Recovery of Copper and Magnetite from Copper Slag Using Concentrated Solar Power (CSP)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Procedure
3. Results
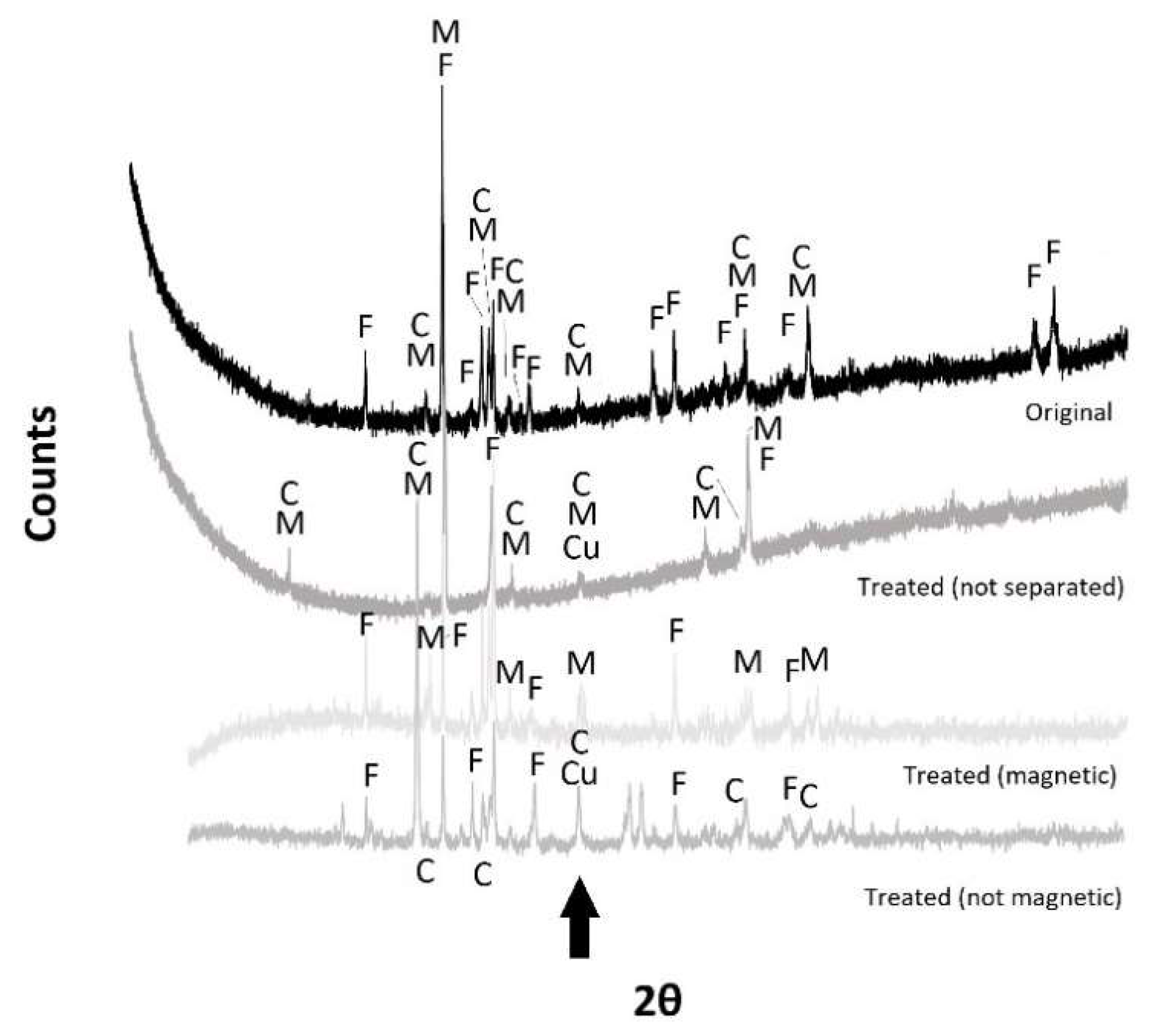
3.1. Macroscopic Analysis
3.2. SEM-EDX
3.3. Size of the Nodules
3.4. Grinding and Magnetic Separation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sancho, J.P.; Verdeja, L.F.; Ballester, A. Metalurgia Extractiva. Volumen II. Procesos de Obtención, 1st ed.; Síntesis: Madrid, Spain, 2000. [Google Scholar]
- Fan, Y.; Shibata, E.; Iizuka, A.; Nakamura, T. Crystallization behaviors of copper smelter slag studied using time-temperature-transformation diagram. Mater. Trans. 2014, 55, 958–963. [Google Scholar] [CrossRef] [Green Version]
- Davenport, W.G.; King, M.; Schlesinger, M.; Biswas, A.K. Extractive Metallurgy of Copper, 4th ed.; Pergamon-Elsevier Science Ltd.: Oxford, UK, 2002. [Google Scholar]
- Nazer, A.; Pavez, O.; Rojas, F.; Aguilar, C. Una revisión de los usos de las escorias de cobre. In Proceedings of the IBEROMET XI. X CONAMET/SAM, Viña del Mar, Chile, 2–5 November 2010. [Google Scholar]
- Coursol, P.; Cardona, N.; Mackey, P.; Bell, S.; Davis, B. Minimization of copper losses in copper smelting slag during electric furnace treatment. JOM 2012, 64, 1305–1313. [Google Scholar] [CrossRef] [Green Version]
- Cardona, N.; Coursol, P.; Vargas, J.; Parra, R. Physical chemistry of copper smelting slags and copper losses at the Paipote smelter. Part 1-Thermodynamic modelling. Can. Metall. Quart. 2011, 50, 318–329. [Google Scholar] [CrossRef]
- Cardona, N.; Coursol, P.; Vargas, J.; Parra, R. Physical chemistry of copper smelting slags and copper losses at the Paipote smelter. Part 2-Characterization of industrial slags. Can. Metall. Quart. 2011, 50, 330–340. [Google Scholar] [CrossRef]
- Madheswaran, C.K.; Ambily, P.S.; Dattatreaya, J.K.; Rajamane, N.P. Studies on use of copper slag as replacement material for river sand in building constructions. J. Inst. Eng. Ser. A 2014, 95, 169–177. [Google Scholar] [CrossRef] [Green Version]
- Nazer, A.; Pavez, O.; Rojas, F. Use of copper slag in cement mortar. REM Rev. Esc. Minas 2012, 65, 87–91. [Google Scholar] [CrossRef] [Green Version]
- Shi, C.; Meyer, C.; Behnood, A. Utilization of copper slag in cement and concrete. Resour. Conserv. Recy. 2008, 52, 1115–1120. [Google Scholar] [CrossRef]
- Potysz, A.; Van Hullebusch, E.D.; Kierczak, J.; Grybos, M.; Lens, P.N.L.; Guibaud, G. Copper metallurgy slags- Current knowledge and fate: A review. Crit. Rev. Env. Sci. Tec. 2015, 45, 2424–2488. [Google Scholar] [CrossRef]
- Palacios, J.; Sánchez, M. Wastes as resources: Update on recovery of valuable metals from copper slags. Miner. Process. Extr. Metall. 2011, 120, 218–223. [Google Scholar] [CrossRef]
- Cendoya, P. Efecto en la resistencia de las escorias de fundición de cobre como agregado fino en el comportamiento resistente del hormigón. Ingeniare Rev. Chil. Ing. 2009, 17, 85–94. [Google Scholar] [CrossRef] [Green Version]
- Murari, K.; Siddique, R.; Jain, K.K. Use of waste copper slag, a sustainable material. J. Mater. Cycles. Waste Manag. 2015, 17, 13–26. [Google Scholar] [CrossRef]
- Kambham, K.; Sangameswaran, S.; Datar, S.R.; Kura, B. Copper slag: Optimization of productivity and consumption for cleaner production in dry abrasive blasting. J. Clean Prod. 2007, 15, 465–473. [Google Scholar] [CrossRef]
- Jiménez-Padilla, B. Armado de Tuberías. FMEC0108, 1st ed.; IC Editorial: Antequera, Spain, 2014. [Google Scholar]
- Biswas, S.; Satapathy, A. Use of copper slag in glass-epoxy composites for improved wear resistance. Waste Manag. Res. 2010, 28, 615–625. [Google Scholar] [CrossRef]
- Pundhir, N.K.S.; Kamaraj, C.; Nanda, P.K. Use of copper slag as construction material in bituminous pavements. J. Sci. Ind. Res. India. 2005, 64, 997–1002. [Google Scholar]
- Busolic, D.; Parada, F.; Parra, R.; Sánchez, M.; Palacios, J.; Hino, M. Recovery of iron from copper flash smelting slags. Miner. Process. Extr. Metall. 2011, 120, 32–36. [Google Scholar] [CrossRef]
- Guo, Z.; Zhu, D.; Pan, J.; Wu, T.; Zhang, F. Improving beneficiation of copper and iron from copper slag by modifying the molten copper slag. Metals 2016, 6, 86. [Google Scholar] [CrossRef] [Green Version]
- Xian-Lin, Z.; De-Qing, Z.; Jian, P.; Teng-Jiao, W. Utilization of waste copper slag to produce directly reduced iron for weathering resistant steel. ISIJ Int. 2015, 55, 1347–1352. [Google Scholar] [CrossRef] [Green Version]
- Heo, J.H.; Chung, Y.; Park, J.H. Recovery of iron and removal of hazardous elements from waste copper slag via a novel aluminothermic smelting reduction (ASR) process. J. Clean. Prod. 2016, 137, 777–787. [Google Scholar] [CrossRef]
- Siwiec, G.; Sozanska, M.; Blacha, L.; Smalcerz, A. Behaviour of iron during reduction of slag obtained from copper flash smelting. Metabk 2015, 54, 113–115. [Google Scholar]
- Sarfo, P.; Wyss, G.; Ma, G.; Das, A.; Young, C. Carbothermal reduction of copper smelter slag for recycling into pig iron and glass. Miner. Eng. 2017, 107, 8–19. [Google Scholar] [CrossRef]
- Liao, Y.; Zhou, J.; Huang, F. Separating and recycling of Fe, Cu, Zn from dumped copper slag by microwave irradiation assisted carbothermic method. J. Residuals Sci. Technool. 2016, 13, S155–S160. [Google Scholar] [CrossRef] [Green Version]
- Li, K.; Ping, S.; Wang, H.; Ni, W. Recovery of iron from copper slag by deep reduction and magnetic beneficiation. Int. J. Min. Met. Mater. 2013, 20, 1035–1041. [Google Scholar] [CrossRef]
- Fernández-González, D.; Ruiz-Bustinza, I.; González-Gasca, C.; Piñuela-Noval, J.; Mochón-Castaños, J.; Sancho-Gorostiaga, J.; Verdeja, L.F. Concentrated solar energy applications in materials science and metallurgy. Sol. Energy 2018, 170, 520–540. [Google Scholar] [CrossRef]
- Murray, J.P.; Flamant, G.; Roos, C.J. Silicon and solar-grade silicon production by solar dissociation of Si3N4. Sol. Energy 2006, 80, 1349–1354. [Google Scholar] [CrossRef]
- Loutzenhiser, P.G.; Tuerk, O.; Steinfeld, A. Production of Si by vacuum carbothermal reduction of SiO2 using concentrated solar energy. JOM 2010, 62, 49–54. [Google Scholar] [CrossRef]
- Murray, J.P. Aluminum production using high-temperature solar process heat. Sol. Energy 1999, 66, 133–142. [Google Scholar] [CrossRef]
- Murray, J.P. Aluminum-silicon carbothermal reduction using high-temperature solar process heat. In Proceedings of the 128th TMS Annual Meeting, San Diego, CA, USA, 28 February–4 March 1999. [Google Scholar]
- Murray, J.P. Solar production of aluminum by direct reduction: Preliminary results for two processes. J. Sol. Energy Eng. 2001, 123, 125–132. [Google Scholar] [CrossRef]
- Lytvynenko, Y.M. Obtaining aluminum by the electrolysis with the solar radiation using. Appl. Sol. Energy 2013, 49, 4–6. [Google Scholar] [CrossRef]
- Epstein, M.; Olalde, G.; Santén, S.; Steinfeld, A.; Wieckert, C. Towards the industrial solar carbothermal production of zinc. J. Sol. Energ. 2008, 130, 014501–014504. [Google Scholar] [CrossRef]
- Fletcher, E.A.; Noring, J.E. High temperature solar electrothermal processing—Zinc from zinc oxide. Energy 1983, 8, 247–254. [Google Scholar] [CrossRef]
- Fletcher, E.A.; Macdonald, F.J.; Kunnerth, D. High temperature solar electrothermal processing—II. Zinc from zinc oxide. Energy 1985, 10, 1255–1272. [Google Scholar] [CrossRef]
- Palumbo, R.D.; Fletcher, E.A. High temperature solar electrothermal processing—III. Zinc from zinc oxide at 1200–1675K using a non-consumable anode. Energy 1988, 13, 319–332. [Google Scholar] [CrossRef]
- Osinga, T.; Frommherz, U.; Steinfeld, A.; Wieckert, C. Experimental investigation of the solar carbothermic reduction of ZnO using a two-cavity solar reactor. J. Sol. Energ. 2004, 126, 633–637. [Google Scholar] [CrossRef]
- Epstein, M.; Ehrensberger, K.; Yogev, A. Ferro-reduction of ZnO using concentrated solar energy. Energy 2004, 29, 745–756. [Google Scholar] [CrossRef]
- Steinfeld, A.; Brack, M.; Meier, A.; Weidenkaff, A.; Wuillemin, D. A solar chemical reactor for co-production of zinc and synthesis gas. Energy 1998, 23, 803–814. [Google Scholar] [CrossRef]
- Wieckert, C.; Palumbo, R.; Frommherz, U. A two-cavity reactor for solar chemical processes: Heat transfer model and application to carbothermic reduction of ZnO. Energy 2004, 29, 771–787. [Google Scholar] [CrossRef]
- Wieckert, C.; Frommherz, U.; Kräupl, S.; Guillot, E.; Olalde, G.; Epstein, M.; Santén, S.; Osinga, T.; Steinfeld, A. A 300 kW solar chemical pilot plant for the carbothermic production of zinc. J. Sol. Energy 2006, 129, 190–196. [Google Scholar] [CrossRef]
- Schunk, L.O.; Lipinski, W.; Steinfeld, A. Heat transfer model of a solar receiver-reactor for the thermal dissociation of ZnO-Experimental validation at 10 kW and scale-up to 1 MW. Chem. Eng. J. 2009, 150, 502–508. [Google Scholar] [CrossRef]
- Villasmil, W.; Brkic, M.; Wuillemin, D.; Meier, A.; Steinfeld, A. Pilot scale demonstration of a 100-kWth solar thermochemical plant for the thermal dissociation of ZnO. J. Sol. Energy 2013, 136, 011016–0111027. [Google Scholar] [CrossRef]
- Koepft, E.; Villasmil, W.; Meier, A. Pilot-scale solar reactor operation and characterization for fuel production via the Zn/ZnO thermochemical cycle. Appl. Energy 2016, 165, 1004–1023. [Google Scholar] [CrossRef]
- Ruiz-Bustinza, I.; Cañadas, I.; Rodríguez, J.; Mochón, J.; Verdeja, L.F.; García-Carcedo, F.; Vázquez, A. Magnetite production from steel wastes with concentrated solar energy. Steel Res. Int. 2013, 84, 207–217. [Google Scholar] [CrossRef]
- Sibieude, F.; Ducarroir, M.; Tofighi, A.; Ambriz, J. High temperature experiments with a solar furnace: The decomposition of Fe3O4, Mn3O4, CdO. Int. J. Hydrogen Energy 1982, 7, 79–88. [Google Scholar] [CrossRef]
- Steinfeld, A.; Fletcher, E.A. Theoretical and experimental investigation of the carbothermic reduction of Fe2O3 using solar energy. Energy 1991, 16, 1011–1019. [Google Scholar] [CrossRef]
- Steinfeld, A.; Kuhn, P.; Karni, J. High-temperature solar thermochemistry: Production of iron and synthesis gas by Fe3O4-reduction with methane. Energy 1993, 18, 239–249. [Google Scholar] [CrossRef]
- Fernández-González, D.; Prazuch, J.; Ruiz-Bustinza, I.; González-Gasca, C.; Piñuela-Noval, J.; Verdeja, L.F. Solar synthesis of calcium aluminates. Sol. Energy 2018, 171, 658–666. [Google Scholar] [CrossRef]
- Mochón, J.; Ruiz-Bustinza, Í.; Vázquez, A.; Fernández, D.; Ayala, J.M.; Barbés, M.F.; Verdeja, L.F. Transformations in the iron-manganese-oxygen-carbon system resulted from treatment of solar energy with high concentration. Steel Res. Int. 2014, 85, 1469–1476. [Google Scholar] [CrossRef]
- Fernández-González, D.; Prazuch, J.; Ruiz-Bustinza, I.; González-Gasca, C.; Piñuela-Noval, J.; Verdeja, L.F. Iron Metallurgy via Concentrated Solar Energy. Metals-Basel 2018, 8, 873. [Google Scholar] [CrossRef] [Green Version]
- Fernández-González, D.; Prazuch, J.; Ruiz-Bustinza, I.; González-Gasca, C.; Piñuela-Noval, J.; Verdeja, L.F. The treatment of Basic Oxygen Furnace (BOF) slag with concentrated solar energy. Sol. Energy 2019, 180, 372–382. [Google Scholar] [CrossRef]
- Fernández-González, D.; Prazuch, J.; Ruiz-Bustinza, I.; González-Gasca, C.; Piñuela-Noval, J.; Verdeja, L.F. Transformations in the Mn-O-Si system using concentrated solar energy. Sol. Energy 2019, 184, 148–152. [Google Scholar] [CrossRef]
- Fernández-González, D.; Prazuch, J.; Ruiz-Bustinza, I.; González-Gasca, C.; Piñuela-Noval, J.; Verdeja, L.F. Transformations in the Si-O-Ca system: Silicon-calcium via solar energy. Sol. Energy 2019, 181, 414–423. [Google Scholar] [CrossRef]
- Fernández-González, D.; Prazuch, J.; Ruiz-Bustinza, I.; González-Gasca, C.; Gómez-Rodríguez, C.; Verdeja, L.F. Treatment of copper slag with concentrated solar energy. In Proceedings of the Environmental Safety-Non-Energy and Raw Materials, I International Conference on Engineering Materials, Safety, Environment and Technology, Zielona Gora, Poland, 3–4 June 2020; Gabryelewicz, I., Wedrychowicz, M., Eds.; University of Zielona Gora: Zielona Gora, Poland, 2021; pp. 39–59. [Google Scholar]
- Winkel, H.E. Thermal Decomposition of Copper Sulfides under Concentrated Irradiation. Ph.D. Thesis, Swiss Federal Institute of Technology Zurich, Zurich, Switzerland, 2006. [Google Scholar]
- Alcock, C.B. Principles of Pyrometallurgy; Academic Press: London, UK, 1976; pp. 15–16. [Google Scholar]
- Ballester, A.; Verdeja, L.F.; Sancho, J.P. Metalurgia Extractiva. Volumen I. Fundamentos, 1st ed.; Síntesis: Madrid, Spain, 2000. [Google Scholar]
- Richardson, F.D.; Jeffers, J.H.E. The Ellingham diagram for metal oxides. In Introduction to Metallurgical Thermodynamics; Hemisphere Publishing Corporation: San Francisco, CA, USA, 1981. [Google Scholar]
- Verdeja, L.F.; Sancho, J.P.; Ballester, A. Refractory and Ceramic Materials, 1st ed.; Síntesis: Madrid, Spain, 2014. [Google Scholar]
- Pero-Sanz, J.A.; Quintana, M.J.; Verdeja, L.F. Solidification and Solid-state Transformations of Metals and Alloys, 1st ed.; Elsevier: Boston, MA, USA, 2017. [Google Scholar]
- Flamant, G.; Ferriere, A.; Laplaze, D.; Monty, D. Solar processing of materials: Opportunities and new frontiers. Sol. Energy 1999, 66, 117–132. [Google Scholar] [CrossRef]
- Alvarez, M.A. Environmental Impact of Plastics. J. Mater. Ed. 2018, 40, 119–124. [Google Scholar]
- Dobiszewska, M. Waste Materials Used in Making Mortar and Concrete. J. Mater. Ed. 2017, 39, 133–156. [Google Scholar]
- Fernández-González, D.; Sancho-Gorostiaga, J.; Piñuela-Noval, J.; Verdeja, L.F. Anodic Lodes and Scrapings as a Source of Electrolytic Manganese. Metals 2018, 8, 162. [Google Scholar] [CrossRef] [Green Version]
- Ordiales, M.; Iglesias, J.; Fernández-González, D.; Sancho-Gorostiaga, J.; Fuentes, A.; Verdeja, L.F. Cold agglomeration of Ultrafine Oxidized Dust (UOD) from ferromanganese and silicomanganese industrial process. Metals 2016, 6, 203. [Google Scholar] [CrossRef] [Green Version]
- Fernández-González, D.; Piñuela-Noval, J.; Verdeja, L.F. Silicomanganese and ferromanganese slags treated with concentrated solar energy. Proceedings 2018, 2, 1450. [Google Scholar] [CrossRef] [Green Version]
- Fernández-González, D. Aplicaciones de la Energía Solar Concentrada en Metalurgia y Ciencia de los Materiales. Ph.D. Thesis, Department of Materials Science and Metallurgical Engineering, University of Oviedo, Oviedo/Uviéu, Asturias, Spain, September 2019. [Google Scholar]












| Material | Process | Temperature | Installation | Researcher |
|---|---|---|---|---|
| Si | Dissociation of Si3N4 by carbothermal reduction of SiO2 under an N2 atmosphere | >1400 °C | OSF | Jean P. Murray, Gilles Flamant, Carolyn J. Roos [28] |
| Si | Carbothermic reduction of SiO2 under vacuum conditions at a high temperature | 1725–2000 °C | PSI | Peter G. Loutzenhiser, Ozan Tuerk, Aldo Steinfeld [29] |
| Al | Production of aluminum via carbothermal reduction | >2000 °C | PSI and OSF | Jean P. Murray [30,31,32] |
| Al | Production of aluminum using both electricity and heat generated using solar energy | ≃1000 °C | UKR | Y. M. Lytvynenko [33] |
| Zn | Production of zinc via CSP to be used in water and carbon dioxide splitting (scaled up to demonstration plant scale [34]) | >1750 °C | Mainly at the PSI, OSF, and WIS | E. A. Fletcher, R. D. Palumbo, T. Osinga, M. Epstein A. Steinfeld, C. Wieckert, L. Schunck, W. Villasmil, E. Koepft, and others [35,36,37,38,39,40,41,42,43,44,45] |
| Fe, Mn, Cd | Treatment of different materials containing iron | >1100 °C | Mainly at the PSI, PSA, OSF, and WIS | F. Sibieude, A. Steinfeld, E. A. Fletcher, I. Ruiz-Bustinza, J. Mochón and others [46,47,48,49] |
| Fe | O | Si | Al | Cu | Ca | Na | S | Others |
|---|---|---|---|---|---|---|---|---|
| 42.82 | 36.15 | 10.36 | 3.24 | 1.84 | 1.76 | 0.99 | 0.52 | 2.32 |
| Sample | Lime | Shutter Opening | Time (min) | Average Incident Radiation (W/m2) | Power (W) |
|---|---|---|---|---|---|
| CuSin1 | No | 80 | 20 | 941.2 | 1129 |
| CuSin2 | No | 88 | 25 | 955.8 | 1262 |
| CuSin3 | No | 41 | 30 | 915.5 | 563 |
| Cusinbonus | No | 60 | 23 | 867.8 | 781 |
| CuCon1 (stop in the middle of the experiment) | Yes | 92 | - | 938 | 1294 |
| CuCon2 | Yes | 100 | 15 | 939.5 | 1409 |
| CuCon3 | Yes | 84 | 20 | 973 | 1226 |
| Element | Original Copper Slag | CuSin1 | CuSin2 | CuSin3 | ||
|---|---|---|---|---|---|---|
| Magnetic | Magnetic | Non-Magnetic | Magnetic | Non-Magnetic | ||
| Cu | 1.84 | 1.34 | 1.29 | 11.72 | 1.27 | 7.09 |
| Fe | 42.82 | 38.53 | 44.75 | 43.51 | 45.45 | 45.34 |
| O | 36.15 | 37.98 | 36.17 | 32.50 | 35.94 | 33.43 |
| Si | 10.36 | 12.28 | 10.39 | 6.20 | 9.85 | 6.90 |
| Al | 3.24 | 5.94 | 2.47 | 1.16 | 2.67 | 1.82 |
| Ca | 1.76 | 0.88 | 1.06 | 1.64 | 1.10 | 2.08 |
| Na | 0.99 | 0.71 | 0.87 | 0.30 | 0.78 | 0.43 |
| K | 0.69 | 0.64 | 0.75 | 0.56 | 0.76 | 0.62 |
| S | 0.52 | 0.25 | 0.63 | 0.88 | 0.60 | 0.58 |
| Others | 1.63 | 1.46 | 1.62 | 1.52 | 1.58 | 1.70 |
| Element | Cubonus | CuCon1 | CuCon2 | CuCon3 | ||
|---|---|---|---|---|---|---|
| Magnetic | Magnetic | Non-Magnetic | Magnetic | Non-Magnetic | Magnetic | |
| Cu | 0.88 | 1.28 | 7.25 | 1.01 | 5.34 | 1.52 |
| Fe | 39.05 | 42.21 | 23.24 | 38.02 | 32.27 | 38.26 |
| O | 37.63 | 34.64 | 35.68 | 37.83 | 34.78 | 37.55 |
| Si | 11.63 | 7.22 | 6.42 | 11.48 | 7.32 | 11.31 |
| Al | 5.80 | 2.85 | 10.02 | 6.50 | 5.24 | 6.31 |
| Ca | 1.93 | 8.52 | 14.29 | 2.05 | 12.24 | 2.22 |
| Na | 0.62 | 0.47 | 0.27 | 0.69 | 0.33 | 0.53 |
| K | 0.67 | 0.62 | 0.46 | 0.67 | 0.46 | 0.65 |
| S | 0.25 | 0.71 | 0.76 | 0.30 | 0.58 | 0.24 |
| Others | 1.53 | 1.47 | 1.61 | 1.45 | 1.44 | 1.42 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-González, D.; Prazuch, J.; Ruiz-Bustinza, Í.; González-Gasca, C.; Gómez-Rodríguez, C.; Verdeja, L.F. Recovery of Copper and Magnetite from Copper Slag Using Concentrated Solar Power (CSP). Metals 2021, 11, 1032. https://doi.org/10.3390/met11071032
Fernández-González D, Prazuch J, Ruiz-Bustinza Í, González-Gasca C, Gómez-Rodríguez C, Verdeja LF. Recovery of Copper and Magnetite from Copper Slag Using Concentrated Solar Power (CSP). Metals. 2021; 11(7):1032. https://doi.org/10.3390/met11071032
Chicago/Turabian StyleFernández-González, Daniel, Janusz Prazuch, Íñigo Ruiz-Bustinza, Carmen González-Gasca, Cristian Gómez-Rodríguez, and Luis Felipe Verdeja. 2021. "Recovery of Copper and Magnetite from Copper Slag Using Concentrated Solar Power (CSP)" Metals 11, no. 7: 1032. https://doi.org/10.3390/met11071032





