Platinum Based Catalysts in the Water Gas Shift Reaction: Recent Advances
Abstract
:1. Introduction
2. Comparative Studies
2.1. Monometallic Catalysts: The Supports
2.1.1. Effect of the Preparation Method of the Ceria Support
2.1.2. Effect of the Preparation Method of the Ceria-Based Solid Solutions and Mixed Oxide Support
2.1.3. Effect of Preparation Method of Supports Different from Ceria
2.1.4. Commercial Supports
2.1.5. Conclusions
| Selected Catalyst (Particle/Crystallite Size) | Preparation Procedure | Operative Condition WGS | CO Conversion (XCO) (Temperature) | Ref. |
|---|---|---|---|---|
| 1wt%Pt/CeO2 (12 nm) | Pt loading by wet impregnation; CeO2 by microwave-assisted hydrothermal synthesis | WHSV = 40,000 mL·g−1·h−1; H2O/CO = 6 | XCO ≈ 97% (T = 360 °C) | [23] |
| 1wt%Pt/CeO2 (not specified) | Wet impregnation + Ar plasma treatment | WHSV = 40,000 mL·g−1·h−1; CO/H2O = 6 | XCO ≈ 97% (T = 280 °C) | [24] |
| 1wt%Pt/CeO2 (14.0 nm) | Pt loading by wet impregnation; CeO2 by supercritical antisolvent process | GHSV = 5000 h−1; H2O/CO = 3 | XCO ≈ 99% (T = 287 °C) | [25] |
| 1wt%Pt/CeO2 (5.8 nm) | Pt loading by wet impregnation; CeO2 by supercritical antisolvent process | WHSV = 1.13 gCO·gcat−1·h−1; H2O/CO = 3 | XCO ≈ 98% (T = 280 °C) | [26] |
| 1wt%Pt/CeO2 (7.9 nm) | Incipient wetness impregnation | GHSV = 45,625 h−1; H2O/(CH4 + CO + CO2) = 2 | XCO ≈ 87% (T = 320 °C) | [27] |
| 1wt%Pt/CeO2 (1.4 nm) | Incipient wetness impregnation | GHSV = 45,625 h−1; H2O/(CH4 + CO + CO2) = 2 | XCO ≈ 82% (T = 320 °C) | [28] |
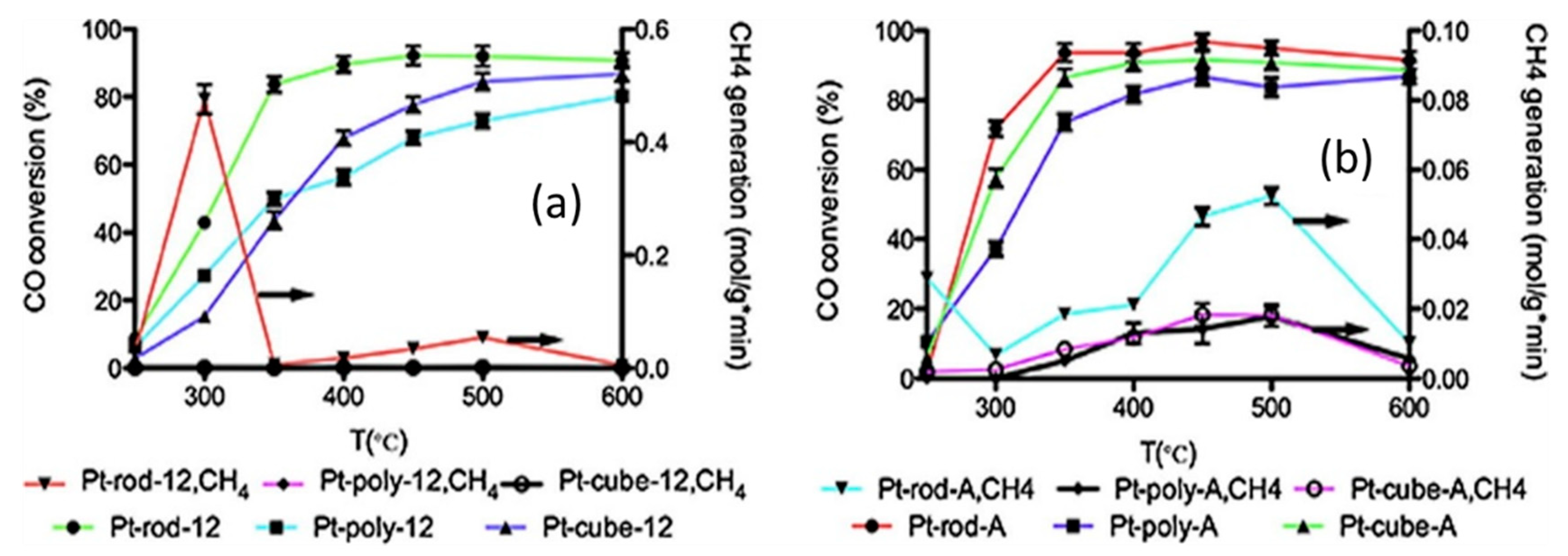
| 1wt%Pt/CeO2 nanorods (1.6 nm) | Pt loading by incipient wetness impregnation; CeO2 by hydrothermal process for 12 h | GHSV = 95,541 h−1; H2O/(CH4 + CO + CO2) = 2 | XCO ≈ 77% (T = 360 °C) | [29] |
| 3wt%Pt/CeO2-nanorod (<2 nm) | Pt by impregnation method | GHSV = 4.6 × 105 h−1; H2O/CO = 2.4 | XCO ≈ 92% (T = 600 °C) | [30] |
| 1wt%Pt/CeO2 nanofibers (4.5 nm) (dnanofibers = 80–120 nm) | Electrospinning technology | WHSV = 60,000 mL·gcat−1·h−1; H2O/CO = 5.3 | XCO ≈ 98% (T = 350 °C) | [31] |
| 1wt% Pt/Ce0.8Zr0.2O2 (1.69 nm) | Incipient wetness impregnation | GHSV = 45,515 h−1; H2O/(CH4 + CO + CO2) = 2 | XCO ≈ 86% (T = 320 °C) | [32] |
| 1Wt%Pt/CeO2/ZrO2 (7.22 nm) | Wet impregnation | GHSV = 5000 h−1; H2O/CO = 5 | Equilibrium CO conversion at 200 °C | [33] |
| 2wt%Pt/Ce0.8Fe0.2/Al2O3 (2.2 nm) | Wet impregnation | GHSV = 4000 h−1; H2O/CO = 6.9 | Equilibrium CO conversion at 280 °C | [34] |
| 1.63wt%Pt/Ce0.4Ti0.6O2 (7.25 nm) | Wet impregnation | GHSV = 3600 h−1; H2O/(CO + CO2) = 4.8 | XCO ≈ 91% (T = 400 °C) | [35] |
| 0.9wt%Pt/CeO2@SiO2-nanotube (3.1 nm) | CeO2 by hydrothermal synthesis method SiO2 shell by modified Stober method | WHSV = 36,000 mL·gcat−1·h−1; H2O/CO = 3 | XCO ≈ 30% (T = 250 °C) | [36] |
| 1wt%Pt/CeO2 (3 nm) | Incipient wetness impregnation | GHSV = 45,515 h−1; H2O/(CH4 + CO + CO2) = 2 | XCO ≈ 88% (T = 320 °C) | [38] |
| 1wt%Pt@Al2O3-nanorods (10.4 nm) | Pt loading by NaBH4 reduction Al2O3 by polymerization | GHSV = 22,500 h−1; H2O/CO = 2 | XCO ≈ 96% (T = 450 °C) | [39] |
| 1Wt%Pt/sZnOspherical morphology (1.5 nm) | Incipient wetness impregnation | GHSV = 9583 h−1; H2O/(CH4 + CO + CO2) = 2 | XCO ≈ 92% (T = 240 °C) | [40] |
| 0.5wt%Pt/Zr0.9Y0.1O1.95 (0.5–2 nm) | Incipient wetness impregnation | GHSV = 120,220 h−1; H2O/CO = 8.7 | XCO ≈ 74% (T = 300 °C) | [41] |
| 1wt%Pt/TiO2/PRGO-5 (11.3 nm) | Incipient wetness impregnation | GHSV = 47,770 h−1; H2O/(CH4 + CO + CO2) = 3.3 | XCO ≈ 81% (T = 280 °C) | [42] |
| 1wt%Pt/ZrO2-monoclinic (10.1 nm) | Incipient wetness impregnation | WHSV = 43,200 mL·gcat−1·h−1; H2O/(CO + CO2) = 3.2 | XCO ≈ 65% (T = 300 °C) | [43] |
2.2. Monometallic Catalysts: The Active Phase
2.2.1. Effect of the Preparation Method
2.2.2. Effect of the Platinum Loading
2.2.3. Comparative Studies between Pt and Rh
2.2.4. Conclusions
| Selected Catalyst (Particle/Crystallite Size) | Preparation Procedure | Operative Condition WGS | CO Conversion/Rate (XCO)/(rCO) (Temperature) | Ref. |
|---|---|---|---|---|
| 1wt%Pt/CeO2 (1.5–2 nm) | Reactive Spray Deposition Technology | GHSV = 8622 h−1; H2O/CO = 3 | Equilibrium CO (T = 350 °C) | [44] |
| 3wt%Pt/CeO2 (1.7 nm) | Pt loading by photochemical method adding PVP and 4-benzyolbenzoic acid; CeO2 by co-electrospinning | WHSV = 1.2 × 105 mL·g−1·h−1; H2O/CO = 5 | XCO ≈ 95% (T = 450 °C) | [45] |
| BaCe0.96Pt0.04O(3-δ) (<100 nm) | Citrate-gel method | GHSV = 5000 h−1; H2O/CO = 4.5 | XCO ≈ 86% (T = 400 °C) | [46] |
| 0.9wt%Pt/CeO2 (0.8 nm) | Hydrothermal method | GHSV = 5000 h−1; H2O/CO = 1 | XCO ≈ 97% (T = 140 °C) | [47] |
| 2wt%Pt/Ce0.75Zr0.25O2 (7.45 nm) | Yolk−shell microspheres formation by a spray pyrolysis process | GHSV = 18,193 h−1; H2O/(CH4 + CO + CO2) = 2 | XCO ≈ 89% (T = 320 °C) | [48] |
| 1.2wt%Pt/La2O3·SiO2 | Incipient wetness impregnation | GHSV = 2.8 × 106 h−1; H2O/CO = 3 | rCO = 350 mol·g−1·min−1 (T = 400 °C) | [49] |
| Pt@TiO2 (1.0 nm) | Yolk-shell nanospheres by a reverse micelle system | WHSV = 40,000 mL·gcat−1·h−1; H2O/CO = 5 | XCO ≈ 99%, T = 260 °C) | [50] |
| 2.2wt%Pt_NaA (not specified) | NaA zeolite by hydrothermal syntesis with conventional heating; Pt loading by encapsulation | GHSV = 6421 h−1; H2O/CO = 2 | XCO ≈ 96% (T = 400 °C) | [51] |
| 0.5wt%Pt/CeO2 (1.3 nm) | By wet impregnation | H2O/(CO + CO2) = 2.5 | XCO ≈ 45% (T = 275 °C) | [52] |
| 3.7wt% Pt/CeO2 (2.3 nm) | Flamespray pyrolisis method | WHSV = 5 × 104 mL·g−1·h−1; H2O/CO = 4 | XCO ≈ 97% (T = 250 °C) | [54] |
| 0.6wt%Pt/La2O3·SiO2 (not specified) | By Incipient wetness impregnation | WHSV = 6–24 × 103 mL·g−1·h−1; H2O/CO = 3 | XCO ≈ 95% (T = 400 °C) | [55] |
2.3. Polymetallic Catalysts and Addition of Promoters: The Active Phase
2.3.1. The Addition of Na, Re, Mo, V and Ni
2.3.2. Comparative Studies between Multiple Metals
| Selected Catalyst (Particle/Crystallite Size) | Preparation Procedure | Operative Condition WGS | CO Conversion/Rate (XCO)/(r) (Temperature) | Ref. |
|---|---|---|---|---|
| 1wt%Pt-2wt%Na/CeO2 (2.2 nm) | By incipient wetness impregnation | GHSV = 45,515 h−1; H2O/(CH4 +CO + CO2) = 2.0 | Equilibrium CO conversion (T = 310 °C) | [56] |
| 0.5wt%Pt–0.5wt%Re/TiO2 (not specified) | By co-impregnation | GHSV = 410,000 h−1; H2O/CO = 2.5 | XCO ≈ 90% (T = 300 °C) | [57] |
| Pt0.25-Mo0.75/C (1.27 nm) | By controlled surface reaction | WHSV = 240,000 mL·gcat−1·min−1; H2O/CO = 2 | r ≈ 10 μmol/gcat·s (T = 300 °C) | [58] |
| 4.3wt%Pt/64.6wt%Mo2C (not specified) | By wet impregnation | GHSV = 125,000 h−1 H2O/(CO + CO2) = 1.23 | XCO = 70% (T = 250 °C) | [60] |
| 4.79wt%VOx-0.49wt%Pt/Al2O3 (not specified) | By wet impregnation | WHSV = 80,000 mL·gcat−1·h−1; H2O/CO = 3 | XCO ≈ 60% (T = 450 °C) | [61] |
| 2.5wt%Pt-2.5wt%Ni/5wt%CeO2/Al2O3 (not specified) | By wetness incipient impregnation | W/FCOin = 20.37 gcat·h/molCO; H2O/CO = 5 | XCO ≈ 80% (T = 750 °C) | [62] |
| 1wt%Pt-1.25wt%K-1.25wt%CeO2/Al2O3 (not specified) | By incipient wetness co-impregnation | WHSV = 24,000 mL·gcat−1·h−1; H2O/(CO + CO2) = 0.67 | XCO ≈ 60% (T = 300 °C) | [64] |
| 1wt%Pt/1wt%Sn/CeZrO4 (9.3 nm) | By wet impregnation | GHSV = 10,000 h−1; H2O/CO = 3.75 | Equilibrium CO conversion (T = 230 °C) | [65] |
| 1wt%Pt-1wt%Re/CeZrO4 (9.4 nm) | By wet impregnation | GHSV = 10,000 h−1; H2O/CO = 3.75 | Equilibrium CO conversion (T = 200 °C) | [66] |
| 2.1wt%Pt–2.1wt%Re/25wt%CeO2/Al2O3 (1.4–5.0 nm) | By the incipient wetness impregnation | WHSV = 400,000 mL·gcat−1·h−1 H2O/(CO + CO2)= 2.2 | XCO ≈ 74% (T = 400 °C) | [67] |
| LaCo0.94Pt0.04O3-δ (75.5 nm) | By pyrolysis | GHSV = 5000 h−1; H2O/(CO + CO2) = 1.8 | XCO ≈ 90% (T = 325 °C) | [69] |
2.3.3. The Addition of Promoters to the Support
2.3.4. Conclusions
| Selected Catalyst (Particle/Crystallite Size) | Preparation Procedure | Operative Condition WGS | CO Conversion/H2 Formation Rate (XCO)/(rH2) (Temperature) | Ref. |
|---|---|---|---|---|
| 2.5wt%Au-2.5wt%Pt/FSM16 (7 nm) | Pt and Au loading by co-impregnation; FSM by surfactant templation | H2O/CO = 0.08 | rH2 = 5 mmol·gcat−1 (T = 77 °C) | [70] |
| 1wt%Pt–40wt%CeO2/C (not specified) | Pt and Ce loading by impregnation under vacuum with acetone | WHSV = 60,000 mL·gcat−1·h−1 H2O/CO = 20.5 | XCO > 70% (T = 300 °C) | [72] |
| 20wt%CeO2/1wt%Pt/Al2O3 (5.7 nm) | By sol-gel synthesis | WHSV = 130,000 mL·gcat−1·h−1 H2O/CO = 10 | XCO ≈ 95% (T = 350 °C) | [73] |
3. Kinetics of Water-Gas Shift Reaction
3.1. Monometallic Catalysts
3.1.1. Pt/Mo2C-Based Catalysts
3.1.2. Pt/CeO2-Based Catalysts
3.1.3. Pt/MnO2-Based and Pt/strontium Hydroxy and Fluorapatite Catalysts
3.1.4. Conclusions
3.2. Polymetallic and Promoted Catalysts
3.2.1. Mo-Promoted Catalysts
3.2.2. Alkali-Promoted Catalysts
3.2.3. Rare Earth and Transition Metals-Promoted Catalysts
3.2.4. Conclusions
4. Reaction Mechanisms
4.1. Monometallic Catalysts Supported on Single and Mixed Oxides
4.1.1. CeO2- and CeO2-TiO2-Supported Platinum Catalysts
4.1.2. Ca and Si Addition to CeO2-Supported Platinum Catalysts
4.1.3. CeO2ZrO2-Supported Platinum Catalysts
4.1.4. CeO2La2O3-Supported Platinum Catalysts
4.1.5. Different-Supported Platinum Catalysts
4.1.6. Conclusions
4.2. Polymetallic and Promoted Catalysts
4.2.1. Pt-Based Bimetallic Catalysts Supported on Different Oxides
4.2.2. Alkali Metals Promotion of Pt-Based Catalysts
4.2.3. Other Metals Promotion of Pt-Based Bimetallic Catalysts
4.2.4. Conclusions
4.3. DFT and Theoretical Studies
4.3.1. Unsupported Monometallic Pt Surface Models
4.3.2. Unsupported Polymetallic Surface Models
4.3.3. TiO2-Supported Pt Models
4.3.4. CeO2-Supported Pt Models
4.3.5. MgO-Supported Pt and Bimetallic Supported Models
4.3.6. Conclusions
5. Deactivation Studies
6. Electrochemical Promotion
7. Pt-Based Catalysts for Medium Temperature Single Stage WGS Process
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Acronym | Extended term |
| ALD | Atomic layer deposition |
| BET | Brunauer–Emmett–Teller |
| CNTs | Carbon nanotubes |
| DFT | Density functional theory |
| DRIFTS | Diffuse reflection infrared Fourier transform spectroscopy |
| EPOC | Electrochemical promotion of catalysis |
| GGA | Generalized gradient approximation |
| GHSV | Gas Hourly Space Velocity |
| HOPG | Highly oriented pyrolytic graphite |
| HTS | High-temperature water-gas shift |
| IGCC | Integrated gasification combined cycle |
| KMC | Kinetic Monte Carlo |
| LDHs | Layered double hydroxides |
| LTS | Low-temperature water-gas shift |
| MCNTs | Multiwalled carbon nanotubes |
| MOF | Metal-organic frameworks |
| NEMCA | Non-Faradaic Electrochemical Modification of Catalytic Activity |
| NPs | Nanoparticles |
| OSC | Oxygen storage capacity |
| PBE | Perdew–Burke–Ernzerhof functional |
| PRGO | Partially reduced graphite oxide |
| PRO | Partially reducible oxide |
| QoIs | Quantities of interest |
| RDS | Rate determining step |
| RSDT | Reactive spray deposition technology |
| SAC | Single-atom catalyst |
| SSA | Specific Surface Area |
| TEM | Transmission Electron Microscopy |
| TMCs | Transition metal carbides |
| TOF | Turnover frequency |
| TPB | The three-phase boundary |
| UPS | UV photoelectron spectroscopy |
| UQ | Uncertainty quantification |
| WGS | Water gas shift |
References
- Jacobs, G.; Davis, B.H. Low Temperature Water-Gas Shift Catalysts; Royal Society of Chemistry: London, UK, 2008; pp. 122–285. [Google Scholar]
- Abe, M.; Iriki, T.; Yoshida, N. Ammonia Production from Soluble-N Fraction of Various Feedstuffs When Incubated with Rumen Fluid. Nihon Chikusan Gakkaiho 1992, 63, 721–727. [Google Scholar] [CrossRef]
- Vu, M.-H.; Sakar, M.; Do, T.-O. Insights into the Recent Progress and Advanced Materials for Photocatalytic Nitrogen Fixation for Ammonia (NH3) Production. Catalysts 2018, 8, 621. [Google Scholar] [CrossRef] [Green Version]
- Patart, G. Procédé de Production d’Alcools, d’Aldéhydes et d’Acides à Partir de Mélanges Gazeux Maintenus sous Pression et Soumis à l’Action d’Agents Catalytiques ou de l’Électricité’. Fr. Pat. Appl. 1922, 540, 543. [Google Scholar]
- Dawood, F.; Anda, M.; Shafiullah, G.M. Hydrogen production for energy: An overview. Int. J. Hydrog. Energy 2020, 45, 3847–3869. [Google Scholar] [CrossRef]
- LeValley, T.L.; Richard, A.R.; Fan, M. The progress in water gas shift and steam reforming hydrogen production technologies—A review. Int. J. Hydrog. Energy 2014, 39, 16983–17000. [Google Scholar] [CrossRef]
- Eigenberger, G. Catalytic Fixed-Bed Reactors. Handb. Heterog. Catal. 2008, 10, 2075–2106. [Google Scholar] [CrossRef]
- Bhaskar, T.; Balagurumurthy, B.; Singh, R.; Poddar, M.K. Thermochemical Route for Biohydrogen Production. In Biohydrogen; Pandey, A., Chang, J.-S., Hallenbeck, P.C., Larroche, C., Eds.; Elsevier: San Diego, CA, USA, 2013; pp. 285–316. [Google Scholar]
- Odabaşı, Ç.; Günay, M.E.; Yıldırım, R. Knowledge extraction for water gas shift reaction over noble metal catalysts from publications in the literature between 2002 and 2012. Int. J. Hydrog. Energy 2014, 39, 5733–5746. [Google Scholar] [CrossRef]
- Bosch, C.; Wild, W. Hydrogen Production. U.S. Patent 1,115,776, 3 November 1914. [Google Scholar]
- Zhu, M.; Wachs, I.E. Iron-Based Catalysts for the High-Temperature Water–Gas Shift (HT-WGS) Reaction: A Review. ACS Catal. 2015, 6, 722–732. [Google Scholar] [CrossRef]
- Dienes, E.K. Low Temperature Shift Reaction Involving a Zinc Oxide-Copper Catalyst. U.S. Patent No. 3,303,001, 7 February 1967. [Google Scholar]
- Gradisher, L.; Dutcher, B.; Fan, M. Catalytic hydrogen production from fossil fuels via the water gas shift reaction. Appl. Energy 2015, 139, 335–349. [Google Scholar] [CrossRef]
- Kam, R.; Scotta, J.; Amal, R.; Selomulya, C. Pyrophoricity and stability of copper and platinum based water-gas shift catalysts during oxidative shut-down/start-up operation. Chem. Eng. Sci. 2010, 65, 6461–6470. [Google Scholar] [CrossRef]
- Pal, D.; Chand, R.; Upadhyay, S.; Mishra, P.K. Performance of water gas shift reaction catalysts: A review. Renew. Sustain. Energy Rev. 2018, 93, 549–565. [Google Scholar] [CrossRef]
- Bunluesin, T.; Gorte, R.; Graham, G. Studies of the water-gas-shift reaction on ceria-supported Pt, Pd, and Rh: Implications for oxygen-storage properties. Appl. Catal. B Environ. 1998, 15, 107–114. [Google Scholar] [CrossRef]
- Palma, V.; Pisano, D.; Martino, M. Comparative Study Between Aluminum Monolith and Foam as Carriers for The Intensification of The CO Water Gas Shift Process. Catalysts 2018, 8, 489. [Google Scholar] [CrossRef] [Green Version]
- Nguyen-Phan, T.-D.; Baber, A.E.; Rodriguez, J.A.; Senanayake, S.D. Au and Pt nanoparticle supported catalysts tailored for H2 production: From models to powder catalysts. Appl. Catal. A Gen. 2016, 518, 18–47. [Google Scholar] [CrossRef] [Green Version]
- Flytzani-Stephanopoulos, M. Supported metal catalysts at the single-atom limit—A viewpoint. Chin. J. Catal. 2017, 38, 1432–1442. [Google Scholar] [CrossRef]
- Hwang, K.-R.; Lee, S.-W.; Lee, D.-W.; Lee, C.-B.; Ji, S.-M.; Park, J.-S. Bi-functional hydrogen membrane for simultaneous chemical reaction and hydrogen separation. Int. J. Hydrog. Energy 2014, 39, 2614–2620. [Google Scholar] [CrossRef]
- Moncada, N.G.; Castaño, M.G.; Ivanova, S.; Centeno, M.Á.; Romero-Sarria, F.; Odriozola, J. New concept for old reaction: Novel WGS catalyst design. Appl. Catal. B Environ. 2018, 238, 1–5. [Google Scholar] [CrossRef]
- Palma, V.; Ruocco, C.; Cortese, M.; Martino, M. Recent Advances in Structured Catalysts Preparation and Use in Water-Gas Shift Reaction. Catalysts 2019, 9, 991. [Google Scholar] [CrossRef] [Green Version]
- Pastor-Pérez, L.; Ramos-Fernández, E.; Sepúlveda-Escribano, A. Effect of the CeO2 synthesis method on the behaviour of Pt/CeO2 catalysis for the water-gas shift reaction. Int. J. Hydrog. Energy 2019, 44, 21837–21846. [Google Scholar] [CrossRef]
- Pastor-Pérez, L.; Belda-Alcázar, V.; Marini, C.; Pastor-Blas, M.M.; Sepúlveda-Escribano, A.; Ramos-Fernandez, E.V. Effect of cold Ar plasma treatment on the catalytic performance of Pt/CeO2 in water-gas shift reaction (WGS). Appl. Catal. B Environ. 2018, 225, 121–127. [Google Scholar] [CrossRef] [Green Version]
- Palma, V.; Pietrosanto, A.; Martino, M.; Reverchon, E.; De Marco, I. Supercritical Antisolvent Process to Produce Cerium Oxide Nanoparticles as a Support for High-Activity Platinum Catalysts. Chem. Eng. Trans. 2017, 57, 967–972. [Google Scholar] [CrossRef]
- Franco, P.; Martino, M.; Palma, V.; Scarpellini, A.; De Marco, I. Pt on SAS-CeO2 nanopowder as catalyst for the CO-WGS reaction. Int. J. Hydrog. Energy 2018, 43, 19965–19975. [Google Scholar] [CrossRef]
- Potdar, H.S.; Jeong, D.-W.; Kim, K.-S.; Roh, H.-S. Synthesis of Highly Active Nano-Sized Pt/CeO2 Catalyst via a Cerium Hydroxy Carbonate Precursor for Water Gas Shift Reaction. Catal. Lett. 2011, 141, 1268–1274. [Google Scholar] [CrossRef]
- Roh, H.-S.; Potdar, H.S.; Jeong, D.-W.; Kim, K.-S.; Shim, J.-O.; Jang, W.-J.; Koo, K.Y.; Yoon, W.L. Synthesis of highly active nano-sized (1 wt.% Pt/CeO2) catalyst for water gas shift reaction in medium temperature application. Catal. Today 2012, 185, 113–118. [Google Scholar] [CrossRef]
- Im, H.B.; Kwon, S.J.; Byun, C.K.; Ahn, H.S.; Koo, K.Y.; Yoon, W.L.; Yi, K.B. Effect of Support Geometry on Catalytic Activity of Pt/CeO2 Nanorods in Water Gas Shift Reaction. Trans. Korean Hydrog. New Energy Soc. 2014, 25, 577–585. [Google Scholar] [CrossRef] [Green Version]
- Mei, Z.; Li, Y.; Fan, M.; Zhao, L.; Zhao, J. Effect of the interactions between Pt species and ceria on Pt/ceria catalysts for water gas shift: The XPS studies. Chem. Eng. J. 2015, 259, 293–302. [Google Scholar] [CrossRef]
- Tang, H.; Sun, H.; Chen, D.; Jiao, X. Fabrication of Pt/CeO2 nanofibers for use in water–gas shift reaction. Mater. Lett. 2012, 77, 7–9. [Google Scholar] [CrossRef]
- Jeong, D.-W.; Potdar, H.S.; Roh, H.-S. Comparative Study on Nano-Sized 1 wt% Pt/Ce0.8Zr0.2O2 and 1 wt% Pt/Ce0.2Zr0.8O2 Catalysts for a Single Stage Water Gas Shift Reaction. Catal. Lett. 2012, 142, 439–444. [Google Scholar] [CrossRef]
- Palma, V.; Pisano, D.; Martino, M.; Ricca, A.; Ciambelli, P. Comparative studies of low temperature water gas shift reaction over platinum based catalysts. Chem. Eng. Trans. 2014, 39, 31–36. [Google Scholar] [CrossRef]
- Castaño, M.G.; Reina, T.R.; Ivanova, S.; Centeno, M.Á.; Odriozola, J. Pt vs. Au in water–gas shift reaction. J. Catal. 2014, 314, 1–9. [Google Scholar] [CrossRef]
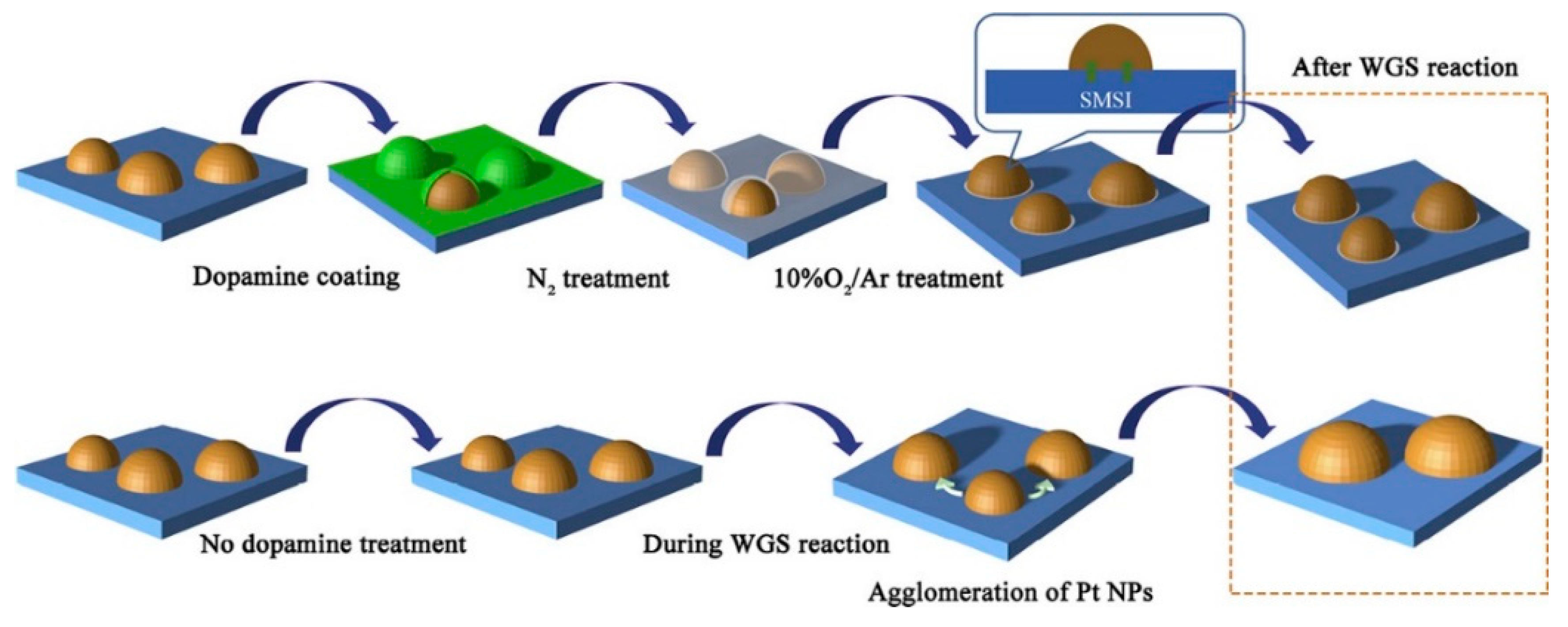
- Zhang, Y.; Cai, J.; Liu, Y.; Wang, X.; Au, C.-T.; Jiang, L. Preparation of sintering-resistant Pt nanocatalysts by dopamine mediation for water-gas shift reaction. Appl. Surf. Sci. 2019, 496, 1–8. [Google Scholar] [CrossRef]
- Wan, C.; Cheng, D.; Chen, F.; Zhan, X. Fabrication of CeO2 nanotube supported Pt catalyst encapsulated with silica for high and stable performance. Chem. Commun. 2015, 51, 9785–9788. [Google Scholar] [CrossRef] [PubMed]
- Stöber, W.; Fink, A.; Bohn, E. Controlled growth of monodisperse silica spheres in the micron size range. J. Colloid Interface Sci. 1968, 26, 62–69. [Google Scholar] [CrossRef]
- Roh, H.-S.; Jeong, D.-W.; Kim, K.-S.; Eum, I.-H.; Koo, K.Y.; Yoon, W.L. Single Stage Water–Gas Shift Reaction Over Supported Pt Catalysts. Catal. Lett. 2010, 141, 95–99. [Google Scholar] [CrossRef]
- Yati, I.; Ridwan, M.; Jeong, G.E.; Lee, Y.; Choi, J.-W.; Yoon, C.W.; Suh, D.J.; Ha, J.-M. Effects of sintering-resistance and large metal-support interface of alumina nanorod-stabilized Pt nanoparticle catalysts on the improved high temperature water gas shift reaction activity. Catal. Commun. 2014, 56, 11–16. [Google Scholar] [CrossRef]
- Subramanian, V.; Potdar, H.S.; Jeong, D.-W.; Shim, J.-O.; Jang, W.-J.; Roh, H.-S.; Jung, U.H.; Yoon, W.L. Synthesis of a Novel Nano-Sized Pt/ZnO Catalyst for Water Gas Shift Reaction in Medium Temperature Application. Catal. Lett. 2012, 142, 1075–1081. [Google Scholar] [CrossRef]
- Martinelli, M.; Jacobs, G.; Graham, U.M.; Shafer, W.D.; Cronauer, D.C.; Kropf, A.J.; Marshall, C.L.; Khalid, S.; Visconti, C.G.; Lietti, L.; et al. Water-gas shift: Characterization and testing of nanoscale YSZ supported Pt catalysts. Appl. Catal. A Gen. 2015, 497, 184–197. [Google Scholar] [CrossRef] [Green Version]
- Kwon, S.J.; Park, J.H.; Koo, K.Y.; Yoon, W.L.; Yi, K.B. Enhanced catalytic performance of Pt/TiO2 catalyst in water gas shift reaction by incorporation of PRGO. Catal. Today 2017, 113–121. [Google Scholar] [CrossRef]
- Franchini, C.A.; De Farias, A.M.D.; Albuquerque, E.; Dos Santos, R.; Fraga, M. Single-stage medium temperature water-gas shift reaction over Pt/ZrO2—Support structural polymorphism and catalyst deactivation. Appl. Catal. B Environ. 2012, 117, 302–309. [Google Scholar] [CrossRef]
- Jain, R.; Maric, R. Synthesis of nano-Pt onto ceria support as catalyst for water–gas shift reaction by Reactive Spray Deposition Technology. Appl. Catal. A Gen. 2014, 475, 461–468. [Google Scholar] [CrossRef]
- Lu, P.; Qiao, B.; Lu, N.; Hyun, D.C.; Wang, J.; Kim, M.J.; Liu, J.; Xia, Y. Photochemical Deposition of Highly Dispersed Pt Nanoparticles on Porous CeO2Nanofibers for the Water-Gas Shift Reaction. Adv. Funct. Mater. 2015, 25, 4153–4162. [Google Scholar] [CrossRef]
- Rajesh, T.; Rajarajan, A.K.; Gopinath, C.S.; Devi, R.N. Evidence of Cationic Pt Active for Water–Gas Shift Reaction: Pt-Doped BaCeO3 Perovskite. J. Phys. Chem. C 2012, 116, 9526–9532. [Google Scholar] [CrossRef]
- Tiwari, R.; Sarkar, B.; Tiwari, R.; Pendem, C.; Sasaki, T.; Saran, S.; Bal, R. Pt nanoparticles with tuneable size supported on nanocrystalline ceria for the low temperature water-gas-shift (WGS) reaction. J. Mol. Catal. A Chem. 2014, 395, 117–123. [Google Scholar] [CrossRef]
- Shim, J.-O.; Hong, Y.J.; Na, H.-S.; Jang, W.-J.; Kang, Y.C.; Roh, H.-S. Highly Active and Stable Pt-Loaded Ce0.75Zr0.25O2 Yolk–Shell Catalyst for Water–Gas Shift Reaction. ACS Appl. Mater. Interfaces 2016, 8, 17239–17244. [Google Scholar] [CrossRef]
- Cornaglia, C.A.; Tosti, S.; Múnera, J.F.; Lombardo, E.A. Optimal Pt load of a Pt/La2O3·SiO2 highly selective WGS catalyst used in a Pd-membrane reactor. Appl. Catal. A Gen. 2014, 486, 85–93. [Google Scholar] [CrossRef]
- Zhao, H.; Yao, S.; Zhang, M.; Huang, F.; Fan, Q.; Zhang, S.; Liu, H.; Ma, D.; Gao, C. Ultra-Small Platinum Nanoparticles Encapsulated in Sub-50 nm Hollow Titania Nanospheres for Low-Temperature Water–Gas Shift Reaction. ACS Appl. Mater. Interfaces 2018, 10, 36954–36960. [Google Scholar] [CrossRef]
- Galeano, Y.M.; Negri, F.; Moreno, M.S.; Múnera, J.; Cornaglia, L.; Tarditi, A.M. Pt encapsulated into NaA zeolite as catalyst for the WGS reaction. Appl. Catal. A Gen. 2019, 572, 176–184. [Google Scholar] [CrossRef]
- Roberts, S.J.; Fletcher, J.V.; Zhou, Y.; Luchters, N.T.; Fletcher, J.C. Water-gas shift of reformate streams over mono-metallic PGM catalysts. Int. J. Hydrog. Energy 2018, 43, 6150–6157. [Google Scholar] [CrossRef]
- Luchters, N.T.; Fletcher, J.; Roberts, S. Variability of Data in High Throughput Experimentation for Catalyst Studies in Fuel Processing. Bull. Chem. React. Eng. Catal. 2017, 12, 106. [Google Scholar] [CrossRef] [Green Version]
- Cavusoglu, G.; Miao, D.; Lichtenberg, H.; Carvalho, H.W.; Xu, H.; Goldbach, A.; Grunwaldt, J.-D. Structure and activity of flame made ceria supported Rh and Pt water gas shift catalysts. Appl. Catal. A Gen. 2015, 504, 381–390. [Google Scholar] [CrossRef]
- Cornaglia, C.A.; Adrover, M.E.; Múnera, J.F.; Pedernera, M.N.; Borio, D.O.; Lombardo, E.A. Production of ultrapure hydrogen in a Pd–Ag membrane reactor using noble metals supported on La–Si oxides. Heterogeneous modeling for the water gas shift reaction. Int. J. Hydrog. Energy 2013, 38, 10485–10493. [Google Scholar] [CrossRef]
- Jeong, D.-W.; Shim, J.-O.; Jang, W.-J.; Roh, H.-S. A Study on Pt-Na/CeO2 Catalysts for Single Stage Water Gas Shift Reaction. Trans. Korean Hydrog. New Energy Soc. 2012, 23, 111–116. [Google Scholar] [CrossRef] [Green Version]
- Azzam, K.G.; Babich, I.V.; Seshan, K.; Mojet, B.L.; Lefferts, L. Stable and Efficient Pt-Re/TiO2 catalysts for Water-Gas-Shift: On the Effect of Rhenium. ChemCatChem 2013, 5, 557–564. [Google Scholar] [CrossRef]
- Ciftci, A.; Ligthart, D.M.; Sen, A.O.; Van Hoof, A.; Friedrich, H.; Hensen, E.J. Pt-Re synergy in aqueous-phase reforming of glycerol and the water–gas shift reaction. J. Catal. 2014, 311, 88–101. [Google Scholar] [CrossRef]
- Sener, C.; Wesley, T.; Alba-Rubio, A.C.; Kumbhalkar, M.D.; Hakim, S.H.; Ribeiro, F.H.; Miller, J.T.; Dumesic, J.A. PtMo Bimetallic Catalysts Synthesized by Controlled Surface Reactions for Water Gas Shift. ACS Catal. 2016, 6, 1334–1344. [Google Scholar] [CrossRef]
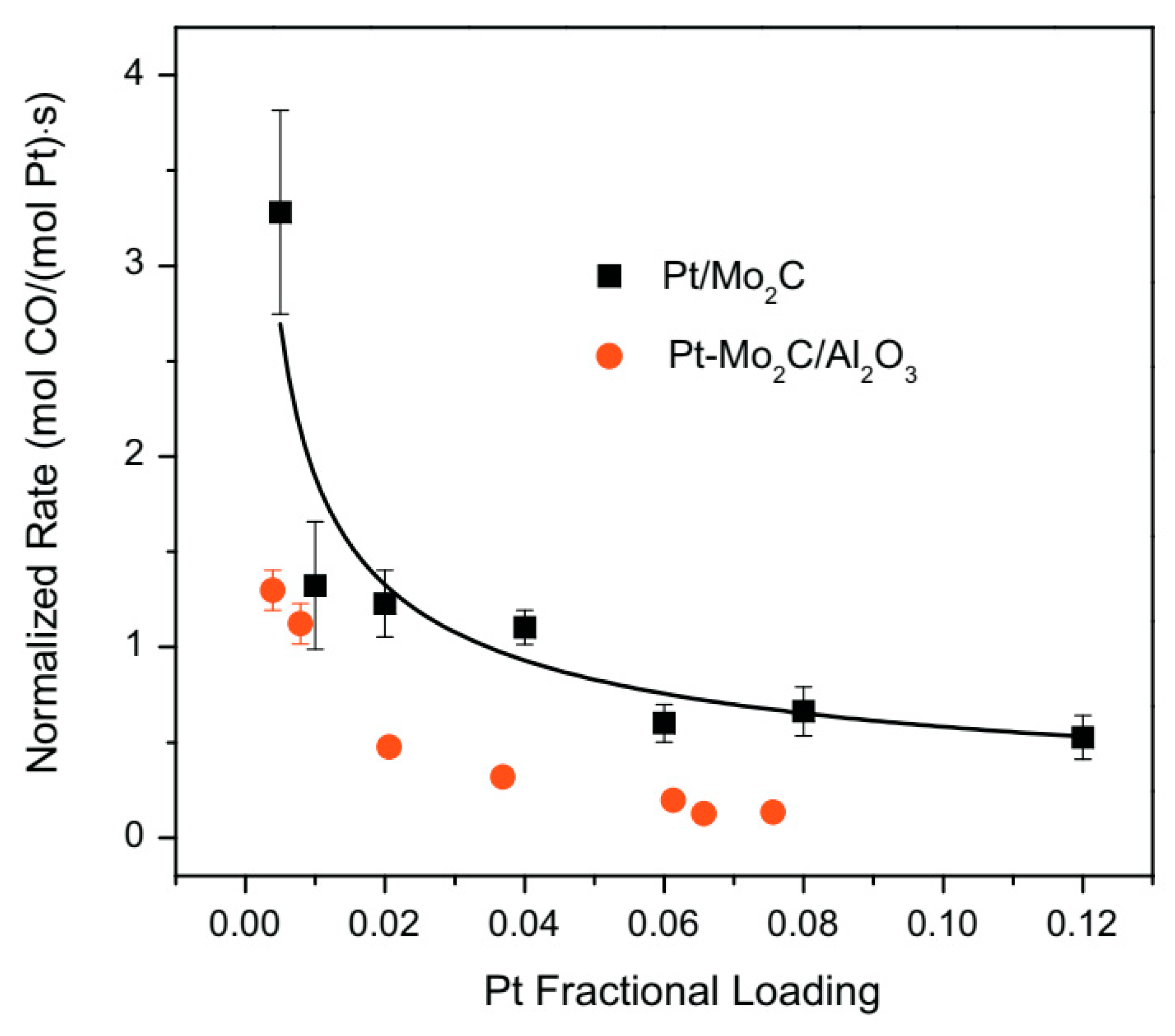
- Osman, A.I.; Abu-Dahrieh, J.K.; Cherkasov, N.; Fernández-García, J.; Walker, D.; Walton, R.I.; Rooney, D.W.; Rebrov, E.V. A highly active and synergistic Pt/Mo2C/Al2O3 catalyst for water-gas shift reaction. Mol. Catal. 2018, 455, 38–47. [Google Scholar] [CrossRef] [Green Version]
- Kokumai, T.M.; Cantane, D.A.; Melo, G.T.; Paulucci, L.B.; Zanchet, D. VO x -Pt/Al2O3 catalysts for hydrogen production. Catal. Today 2017, 289, 249–257. [Google Scholar] [CrossRef]
- Chein, R.; Lin, Y.; Chen, Y.; Chyou, Y.; Chung, J. Study on water–gas shift reaction performance using Pt-based catalysts at high temperatures. Int. J. Hydrog. Energy 2014, 39, 18854–18862. [Google Scholar] [CrossRef]
- Wang, T.; Porosoff, M.D.; Chen, J. Effects of oxide supports on the water-gas shift reaction over PtNi bimetallic catalysts: Activity and methanation inhibition. Catal. Today 2014, 233, 61–69. [Google Scholar] [CrossRef]
- Günay, M.E.; Akpinar, F.; Önsan, Z.I.; Yıldırım, R.; Yıldırım, R. Investigation of water gas-shift activity of Pt–MOx–CeO2/Al2O3 (M = K, Ni, Co) using modular artificial neural networks. Int. J. Hydrog. Energy 2012, 37, 2094–2102. [Google Scholar] [CrossRef]
- Palma, V.; Martino, M.; Pisano, D.; Ciambelli, P. Catalytic Activities of Bimetallic Catalysts for Low Temperature Water Gas Shift Reaction. Chem. Eng. Trans. 2016, 52, 481–486. [Google Scholar] [CrossRef]
- Palma, V.; Martino, M. Pt-Re Based Catalysts for the Realization of a Single Stage Water Gas Shift Process. Chem. Eng. Trans. 2017, 57, 1657–1662. [Google Scholar] [CrossRef]
- Izquierdo, U.; Neuberg, S.; Pecov, S.; Pennemann, H.; Zapf, R.; Wichert, M.; Barrio, V.; Cambra, J.F.; Kolb, G. Hydrogen production with a microchannel heat-exchanger reactor by single stage water-gas shift; catalyst development. Chem. Eng. J. 2017, 313, 1494–1508. [Google Scholar] [CrossRef]
- Rajesh, T.; Devi, R.N. Resistance to ionic Pt insertion in oxygen excess LaMnO3 perovskite lattices and its effect in water gas shift reaction. J. Mol. Catal. A Chem. 2014, 395, 534–542. [Google Scholar] [CrossRef]
- Rajesh, T.; Upadhyay, A.; Sinha, A.K.; Deb, S.K.; Devi, R.N. Effect of Pt incorporation in LaBO3 (B = Mn, Fe, Co) perovskites on water gas shift activity. J. Mol. Catal. A Chem. 2014, 395, 506–513. [Google Scholar] [CrossRef]
- Mohamed, M.M.; Khairou, K. Fabrication and characterization of bimetallic Pt–Au nanowires supported on FSM-16 and their catalytic activities toward water–gas shift reaction. J. Colloid Interface Sci. 2011, 354, 100–108. [Google Scholar] [CrossRef]
- Buitrago-Sierra, R.; Ruiz-Martinez, J.; Silvestre-Albero, J.; Sepúlveda-Escribano, A.; Rodriguez-Reinoso, F. Water gas shift reaction on carbon-supported Pt catalysts promoted by CeO2. Catal. Today 2012, 180, 19–24. [Google Scholar] [CrossRef]
- Zugic, B.; Bell, D.C.; Flytzani-Stephanopoulos, M. Activation of carbon-supported platinum catalysts by sodium for the low-temperature water-gas shift reaction. Appl. Catal. B Environ. 2014, 144, 243–251. [Google Scholar] [CrossRef]
- Meira, D.; Ribeiro, R.; Mathon, O.; Pascarelli, S.; Bueno, J.; Zanchet, D. Complex interplay of structural and surface properties of ceria on platinum supported catalyst under water gas shift reaction. Appl. Catal. B Environ. 2016, 197, 73–85. [Google Scholar] [CrossRef]
- Mao, X.; Foucher, A.C.; Stach, E.A.; Gorte, R.J. “Intelligent” Pt Catalysts Based on Thin LaCoO3 Films Prepared by Atomic Layer Deposition. Inorganics 2019, 7, 113. [Google Scholar] [CrossRef] [Green Version]
- Brandt, A.J.; Maddumapatabandi, T.D.; Shakya, D.M.; Xie, K.; Seuser, G.S.; Farzandh, S.; Chen, D.A. Water-gas shift activity on Pt-Re surfaces and the role of the support. J. Chem. Phys. 2019, 151, 234714. [Google Scholar] [CrossRef]
- Schweitzer, N.M.; Schaidle, J.A.; Ezekoye, O.K.; Pan, X.; Linic, S.; Thompson, L.T. High Activity Carbide Supported Catalysts for Water Gas Shift. J. Am. Chem. Soc. 2011, 133, 2378–2381. [Google Scholar] [CrossRef] [PubMed]
- Wyvratt, B.M.; Gaudet, J.R.; Thompson, L.T. Effects of passivation on synthesis, structure and composition of molybdenum carbide supported platinum water–gas shift catalysts. J. Catal. 2015, 330, 280–287. [Google Scholar] [CrossRef] [Green Version]
- Wang, G.; Schaidle, J.A.; Katz, M.B.; Li, Y.; Pan, X.Q.; Thompson, L. Alumina supported Pt–Mo2C catalysts for the water–gas shift reaction. J. Catal. 2013, 304, 92–99. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Ramírez, P.J.; Gutierrez, R.A. Highly active Pt/MoC and Pt/TiC catalysts for the low-temperature water-gas shift reaction: Effects of the carbide metal/carbon ratio on the catalyst performance. Catal. Today 2017, 289, 47–52. [Google Scholar] [CrossRef]
- Wen, C.; Zhu, Y.; Ye, Y.; Zhang, S.; Cheng, F.; Liu, Y.; Wang, P.; Tao, D.F. (Feng) Water–Gas Shift Reaction on Metal Nanoclusters Encapsulated in Mesoporous Ceria Studied with Ambient-Pressure X-ray Photoelectron Spectroscopy. ACS Nano 2012, 6, 9305–9313. [Google Scholar] [CrossRef]
- Jeong, D.-W.; Jang, W.-J.; Shim, J.-O.; Han, W.-B.; Kim, H.-M.; Lee, Y.-L.; Bae, J.W.; Roh, H.-S. Optimization of a highly active nano-sized Pt/CeO2 catalyst via Ce(OH)CO3 for the water-gas shift reaction. Renew. Energy 2015, 79, 78–84. [Google Scholar] [CrossRef]
- Jeong, D.-W.; Potdar, H.S.; Shim, J.-O.; Jang, W.-J.; Roh, H.-S. H2 production from a single stage water–gas shift reaction over Pt/CeO2, Pt/ZrO2, and Pt/Ce(1−x)Zr(x)O2 catalysts. Int. J. Hydrog. Energy 2013, 38, 4502–4507. [Google Scholar] [CrossRef]
- Torrente-Murciano, L.; García-García, F. Effect of nanostructured support on the WGSR activity of Pt/CeO2 catalysts. Catal. Commun. 2015, 71, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Deal, J.W.; Le, P.; Corey, C.B.; More, K.L.; West, C.W. Water-gas shift reaction on alumina-supported Pt-CeO catalysts prepared by supercritical fluid deposition. J. Supercrit. Fluids 2017, 119, 113–121. [Google Scholar] [CrossRef] [Green Version]
- Shan, J.; Nguyen, L.; Zhang, S.; Tao, F.-F. Water–Gas Shift on Pd/α-MnO2 and Pt/α-MnO2. Catal. Lett. 2015, 145, 1571–1580. [Google Scholar] [CrossRef]
- Kuai, L.; Liu, S.; Cao, S.; Ren, Y.; Kan, E.; Zhao, Y.; Yu, N.; Li, F.; Li, X.; Wu, Z.; et al. Atomically Dispersed Pt/Metal Oxide Mesoporous Catalysts from Synchronous Pyrolysis–Deposition Route for Water–Gas Shift Reaction. Chem. Mater. 2018, 30, 5534–5538. [Google Scholar] [CrossRef]
- De León, J.D.; Loera-Serna, S.; Zepeda, T.; Domínguez, D.; Pawelec, B.; Venezia, A.; Fuentes-Moyado, S. Noble metals supported on binary γ-Al2O3-α-Ga2O3 oxide as potential low-temperature water-gas shift catalysts. Fuel 2020, 266, 117031. [Google Scholar] [CrossRef]
- Miao, D.; Cavusoglu, G.; Lichtenberg, H.; Yu, J.; Xu, H.; Grunwaldt, J.-D.; Goldbach, A. Water-gas shift reaction over platinum/strontium apatite catalysts. Appl. Catal. B Environ. 2017, 202, 587–596. [Google Scholar] [CrossRef]
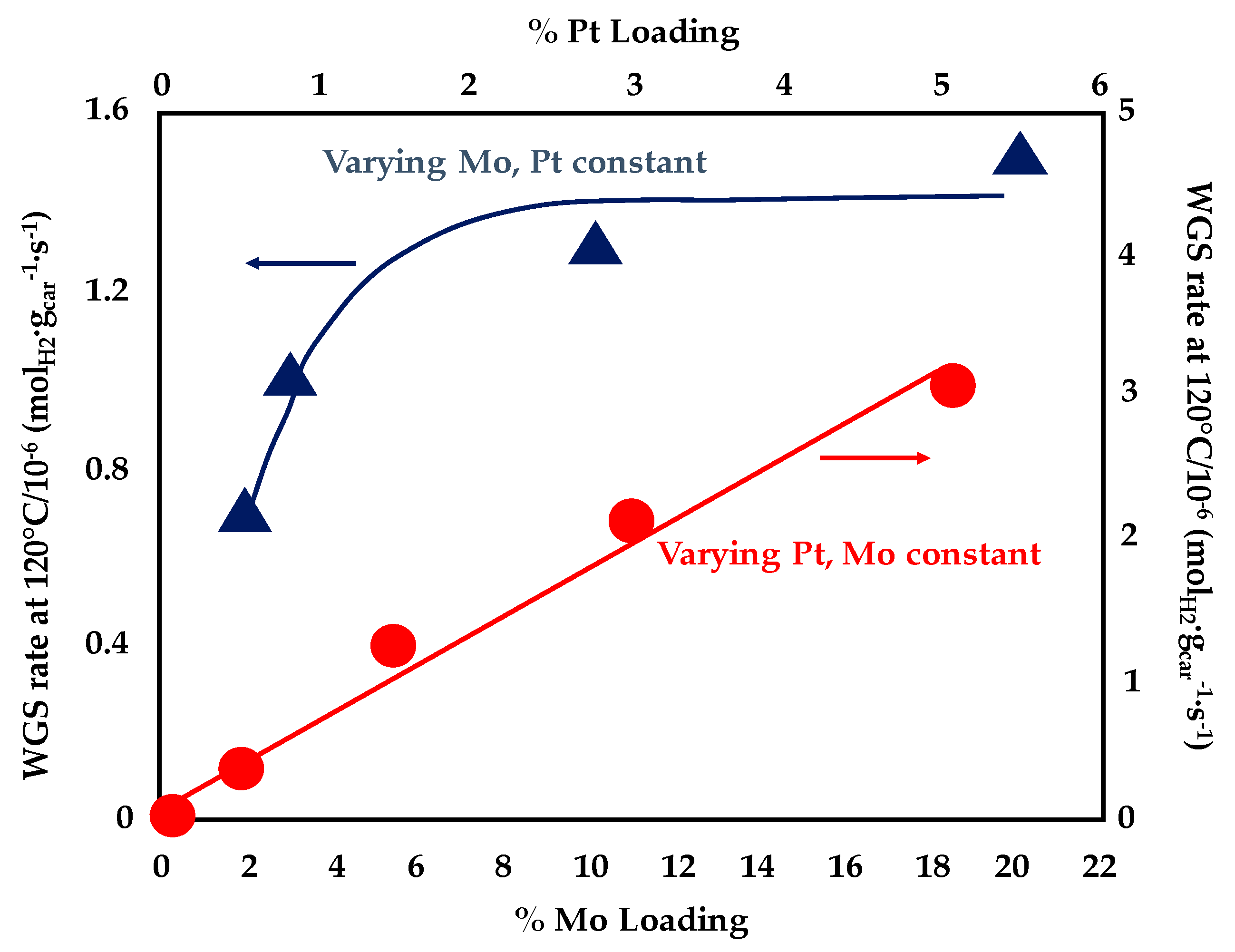
- Williams, W.D.; Bollmann, L.; Miller, J.T.; Delgass, W.N.; Ribeiro, F.H. Effect of molybdenum addition on supported platinum catalysts for the water–gas shift reaction. Appl. Catal. B Environ. 2012, 125, 206–214. [Google Scholar] [CrossRef]
- Xu, X.; Fu, Q.; Bao, X. MoOx-promoted Pt catalysts for the water gas shift reaction at low temperatures. Chin. J. Catal. 2015, 36, 750–756. [Google Scholar] [CrossRef]
- Sabnis, K.D.; Akatay, M.C.; Cui, Y.; Sollberger, F.G.; Stach, E.A.; Miller, J.T.; Delgass, W.N.; Ribeiro, F.H. Probing the active sites for water–gas shift over Pt/molybdenum carbide using multi-walled carbon nanotubes. J. Catal. 2015, 330, 442–451. [Google Scholar] [CrossRef] [Green Version]
- Yang, M.; Flytzani-Stephanopoulos, M. Design of single-atom metal catalysts on various supports for the low-temperature water-gas shift reaction. Catal. Today 2017, 298, 216–225. [Google Scholar] [CrossRef]
- Wang, Y.; Zhai, Y.; Pierre, D.; Flytzani-Stephanopoulos, M. Silica-encapsulated platinum catalysts for the low-temperature water-gas shift reaction. Appl. Catal. B Environ. 2012, 127, 342–350. [Google Scholar] [CrossRef]
- Zhu, X.; Shen, M.; Lobban, L.L.; Mallinson, R.G. Structural effects of Na promotion for high water gas shift activity on Pt–Na/TiO2. J. Catal. 2011, 278, 123–132. [Google Scholar] [CrossRef]
- Zugic, B.; Zhang, S.; Bell, D.C.; Tao, F.; Flytzani-Stephanopoulos, M. Probing the Low-Temperature Water–Gas Shift Activity of Alkali-Promoted Platinum Catalysts Stabilized on Carbon Supports. J. Am. Chem. Soc. 2014, 136, 3238–3245. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Liu, J.; Lee, S.; Zugic, B.; Huang, J.; Allard, L.F.; Flytzani-Stephanopoulos, M. A Common Single-Site Pt(II)–O(OH)x–Species Stabilized by Sodium on “Active” and “Inert” Supports Catalyzes the Water-Gas Shift Reaction. J. Am. Chem. Soc. 2015, 137, 3470–3473. [Google Scholar] [CrossRef]
- Xie, H.; Lu, J.; Shekhar, M.; Elam, J.W.; Delgass, W.N.; Ribeiro, F.H.; Weitz, E.; Poeppelmeier, K.R. Synthesis of Na-Stabilized Nonporous t-ZrO2 Supports and Pt/t-ZrO2 Catalysts and Application to Water-Gas-Shift Reaction. ACS Catal. 2012, 3, 61–73. [Google Scholar] [CrossRef]
- Pazmiño, J.H.; Shekhar, M.; Williams, W.D.; Akatay, M.C.; Miller, J.T.; Delgass, W.N.; Ribeiro, F.H. Metallic Pt as active sites for the water–gas shift reaction on alkali-promoted supported catalysts. J. Catal. 2012, 286, 279–286. [Google Scholar] [CrossRef]
- Kusche, M.; Bustillo, K.; Agel, F.; Wasserscheid, P. Highly Effective Pt-Based Water–Gas Shift Catalysts by Surface Modification with Alkali Hydroxide Salts. ChemCatChem 2015, 7, 766–775. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panagiotopoulou, P.; Kondarides, D.I. Effects of promotion of TiO2 with alkaline earth metals on the chemisorptive properties and water–gas shift activity of supported platinum catalysts. Appl. Catal. B Environ. 2011, 101, 738–746. [Google Scholar] [CrossRef]
- Kim, Y.-T.; You, S.J.; Park, E.D. Water–gas shift reaction over Pt and Pt–CeOx supported on CexZr1−xO2. Int. J. Hydrog. Energy 2012, 37, 1465–1474. [Google Scholar] [CrossRef]
- Carta, D.; Montini, T.; Casula, M.F.; Monai, M.; Bullita, S.; Fornasiero, P.; Corrias, A. The water gas shift reaction over Pt–CeO2 nanoparticles confined within mesoporous SBA-16. J. Mater. Chem. A 2017, 5, 20024–20034. [Google Scholar] [CrossRef] [Green Version]
- Mahadevaiah, N.; Singh, P.; Mukri, B.D.; Parida, S.K.; Hegde, M.S. Ce0.67Fe0.33O2−δ and Ce0.65Fe0.33Pt0.02O2−δ: New water gas shift (WGS) catalysts. Appl. Catal. B Environ. 2011, 108, 117–126. [Google Scholar] [CrossRef]
- Shinde, V.M.; Madras, G. Low temperature CO oxidation and water gas shift reaction over Pt/Pd substituted in Fe/TiO2 catalysts. Int. J. Hydrog. Energy 2012, 37, 18798–18814. [Google Scholar] [CrossRef]
- Shinde, V.M.; Madras, G. A Single-Stage Water-Gas Shift Reaction over Highly Active and Stable Si- and Al-Substituted Pt/CeO2 Catalysts. ChemCatChem 2012, 4, 1968–1978. [Google Scholar] [CrossRef]
- Petallidou, K.C.; Efstathiou, A.M. Low-temperature water-gas shift on Pt/Ce1−xLaxO2−δ: Effect of Ce/La ratio. Appl. Catal. B Environ. 2013, 140, 333–347. [Google Scholar] [CrossRef]
- Palma, V.; Gallucci, F.; Pullumbi, P.; Ruocco, C.; Meloni, E.; Martino, M. Pt/Re/CeO2 Based Catalysts for CO-Water–Gas Shift Reaction: From Powders to Structured Catalyst. Catalysts 2020, 10, 564. [Google Scholar] [CrossRef]
- Ratnasamy, C.; Wagner, J.P. Water Gas Shift Catalysis. Catal. Rev. 2009, 51, 325–440. [Google Scholar] [CrossRef]
- Ribeiro, M.; Jacobs, G.; Linganiso, L.; Azzam, K.G.; Graham, U.M.; Crocker, M. Low Temperature Water Gas Shift: Evaluation of Pt/HfO2 and Correlation between Reaction Mechanism and Periodic Trends in Tetravalent (Ti, Zr, Hf, Ce, Th) Metal Oxides. ACS Catal. 2011, 1, 1375–1383. [Google Scholar] [CrossRef]
- Rodríguez, J.A.; Hanson, J.C.; Stacchiola, D.J.; Senanayake, S.D. In situ/operando studies for the production of hydrogen through the water-gas shift on metal oxide catalysts. Phys. Chem. Chem. Phys. 2013, 15, 12004. [Google Scholar] [CrossRef]
- Petallidou, K.C.; Polychronopoulou, K.; Boghosian, S.; García-Rodríguez, S.; Efstathiou, A.M. Water–Gas Shift Reaction on Pt/Ce1–xTixO2−δ: The Effect of Ce/Ti Ratio. J. Phys. Chem. C 2013, 117, 25467–25477. [Google Scholar] [CrossRef]
- Yao, S.Y.; Xu, W.Q.; Johnston-Peck, A.C.; Zhao, F.Z.; Liu, Z.; Luo, S.; Senanayake, S.D.; Martínez-Arias, A.; Liu, W.; Rodriguez, J.A. Morphological effects of the nanostructured ceria support on the activity and stability of CuO/CeO2 catalysts for the water-gas shift reaction. Phys. Chem. Chem. Phys. 2014, 16, 17183–17195. [Google Scholar] [CrossRef]
- Barrio, L.; Zhou, G.; González, I.D.; Estrella, M.A.; Hanson, J.; Rodriguez, J.A.; Yerga, R.M.N.; Fierro, J.L.G. In situ characterization of Ptcatalysts supported on ceria modified TiO2 for the WGS reaction: Influence of ceria loading. Phys. Chem. Chem. Phys. 2012, 14, 2192–2202. [Google Scholar] [CrossRef]
- Jain, R.; Poyraz, A.S.; Gamliel, D.P.; Valla, J.; Suib, S.L.; Maric, R. Comparative study for low temperature water-gas shift reaction on Pt/ceria catalysts: Role of different ceria supports. Appl. Catal. A Gen. 2015, 507, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.M.; Kim, G.J.; Lee, S.H.; Hwang, I.H.; Hong, S.C.; Kim, S.S. Catalytic Performance of Ce0.6Y0.4O2-Supported Platinum Catalyst for Low-Temperature Water-Gas Shift Reaction. ACS Omega 2018, 3, 3156–3163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalamaras, C.; Gonzalez, I.D.; Navarro, R.M.; Fierro, J.L.G.; Efstathiou, A.M. Effects of Reaction Temperature and Support Composition on the Mechanism of Water–Gas Shift Reaction over Supported-Pt Catalysts. J. Phys. Chem. C 2011, 115, 11595–11610. [Google Scholar] [CrossRef]
- Luo, S.; Barrio, L.; Nguyen-Phan, T.-D.; Vovchok, D.; Johnston-Peck, A.C.; Xu, W.; Stach, E.A.; Rodriguez, J.A.; Senanayake, S.D. Importance of Low Dimensional CeOx Nanostructures in Pt/CeOx–TiO2 Catalysts for the Water–Gas Shift Reaction. J. Phys. Chem. C 2017, 121, 6635–6642. [Google Scholar] [CrossRef]
- Luo, S.; Nguyen-Phan, T.-D.; Johnston-Peck, A.C.; Barrio, L.; Sallis, S.; Arena, D.A.; Kundu, S.; Xu, W.; Piper, L.F.; Stach, E.A.; et al. Hierarchical Heterogeneity at the CeOx–TiO2Interface: Electronic and Geometric Structural Influence on the Photocatalytic Activity of Oxide on Oxide Nanostructures. J. Phys. Chem. C 2015, 119, 2669–2679. [Google Scholar] [CrossRef]
- Linganiso, L.Z.; Jacobs, G.; Azzam, K.G.; Graham, U.M.; Davis, B.H.; Cronauer, D.; Kropf, A.J.; Marshall, C.L. Low-temperature water–gas shift: Strategy to lower Pt loading by doping ceria with Ca2+ improves formate mobility/WGS rate by increasing surface O-mobility. Appl. Catal. A Gen. 2011, 394, 105–116. [Google Scholar] [CrossRef]
- Hwang, K.-R.; Park, J.-S.; Ihm, S.-K. Si-modified Pt/CeO2 catalyst for a single-stage water-gas shift reaction. Int. J. Hydrog. Energy 2011, 36, 9685–9693. [Google Scholar] [CrossRef]
- Vignatti, C.; Avila, M.; Apesteguia, C.; Garetto, T. Catalytic and DRIFTS study of the WGS reaction on Pt-based catalysts. Int. J. Hydrog. Energy 2010, 35, 7302–7312. [Google Scholar] [CrossRef]
- Vignatti, C.; Avila, M.; Apesteguia, C.; Garetto, T. Study of the water-gas shift reaction over Pt supported on CeO2–ZrO2 mixed oxides. Catal. Today 2011, 171, 297–303. [Google Scholar] [CrossRef]
- Kalamaras, C.; Dionysiou, D.D.; Efstathiou, A.M. Mechanistic Studies of the Water–Gas Shift Reaction over Pt/CexZr1–xO2 Catalysts: The Effect of Pt Particle Size and Zr Dopant. ACS Catal. 2012, 2, 2729–2742. [Google Scholar] [CrossRef]
- Pinaeva, L.; Sadovskaya, E.; Ivanova, Y.; Kuznetsova, T.; Prosvirin, I.P.; Sadykov, V.; Schuurman, Y.; Van Veen, A.; Mirodatos, C. Water gas shift and partial oxidation of CH4 over CeO2–ZrO2(–La2O3) and Pt/CeO2–ZrO2(–La2O3): Performance under transient conditions. Chem. Eng. J. 2014, 257, 281–291. [Google Scholar] [CrossRef]
- Petallidou, K.C.; Boghosian, S.; Efstathiou, A.M. Low-temperature water–gas shift on Pt/Ce0.5La0.5O2−δ: Effect of support synthesis method. Catal. Today 2015, 242, 153–167. [Google Scholar] [CrossRef]
- Leet, J.S.; Kim, S.; Society, T.A.C. Synthesis and Characterization of Ce1−xGdxO2-δ Nanorods. J. Am. Ceram. Soc. 2007, 90, 661–663. [Google Scholar]
- Petallidou, K.; Polychronopoulou, K.; Fierro, J.; Efstathiou, A.M. Low-temperature water-gas shift on Pt/Ce0.8La0.2O2−δ–CNT: The effect of Ce0.8La0.2O2−δ/CNT ratio. Appl. Catal. A Gen. 2015, 504, 585–598. [Google Scholar] [CrossRef]
- Petallidou, K.C.; Kalamaras, C.; Efstathiou, A.M. The effect of La3+, Ti4+ and Zr4+ dopants on the mechanism of WGS on ceria-doped supported Pt catalysts. Catal. Today 2014, 228, 183–193. [Google Scholar] [CrossRef]
- Linganiso, L.Z.; Pendyala, V.R.R.; Jacobs, G.; Crocker, M.; Cronauer, D.C.; Kropf, A.J.; Marshall, C.L. Low-Temperature Water–Gas Shift: Doping Ceria Improves Reducibility and Mobility of O-Bound Species and Catalyst Activity. Catal. Lett. 2011, 141, 1723–1731. [Google Scholar] [CrossRef]
- Kalamaras, C.; Petallidou, K.C.; Efstathiou, A.M. The effect of La3+-doping of CeO2 support on the water-gas shift reaction mechanism and kinetics over Pt/Ce1−xLaxO2−δ. Appl. Catal. B Environ. 2013, 136, 225–238. [Google Scholar] [CrossRef]
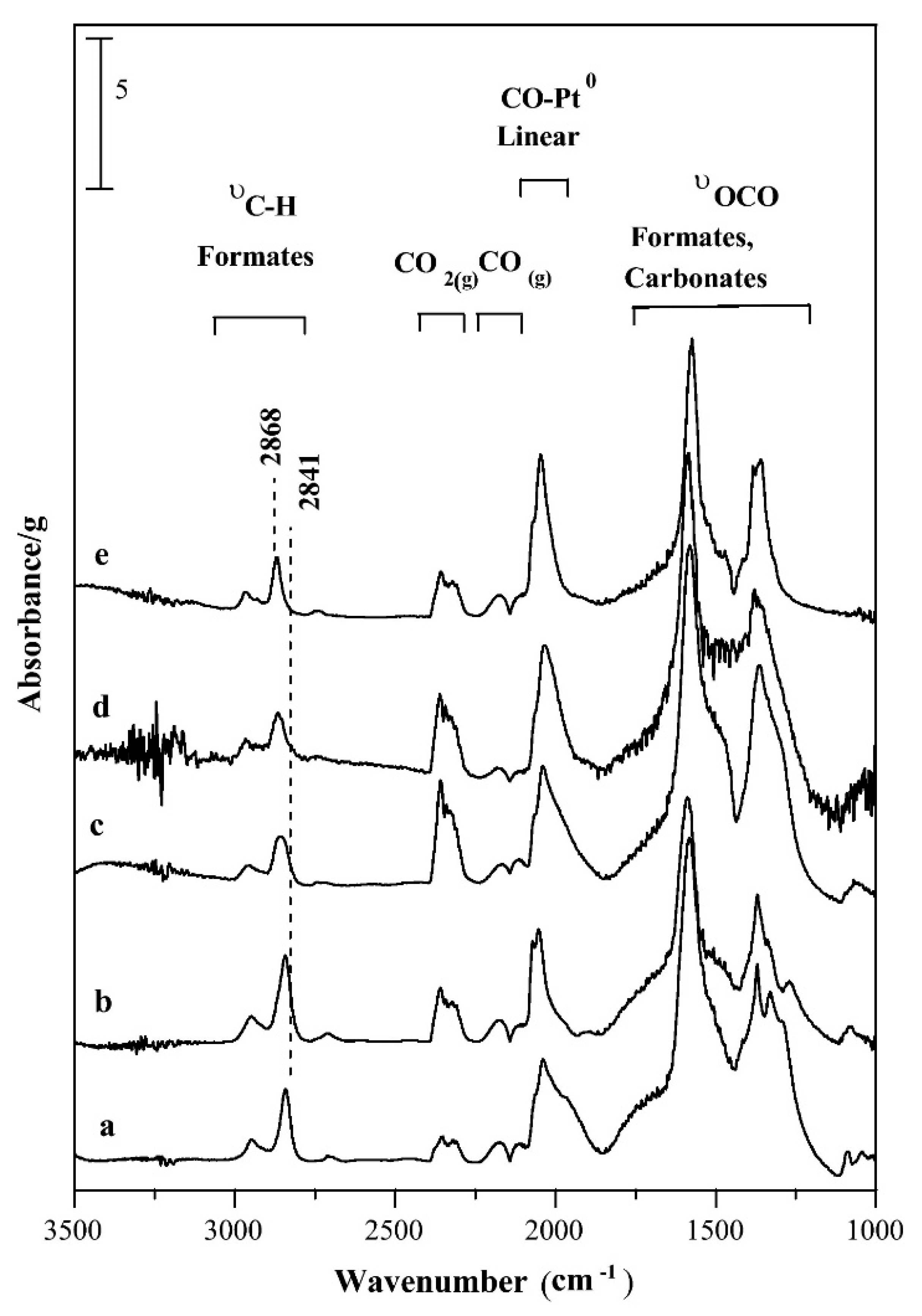
- Busca, G.; Finocchio, E.; Escribano, V.S. Infrared studies of CO oxidation by oxygen and by water over Pt/Al2O3 and Pd/Al2O3 catalysts. Appl. Catal. B Environ. 2012, 113, 172–179. [Google Scholar] [CrossRef]
- Sabnis, K.D.; Cui, Y.; Akatay, M.C.; Shekhar, M.; Lee, W.-S.; Miller, J.T.; Delgass, W.N.; Ribeiro, F.H. Water–gas shift catalysis over transition metals supported on molybdenum carbide. J. Catal. 2015, 331, 162–171. [Google Scholar] [CrossRef] [Green Version]
- Miao, D.; Goldbach, A.; Xu, H. Platinum/Apatite Water-Gas Shift Catalysts. ACS Catal. 2016, 6, 775–783. [Google Scholar] [CrossRef]
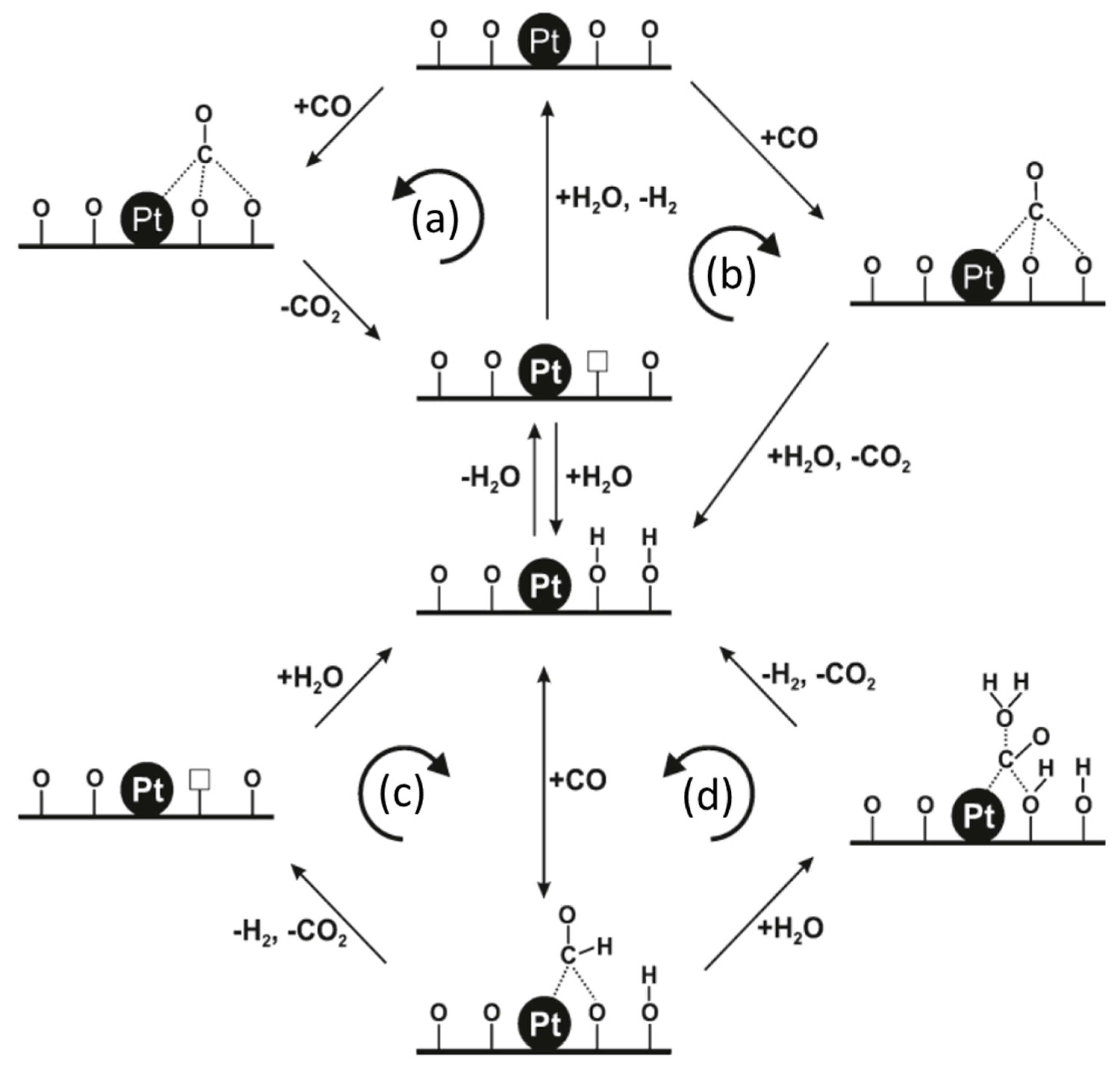
- Ding, K.; Gulec, A.; Johnson, A.M.; Schweitzer, N.M.; Stucky, G.D.; Marks, L.D.; Stair, P.C. Identification of active sites in CO oxidation and water-gas shift over supported Pt catalysts. Science 2015, 350, 189–192. [Google Scholar] [CrossRef] [Green Version]
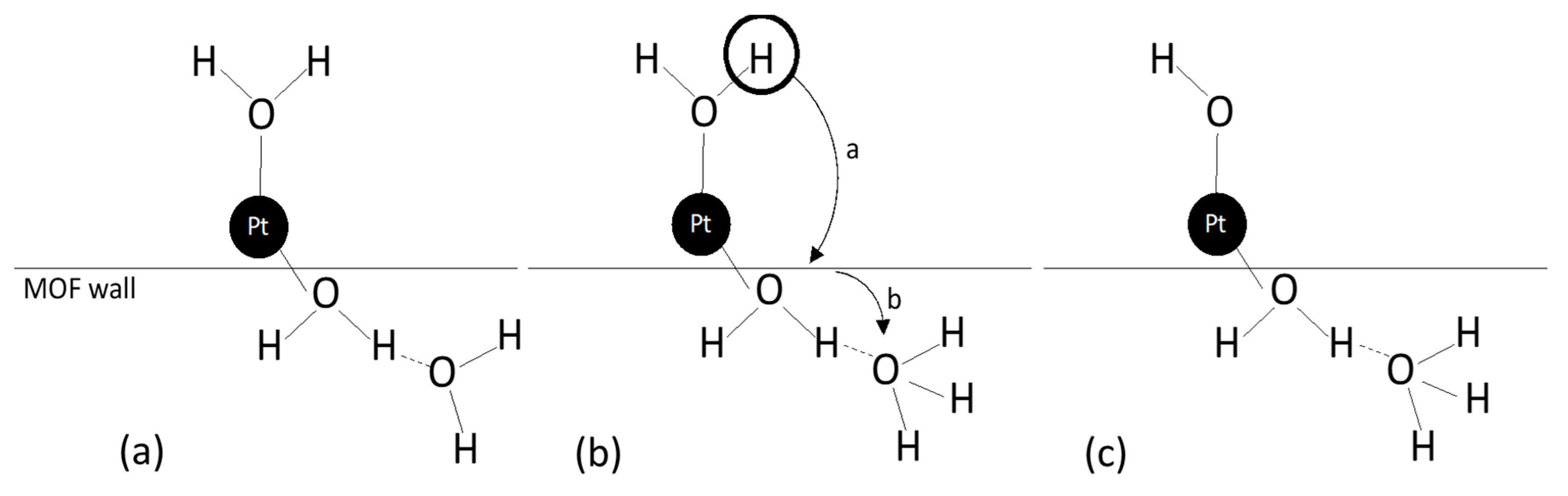
- Rivero-Crespo, M.; Mon, M.; Ferrando-Soria, J.; Lopes, C.W.; Boronat, M.; Leyva–Pérez, A.; Corma, A.; Hernandez-Garrido, J.C.; López-Haro, M.; Calvino, J.J.; et al. Confined Pt11+ Water Clusters in a MOF Catalyze the Low-Temperature Water-Gas Shift Reaction with both CO2 Oxygen Atoms Coming from Water. Angew. Chem. Int. Ed. 2018, 57, 17094–17099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.; Lin, J.; Li, L.; Qiao, B.; Liu, J.; Su, Y.; Wang, X. Identifying Size Effects of Pt as Single Atoms and Nanoparticles Supported on FeOx for the Water-Gas Shift Reaction. ACS Catal. 2018, 8, 859–868. [Google Scholar] [CrossRef]
- Flaherty, D.W.; Yu, W.-Y.; Pozun, Z.; Henkelman, G.; Mullins, C.B. Mechanism for the water–gas shift reaction on monofunctional platinum and cause of catalyst deactivation. J. Catal. 2011, 282, 278–288. [Google Scholar] [CrossRef]
- Gerceker, D.; Motagamwala, A.H.; Rivera-Dones, K.R.; Miller, J.B.; Huber, G.W.; Mavrikakis, M.; Dumesic, J.A. Methane Conversion to Ethylene and Aromatics on PtSn Catalysts. ACS Catal. 2017, 7, 2088–2100. [Google Scholar] [CrossRef]
- Duke, A.S.; Xie, K.; Brandt, A.J.; Maddumapatabandi, T.D.; Ammal, S.C.; Heyden, A.; Monnier, J.R.; Chen, D.A. Understanding Active Sites in the Water–Gas Shift Reaction for Pt–Re Catalysts on Titania. ACS Catal. 2017, 7, 2597–2606. [Google Scholar] [CrossRef]
- Aragão, I.B.; Ro, I.; Liu, Y.; Ball, M.; Huber, G.W.; Zanchet, D.; Dumesic, J.A. Catalysts synthesized by selective deposition of Fe onto Pt for the water-gas shift reaction. Appl. Catal. B Environ. 2018, 222, 182–190. [Google Scholar] [CrossRef]
- Xu, W.; Si, R.; Senanayake, S.D.; Llorca, J.; Idriss, H.; Stacchiola, D.J.; Hanson, J.C.; Rodriguez, J.A. In situ studies of CeO2-supported Pt, Ru, and Pt–Ru alloy catalysts for the water–gas shift reaction: Active phases and reaction intermediates. J. Catal. 2012, 291, 117–126. [Google Scholar] [CrossRef]
- Anil, C.; Madras, G. Catalytic behaviour of Mn2.94M0.06O4-δ (M = Pt, Ru and Pd) catalysts for low temperature water gas shift (WGS) and CO oxidation. Int. J. Hydrog. Energy 2020, 45, 10461–10474. [Google Scholar] [CrossRef]
- Rajesh, T.; Devi, R.N. Role of Oxygen Vacancies in Water Gas Shift Reaction: Activity Study on BaCe0.98–xYxPt0.02O3−δ Perovskites. J. Phys. Chem. C 2014, 118, 20867–20874. [Google Scholar] [CrossRef]
- Cybulskis, V.J.; Wang, J.; Pazmiño, J.H.; Ribeiro, F.H.; Delgass, W.N. Isotopic transient studies of sodium promotion of Pt/Al2O3 for the water–gas shift reaction. J. Catal. 2016, 339, 163–172. [Google Scholar] [CrossRef] [Green Version]
- Faust, M.; Dinkel, M.; Bruns, M.; Bräse, S.; Seipenbusch, M.; Bräse, S. Support Effect on the Water Gas Shift Activity of Chemical Vapor Deposition-Tailored-Pt/TiO2 Catalysts. Ind. Eng. Chem. Res. 2017, 56, 3194–3203. [Google Scholar] [CrossRef]
- Martinelli, M.; Jacobs, G.; Shafer, W.D.; Crocker, M. Effect of alkali on C H bond scission over Pt/YSZ catalyst during water-gas-shift, steam-assisted formic acid decomposition and methanol steam reforming. Catal. Today 2017, 291, 29–35. [Google Scholar] [CrossRef]
- Gao, P.; Graham, U.M.; Shafer, W.D.; Linganiso, L.Z.; Jacobs, G.; Crocker, M. Nanostructure and kinetic isotope effect of alkali-doped Pt/silica catalysts for water-gas shift and steam-assisted formic acid decomposition. Catal. Today 2016, 272, 42–48. [Google Scholar] [CrossRef]
- Watson, C.D.; Martinelli, M.; Cronauer, D.C.; Kropf, A.J.; Marshall, C.L.; Jacobs, G. Low temperature water-gas shift: Optimization of K loading on Pt/m-ZrO2 for enhancing CO conversion. Appl. Catal. A Gen. 2020, 598, 117572. [Google Scholar] [CrossRef]
- Kaftan, A.; Kusche, M.; Laurin, M.; Wasserscheid, P.; Libuda, J.; Libuda, J. KOH-promoted Pt/Al2O3 catalysts for water gas shift and methanol steam reforming: An operando DRIFTS-MS study. Appl. Catal. B Environ. 2017, 201, 169–181. [Google Scholar] [CrossRef]
- Hwang, K.-R.; Ihm, S.-K.; Park, S.-C.; Park, J.S. Pt/ZrO2 catalyst for a single-stage water-gas shift reaction: Ti addition effect. Int. J. Hydrog. Energy 2013, 38, 6044–6051. [Google Scholar] [CrossRef]
- Ammal, S.C.; Heyden, A. Understanding the Nature and Activity of Supported Platinum Catalysts for the Water–Gas Shift Reaction: From Metallic Nanoclusters to Alkali-Stabilized Single-Atom Cations. ACS Catal. 2019, 9, 7721–7740. [Google Scholar] [CrossRef]
- Stamatakis, M.; Chen, Y.; Vlachos, D.G. First-Principles-Based Kinetic Monte Carlo Simulation of the Structure Sensitivity of the Water–Gas Shift Reaction on Platinum Surfaces. J. Phys. Chem. C 2011, 115, 24750–24762. [Google Scholar] [CrossRef]
- Fajín, J.L.; Cordeiro, M.N.D. Probing the efficiency of platinum nanotubes for the H2 production by water gas shift reaction: A DFT study. Appl. Catal. B Environ. 2020, 263, 118301. [Google Scholar] [CrossRef]
- Lin, C.-H.; Chen, C.-L.; Wang, J.-H. Mechanistic Studies of Water–Gas-Shift Reaction on Transition Metals. J. Phys. Chem. C 2011, 115, 18582–18588. [Google Scholar] [CrossRef]
- Clay, J.P.; Greeley, J.; Ribeiro, F.H.; Delgass, W.N.; Schneider, W.F. DFT comparison of intrinsic WGS kinetics over Pd and Pt. J. Catal. 2014, 320, 106–117. [Google Scholar] [CrossRef] [Green Version]
- Lian, X.; Guo, W.; Shu, J.; Zhang, X.; Liu, Z.; Zhang, Y.; Liu, R. A density functional theory study of the water–gas shift reaction promoted by Pt-based catalysts. Theor. Chem. Acc. 2015, 134. [Google Scholar] [CrossRef]
- Guo, L.; Li, A.; An, X.; Cao, Z.; Liu, N. Catalytic activity of TM@Cu12 core–shell nanoclusters for water gas shift reaction. Int. J. Hydrog. Energy 2015, 40, 8330–8340. [Google Scholar] [CrossRef]
- Fang, L.; Chen, T.; Meng, Y.; Wang, Y.; Xue, J.; Ni, Z.; Xia, S. Water-gas shift reaction catalyzed by Au32M6 (M = Cu, Pt, Pd, Rh, Ir) core-shell nanoclusters: A density functional theory study. Mol. Catal. 2020, 483, 110757. [Google Scholar] [CrossRef]
- Cao, Z.; Guo, L.; Liu, N. A Theoretical Study of the Water–Gas-Shift Reaction on Cu6TM (TM = Co, Ni, Cu, Rh, Pd, Ag, Ir, Pt, Au) Clusters. J. Clust. Sci. 2015, 27, 523–535. [Google Scholar] [CrossRef]
- Xue, J.-L.; Fang, L.; Luo, W.; Meng, Y.; Chen, T.; Xia, S.; Ni, Z.-M. Density functional study of water gas shift reaction catalyzed by Cu-Pt-Au ternary alloy. J. Fuel Chem. Technol. 2019, 47, 688–696. [Google Scholar] [CrossRef]
- Xia, S.; Fang, L.; Meng, Y.; Zhang, X.; Zhang, L.; Yang, C.; Ni, Z. Water-gas shift reaction catalyzed by layered double hydroxides supported Au-Ni/Cu/Pt bimetallic alloys. Appl. Catal. B Environ. 2020, 272, 118949. [Google Scholar] [CrossRef]
- Williams, W.D.; Greeley, J.; Delgass, W.N.; Ribeiro, F.H. Water activation and carbon monoxide coverage effects on maximum rates for low temperature water-gas shift catalysis. J. Catal. 2017, 347, 197–204. [Google Scholar] [CrossRef] [Green Version]
- Ammal, S.C.; Heyden, A. Nature of Ptn/TiO2(110) Interface under Water-Gas Shift Reaction Conditions: A Constrained ab Initio Thermodynamics Study. J. Phys. Chem. C 2011, 115, 19246–19259. [Google Scholar] [CrossRef]
- Blöchl, P.E. Projector augmented-wave method. Phys. Rev. B 1994, 50, 17953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azzam, K.; Babich, I.; Seshan, K.; Lefferts, L. Role of Re in Pt–Re/TiO2 catalyst for water gas shift reaction: A mechanistic and kinetic study. Appl. Catal. B Environ. 2008, 80, 129–140. [Google Scholar] [CrossRef]
- Ammal, S.C.; Heyden, A. Origin of the unique activity of Pt/TiO2 catalysts for the water–gas shift reaction. J. Catal. 2013, 306, 78–90. [Google Scholar] [CrossRef]
- Stegelmann, C.; Andreasen, A.; Campbell, C.T. Degree of Rate Control: How Much the Energies of Intermediates and Transition States Control Rates. J. Am. Chem. Soc. 2009, 131, 8077–8082. [Google Scholar] [CrossRef]
- Grabow, L.C.; Gokhale, A.A.; Evans, S.T.; Dumesic, J.A.; Mavrikakis, M. Mechanism of the Water Gas Shift Reaction on Pt: First Principles, Experiments, and Microkinetic Modeling. J. Phys. Chem. C 2008, 112, 4608–4617. [Google Scholar] [CrossRef]
- Ammal, S.C.; Heyden, A. Water–Gas Shift Catalysis at Corner Atoms of Pt Clusters in Contact with a TiO2 (110) Support Surface. ACS Catal. 2014, 4, 3654–3662. [Google Scholar] [CrossRef]
- Walker, E.; Ammal, S.C.; Terejanu, G.A.; Heyden, A. Uncertainty Quantification Framework Applied to the Water–Gas Shift Reaction over Pt-Based Catalysts. J. Phys. Chem. C 2016, 120, 10328–10339. [Google Scholar] [CrossRef]
- Hammer, B.; Hansen, L.B.; Nørskov, J.K. Improved adsorption energetics within density-functional theory using revised Perdew-Burke-Ernzerhof functionals. Phys. Rev. B 1999, 59, 7413–7421. [Google Scholar] [CrossRef] [Green Version]
- Heyd, J.; Scuseria, G.E.; Ernzerhof, M. Hybrid functionals based on a screened Coulomb potential. J. Chem. Phys. 2003, 118, 8207. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Truhlar, D.G. A new local density functional for main-group thermochemistry, transition metal bonding, thermochemical kinetics, and noncovalent interactions. J. Chem. Phys. 2006, 125, 194101. [Google Scholar] [CrossRef] [Green Version]
- Plessis, S.; Carrasco, N.; Pernot, P. Knowledge-based probabilistic representations of branching ratios in chemical networks: The case of dissociative recombinations. J. Chem. Phys. 2010, 133, 134110. [Google Scholar] [CrossRef] [PubMed]
- Kullback, S.; Leibler, R.A. On Information and Sufficiency. Ann. Math. Stat. 1951, 22, 79–86. [Google Scholar] [CrossRef]
- Walker, E.; Mitchell, D.; Terejanu, G.A.; Heyden, A. Identifying Active Sites of the Water–Gas Shift Reaction over Titania Supported Platinum Catalysts under Uncertainty. ACS Catal. 2018, 8, 3990–3998. [Google Scholar] [CrossRef] [Green Version]
- Ammal, S.C.; Heyden, A. Water-Gas Shift Activity of Atomically Dispersed Cationic Platinum versus Metallic Platinum Clusters on Titania Supports. ACS Catal. 2016, 7, 301–309. [Google Scholar] [CrossRef]
- Ammal, S.C.; Heyden, A. Titania-Supported Single-Atom Platinum Catalyst for Water-Gas Shift Reaction. Chem. Ing. Tech. 2017, 89, 1343–1349. [Google Scholar] [CrossRef]
- Rodriguez, J.A.; Grinter, D.C.; Liu, Z.; Palomino, R.M.; Senanayake, S.D. Ceria-based model catalysts: Fundamental studies on the importance of the metal–ceria interface in CO oxidation, the water–gas shift, CO2 hydrogenation, and methane and alcohol reforming. Chem. Soc. Rev. 2017, 46, 1824–1841. [Google Scholar] [CrossRef]
- Vecchietti, J.; Bonivardi, A.; Xu, W.; Stacchiola, D.J.; Delgado, J.J.; Calatayud, M.; Collins, S.E. Understanding the Role of Oxygen Vacancies in the Water Gas Shift Reaction on Ceria-Supported Platinum Catalysts. ACS Catal. 2014, 4, 2088–2096. [Google Scholar] [CrossRef]
- Bruix, A.; Rodríguez, J.A.; Ramirez, P.J.; Senanayake, S.D.; Evans, J.; Park, J.B.; Stacchiola, D.J.; Liu, P.; Hrbek, J.; Illas, F. A New Type of Strong Metal–Support Interaction and the Production of H2 through the Transformation of Water on Pt/CeO2(111) and Pt/CeOx/TiO2(110) Catalysts. J. Am. Chem. Soc. 2012, 134, 8968–8974. [Google Scholar] [CrossRef]
- Zhou, Y.; Perket, J.M.; Zhou, J. Growth of Pt Nanoparticles on Reducible CeO2(111) Thin Films: Effect of Nanostructures and Redox Properties of Ceria. J. Phys. Chem. C 2010, 114, 11853–11860. [Google Scholar] [CrossRef]
- Senanayake, S.D.; Rodriguez, J.A.; Stacchiola, D.J. Electronic Metal–Support Interactions and the Production of Hydrogen through the Water-Gas Shift Reaction and Ethanol Steam Reforming: Fundamental Studies with Well-Defined Model Catalysts. Top. Catal. 2013, 56, 1488–1498. [Google Scholar] [CrossRef]
- Aranifard, S.; Ammal, S.C.; Heyden, A. Nature of Ptn/CeO2 (111) Surface under Water–Gas Shift Reaction Conditions: A Constrained ab Initio Thermodynamics Study. J. Phys. Chem. C 2012, 116, 9029–9042. [Google Scholar] [CrossRef]
- Aranifard, S.; Ammal, S.C.; Heyden, A. On the Importance of the Associative Carboxyl Mechanism for the Water-Gas Shift Reaction at Pt/CeO2 Interface Sites. J. Phys. Chem. C 2014, 118, 6314–6323. [Google Scholar] [CrossRef]
- Aranifard, S.; Ammal, S.C.; Heyden, A. On the importance of metal–oxide interface sites for the water–gas shift reaction over Pt/CeO2 catalysts. J. Catal. 2014, 309, 314–324. [Google Scholar] [CrossRef]
- Carrasquillo-Flores, R.; Gallo, J.M.R.; Hahn, K.R.; Dumesic, J.A.; Mavrikakis, M. Density Functional Theory and Reaction Kinetics Studies of the Water-Gas Shift Reaction on Pt-Re Catalysts. ChemCatChem 2013, 5, 3690–3699. [Google Scholar] [CrossRef]
- Wang, Y.-X.; Wang, G.-C. A systematic theoretical study of the water gas shift reaction on the Pt/ZrO2 interface and Pt(111) face: Key role of a potassium additive. Catal. Sci. Technol. 2020, 10, 876–892. [Google Scholar] [CrossRef]
- Ghanekar, P.; Kubal, J.; Cui, Y.; Mitchell, G.; Delgass, W.N.; Ribeiro, F.; Greeley, J. Catalysis at Metal/Oxide Interfaces: Density Functional Theory and Microkinetic Modeling of Water Gas Shift at Pt/MgO Boundaries. Top. Catal. 2020, 1–15. [Google Scholar] [CrossRef]
- Rajesh, T.; Devi, R.N. Pt States in BaCe0.98Pt0.02O3−δ during Start Up and Shut Down Operations Under Different Conditions: Stability and Activity of Ionic Pt in Water Gas Shift Reaction. Catal. Lett. 2014, 144, 2227–2232. [Google Scholar] [CrossRef]
- Ribeiro, R.; Meira, D.; Rodella, C.; Oliveira, D.; Bueno, J.M.C.; Zanchet, D. Probing the stability of Pt nanoparticles encapsulated in sol–gel Al2O3 using in situ and ex situ characterization techniques. Appl. Catal. A Gen. 2014, 485, 108–117. [Google Scholar] [CrossRef]
- Liu, B.; Goldbach, A.; Xu, H. Sour water–gas shift reaction over Pt/CeO2 catalysts. Catal. Today 2011, 171, 304–311. [Google Scholar] [CrossRef]
- Liu, B.; Xu, H.; Zhang, Z. Platinum based core–shell catalysts for sour water–gas shift reaction. Catal. Commun. 2012, 26, 159–163. [Google Scholar] [CrossRef]
- Neto, R.C.; Monteiro, I.; Maximino, R.; De Azevedo, J.T. Longevity test for a Water–Gas Shift catalyst. Int. J. Hydrog. Energy 2014, 39, 5242–5247. [Google Scholar] [CrossRef]
- Silva, L.P.; Terra, L.E.; Coutinho, A.C.; Passos, F.B. Sour water–gas shift reaction over Pt/CeZrO2 catalysts. J. Catal. 2016, 341, 1–12. [Google Scholar] [CrossRef]
- Silva, L.; Luis, E.; Ana, C. Sulphur-Tolerant Catalyst for Use in Water-Gas Shift Reactions, and Water-Gas Shift Process. Patent WO2016197211Al, 15 December 2016. [Google Scholar]
- Silva, L.P.; Freitas, M.M.; Santos, R.M.; Perez, G.; Terra, L.E.; Coutinho, A.C.; Passos, F.B. The effect of metal type on the sulfur tolerance of catalysts supported on niobia for sour water-gas shift reaction. Int. J. Hydrog. Energy 2018, 43, 3190–3202. [Google Scholar] [CrossRef]
- Lombardo, E.; Cornaglia, C.; Múnera, J. Development of an active, selective and durable water-gas shift catalyst for use in membrane reactors. Catal. Today 2016, 259, 165–176. [Google Scholar] [CrossRef]
- Kambolis, A.; Lizarraga, L.; Tsampas, M.N.; Burel, L.; Rieu, M.; Viricelle, J.-P.; Vernoux, P. Electrochemical promotion of catalysis with highly dispersed Pt nanoparticles. Electrochem. Commun. 2012, 19, 5–8. [Google Scholar] [CrossRef]
- Hajar, Y.M.; Venkatesh, B.; Baranova, E.A. Electrochemical Promotion of Nanostructured Palladium Catalyst for Complete Methane Oxidation. Catalysts 2019, 9, 48. [Google Scholar] [CrossRef] [Green Version]
- González-Cobos, J.; Valverde, J.L.; De Lucas-Consuegra, A. Electrochemical vs. chemical promotion in the H2 production catalytic reactions. Int. J. Hydrog. Energy 2017, 42, 13712–13723. [Google Scholar] [CrossRef]
- Souentie, S.; Lizarraga, L.; Kambolis, A.; Alves-Fortunato, M.; Valverde, J.L.; Vernoux, P. Electrochemical promotion of the water–gas shift reaction on Pt/YSZ. J. Catal. 2011, 283, 124–132. [Google Scholar] [CrossRef]
- Sekine, Y.; Yamagishi, K.; Nogami, Y.; Manabe, R.; Oshima, K.; Ogo, S. Low Temperature Catalytic Water Gas Shift in an Electric Field. Catal. Lett. 2016, 146, 1423–1428. [Google Scholar] [CrossRef]
- Cui, X.; Su, H.-Y.; Chen, R.; Yu, L.; Dong, J.; Mac, C.; Wang, S.; Li, J.-F.; Yang, F.; Xiao, J.; et al. Room-temperature electrochemical water–gas shift reaction for high purity hydrogen production. Nat. Commun. 2019, 10, 86. [Google Scholar] [CrossRef] [Green Version]
- Ladebeck, J.R.; Wagner, J.P. Catalyst development for water-gas shift. Handb. Fuel Cells 2010, 3, 190–201. [Google Scholar]
- Makets Insider. Available online: https://markets.businessinsider.com/commodities (accessed on 12 June 2020).
- Tonkovich, A.; Zilka, J.; Lamont, M.; Wang, Y.; Wegeng, R. Microchannel reactors for fuel processing applications. I. Water gas shift reactor. Chem. Eng. Sci. 1999, 54, 2947–2951. [Google Scholar] [CrossRef]

















| Catalyst | TOF (s−1) | Ea (kJ·mol−1) | |
|---|---|---|---|
| 240 °C | 280 °C | ||
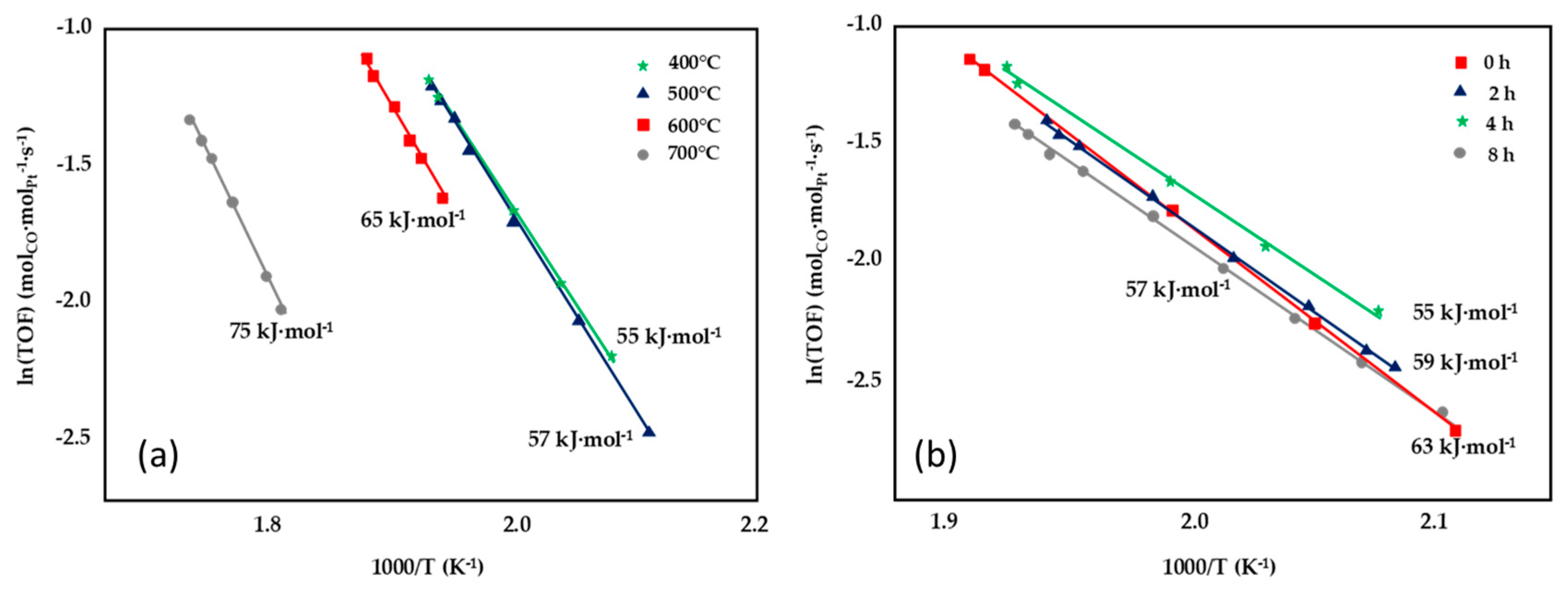
| Pt/CeO2 | 0.38 | 1.30 | 55 |
| Pt/Ce0.8Zr0.2O2 | 0.19 | 0.70 | 57 |
| Pt/Ce0.6Zr0.4O2 | 0.14 | 0.49 | 59 |
| Pt/Ce0.4Zr0.6O2 | 0.09 | 0.36 | 65 |
| Pt/Ce0.2Zr0.8O2 | 0.06 | 0.22 | 72 |
| Pt/ZrO2 | 0.05 | 0.14 | 83 |
| Support | Pt Content (wt%) | Reaction Orders | Ea (kJ·mol−1) | TOF at 270 °C/× 10−3 molH2·(Surface molPt)−1·s−1 | |||
|---|---|---|---|---|---|---|---|
| CO | H2O | CO2 | H2 | ||||
| SiO2 | 4.3 | 0.1 | 0.6 | 0.0 | −0.1 | 74 | 85 |
| 1.2Mo/SiO2 | 1.9 | 0.1 | 0.8 | 0.1 | −0.2 | 48 | 260 |
| 4.2Mo/SiO2 | 1.8 | 0.5 | 0.8 | −0.1 | −0.3 | 50 | 100 |
| 9.0Mo/SiO2 | 1.8 | 0.8 | 0.3 | −0.4 | −0.1 | 42 | 28 |
| Al2O3 | 2.6 | 0.1 | 0.6 | 0.0 | −0.4 | 82 | 17 |
| 0.63Mo/Al2O3 | 1.8 | 0.0 | 0.8 | 0.0 | −0.2 | 44 | 150 |
| 1.4Mo/Al2O3 | 2.1 | 0.0 | 0.8 | −0.1 | −0.2 | 47 | 220 |
| 3.4Mo/Al2O3 | 2.2 | 0.1 | 0.8 | −0.1 | −0.2 | 48 | 150 |
| 7.5Mo/Al2O3 | 1.7 | 0.3 | 0.5 | −0.1 | −0.3 | 58 | 54 |
| 10.7Mo/Al2O3 | 1.7 | 0.8 | 0.5 | −0.7 | −0.5 | 63 | 5 |
| CO Path (Support) | H2O Path | CO Path (Pt Particles) |
|---|---|---|
| CO + * = CO* CO* + O* → CO2 + 2* * + Obulk = O* * + mCO ⟷ (CO)m* | H2O* + * → OH* + H* 2OH* → H2O + O* + * 2H* → H2 H2 + O* → OH* + H* | PtO + CO ⟷ CO2 + Pt Pt + O* ⟷ PtO + * |
| Mechanism | Reaction Temperature (°C) | Catalysts (Pt Particles Size) | Ref. |
|---|---|---|---|
| Associative | 200–450 | Pt/HfO2 (~2 nm) | [109] |
| Redox | 200–350 | Pt/CeO2-TiO2 (1.1–2.0 nm) | [111] |
| Associative | 150–450 | Pt/CeO2 (sol-gel method) (~0.5–2 nm) | [114] |
| Redox | 200–400 | Pt/Ce0.6Y0.4O2 (~2.3–3.4 nm) | [115] |
| Redox + associative | 200–330 | Pt/CeO2, Pt/CeO2-TiO2 (1.8–2 nm) | [116] |
| Redox | 200–330 | Pt/TiO2 (1.9 nm) | [116] |
| Redox | 100–400 | Pt/CeO2-TiO2 | [117] |
| Associative | 200–300 | CexCa1−xOy (~1.5 nm) | [119] |
| Associative | 270 | Pt/S5C95 (not specified) | [121] |
| Associative | 250 | Pt/CeO2, Pt/CeO2-ZrO2, Pt/ZrO2 (not specified) | [122] |
| Redox (different carbonate species) | 200–300 | Pt/CeO2-ZrO2 (1.9–2.4 nm) | [123] |
| Proposed mechanism (Table 7) | - | Pt/CeO2–ZrO2(−La2O3) (not specified) | [124] |
| Associative | 250–350 | Pt/Ce0.5La0.5O2−∂ (1.2–1.5 nm) | [125] |
| Redox (La-rich) + associative (Ce-rich) | 200–350 | Pt/Ce1−xLaxO2−∂ (1.0–1.4 nm) | [126] |
| Redox + associative | 300 | Pt/(100−x)wt%Ce0.8La0.2O2−∂-xwt%CNT (5.6–14.4 nm) | [127] |
| Associative | 200–300 | Pt/CexMe1−xO2 (Me = Ba, La, Y, Hf, Zn) (not specified) | [128] |
| Redox + associative | 250–300 | Pt/Ce0.8Ti0.2O2−δ (Pt ~ 1.7 nm) | [129] |
| Redox + associative | 250–300 | Pt/Ce1−xLaxO2−∂ (1.0–1.2 nm) | [130] |
| CO preferential oxidation onto Ptn+ sites in Al2O3 defects | 227–400 | Pt/Al2O3 (not specified) | [131] |
| CO activation on Pt-NP H2O activation on Mo2C | 110–140 | Pt/Mo2C (not specified) | [132] |
| Associative | 250–450 | Pt/HAP (0.8–1.9 nm) | [133] |
| CO preferential oxidation onto Pt-NPs sites | 100–400 | Pt/HZSM-5 (Pt-NPs (not-specified) and SAC) | [134] |
| CO activation through the mechanism proposed in Figure 20 | 50–150 | Pt/MOF (SAC) | [135] |
| Redox (Pt-SAC) + associative (Pt-NPs) | 150–300 | Pt/FeOx (2.1 nm and SAC) | [136] |
| Associative (OCOH intermediate) | 252–402 | Pt(111) single crystal (SAC) | [137] |
| Mechanism | Reaction Temperature (°C) | Catalysts (Metal Particles Size) | Ref. |
|---|---|---|---|
| Associative | 350 | Fe-Pt/SiO2 (not specified) | [140] |
| Associative (HCOO-) formates | 100–350 | Pt/CeO2 (Pt ~2.5 nm) Ru/CeO2 (Ru ~1.5 nm) Pt-Ru/CeO2 (Pt-Ru alloy of ~2 nm) | [141] |
| Redox | 20–220 | Pt substituted Mn3O4 (12–22 nm) | [142] |
| Associative | 250–400 | Na doped Pt/YSZ (Pt ~1.5 nm) | [146] |
| Associative | 200–350 | 2%Pt/2.5%Na/SiO2 (Pt ~1–4 nm) | [147] |
| Associative | 260–300 | 2.6wt%K-Pt/ZrO2 (Pt ~3.6 nm) | [148] |
| Associative | 150–350 | KOH-coated Pt/Al2O3 (Pt ~5 nm) | [149] |
| Associative with red-ox regeneration | 300 | Pt/Ti[20]/ZrO2 (not specified) | [150] |
| Reaction | Elementary Reactions | Mechanism |
|---|---|---|
| 1 | CO* + O* → CO2(g) | Redox |
| 2 | CO* + OH* → COOH* → CO2(g) + H* | Carboxyl |
| 3 | CO* + H* + O* → CHO* + O* → HCOO** → CO2(g) + H* | Formate |
| Elementary Reaction | Ea (eV) | ΔE (eV) |
|---|---|---|
| H2O + * = H* + OH* | 0.11 | −0.14 |
| CO* + OH* = COOH* + * | 0.34 | −0.17 |
| COOH* + OH* = H2O* + CO2* | 6.03 | −1.09 |
| 2H* = H2* + 2* | 0.87 | 0.43 |
| Reaction | RDS | Mechanism | Ea (eV) | ΔE (eV) | k (cm3/mol·s) |
|---|---|---|---|---|---|
| 1 | CO* + O* = CO2* + * | Redox | 4.84 | −0.05 | 8.30 × 1012 |
| 2 | CO* + OH* = COOH* + * | Carboxyl | 3.15 | 0.48 | 1.99 × 1011 |
| 3 | HCOO* + * = CO2* + H* | Formate | 3.09 | −0.24 | 7.03 × 1012 |
| Catalyst | Temperature | Sour Conditions | Stability Test | Ref. |
|---|---|---|---|---|
| BaCe0.98Pt0.2O3−∂ | 300–400 °C | - | 5 h | [191] |
| Pt/CeO2 | 400 °C | 20 ppm H2S | 300 h | [193] |
| Pt@CeO2; Pt@CeO0.67Zr0.33O2 | 450 °C | 20 ppm H2S | 130 h | [194] |
| Pt-based commercial catalyst | 330 °C | 1 ppm H2S | 100 h | [195] |
| Pt on γ-Al2O3, ZrO2, CeO2, Ce0.75Zr0.25O2, Ce0.25Zr0.75O2 | 300 °C | 50 ppm H2S | 20 h | [196] |
| Pt/Nb2O5 | 300 °C | 50 ppm H2S | 2.5 h | [197] |
| Pt/Nb2O5 | 300 °C | 50–1000 ppm H2S | 4 h | [198] |
| Pt(0.6)/La2O3(27)SiO2 | 400 °C | - | 155 h | [199] |
| Selected Catalyst | Preparation Procedure | Operative Condition WGS | CO Conversion (XCO)/Current Density (Temperature) | Ref. |
|---|---|---|---|---|
| Pt/8mol%Y2O3-stabilized-ZrO2 metal loading = 0.83 mg Pt/cm2 | Pt loading by calcination of organometallic paste on the inner and on the outer side of a closed-end tube of Y2O3-stabilized-ZrO2 | GHSV = 1500 h−1; PH2O = 3.1 kPa; PCO = 0.1 kPa η = −1.5 V | XCO ≈ 95% (T = 350 °C) | [203] |
| 1 wt%Pt/10 mol%La–ZrO2 | La–ZrO2By polymerized complex method Pt loading by impregnation | WHSV = 117,000 ml·gcat−1·h−1 H2O/CO = 1 I = 11 mA | XCO ≈ 70% (T = 500 °C) | [204] |
| Pt2.7Cu@CNTs | By impregnation method | WHSV = 333,333 mlCO·gcat−1·h−1 P = 1 bar η = 0.6 V | Current density = 70 mA/cm2 (T = 25 °C) | [205] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palma, V.; Ruocco, C.; Cortese, M.; Renda, S.; Meloni, E.; Festa, G.; Martino, M. Platinum Based Catalysts in the Water Gas Shift Reaction: Recent Advances. Metals 2020, 10, 866. https://doi.org/10.3390/met10070866
Palma V, Ruocco C, Cortese M, Renda S, Meloni E, Festa G, Martino M. Platinum Based Catalysts in the Water Gas Shift Reaction: Recent Advances. Metals. 2020; 10(7):866. https://doi.org/10.3390/met10070866
Chicago/Turabian StylePalma, Vincenzo, Concetta Ruocco, Marta Cortese, Simona Renda, Eugenio Meloni, Giovanni Festa, and Marco Martino. 2020. "Platinum Based Catalysts in the Water Gas Shift Reaction: Recent Advances" Metals 10, no. 7: 866. https://doi.org/10.3390/met10070866










