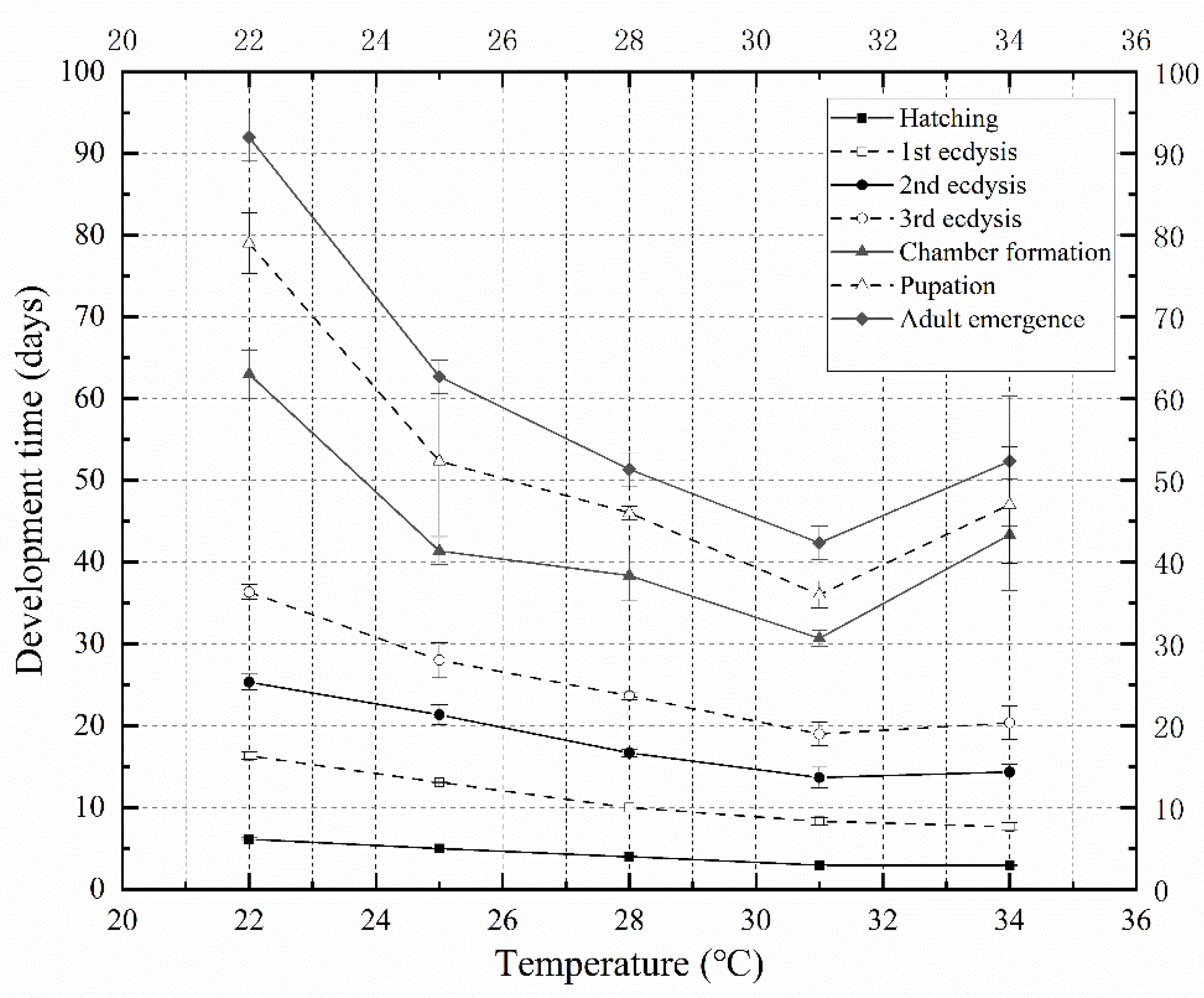
The development rate of
N. ruficollis accelerated with an increasing temperature between 22 and 31 °C, while the overall development rate decreased at 34 °C. At 31 °C, the minimum development duration of
N. ruficollis from egg to adult was 42.33 days, while it was 52.33 days at 34 °C, which was an increase compared to that observed at 31 °C. This indicates that 34 °C might be the maximum tolerance temperature of
N. ruficollis. This was consistent with the upper lethal developmental threshold temperature (34.94 °C) inferred by the Curvilinear crystal model (Optim SSI). Compared with D
0 (14.51 °C) obtained by the revised linear regression model proposed by Ikemoto and Takai [
45], a similar
TL (14.61 °C) was estimated by a nonlinear model of Shi et al. [
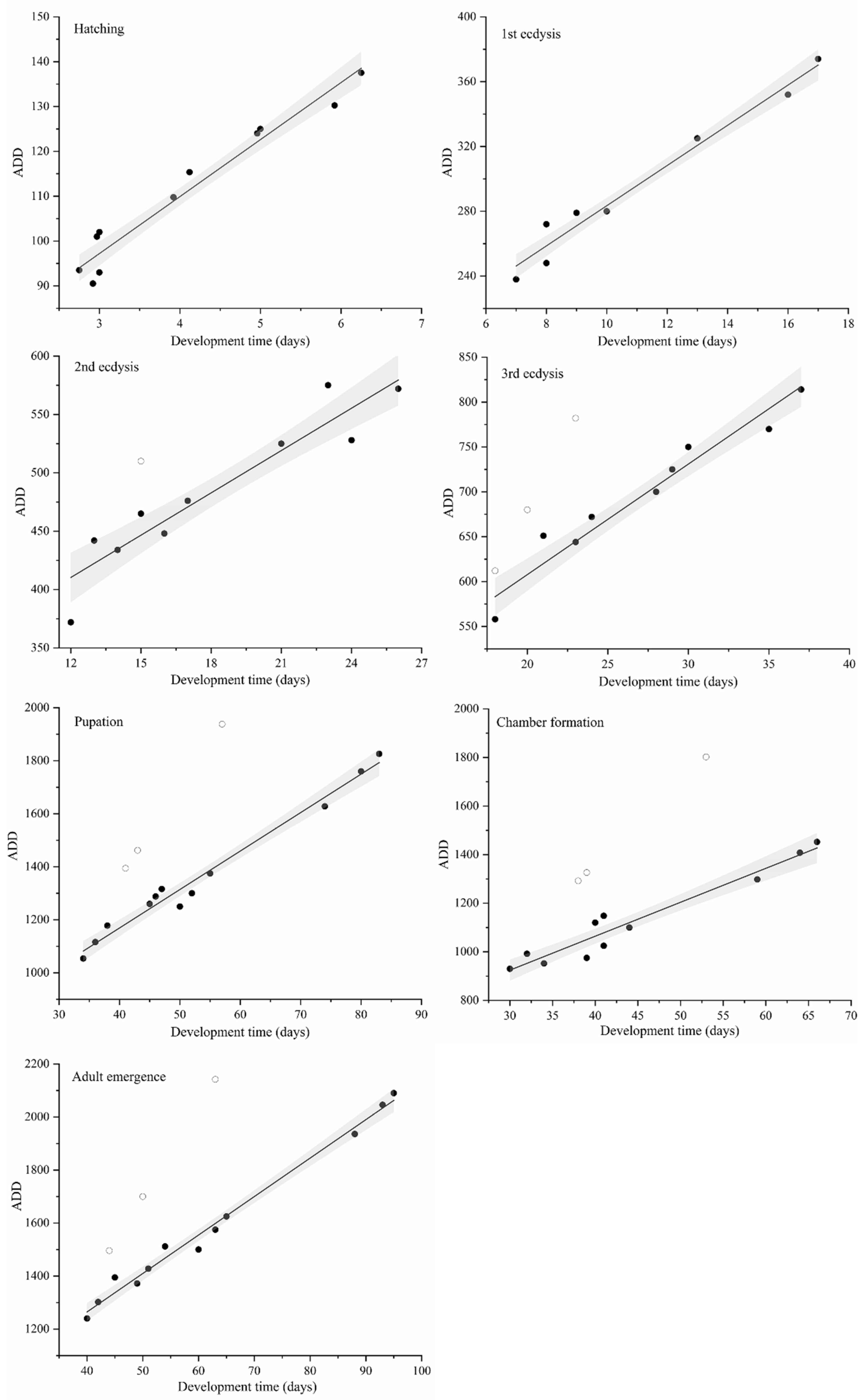
46] (the variation was less than 0.7%). The development rate of theses insects generated a bell-shaped curve. The linear thermal summation model is only applicable to the linear range of growth rate change and is not applicable when the temperature change is close to the lower and upper developmental threshold. This study adopted the linear model proposed by Ikemoto and Takai [
45], who put forward their opinions on model modification. Data points at critical temperatures could not be included in the linear array of points, which results in an uncertainty in the optimum temperature range as well as an unreliable estimation of parameters [
45]. Therefore, nonlinear data that are outside the scope of the linear model should be removed. Thus, in this study, the nonlinear data at 34°C were removed during the linear regression analysis according to the recommendation of Ikemoto and Takai [
45]. The curvilinear modeling was able to simulate the growth rate of
N. ruficollis at 22–34 °C, including the nonlinear relationship at 34 °C; it can therefore be used to calculate lower and upper thermal thresholds as well as the optimum temperature for development. Nonlinear models have been studied extensively in recent years. The SSI model selected in this study can calculate the optimal temperature when the enzyme activity is the highest (the probability of an enzyme being in its active state is maximal), but not the optimum temperature when the growth rate is the fastest.
Although many studies have emphasized the value of
N. ruficollis in forensic investigations, no development data about this species were available. Compared to other sarcosaprophagous beetles, the development time of
N. ruficollis is much longer. For example, at 25 °C, the development times for
Euspilotus azureus (Sahlberg, 1823) (Coleoptera: Histeridae) [
30],
Thanatophilus sinuatus (Fabricius, 1775) (Coleoptera: Silphidae) [
48],
Dermestes frischii (Kugelan, 1792) (Coleoptera: Dermestidae) [
27], and
Omosita colon (Linnaeus, 1758) (Coleoptera: Nitidulidae) [
26] from eggs to eclosion are 31.5, 24.8, 36.0, and 34.9 days, respectively, while
N. ruficollis requires 62.7 days to complete one generation development at this temperature. Compared to the closely related species of
N. rufipes, the development duration of
N. rufipes from egg to adult at 25 °C is 66.2 days [
43], which is similar to the development time of
N. ruficollis.
Necrobia ruficollis and
N. rufipes are related species, and are very similar in morphological, genetic, and ecological attributes, which may explain the similarity in their development data.
Necrobia ruficollis is one of the last insect species to arrive at the corpse. When the immature
N. ruficollis is prevalent on the corpse, most of the flies that colonize the cadaver early, such as blow flies, have accomplished one generation of development and have left the corpse. As a result, research on
N. ruficollis can improve the accuracy of PMI estimation in the “post-fly stage” and provide a scientific basis for PMI estimations for highly decomposed and skeletonized corpses. In addition, the immature stage of
N. ruficollis is longer than that of flies. The longer development time of
N. ruficollis can further expand the estimation range of the PMI and corroborate the indexes of flies and other beetles, possibly obtaining more accurate PMI results. Simultaneously, in many succession experiments around the world, it was found that the time for
N. ruficollis to colonize a corpse was different with the seasons [
15,
16,
49], which means that it is of great importance to determine the colonization time in different seasons in corresponding areas.
Sarcosaprophagous beetles have a longer life history and their reproduction rates are far lower than that of flies; it is thus difficult to collect enough eggs for continuous and intensive sampling in a short time. Considering that the study required a high sample size, in this study, in vivo photography was applied at specific times to obtain developmental and morphological data. To reduce disruption to larvae, we were very careful during in vivo photography and the shooting time was also shortened as much as possible, but there still exists the risk that the data obtained in this study differs from the growth and development that would be observed in the natural environment. In particular, the research of Frątczak-Łagiewska and Matuszewski [
34] pointed out that the larval size from in vivo measurements would be smaller, and the data obtained through in vivo measurements might overestimate the actual age of larvae. In future research, a sampling method with the minimum impact on larval development should be investigated, and the effects of in vivo measurement on larval development should be further studied.
In most forensic entomology cases, the temperature experienced by insects fluctuates rather than remaining constant [
6]. Hence, theoretically, the age of immature stage should be estimated using development data obtained under fluctuating temperatures. However, fluctuation of temperatures is hard to model, as they can manifest in a variety of ways even with the same average temperature. In practice, forensic entomologists usually average the fluctuating environmental temperature to which the corpses are subjected, and then used the data of same constant temperature to estimate the PMI. Several studies have been conducted on the effects of constant and fluctuating temperatures on insect development, and different researchers hold different opinions. Milosavljevic et al. [
50] and McCalla et al. [
51] found that compared with a constant temperature, a fluctuating temperature had a significant impact on the development rates of Encyrtidae and Eulophidae. Parasitoids reared under fluctuating profiles at low average temperatures developed faster and survived for longer when compared to those reared under constant temperatures. In contrast, high average fluctuating temperatures produced parasitoids with an extended developmental period and reduced longevity. Wu et al. studied the development duration of
Trichogramma dendrolimi (Matsumura, 1926) (Hymenoptera: Trichogrammatidae) and
Diaphania indica (Saunders, 1851) (Lepidoptera: Crambidae) under various constant and fluctuating temperature regimes, and results revealed that a fluctuating temperature did not influence the instantaneous rate of development (the variation is less than 4%), and the thermal inputs for completing development did not lead to significant differences, or were similar between constant and fluctuating temperature regimes [
52]. In the study of the influence of fluctuating temperature on Coleoptera, Hagstrum and Leach [
53] found that development time of
Tribolium castancum (Herbst,1797) (Coleoptera: Tenebrionidae) and
Trogoderma iuclusum (LeConte, 1854) (Coleoptera: Dermestidae) was reduced by about 9–12% when beetles were exposed to sinusoidal fluctuating temperatures rather than a constant temperature. However,
Sitophilus oryzac (L. 1763) (Coleoptera: Curculionidae) showed no significant difference in the effect of the two temperature conditions on development time. Another study by Hagstrum and Milliken [
54] showed that developmental time data collected at constant temperatures poorly predicted developmental times of red flour beetle,
T. castaneum, and 16 other species at fluctuating temperatures over a broad range of mean temperatures or amplitudes of fluctuating temperatures. Developmental times at constant temperatures tended to be shorter above 25–30 °C temperature range and longer below this range than at fluctuating temperatures with the same means. In forensic entomology, Byrd and Butler have carried out studies on the development of flies of forensic significance under fluctuating temperature conditions, including
Chrysomya rufifacies (Macquart, 1844) (Diptera: Calliphoridae) [
55],
Cochliomyia macellaria (Fabricius, 1775) (Diptera: Calliphoridae) [
56], and
Sarcophaga haemorrhoidalis (Fallen, 1871) (Diptera: Sarcophagidae) [
57]. In this paper, we provide the development data of
N. ruficollis under constant temperature, which may differ from the development pattern of this species under natural conditions, highlighting a limitation of this study in forensic medicine practice. Future studies should compare the development rates of the species at different fluctuations and constant temperatures to enable more accurate PMI.
In addition, our choice of experimental temperature range has limitations. According to our previous studies on insect succession in the field and in other areas, we found that the
N. ruficollis tends to appear in warm seasons [
15,
16]. In addition, Hu et al. [
43] found that the death rate of its closely related species
N. rufipes at the constant temperatures of 19 °C was 100%. Considering that
N. rufipes and
N. ruficollis are usually present in cadavers at the same time, the same thermal biology characteristic may apply. Therefore, the experimental temperature of 19 °C or lower was not set in this study. However, we should take into account that the
N. ruficollis and
N. rufipes are not the same species, and there may be significant differences in the developmental threshold temperatures, which will be verified by further experiments in the future.