1. Introduction
2024 aluminum alloy is a common feature in various industrial applications, such as aeronautics, aviation, automotive, and architecture, as a result of its high strength, light weight, good surface properties, and sufficient corrosion resistance [
1]. An adequate bonding process to join similar or dissimilar materials is commonly required to enable the application of an aluminum alloy. Among the joining techniques for aluminum alloys, tungsten inert gas (TIG) welding, friction stir (FS) welding, laser welding, and brazing are used especially frequently for the manufacturing of aluminum products. However, the difficulties encountered in joining aluminum alloys by these methods remain to be resolved [
2]. TIG welding is known to produce a large deformation, cause parent material fusion issues, be susceptible to porosity [
3], and create a severe heat-affected zone. Investigations suggest that these problems easily cause failures in aluminum alloy workpieces [
4]; moreover, the slow welding speed increases the production cost [
5]. Apart from these issues, TIG welding does not offer operation under vacuum conditions [
6], and the workpieces are bonded by docking rather than face-to-face bonding. Reactive metals usually do not heterogeneously bond by TIG welding [
7].
As one of the alternatives to TIG welding, advanced FS welding mitigates some of the shortcomings of TIG welding, laser welding, and other types of fusion welding methods. Yet, this technology faces considerable problems, such as the formation of keyhole-induced pores in the joint region [
8]. As a result, the workpieces must be patched or mechanically repaired. Therefore, the workpieces need to be fixed very tightly during the FS welding process and are only able to connect horizontally [
9]. In addition, the weld channel zone produces large unpredictable oxide films composed of onion rings and brittle intermetallic compounds, weakening the joints [
10] and causing wear and deformation to the friction stirring tool [
11]. Brazing, wherein metals are bonded together through the insertion of a filler metal with a melting point lower than that of aluminum alloy, has been popularly employed to bonded aluminum alloys. Brazing seems to be ideally suited for the joining of dissimilar metals and only requires a low process temperature; however, the AA2024 alloy has a low solidus temperature of 502 °C, making it difficult to braze with the conventional Al-12Si filler with a eutectic point of 577 °C.
Diffusion bonding is another type of solid-state joining method that allows the joining of metal workpieces through heating at a temperature below the melting point under adequate pressure. In contrast to the abovementioned bonding methods, diffusion bonding of aluminum alloys can be conducted without involving the liquid phase, and no additional filler metals are required. Diffusion-bonded joints usually most nearly resemble the mechanical and metallurgical properties of the parent metal, and the process has little contamination [
12]. In a previous study by Burkhart et al., the diffusion-bonding technique was successfully applied to aerospace superplastic titanium alloy structures [
13]. Bhatta et al. investigated the optimum conditions required for high-quality diffusion bonding to apply it to superplastic aluminum alloys [
14]. Especially with the advent of the technology of aluminum alloys in high-tech industrial applications, more emphasis has been placed on developing high-quality welding methods for aluminum alloys. Diffusion bonding has a few inherent advantages over conventional welding due to its high welding capability, high flexibility, and low deformation, which does not involve any thermal cracking [
15]. With great potential for modern power devices that require large exchange systems [
16,
17], diffusion-bonded joints with a large area range are becoming increasingly important to improve the reliability of heat-sensitive components in portable devices. However, diffusion bonding of aluminum alloys has always faced several challenges. Despite the high weldability properties of the alloy, it also faces limitations such as oxidation issues during bonding. Huang et al. used an organic surface coating to prevent the oxidation of superplastic 7075 aluminum alloy during diffusion bonding. The treated joints achieved an optimum shear strength of over 200 MPa, which was near that of the parent material [
18].
Due to the melting point of aluminum at about 661 °C, the thermal stability of its alloy usually covers a temperature range substantially lower than other higher melting point materials. As a result, the property variation of aluminum alloys at elevated temperatures is highly influenced by their initial microstructure. Strength degradation related to grain growth can be severe in diffusion-bonded aluminum alloy joints due to the exposure to high process temperatures between 0.5 and 0.8 Tm (Tm = absolute melting temperature). To our knowledge, how the solid-state diffusion-bonding parameters affect the mechanical properties of homogenous AA2024 alloys has not been studied. Therein, the effects of post-weld heat treatments for AA2024 alloys after diffusion bonding were investigated and are presented in this study.
3. Results
The microstructure of the AA2024-T351 specimens before diffusion bonding (as-received) is shown in
Figure 3. Precipitates appeared within the matrix of the alloy, as shown in
Figure 3a. The compositions of the precipitates were about Al:Cu = 66.3:33.7 (at.%) and were identified as Al
2Cu intermetallic compounds by EDX spectrometry. The optical microscopy (OM), shown in
Figure 3b, also revealed that the as-received AA2024-T351 specimens had an equiaxial grain distribution with an average size of about 75 μm.
The joint interfaces of the AA2024/AA2024 diffusion bonded at 490 °C under 5 MPa for different bonding periods are shown in
Figure 4. After bonding for 30 min, voids started to appear at the interface, as shown in
Figure 4a. The corresponding optical microscopy (OM) image in
Figure 4e shows that the compressive stress of 5 MPa by the bonding process was sufficient to start to transform the original equiaxial coarse grains to slender grains. After extending the bonding periods from 30 min to 60 min, the voids at the interface disappeared, as shown in
Figure 4b. However, a few slender precipitates began to form at the AA2024/AA2024 bonding interface after the diffusion-bonding process. After 120 min of bonding, the interfacial precipitates started to form coarse slender particles, as shown in
Figure 4g. These slender grains within the AA2024 matrix grew with a prolonged bonding period. After 240 min of diffusion bonding at 490 °C, the coarse slender Al
2Cu precipitates remained at the void-free interface of the diffusion-bonded AA2024/AA2024 joint, as shown in
Figure 4h. However, the slender grains in the AA2024 matrix recrystallized into numerous approximately equiaxial grains with an average grain size of 32 μm, which was much finer than that of the as-received AA2024-T351 specimen before diffusion bonding.
The shear strength of the AA2024/AA2024 joints after diffusion bonding at 490 °C under the bonding pressure of 5 MPa is summarized in
Figure 5. The shear strength increased from 62.5 MPa to 79.7 MPa by lengthening the bonding time from 30 min to 120 min; it reached its maximum of 81.2 MPa at a bonding time of 240 min. On the other hand, decreasing the bonding temperature to 470 °C for 30 min of bonding resulted in a large drop in the shear strength from 62.5 MPa to 42.8 MPa. Although the bonding strength was improved to 68.7 MPa by extending the bonding period to 240 min, further decreasing the bonding temperature to 460 °C and 450 °C for 30 min continued to degrade the shear strength of the AA2024/AA2024 joints, to values of 41.3 MPa and 40.5 MPa, respectively. At lower temperatures, the shear strength was slightly improved by extending the bonding period. Diffusion bonding at 440 °C for 30 min resulted in a relatively low strength of 32.9 MPa. At this temperature, lengthening the bonding period to 240 min could not increase the shear strength as compared to the joints that were diffusion bonded at higher temperatures for a similar period of time.
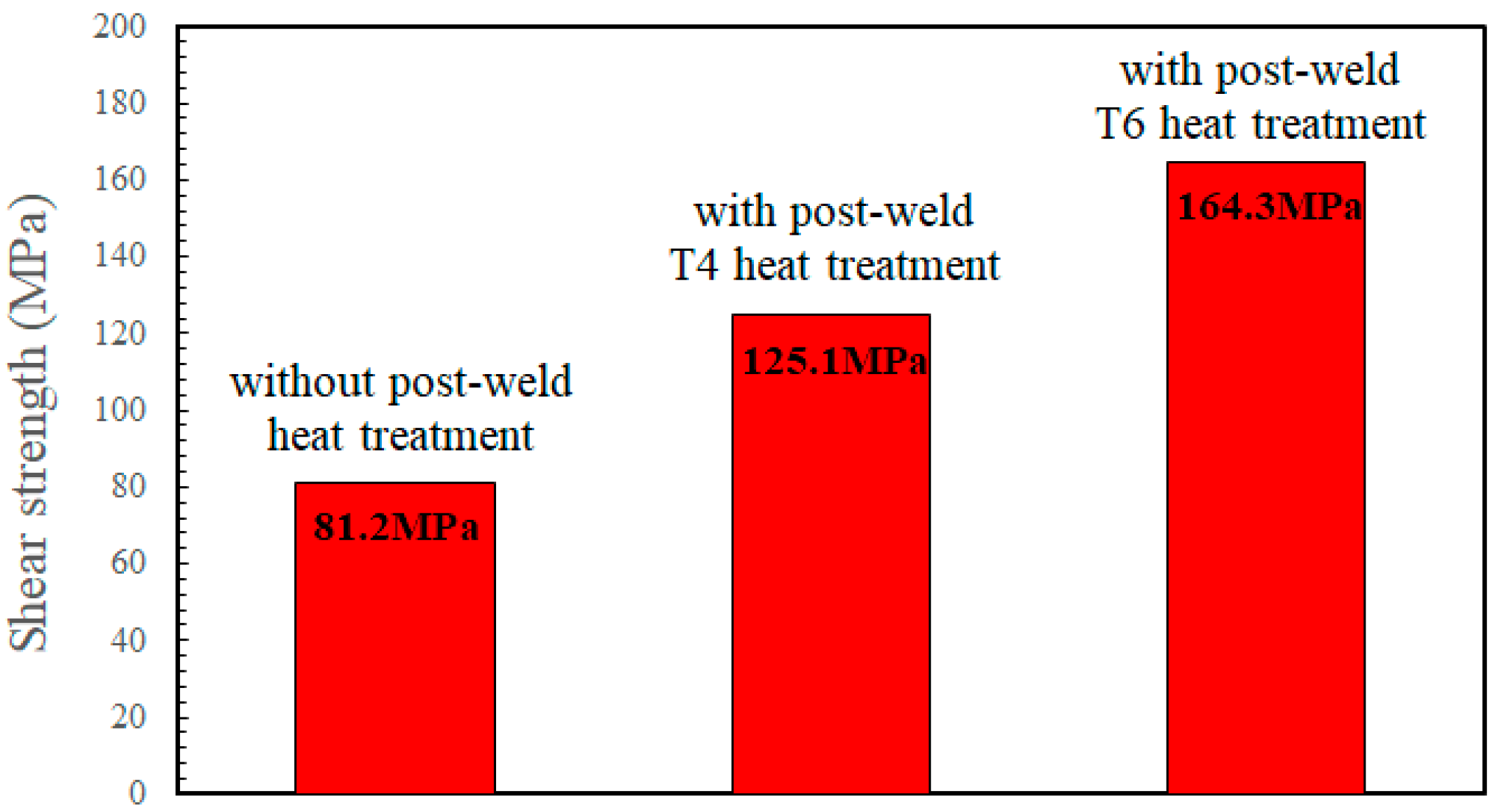
The AA2024/AA2024 joint diffusion bonded at 490 °C for 240 min was further heat treated under the T4 and T6 conditions, respectively, to improve the bonding performance.
Figure 6 shows that the T4 heat-treated (solid solution at 495 °C for 1 h and then water quenched) specimen exhibited an obvious improvement in shear strength, from 81.2 MPa to 125.1 MPa. Further aging at 191 °C for 12 h (T6 heat treatment) resulted in an optimum strength of 164.3 MPa, which was approximately 58% of the shear strength (283 MP) of the T6 heat-treated AA2024 alloy [
20]. Saleh [
21] diffusion bonded 2014 aluminum alloys using copper as an interlayer. The best tensile strength of 188.36 MPa was obtained under a joint pressure of 4 MPa at 475 °C. The study of Venugopal et. al. [
22] pointed out that a higher bonding shear strength of diffusion bonded 5083 aluminum alloy could be obtained under higher temperatures and at higher bonding pressure. A shear strength of 119.46 MPa was obtained by diffusion bonding at 520 °C with a bonding pressure of 10 MPa for 30 min. The bonding strength of about 83–114 MPa was obtained in the study of Zinong et al. [
23], who used a large-deformation hot-roll joining method to join 1060 aluminum alloy at a high temperature of 550–600 °C.
The SEM images in
Figure 7a,c and the OM images in
Figure 7b,d reveal the microstructure of the AA2024/AA2024 joint after the post-weld T4 and T6 heat treatments. The slender coarse precipitates formed at the bonding interface in
Figure 7b disappeared after the heat treatments and were replaced by larger grains (>150 μm) in the AA2024 matrix. Only a few voids were observed at the interface of the T4 heat-treated joint. Other than the obvious improvements in the bonding strength after the post-weld T4 and T6 heat treatments, the microhardness of the AA2024/AA2024 joints across the bonding interface was measured, as plotted in
Figure 8b,c, which was higher than the hardness for the as-bonded AA2024/AA2024 joints (
Figure 8a). For the T6 heat-treated AA2024/AA2024 joint, the measured microhardness was more or less the same as the T4 heat-treated AA2024/AA2024 joint. The microhardness at the interface after diffusion bonding was about 106.3 Hv at a bonding temperature of 490 °C for 240 min, which was similar to the hardness values of the substrate. After T4 heat treatment, the hardness at the interface increased to 138.3 Hv. The hardness in the substrate also increased to about the same hardness value. However, compared with the as-bonded or T4 heat-treated specimens, the difference between the two measured values of hardness after the T6 heat treatment was slightly larger, and the hardness distribution within the substrate was less uniform.
The doubling of the shear strength from 81.2 MPa to 164.3 MPa after the T6 heat treatment for the AA2024/AA2024 joint compared with the as-bonded AA2024/AA2024 joint diffusion bonded at 490 °C for 240 min was attributed to the precipitation hardening of the AA2024 matrix due to the aging effect of the T6 heat treatment, which was consistent with the microhardness measurements of the specimens. As shown in
Figure 8c, the microhardness of the AA2024/AA2024 joint reached its maximum value at approximately 160 Hv after the T6 heat treatment at 191 °C for 12 h.
The fractured surfaces after the shear tests for the AA2024/AA2024 joints before and after the post-weld heat treatments are shown in
Figure 9. Both the as-bonded and the post-weld heat-treated specimens fractured along the bonding interfaces.
Figure 9a shows an obvious shear band characteristic in the fractography of the as-bonded AA2024/AA2024 joint. The appearance of such a shear band structure on the fracture surface of the as-bonded AA2024/AA2024 joints can be correlated with the softening effect of the precipitation-free zone near the slender coarse Al
2Cu precipitates grown at the bonding interface. The existence of these coarse precipitates can cause severe crack issues along the interface, leading to local peeling at the joints. In contrast, the post-weld T4 and T6 heat treatments caused the formation of dimple structures in the AA2024/AA2024 joints, as shown in
Figure 9b,c. The T4 and T6 post-weld heat treatments significantly improved the mechanical properties of the diffusion bonded AA2024/AA2024 joints by the dissolution of the slender coarse Al
2Cu precipitates formed at the as-bonded interfaces. Furthermore, the aging process of T6 heat treatment created precipitates in the AA2024/AA2024 matrix, contributing a strengthening effect to the overall microstructure of the AA2024/AA2024 joints. Combining the dissolution effect and the precipitation hardening of the AA2024 matrix by the post-weld T6 heat treatment, the shear strength of the joint was successfully optimized up to 164.3 MPa.