The Manufacture of Synthetic Rutile by Solvent Extraction of Tri-Alkyl Phosphine Oxide from HCl Leaching Solution of Soda-Roasted Ilmenite Ore
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Procedure
2.2.1. Preparation of Feed Solution for Solvent Extraction of Ti
2.2.2. Solvent Extraction Experiment
2.2.3. Hydrolysis
3. Results and Discussions
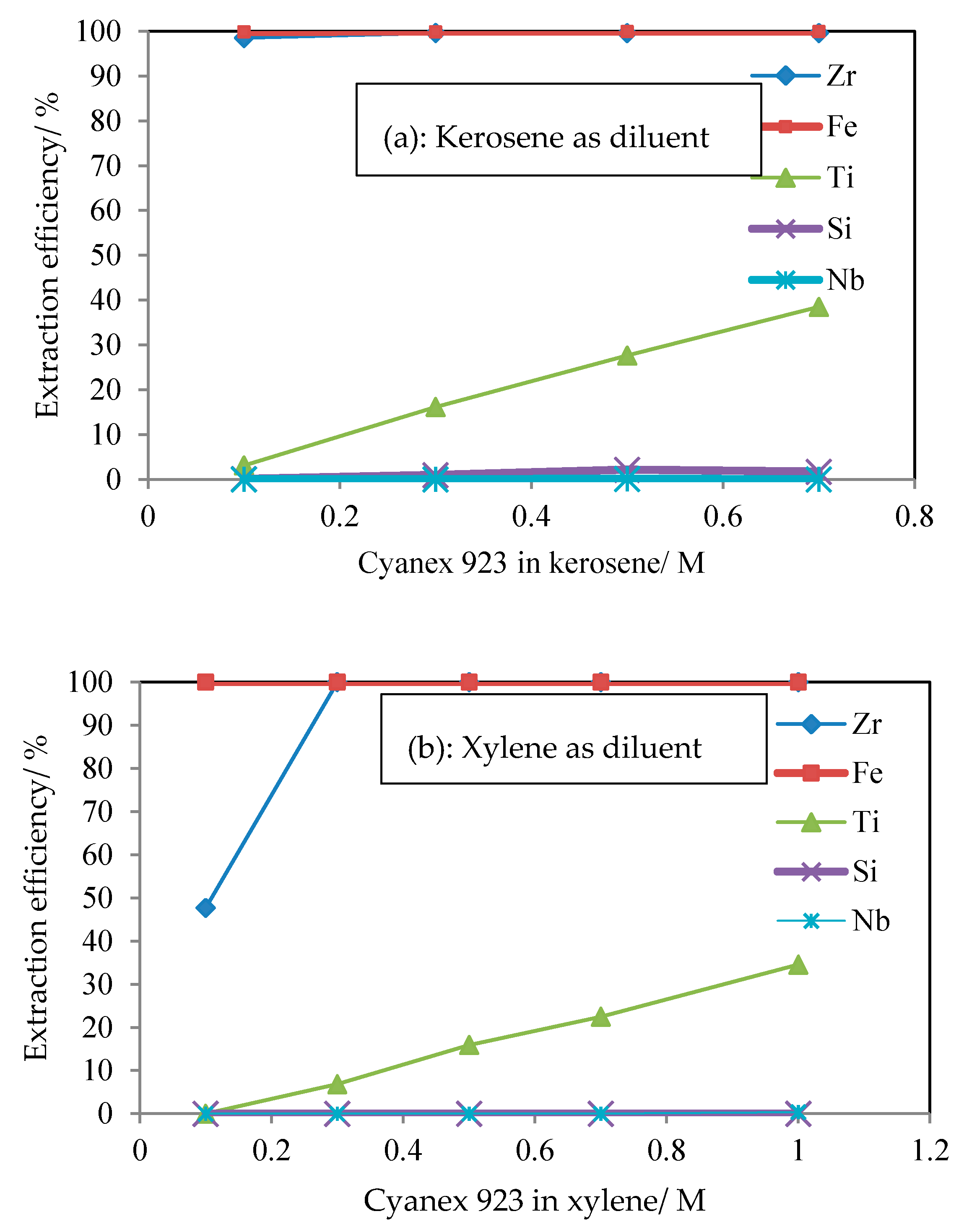
3.1. Effect of Diluents on Extraction of Ti by Cyanex 923
3.2. Extraction of O/A Ratio
3.3. Effect of Stripping Solution for Selective Recovery of Ti
3.4. Stripping of O/A Ratio
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wang, C.P.; Wang, H.Z.; Ruan, G.L.; Wang, S.H.; Xiao, Y.X.; Jiang, L.D. Applications and prospects of titanium and its alloys in seawater desalination industry. Mater. Sci. Eng. 2019, 688, 033036. [Google Scholar] [CrossRef]
- Veiga, C.; Davim, J.P.; Loureiro, A.J.R. Properties and applications of titanium alloys: A brief review. Rev. Adv. Mater. Sci. 2012, 32, 14–34. [Google Scholar]
- Zhang, W.; Zhu, Z.; Cheng, C.Y. A literature review of titanium metallurgical processes. Hydrometallurgy 2011, 108, 177–188. [Google Scholar]
- Shin, D.J.; Joo, S.-H.; Lee, D.S.; Kang, J.S.; Kim, M.-S.; Park, J.-T.; Min, D.J.; Shin, S.M. Leaching Behavior of Titanium from Na2TiO3. Mater. Trans. 2020, 61, 150–155. [Google Scholar]
- Bedinger, G.M. Mineral Commodity Summaries: Titanium Mineral Concentrates; U.S. Geological Survey: Washington, DC, USA, 2013; p. 174175. Available online: http://minerals.usgs.gov/minerals/pubs/commodity/titanium/mcs-2013-timin.pdf (accessed on 1 February 2020).
- Becher, R.G.; Canning, R.G.; Goodheart, B.A.; Uusna, S. A new process for upgrading Hmenitic mineral sands. Proc. Aust. Inst. Min. Metall. 1965, 21, 21–44. [Google Scholar]
- Hoecker, W. Process for the Production of Synthetic Rutile. European Patent EP0612854, 31 August 1994. [Google Scholar]
- Guéguin, M.; Cardarelli, F. Chemistry and mineralogy of titania-rich slags. Part 1—Hemo-ilmenite, sulphate, and upgraded titania slags. Extr. Miner. Process. Metall. Rev. 2007, 28, 1–58. [Google Scholar]
- Akhtar, M.K.; Vemury, S.; Pratsinis, S.E. Competition between TiCl4 hydrolysis and oxidation and its effect on product TiO2 powder. AIChE J. 1994, 40, 1183–1192. [Google Scholar]
- Kroll, W. The Production of Ductile Titanium. Trans. Electrochem. Soc. 1940, 78, 35–47. [Google Scholar] [CrossRef]
- Kang, J.; Moon, H.; Kim, M.-S.; Okabe, T.H. Production of High-grade Titanium Dioxide from Titanium Ore Using Titanium Scrap and Iron Chloride Waste. Met. Mater. Int. 2019, 25, 257–267. [Google Scholar] [CrossRef]
- Habashi, F. Ilmenite for pigment and metal production. Interdiscip. J. Chem. 2016, 1, 28–33. [Google Scholar]
- Habashi, F.; Kamaleddine, F.; Bourricaudy, E. A New Process to Upgrade Ilmenite to Synthetic Rutile Proceedings Conference of Metallurgists, Canadian Institute of Mining, Metallurgy, and Petroleum, Montreal. Metall 2015, 69, 27–30. [Google Scholar]
- Zhang, J.; Zhu, Q.; Xie, Z.; Li, H. Influence of redox pretreatment on the pulverization of panzhihua ilmenite during hydrochloric acid leaching. Hydrometallurgy 2015, 157, 226–233. [Google Scholar] [CrossRef]
- Guo, T.; Liu, S.; Jiang, T.; Qiu, G.; Chen, F. A process for producing synthetic rutile from Panzhihua titanium slag. Hydrometallurgy 2014, 147–148, 134–141. [Google Scholar] [CrossRef]
- Jia, L.; Liang, B.; Lü, L.; Yuan, S.; Zheng, L.; Wang, X.; Li, C. Beneficiation of titania by sulfuric acid pressure leaching of Panzhihua ilmenite. Hydrometallurgy 2014, 59, 92–98. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, L.; Qi, T.; Chen, D.; Zhao, H.; Liu, Y. A novel method to extract iron, titanium, vanadium, and chromium from high-chromium vanadium-bearing titanomagnetite concentrates. Hydrometallurgy 2014, 149, 106–109. [Google Scholar] [CrossRef]
- Zhu, Q.; Zhang, J.; Li, H. Influence of phase and microstructure on the rate of hydrochloric acid leaching in pretreated Panzhihua ilmenite. Particuology 2014, 14, 83–90. [Google Scholar]
- Janssen, A.; Putnis, A. Processes of oxidation and HCl-leaching of Tellnes ilmenite. Hydrometallurgy 2011, 109, 194–201. [Google Scholar]
- Zhang, L.; Hu, H.; Liao, Z.; Chen, Q.; Tan, J. Hydrochloric acid leaching behavior of different treated Panxi ilmenite concentrations. Hydrometallurgy 2011, 107, 40–47. [Google Scholar] [CrossRef]
- Saji, J.; Reddy, M.L.P. Selective Extraction and Separation of Titanium(IV) from Multivalent Metal Chloride Solutions Using 2-Ethylhexyl Phosphonic Acid Mono-2-ethylhexyl Ester. Sep. Sci. Technol. 2003, 38, 427–441. [Google Scholar]
- Borsalani, A.; Ghahremani, H.; Seyfi, S.; Abdi, M. Solvent Extraction of tetravalent titanium from chloride and nitratesolutions by 2-ethylhexyl phosphonic acid mono-2-ethylhexyl ester(EHEHPA) in Kerosene. Chem. Sin. 2011, 2, 204–211. [Google Scholar]
- Saji John, K.; Rao, T.P.; Ramamohan, T.R.; Reddy, M.L.P. Solvent extraction of tetravalent titanium from acidic chloride solutions by 2-ethylhexyl phosphonic acid mono-2-ethylhexyl ester. Hydrometallurgy 1999, 53, 245–253. [Google Scholar] [CrossRef]
- Biswas, R.K.; Begum, D.A. Solvent extraction of tetravalent titanium from chloride solution by di-2-ethylhexyl phosphoric acid in kerosene. Hydrometallurgy 1998, 49, 263–274. [Google Scholar] [CrossRef]
- Awwada, N.S.; Ibrahium, H.A. Kinetic extraction of titanium (IV) from chloride solution containing Fe(III), Cr(III) and V(V) using the single drop technique. J. Environ. Chem. Eng. 2013, 1, 65–72. [Google Scholar]
- SINGH, R.K.; DHADKE, P.M. Extraction and separation of titanium(IV) with D2EHPA and PC-88A from aqueous perchloric acid solutions. J. Serb. Chem. Soc. 2002, 67, 507–521. [Google Scholar]
- John, K.S.; Saji, J.; Reddy, M.L.P.; Ramamohan, T.R.; Rao, T.P. Solvent extraction of titanium(IV) from acidic chloride solutions by Cyanex 923. Hydrometallurgy 1999, 51, 9–18. [Google Scholar] [CrossRef]
- MAO, X.H.; LIU, D.J. Solvent Extraction Separation of Titanium(IV) and Iron(III) from Acid Chloride Solutions by Trioctylphosphine Oxide. Asian J. Chem. 2013, 25, 4753–4756. [Google Scholar]
- Allal, K.M.; Hauchard, D.; Stambouli, M.; Pareau, D.; Durand, G. Solvent extraction of titanium by tributylphosphate, trioctylphosphine oxide and decanol from chloride media. Hydrometallurgy 1997, 45, 113–128. [Google Scholar] [CrossRef]
- Seyfi, S.; Abdi, M. Extraction of titanium (IV) from acidic media by tri-n-butyl phosphate in kerosene. Miner. Eng. 2009, 22, 116–118. [Google Scholar]
- Baba, A.A.; Adekola, F.A.; Arodola, O.A.; Ibrahim, L.; Bale, R.B.; Ghosh, M.K.; Sheik, A.R. Simultaneous recovery of total iron and titanium from ilmenite ore by hydrometallurgical processing. Metall. Mater. Eng. 2012, 18, 67–78. [Google Scholar]
- Zaki, S.A. Preparation of pigment grade TiO2 from Rosetta ilmenite via selective TOPO extraction of titanium from its hydrochloric acid solution. Inorg. Chem. 2014, 9, 085–094. [Google Scholar]
- Middlemas, S.; Fang, Z.Z.; Fan, P. A new method for production of titanium dioxide pigment. Hydrometallurgy 2013, 131–132, 107–113. [Google Scholar]
- Wang, X.; Liang, B.; Lü, L.; Wu, P.; Li, C.; Ma, J. Simultaneous oxidation and extraction of iron from simulated ilmenite hydrochloric acid leachate. Hydrometallurgy 2012, 129–130, 105–110. [Google Scholar]
- Kang, J.S.; Okabe, T.H. Removal of Iron from Titanium Ore through Selective Chlorination Using Magnesium Chloride. Mater. Trans. 2013, 54, 1444–1453. [Google Scholar]
- Sato, T. Liquid-Liquid Extraction of Titanium (IV) from Hydrochloric Acid Solution by Di-(2-Ethylhexyl)-Phosphoric Acid. J. Min. Mater. Process.Inst. Jpn. 2003, 119, 175–181. [Google Scholar]
- Saji, J.; John, K.S.; Reddy, M.L.P. Liquid-liquid extraction of tetravalent titanium from acidic chloride solutions by bis(2,4,4-trimethylpentyl)phosphinicacid. Solvent Extrac. Ion Exchan. 2000, 18, 877–894. [Google Scholar]
- Remya, P.N.; Reddy, M.L.P. Solvent extraction separation of titanium(IV), vanadium(V) and iron(III) from simulated waste chloride liquors of titanium minerals processing industry by the trialkylphosphine oxide Cyanex 923. J. Chem. Technol. Biotechnol. 2004, 79, 734–741. [Google Scholar]





| Element | Ti | Si | Nb | Fe | Zr |
|---|---|---|---|---|---|
| Feed solution | 32,050 | 110 | 88 | 2614 | 130 |
| Cyanex 923, M | 0.1 | 0.3 | 0.5 | 0.7 | 1 |
|---|---|---|---|---|---|
| Kerosene | 440 | 1540 | 1900 | 2200 | - |
| Xylene | - | 3060 | 4950 | 5490 | 6870 |
| O/A | Element | Ti | Si | Nb | Fe | Zr |
|---|---|---|---|---|---|---|
| 3 | 1 stage | 26,700 | 109 | 84 | 4 | 2 |
| 2 stage | 11,200 | 104 | 82 | N.D | N.D | |
| 3 stage | 1160 | 102 | 81 | N.D | N.D | |
| 4 stage | 130 | 101 | 80 | N.D | N.D | |
| 5 | 1 stage | 6250 | 107 | 81 | 11 | 5 |
| 2 stage | 420 | 102 | 72 | N.D | N.D |
| Stripping Solution | Ti | Si | Nb | Zr | Fe |
|---|---|---|---|---|---|
| Distilled water 1st | 12,460 | 1.189 | 2.9 | N.D | 2.77 |
| Distilled water 2nd | 5100 | 0.19 | 2.6 | N.D | 240 |
| 0.1 M HCl 1st | 12,190 | 1.26 | 2.9 | N.D | 2.09 |
| 0.1 M HCl 2nd | 5190 | 0.19 | 2.9 | N.D | 108 |
| 0.5 M HCl 1st | 11,230 | 1.25 | 2.8 | N.D | 1.9 |
| 0.5 M HCl 2nd | 5015 | 0.3 | 0 | N.D | 7.94 |
| 1 M HCl 1st | 10,320 | 0.9 | 2.8 | N.D | N.D |
| 1 M HCl 2nd | 5000 | 0.1 | 2.9 | N.D | N.D |
| 0.1 M NaCl 1st | 8411 | 0.8 | 2.8 | N.D | 2.5 |
| 0.5 M NaCl 1st | 6506 | 0.8 | 2.4 | N.D | 2.5 |
| 1 M NaCl 1st | 4511 | 1.1 | 2.4 | N.D | 2.4 |
| 1 M NaCl 2nd | 4667 | 0.4 | 2.1 | N.D | 2.6 |
| Theoretical metal ion in the loaded organic phase | 10,640 | 3 | 3 | 42 | 871 |
| O/A | Element | Ti | Si | Nb | Fe | Zr |
|---|---|---|---|---|---|---|
| 2 (Counter current stripping) | 1 stage | 13,040 | 3.2 | 2.7 | N.D | N.D |
| 2 stage | 7950 | 1.0 | N.D | N.D | N.D | |
| 3 stage | 5400 | N.D | N.D | N.D | N.D | |
| 4 stage | 3120 | N.D | N.D | N.D | N.D | |
| 1 (Cross stripping) | 1 st | 2690 | N.D | N.D | N.D | N.D |
| 2 nd | 490 | N.D | N.D | N.D | N.D |
| Element | Ti(TiO2) | Si(SiO2) | Nb | Fe | Zr | Na | Al | Mg | Mn | Ca | V |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Purity of TiO2 | 99.9 | 0.03 | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joo, S.-H.; Shin, D.J.; Lee, D.; Kang, J.; Kim, M.-s.; Jeon, H.; Park, J.-T.; Shin, S.M. The Manufacture of Synthetic Rutile by Solvent Extraction of Tri-Alkyl Phosphine Oxide from HCl Leaching Solution of Soda-Roasted Ilmenite Ore. Metals 2020, 10, 588. https://doi.org/10.3390/met10050588
Joo S-H, Shin DJ, Lee D, Kang J, Kim M-s, Jeon H, Park J-T, Shin SM. The Manufacture of Synthetic Rutile by Solvent Extraction of Tri-Alkyl Phosphine Oxide from HCl Leaching Solution of Soda-Roasted Ilmenite Ore. Metals. 2020; 10(5):588. https://doi.org/10.3390/met10050588
Chicago/Turabian StyleJoo, Sung-Ho, Dong Ju Shin, Dongseok Lee, Jungshin Kang, Min-seuk Kim, Hoseok Jeon, Jin-Tae Park, and Shun Myung Shin. 2020. "The Manufacture of Synthetic Rutile by Solvent Extraction of Tri-Alkyl Phosphine Oxide from HCl Leaching Solution of Soda-Roasted Ilmenite Ore" Metals 10, no. 5: 588. https://doi.org/10.3390/met10050588




