How Effective Are Push–Pull Semiochemicals as Deterrents for Bark Beetles? A Global Meta-Analysis of Thirty Years of Research
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
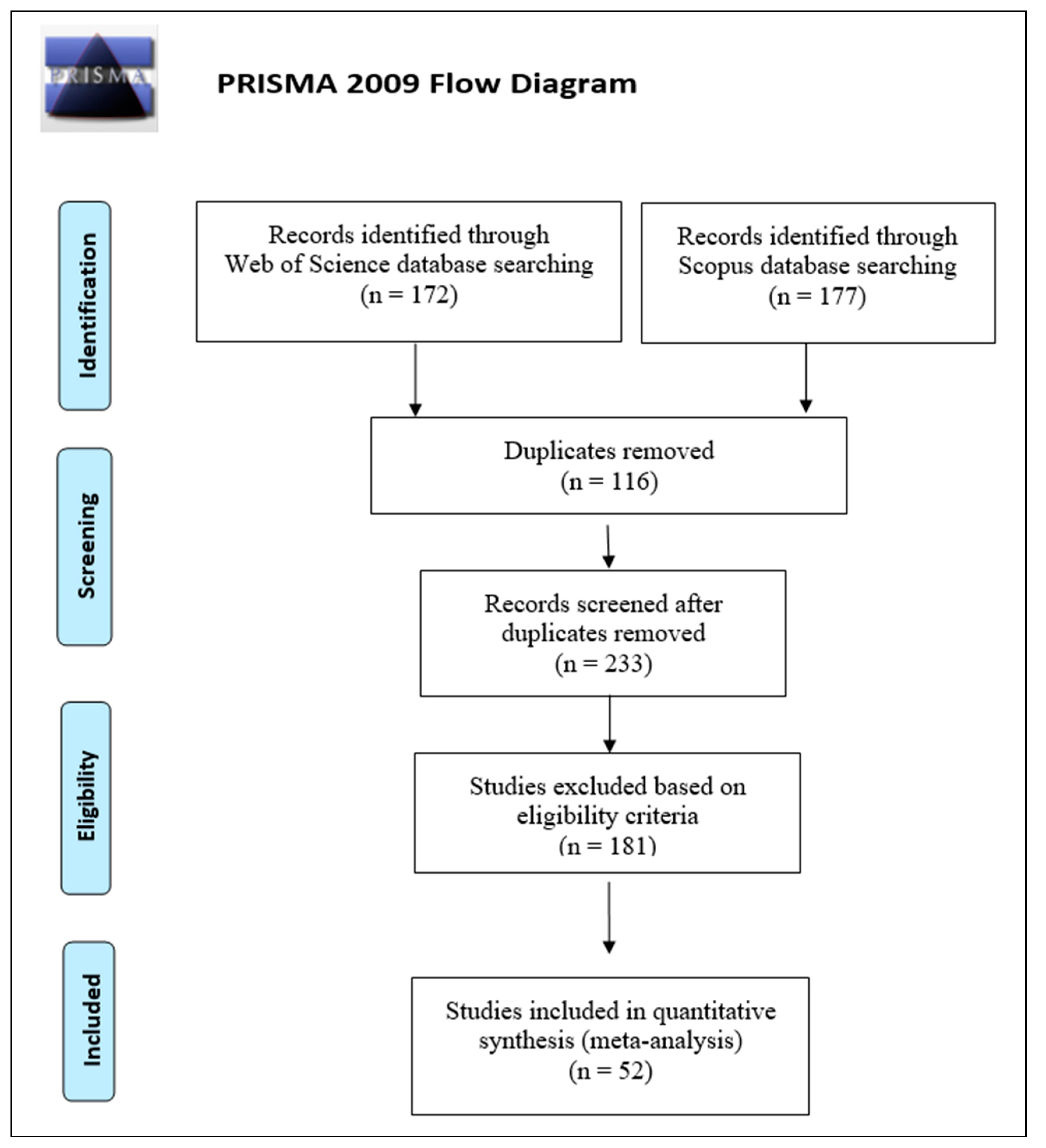
2.1. Database Search and Selection Criteria
2.2. Study Selection
- The study contained at least one species of Dendroctonus or Ips as the target.
- The study focused on the push–pull semiochemical technique; any study dealing with pull-alone, push-alone, insecticide use, and other control methods was excluded.
- The results were reported as means, variance (standard deviation or standard error), sample size, and other relevant statistical information to allow the calculation of the effect size.
2.3. Data Extraction
2.4. Meta-Analysis
3. Results
3.1. Metadata
3.2. The Overall Effect of Push–Pull Semiochemical Treatments on Dendroctonus and Ips
3.3. Effect of Push–Pull Semiochemical Treatment on Attack and Attraction of Dendroctonus and Ips Species
3.4. Effect of Push–Pull Semiochemical Treatments on Dendroctonus and Ips Species
3.5. Effect of Push–Pull Semiochemical Treatment Sources on Dendroctonus and Ips
3.6. Effect of Geographical Regions on Push–Pull Semiochemical Treatments in Dendroctonus and Ips
4. Discussion
5. Limitations and Suggestions
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kirkendall, L.R.; Biedermann, P.H.; Jordal, B.H. Evolution and diversity of bark and ambrosia beetles. In Bark Beetles; Elsevier: Amsterdam, The Netherlands, 2015; pp. 85–156. [Google Scholar]
- Raffa, K.F.; Grégoire, J.-C.; Staffan Lindgren, B. Chapter 1—Natural history and ecology of Bark Beetles. In Bark Beetles; Vega, F.E., Hofstetter, R.W., Eds.; Academic Press: San Diego, CA, USA, 2015; pp. 1–40. [Google Scholar] [CrossRef]
- Gandhi, K.J.; Hofstetter, R. Introduction: Bark beetles, management, and climate change. In Bark Beetle Management, Ecology, and Climate Change; Elsevier: Amsterdam, The Netherlands, 2022; pp. xix–xxvi. [Google Scholar]
- Morris, J.L.; Cottrell, S.; Fettig, C.J.; DeRose, R.J.; Mattor, K.M.; Carter, V.A.; Clear, J.; Clement, J.; Hansen, W.D.; Hicke, J.A. Bark beetles as agents of change in social–ecological systems. Front. Ecol. Environ. 2018, 16, S34–S43. [Google Scholar] [CrossRef]
- Gillette, N.E.; Fettig, C.J. Semiochemicals for bark beetle (Coleoptera: Curculionidae) management in western North America: Where do we go from here? Can. Entomol. 2021, 153, 121–135. [Google Scholar] [CrossRef]
- Grégoire, J.-C.; Raffa, K.F.; Lindgren, B.S. Chapter 15—Economics and politics of Bark Beetles. In Bark Beetles; Vega, F.E., Hofstetter, R.W., Eds.; Academic Press: San Diego, CA, USA, 2015; pp. 585–613. [Google Scholar] [CrossRef]
- Hlásny, T.; König, L.; Krokene, P.; Lindner, M.; Montagné-Huck, C.; Müller, J.; Qin, H.; Raffa, K.F.; Schelhaas, M.-J.; Svoboda, M.; et al. Bark beetle outbreaks in Europe: State of knowledge and ways forward for management. Curr. For. Rep. 2021, 7, 138–165. [Google Scholar] [CrossRef]
- Pan, Q.; Fang, J.; Zhang, S.; He, Y.; Wang, Y. Biorhythm paralleled release of pheromone by photothermal conversion for Long-term bark beetle control. Chem. Eng. J. 2022, 440, 135933. [Google Scholar] [CrossRef]
- Wylie, F.; Peters, B.; DeBaar, M.; King, J.; Fitzgerald, C. Managing attack by bark and ambrosia beetles (Coleoptera: Scolytidae) in fire-damaged Pinus plantations and salvaged logs in Queensland, Australia. Aust. For. 1999, 62, 148–153. [Google Scholar] [CrossRef]
- Gillette, N.E.; Munson, A.S. Semiochemical sabotage: Behavioral chemicals for protection of Western conifers from Bark Beetles. In Proceedings of the Western Bark Beetle Research Group: A Unique Collaboration with Forest Health Protection—Proceedings of a Symposium at the 2007 Society of American Foresters Conference, Portland, Oregon, 23–28 October 2007; p. 85. [Google Scholar]
- Sharma, A.; Sandhi, R.K.; Reddy, G.V. A review of interactions between insect biological control agents and semiochemicals. Insects 2019, 10, 439. [Google Scholar] [CrossRef]
- Yew, J.Y.; Chung, H. Insect pheromones: An overview of function, form, and discovery. Prog. Lipid Res. 2015, 59, 88–105. [Google Scholar] [CrossRef]
- Smart, L.E.; Aradottir, G.I.; Bruce, T.J.A. Chapter 6—Role of semiochemicals in integrated pest management. In Integrated Pest Management; Abrol, D.P., Ed.; Academic Press: San Diego, CA, USA, 2014; pp. 93–109. [Google Scholar] [CrossRef]
- Cook, S.M.; Khan, Z.R.; Pickett, J.A. The use of push-pull strategies in integrated pest management. Annu. Rev. Entomol. 2007, 52, 375–400. [Google Scholar] [CrossRef]
- El-Ghany, N. Semiochemicals for controlling insect pests. J. Plant Prot. Res. 2019, 59, 1–11. [Google Scholar] [CrossRef]
- Flechtmann, C.; Dalusky, M.; Berisford, C. Bark and ambrosia beetle (Coleoptera: Scolytidae) responses to volatiles from aging loblolly pine billets. Environ. Entomol. 1999, 28, 638–648. [Google Scholar] [CrossRef]
- Verhoef, H.A.; Nagelkerke, C. Formation and ecological significance of aggregations in Collembola. Oecologia 1977, 31, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.H.; Schlyter, F. Olfactory recognition and behavioural avoidance of angiosperm nonhost volatiles by conifer-inhabiting bark beetles. Agric. For. Entomol. 2004, 6, 1–20. [Google Scholar] [CrossRef]
- Mashaly, A.; Ali, M.; Al-Khalifa, M. Chapter 6 Trail pheromones in pest control. In New Perspectives in Plant Protection; Bandani, A.R., Ed.; IntechOpen: London, UK, 2012; pp. 121–138. [Google Scholar] [CrossRef]
- Progar, R.A.; Gillette, N.; Fettig, C.J.; Hrinkevich, K. Applied chemical ecology of the mountain pine beetle. For. Sci. 2014, 60, 414–433. [Google Scholar] [CrossRef]
- Cale, J.A.; Taft, S.; Najar, A.; Klutsch, J.G.; Hughes, C.C.; Sweeney, J.D.; Erbilgin, N. Mountain pine beetle (Dendroctonus ponderosae) can produce its aggregation pheromone and complete brood development in naïve red pine (Pinus resinosa) under laboratory conditions. Can. J. For. Res. 2016, 45, 1873–1877. [Google Scholar] [CrossRef]
- Sullivan, B.T.; Brownie, C. The role of wind and semiochemicals in mediating switching behavior in the Southern pine beetle (Coleoptera: Curculionidae: Scolytinae). Environ. Entomol. 2022, 51, 340–350. [Google Scholar] [CrossRef]
- Schlyter, F. Semiochemical diversity in practice: Antiattractant semiochemicals reduce bark beetle attacks on standing trees—A first meta-analysis. Psyche 2012, 2012, 268621. [Google Scholar] [CrossRef]
- Raffa, K.F.; Andersson, M.N.; Schlyter, F. Chapter one—Host selection by bark beetles: Playing the odds in a high-stakes game. In Advances in Insect Physiology; Tittiger, C., Blomquist, G.J., Eds.; Academic Press: San Diego, CA, USA, 2016; Volume 50, pp. 1–74. [Google Scholar]
- Perkins, D.L.; Jorgensen, C.L.; Rinella, M.J. Verbenone decreases whitebark pine mortality throughout a mountain pine beetle outbreak. For. Sci. 2015, 61, 747–752. [Google Scholar] [CrossRef]
- Hunter, J.E.; Schmidt, F.L. Methods of Meta-Analysis: Correcting Error and Bias in Research Findings; Sage: Washington, DC, USA, 2004. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- Grover, S.; Basu, S. Measuring student learning in introductory block-based programming: Examining misconceptions of loops, variables, and boolean logic. In Proceedings of the 2017 ACM SIGCSE Technical Symposium on Computer Science Education, Seattle, WA, USA, 8–11 March 2017; pp. 267–272. [Google Scholar]
- Rohatgi, A. WebPlotDigitizer User Manual Version 3.4. 2014, pp. 1–18. Available online: http://Arohatgi.Info/WebPlotDigitizer/App (accessed on 27 September 2022).
- Lajeunesse, M.J.; Forbes, M.R. Variable reporting and quantitative reviews: A comparison of three meta-analytical techniques. Ecol. Lett. 2003, 6, 448–454. [Google Scholar] [CrossRef]
- Gurevitch, J.; Hedges, L.V. Statistical issues in ecological meta-analyses. Ecology 1999, 80, 1142–1149. [Google Scholar] [CrossRef]
- Hedges, L.V.; Gurevitch, J.; Curtis, P.S. The meta-analysis of response ratios in experimental ecology. Ecology 1999, 80, 1150–1156. [Google Scholar] [CrossRef]
- Lin, L.; Aloe, A.M. Evaluation of various estimators for standardized mean difference in meta-analysis. Stat. Med. 2021, 40, 403–426. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, J.O.; Adhikari, N.K.J.; Beyene, J. The ratio of means method as an alternative to mean differences for analyzing continuous outcome variables in meta-analysis: A simulation study. BMC Med. Res. Methodol. 2008, 8, 32. [Google Scholar] [CrossRef]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.; Rothstein, H.R. Introduction to Meta-Analysis; John Wiley & Sons: Hoboken, NJ, USA, 2021; pp. 1–37. [Google Scholar]
- Viechtbauer, W. Conducting meta-analyses in R with the metafor package. J. Stat. Softw. 2010, 36, 1–48. [Google Scholar] [CrossRef]
- Schwarzer, G. meta: An R package for meta-analysis. R News 2007, 7, 40–45. [Google Scholar]
- Wickham, H. Data analysis. In ggplot2; Springer: Berlin/Heidelberg, Germany, 2016; pp. 189–201. [Google Scholar]
- Byers, J.A.; Maoz, Y.; Cohen, B.; Golani, M.; Fefer, D.; Levi-Zada, A. Protecting avocado trees from ambrosia beetles by repellents and mass trapping (push–pull): Experiments and simulations. J. Pest Sci. 2021, 94, 991–1002. [Google Scholar] [CrossRef]
- Jones, K.L.; Evenden, M.L. Effect of semiochemical exposure on flight propensity and flight capacity of Dendroctonus ponderosae in laboratory bioassays. Arthropod-Plant Interact. 2021, 15, 551–562. [Google Scholar] [CrossRef]
- Seybold, S.J.; Bentz, B.J.; Fettig, C.J.; Lundquist, J.E.; Progar, R.A.; Gillette, N.E. Management of western North American bark beetles with semiochemicals. Annu. Rev. Entomol. 2018, 63, 407–432. [Google Scholar] [CrossRef]
- Kegley, S.; Gibson, K. Individual-Tree Tests of Verbenone and Green-Leaf Volatiles to Protect Lodgepole, Whitebark and Ponderosa Pines, 2004–2007; Forest Health Protection Numbered Report 09-03; USDA Forest Service, Forest Health Protection: Missoula, MT, USA, 2009. [Google Scholar]
- Domínguez-Sánchez, B.; Macías-Sámano, J.E.; Ramírez-Marcial, N.; León-Cortés, J.L. Kairomonal response of coleopterans associated with Dendroctonus frontalis and two Ips species (Coleoptera: Curculionidae) in forest of Chiapas, Mexico. Rev. Mex. De Biodivers. 2008, 79, 175–183. [Google Scholar]
- Gillette, N.; Stein, J.; Owen, D.; Webster, J.; Fiddler, G.; Mori, S.; Wood, D. Verbenone-releasing flakes protect individual Pinus contorta trees from attack by Dendroctonus ponderosae and Dendroctonus valens (Coleoptera: Curculionidae, Scolytinae). Agric. For. Entomol. 2006, 8, 243–251. [Google Scholar] [CrossRef]
- Cardinal, E.; Shepherd, B.; Krakowski, J.; Schwarz, C.J.; Stirrett-Wood, J. Verbenone and green-leaf volatiles reduce whitebark pine mortality in a northern range-expanding mountain pine beetle outbreak. Can. J. For. Res. 2022, 52, 158–168. [Google Scholar] [CrossRef]
- Bedard, W.D.; Tilden, P.E.; Lindahl, K.Q.; Wood, D.L.; Rauch, P.A. Effects of verbenone and trans-verbenol on the response of Dendroctonus brevicomis to natural and synthetic attractant in the field. J. Chem. Ecol. 1980, 6, 997–1013. [Google Scholar] [CrossRef]
- Liu, Z.; Xin, Y.; Xu, B.; Raffa, K.F.; Sun, J. Sound-triggered production of antiaggregation pheromone limits overcrowding of Dendroctonus valens attacking pine trees. Chem. Senses 2017, 42, 59–67. [Google Scholar] [PubMed]
- Seybold, S.J.; Fettig, C.J. Managing bark and ambrosia beetles (Coleoptera: Curculionidae: Scolytinae) with semiochemicals. Can. Entomol. 2021, 153, 4–12. [Google Scholar] [CrossRef]
- Sullivan, B.T.; Clarke, S.R. Semiochemicals for management of the southern pine beetle (Coleoptera: Curculionidae: Scolytinae): Successes, failures, and obstacles to progress. Can. Entomol. 2021, 153, 36–61. [Google Scholar] [CrossRef]
- Sullivan, B.T.; Munro, H.L.; Shepherd, W.P.; Gandhi, K.J.K. 4-allylanisole as a lure adjuvant for Dendroctonus frontalis (Coleoptera: Curculionidae: Scolytinae) and two associated beetles. J. Appl. Entomol. 2022, 146, 813–822. [Google Scholar] [CrossRef]
- Stadelmann, G.; Bugmann, H.; Meier, F.; Wermelinger, B.; Bigler, C. Effects of salvage logging and sanitation felling on bark beetle (Ips typographus L.) infestations. For. Ecol. Manag. 2013, 305, 273–281. [Google Scholar] [CrossRef]
- Huber, D.P.; Fettig, C.J.; Borden, J.H. Disruption of coniferophagous bark beetle (Coleoptera: Curculionidae: Scolytinae) mass attack using angiosperm nonhost volatiles: From concept to operational use. Can. Entomol. 2021, 153, 19–35. [Google Scholar] [CrossRef]
- Schlyter, F.; Jakuš, R.; Han, F.-Z.; Ma, J.-H.; Kalinová, B.; Mezei, P.; Sun, J.-H.; Ujhelyiová, L.; Zhang, Q.-H. Reproductive isolation of Ips nitidus and I. shangrila in mountain forests of Western China: Responses to chiral and achiral candidate pheromone components. J. Chem. Ecol. 2015, 41, 678–688. [Google Scholar] [CrossRef]
- Gaylord, M.L.; Audley, J.P.; McMillin, J.D.; Fettig, C.J. Acetophenone and green leaf volatiles do not enhance the efficacy of verbenone for inhibiting attraction of Ips pini (Coleoptera: Curculionidae) to pheromone-baited traps in northern Arizona. J. Econ. Entomol. 2023, 116, 632–636. [Google Scholar] [CrossRef]
- Fettig, C.J.; Burnside, R.E.; Hayes, C.J.; Kruse, J.J.; Lisuzzo, N.J.; McKelvey, S.R.; Mori, S.R.; Nickel, S.K.; Schultz, M.E. Factors influencing northern spruce engraver colonization of white spruce slash in interior Alaska. For. Ecol. Manag. 2013, 289, 58–68. [Google Scholar] [CrossRef]
- Miller, D.; Borden, J. The use of monoterpenes as kairomones by Ips latidens (LeConte)(Coleoptera: Scolytidae). Can. Entomol. 1990, 122, 301–307. [Google Scholar] [CrossRef]
- Gitau, C.W.; Bashford, R.; Carnegie, A.J.; Gurr, G.M. A review of semiochemicals associated with bark beetle (Coleoptera: Curculionidae: Scolytinae) pests of coniferous trees: A focus on beetle interactions with other pests and their associates. For. Ecol. Manag. 2013, 297, 1–14. [Google Scholar] [CrossRef]
- Jactel, H.; Van Halder, I.; Menassieu, P.; Zhang, Q.H.; Schlyter, F. Non-host volatiles disrupt the response of the stenographer bark beetle, Ips sexdentatus (Coleoptera: Scolytidae), to pheromone-baited traps and maritime pine logs. Integr. Pest Manag. Reviews 2001, 6, 197–207. [Google Scholar] [CrossRef]
- Yejing, L.; Kong, X.; Sufang, Z.; Wang, H.; Zhang, Z.; Li, C.; Jiao, X.; Huo, T. Allelochemical effects on aggregation behaviors of Ips subelongatus (Coleoptera: Scolytidae). Sci. Silvae Sin. 2016, 52, 107–114. [Google Scholar]
- Shea, P.J.; Neustein, M. Protection of a rare stand of Torrey pine from Ips paraconfusus. In Application of Semiochemicals for Management of Bark Beetle Infestations: Proceedings of an Informal Conference; Department of Agriculture Forest Service: Ogden, UT, 1995; pp. 39–43. [Google Scholar]
- Dickens, J.; Billings, R.; Payne, T. Green leaf volatiles interrupt aggregation pheromone response in bark beetles infesting southern pines. Experientia 1992, 48, 523–524. [Google Scholar] [CrossRef]
- Birgersson, G.; Dalusky, M.J.; Espelie, K.E.; Berisford, C.W. Pheromone production, attraction, and interspecific inhibition among four species of Ips bark beetles in the Southeastern USA. Psyche 2012, 2012, 532652. [Google Scholar] [CrossRef]
- Sullivan, B.T. Host odour alpha-pinene increases or reduces response of Ips avulsus (Coleoptera: Curculionidae: Scolytinae) to its aggregation pheromone, depending on separation of release points. Can. Entomol. 2023, 155, e4. [Google Scholar] [CrossRef]
- Wu, C.X.; Liu, F.; Zhang, S.F.; Kong, X.B.; Zhang, Z. Semiochemical regulation of the intraspecific and interspecific behavior of Tomicus yunnanensis and Tomicus minor during the shoot-feeding phase. J. Chem. Ecol. 2019, 45, 227–240. [Google Scholar] [CrossRef]
- Kandasamy, D.; Gershenzon, J.; Hammerbacher, A. Volatile organic compounds emitted by fungal associates of conifer bark beetles and their potential in bark beetle control. J. Chem. Ecol. 2016, 42, 952–969. [Google Scholar] [CrossRef]
- Fettig, C.J.; Audley, J.P.; Homicz, C.S.; Progar, R.A. Applied chemical ecology of the western pine beetle, an important pest of ponderosa pine in western north America. Forests 2023, 14, 757. [Google Scholar] [CrossRef]
- Gaylord, M.L.; McKelvey, S.R.; Fettig, C.J.; McMillin, J.D. Verbenone inhibits attraction of Ips pini (Coleoptera: Curculionidae) to pheromone-baited traps in northern Arizona. J. Econ. Entomol. 2020, 113, 3017–3020. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Martínez, G.; Mehmel, C.J.; González-Gaona, E.; Mori, S.R.; López-Hernández, J.A.; Monárrez-González, J.C.; García-Rodríguez, J.L.; Mejía-Bojórquez, J.M.; Gillette, N.E. 3-Methylcyclohex-2-en-1-one reduces the aggregation of Dendroctonus pseudotsugae barragani and corresponding mortality of Pseudotsuga menziesii in northern Mexico. Agric. For. Entomol. 2022, 24, 344–354. [Google Scholar] [CrossRef]
- Aukema, B.H.; Raffa, K.F. Selective manipulation of predators using pheromones: Responses to frontalin and ipsdienol pheromone components of bark beetles in the Great Lakes region. Agric. For. Entomol. 2005, 7, 193–200. [Google Scholar] [CrossRef]
- Lindmark, M.; Ganji, S.; Wallin, E.A.; Schlyter, F.; Unelius, C.R. Semiochemicals produced by fungal bark beetle symbiont Endoconidiophora rufipennis and the discovery of an anti-attractant for Ips typographus. PLoS ONE 2023, 18, e0283906. [Google Scholar] [CrossRef]
- Jactel, H.; Brockerhoff, E.G. Tree diversity reduces herbivory by forest insects. Ecol. Lett. 2007, 10, 835–848. [Google Scholar] [CrossRef]
- Jactel, H.; Birgersson, G.; Andersson, S.; Schlyter, F. Non-host volatiles mediate associational resistance to the pine processionary moth. Oecologia 2011, 166, 703–711. [Google Scholar] [CrossRef]
- Erbilgin, N.; Raffa, K.F. Opposing effects of host monoterpenes on responses by two sympatric species of bark beetles to their aggregation pheromones. J. Chem. Ecol. 2000, 26, 2527–2548. [Google Scholar] [CrossRef]





| Categorical Variable | Dendroctonus | Ips | ||||
|---|---|---|---|---|---|---|
| k | Qb | p-Value | k | Qb | p-Value | |
| Genus | 624 | - | - | 239 | - | - |
| Species | 624 | 148.11 | <0.0001 | 239 | 99.55 | <0.0001 |
| Treatment region | 624 | 40.34 | <0.0001 | 239 | 24.99 | 0.0008 |
| Treatment Source | 621 | 345.86 | <0.0001 | 239 | 15.68 | 0.0282 |
| Reduction (Repellence response) | 624 | 32.41 | <0.0001 | 239 | 6.89 | 0.075 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Afzal, S.; Nahrung, H.F.; Lawson, S.A.; Hayes, R.A. How Effective Are Push–Pull Semiochemicals as Deterrents for Bark Beetles? A Global Meta-Analysis of Thirty Years of Research. Insects 2023, 14, 812. https://doi.org/10.3390/insects14100812
Afzal S, Nahrung HF, Lawson SA, Hayes RA. How Effective Are Push–Pull Semiochemicals as Deterrents for Bark Beetles? A Global Meta-Analysis of Thirty Years of Research. Insects. 2023; 14(10):812. https://doi.org/10.3390/insects14100812
Chicago/Turabian StyleAfzal, Somia, Helen F. Nahrung, Simon A. Lawson, and Richard Andrew Hayes. 2023. "How Effective Are Push–Pull Semiochemicals as Deterrents for Bark Beetles? A Global Meta-Analysis of Thirty Years of Research" Insects 14, no. 10: 812. https://doi.org/10.3390/insects14100812
APA StyleAfzal, S., Nahrung, H. F., Lawson, S. A., & Hayes, R. A. (2023). How Effective Are Push–Pull Semiochemicals as Deterrents for Bark Beetles? A Global Meta-Analysis of Thirty Years of Research. Insects, 14(10), 812. https://doi.org/10.3390/insects14100812






