1. Introduction
The plastics industry has expanded significantly over the last seven decades, both in terms of production and utilization in nearly all aspects of our everyday life. This is because plastics have evolved exceptional characteristics over time, including affordability, stability, and durability [
1,
2,
3]. Synthetic plastics are made from various hydrocarbons and petroleum derivatives and have a larger molecular weight because of small, repeated monomer units [
4,
5]. Among them, polyethylene (PE) alone makes up 64% of all plastics [
6,
7]. The plastics industry is expected to expand in the upcoming years, reaching an estimated value of about
$1050 billion by 2033. According to current growth trends, the production of plastics is expected to double within the next twenty years [
8,
9].
Pollution from plastic waste is now widely recognized as a serious environmental problem. The main issue with plastics is that they are extremely long-lasting and can take hundreds of years to degrade [
10,
11]. However, only around half of all plastic waste produced has been either recycled or dumped in landfills [
12]. A significant amount of the leftover plastic trash pollutes continents, oceans, and every part of the globe, transforming it into a ‘Plastic World’ [
13,
14]. During disposal, plastic debris release chemicals that are capable of leaching into the soil and contaminating the groundwater and soil [
15,
16]. The deposition of this debris in soil can lead to changes in drainage patterns, disruption of soil fauna, degradation of soil quality, and ultimately reduced agricultural yields. Moreover, microplastics (MPs) that are produced during plastic degradation have a size of less than 5 mm and have the potential to cause eco-toxicological effects [
17,
18,
19,
20]. Additionally, fibrous MPs have the potential to cause health impacts like carcinogenicity and mutagenicity because they can be breathed and may accumulate in the lungs [
20,
21].
Conventional plastic management approaches mainly rely on certain disposal methods mainly including landfilling, incineration, and recycling. In the early 2000s, the majority of plastic waste was managed through landfilling (65–70%) and incineration (20–25%), while only about 10% was recycled. However, this distribution varies by country, depending on factors such as the standard of living and population [
22,
23]. Despite these methods, a significant portion of plastic waste produced remains unattended. Moreover, landfills and incineration can lead to serious environmental problems through emission of hazardous pollutants, including dioxins, metals, carbon monoxide (CO), furans, and volatile organic compounds [
24]. These ecological constraints make these approaches less effective in addressing this global issue [
24,
25].
In light of these problems, it is vital to seek environmentally friendly alternatives to tackle this issue, such as biodegradation [
2,
26]. Plastic biodegradation is an eco-friendly and cost-effective approach to combatting plastic pollution [
27]. Biodegradation refers to the process of physical and chemical changes occurring within a substance because of the activity of microorganisms [
28]. According to recent research, the larvae of the Greater Wax Moth,
Galleria mellonella L. (Lepidoptera: Pyralidae), have been found to be capable of biodegrading low-density PEs [
29,
30].
Galleria mellonella is a serious insect pest of the beekeeping industry due to its destructive feeding habits by tunneling in the comb and feeding on pollen, honey, beeswax, and brood [
31,
32]. In addition to being a major pest in apiculture, its larvae can also feed on PE. Its larvae break down PE and convert it to ethylene glycol. PE degradation by
G. mellonella larvae is attributed to the similar chemical structure of beeswax and PE. Beeswax is made of long-chain hydrocarbons, a complex mixture of lipid compounds that include alkanes, alkenes, fatty acids, and esters. As in PE, the most common hydrocarbon bond is CH
2-CH
2 [
33,
34]. The metabolic pathways involved in the degradation of long-chain hydrocarbons are expected to play an important role in the degradation of PE, which is composed of a long aliphatic chain [
35]. Since
G. mellonella larvae feed on and metabolizes long-chain hydrocarbons from beeswax, they may also potentially metabolize PE.
One hundred larvae of
G. mellonella inflicted 92 mg weight loss in PE even after 12 h [
33]. Gut enzymes and gut microbiota play a key role in PE degradation. The saliva of
G. mellonella larvae contains two enzymes from the phenol oxidase family, demonstrating their capability to effectively degrade polyethylene [
36]. Yang et al. [
37] isolated two bacterial strains,
Enterobacter asburiae YT1 and
Bacillus sp. YP1, from the gut microbiota of
G. mellonella larvae consuming PE. Approximately 6% and 11% of PE film was degraded by YT1 and YP1, respectively, after 60-day incubation. Likewise, different species of the genus
Enterobacter and
Acinetobacter, isolated from the guts of
G. mellonella, were found to be degrading PE [
38,
39].
The current study is novel in terms of evaluating the feeding efficiency of G. mellonella larvae on polyethylene (PE) of four different densities (1 mm, 2 mm, 3 mm, and 4 mm) with and without co-diet supplementation (wheat germ plus honey and beeswax) at three specific time intervals (24, 48, and 72 h). Weight loss in both G. mellonella larvae and PE, reduction in PE surface area, and diet consumption were investigated. This study advances our understanding relating to the biodegradation potential of G. mellonella larvae across different PE densities and explores effective combinations of co-diet supplementation.
4. Discussion
Polyethylene is the most widely used synthetic polymer globally. However, its resistance to natural degradation has led to its accumulation in landfills and the environment [
45,
46,
47]. Consequently, there is a significant need to identify effective methods to expedite its biodegradation process, which has been a focal point of extensive research in recent years. Recently, the larvae of the
G. mellonella, a significant pest in the bee industry, have been recognized as ‘plastivores’ [
48,
49]. These larvae exhibit the remarkable ability to consume and metabolize PE [
50]. In our study, we used PEs of different densities alone and in combination with beeswax and wheat germ + honey against the larvae of the
G. mellonella. The results show that the highest weight loss of PE and larvae was observed when larvae were provided with PE alone. A significant reduction in the PE mass was also observed when larvae fed on PE in combination with beeswax, while the larvae experienced minimal weight loss. The larvae exhibited a higher PE consumption with a thickness of 1 mm, indicating that the smaller the density of PE, the greater the consumed area, while the lowest consumption of PE was with a density of 4 mm. The biodegradation level was notably higher at 24 h.
The biodegradation of PE could be attributed to the gut microbiota of
G. mellonella larvae, which contain bacteria and fungi from various genera. Noel et al. [
51] observed that the addition of low-density polyethylene does not significantly alter the bacterial microbiota of
G. mellonella larvae at either the community or taxonomic levels, indicating the resilience of the bacterial microbiome. Similarly, Yang et al. [
14] isolated two bacterial strains,
Enterobacter asburiae YT1 and
Bacillus sp. YP1, from the gut microbiota of
G. mellonella larvae consuming PE. Likewise, bacteria from genus
Acinetobacter and
Enterobacter sp. D1, were isolated from the guts of
G. mellonella, possessing the ability to degrade PE [
33,
34]. On the other hand, the fungus
Aspergillus flavus was also found to degrade high-density MP particles into low-molecular-weight MP particles [
52]. Researchers also accept that the saliva of
G. mellonella larvae exhibits the ability to oxidize and depolymerize polyethylene. Within the saliva, two enzymes from the phenol oxidase family have been identified, demonstrating their capability to effectively degrade polyethylene [
36].
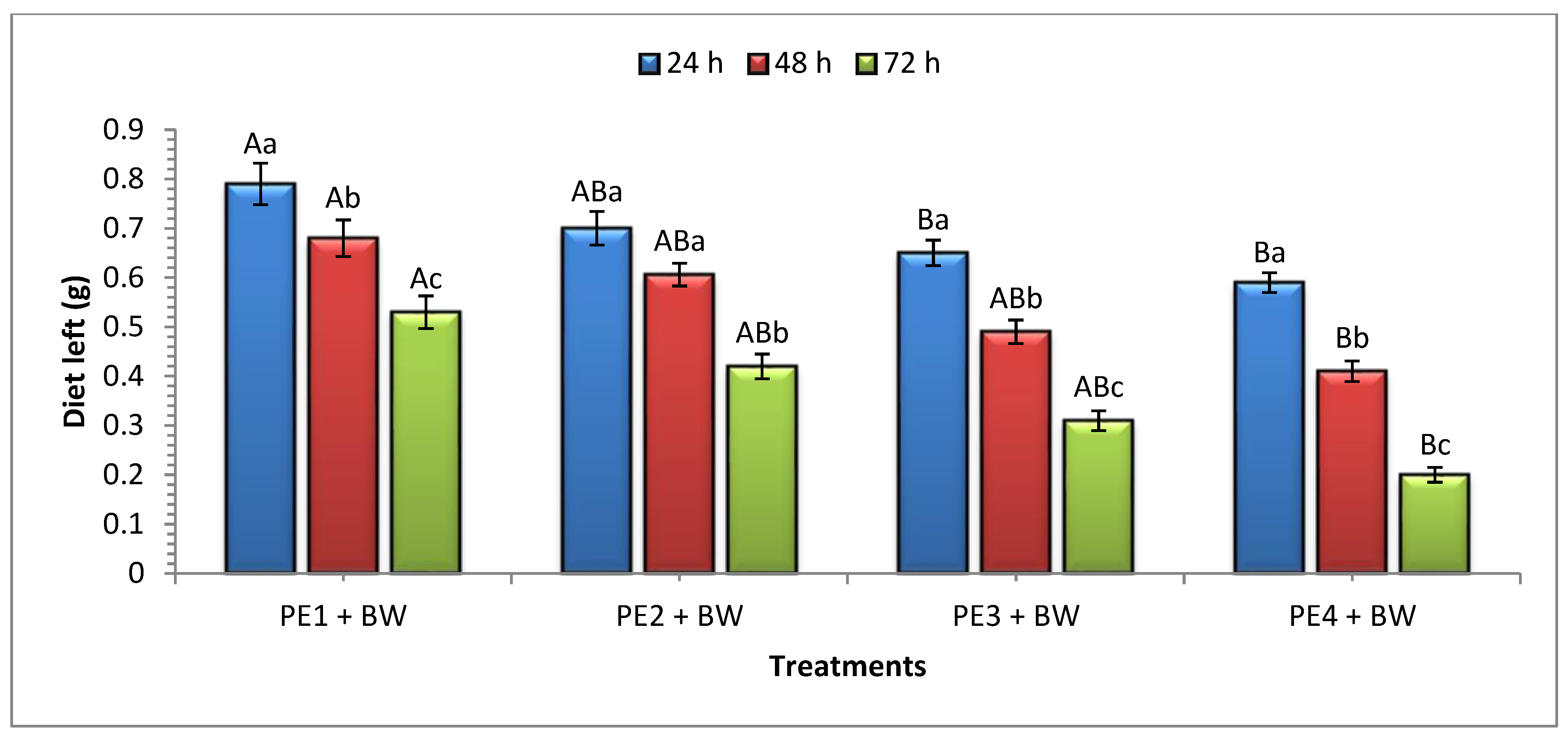
In our study, when examining the consumption rate on the first day of the experiment, it was observed that ten larvae on 1 mm plastic (PE
1) consumed 9.98 mg of PE, resulting in a larval weight loss of 27.29 mg. In comparison, 5.87 mg of PE was consumed in combination with beeswax (PE
1 + BW), leading to a larval weight loss of 9.59 mg in 24 h. In a comparable study by Bombelli et al. [
33], it was noted that 100
G. mellonella larvae could biodegrade 92 mg of polyethylene in 12 h. Despite extending the duration to 24 h in our study, we utilized only 10 larvae and achieved a greater amount of biodegraded PE. Alkassab et al. [
53] reported that 10 larvae of
G. mellonella could degrade 0.0210 mg in 12 h at 25 °C. Mandal and Vishwakarma [
54] demonstrated that 480 larvae of the
G. mellonella, conditioned at a temperature of 27 °C biodegraded 0.173 mg of PE over a period of 7 days. In their study, the biodegradation efficiency was lower compared to the findings in our research, even though the treatment duration was longer. They also noted that there was no correlation between relative humidity and larval growth.
However, our research revealed that
G. mellonella larvae exhibited a higher biodegradation rate of low-density PE at 24 h compared to 48 and 72 h. This difference could be attributed to the notable number of larvae in Petri dishes at 24 h, where they actively fed. This behavior differed from the observations at 48 and 72 h when larvae were dispersed and did not consume PE in the same manner. Kwadha et al. [
55] noted that larvae tend to feed more when they are in close proximity. Additionally, the larvae observed at 24 h were more active compared to those at 48 and 72 h, as they were entering the pupation stage [
56]. In comparison to these studies, our research demonstrated better results in terms of weight loss in low density polyethylene (LDPE) biodegradation, and the duration of the study was relatively shorter. Additionally, temperature emerged as a significant factor influencing LDPE biodegradation. Alkassab et al. [
53] conducted experiments with
G. mellonella larvae at 25 °C and 35 °C, highlighting that the optimal temperature for biodegradation is 25 °C. According to Rodríguez Vega [
56],
G. mellonella larvae exhibit normal development at a temperature of 25 °C, with a lifespan of around 28 days. Deviations from this temperature can affect their activity and lifespan: lower temperatures slow down activity and prolong lifespan, while higher temperatures accelerate activity and significantly shorten lifespan. Consequently, our research was conducted at a temperature of 25 °C.
In our study involving the co-diet supplementation of
G. mellonella, we employed beeswax, wheat germ, and honey. In previous studies, co-diet supplementation with beeswax resulted in a higher PE consumption [
54,
57]. This aligns with our findings, as the treatment with beeswax yielded better results, possibly because under natural conditions, these larvae typically feed on beeswax, and their bodies are accustomed to it [
55]. Furthermore, it was observed that the growth of
G. mellonella was unaffected by the ingestion of polyethylene because the composition and chemical structure of LDPE closely resemble those of beeswax [
33]. For future research projects, evaluations of the pupation rate and survival of tested insects could offer additional insights into the ecology of insect species when exposed to polyethylene (PE). Investigating these factors would improve our understanding of how PE affects both pupation and survival rates of tested insects.
Decisively, it has become an established fact that using insects to degrade plastics does not create secondary pollution, and these insects can latterly also be used as poultry and fish feed, providing both practical and economic benefits. However, using insects for plastic degradation is still an emerging field, and the current findings are not yet ready for large-scale application. Future research should focus on high-throughput sequencing to analyze the microbes in the insects’ guts, understanding how they break down plastics, and discovering new ways to improve this process. Moreover, policymakers must support the integration of researcher from diverse fields such as the environment and poultry and fish industries to obtain greater and sustainable benefits via insect rearing on plastics.