Effect of Surface-Active Element Oxygen on Heat and Mass Transfer in Laser Welding of Dissimilar Metals: Numerical and Experimental Study
Abstract
:1. Introduction
2. Experimental Procedure
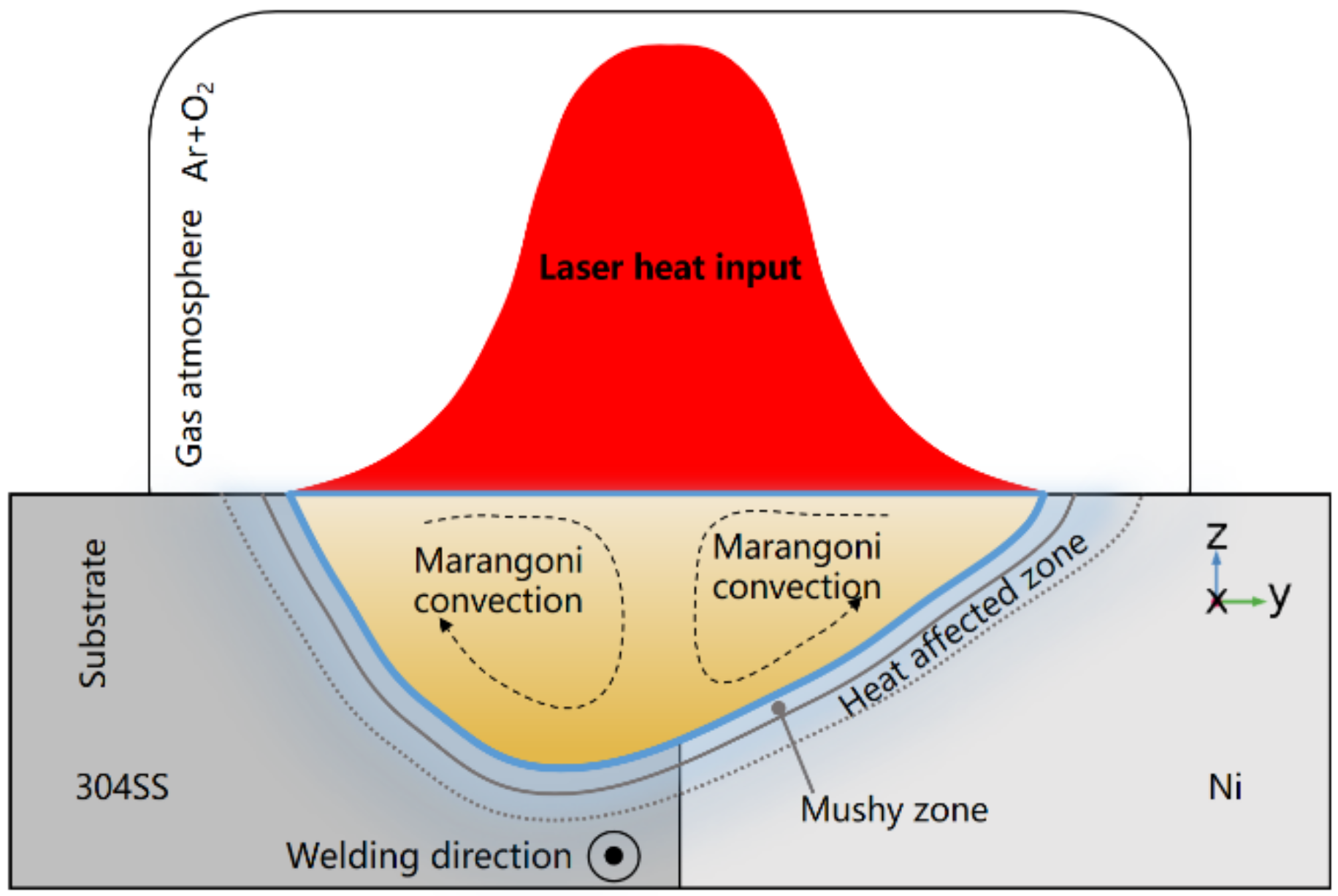
3. Mathematical Model
- The distribution of the incident flux of the laser beam is Gaussian;
- Liquid metal flow inside the molten pool is Newtonian, laminar, and incompressible, and the Boussinesq approximation can be employed [12];
- The mushy zone, where the temperature is between solidus and liquidus, is assumed to be a porous medium with isotropic permeability [30];
- Porous medium flow is assumed in the mushy zone and described by a Carmen–Kozeny relation [30];
- The effective absorption coefficient of the laser heat source is assumed to be the function of wavelength and substrate resistivity [31].
3.1. Governing Equations
3.2. Boundary Conditions
4. Results and Discussion
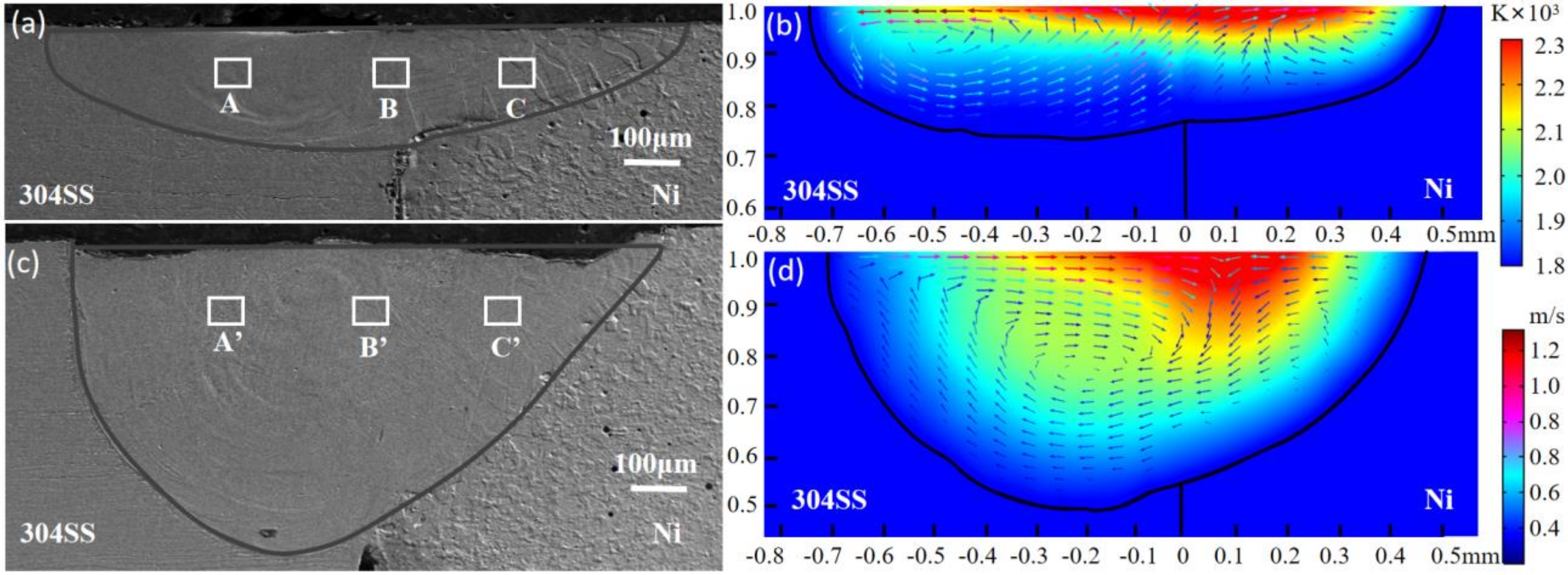
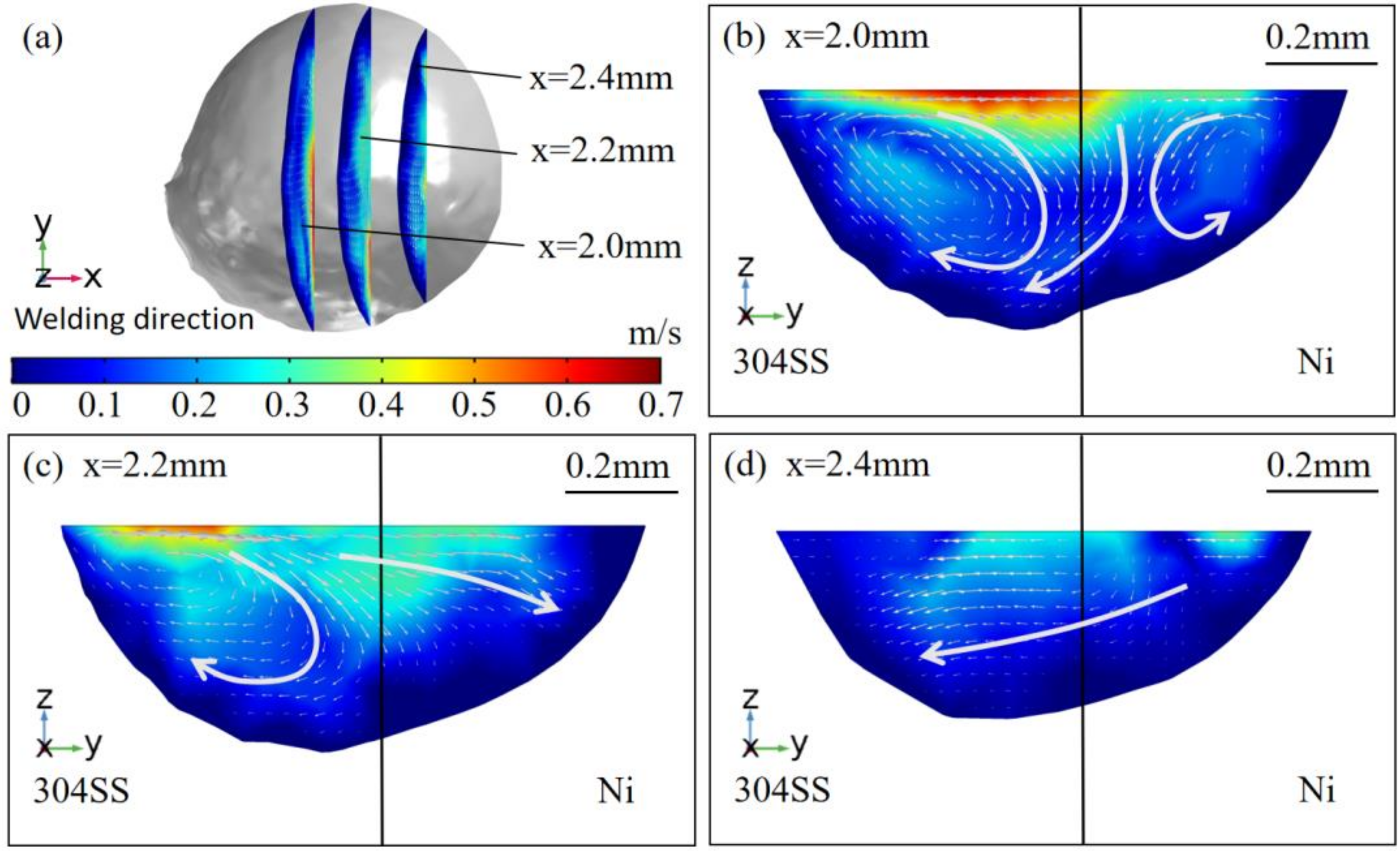
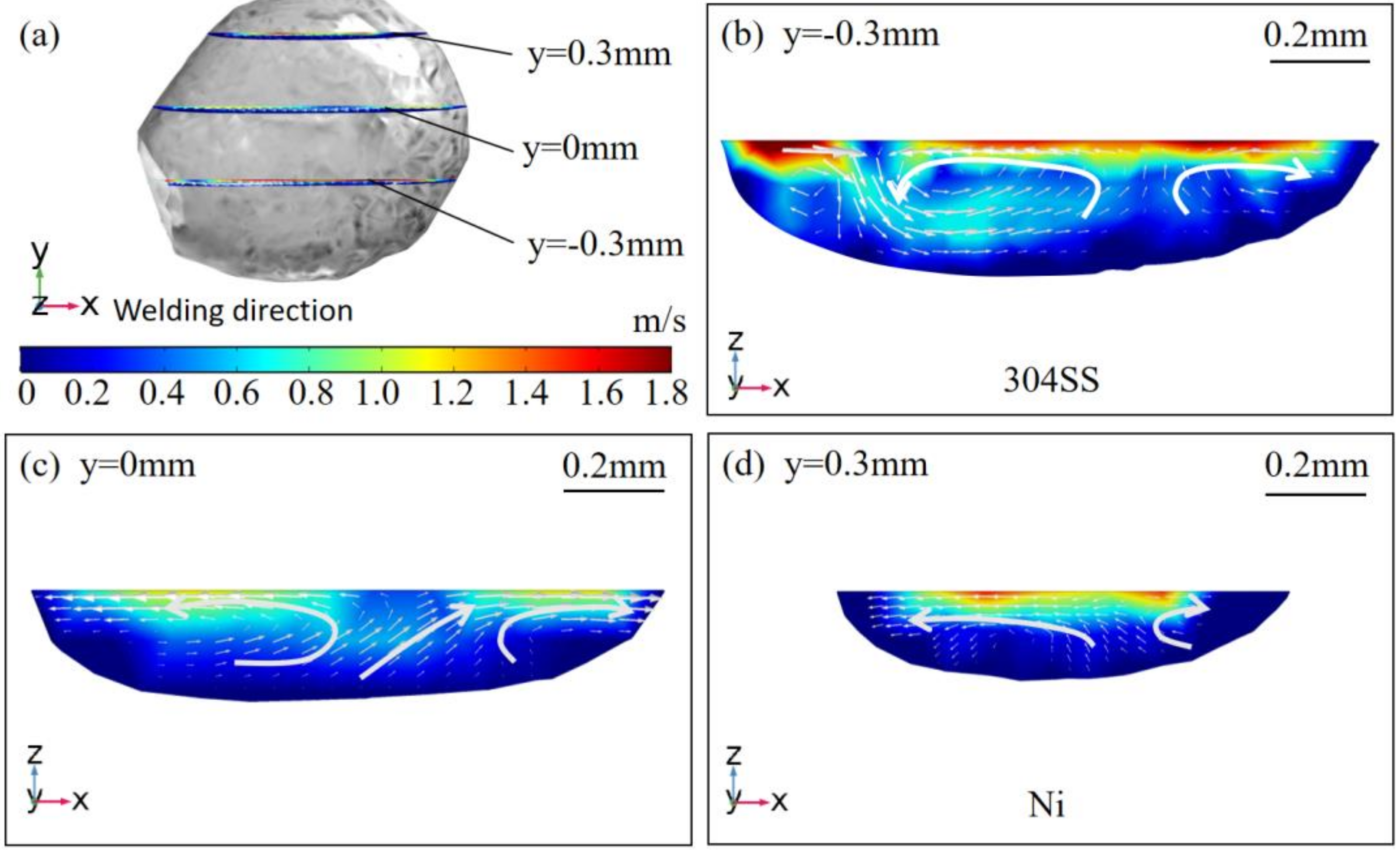
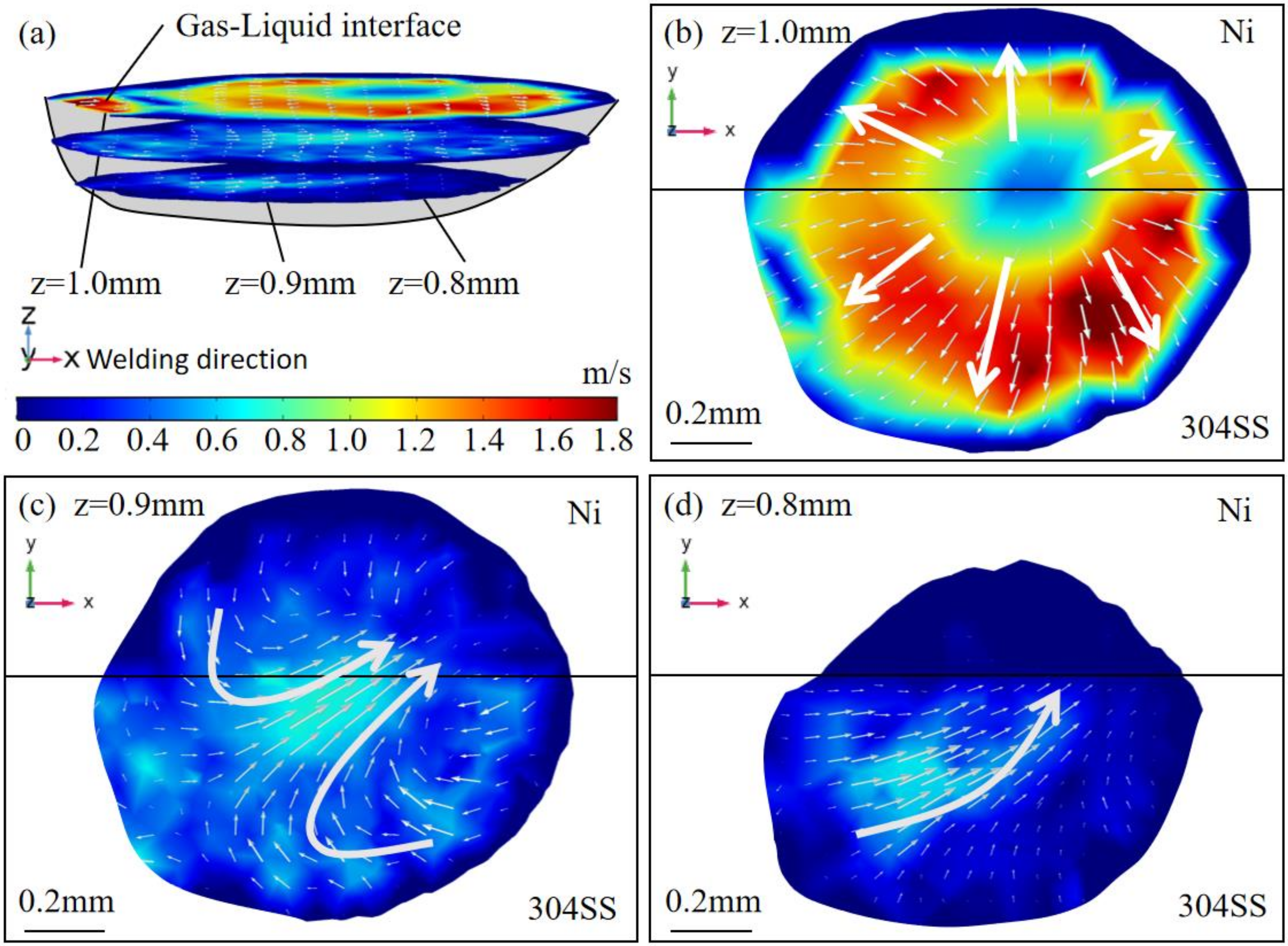
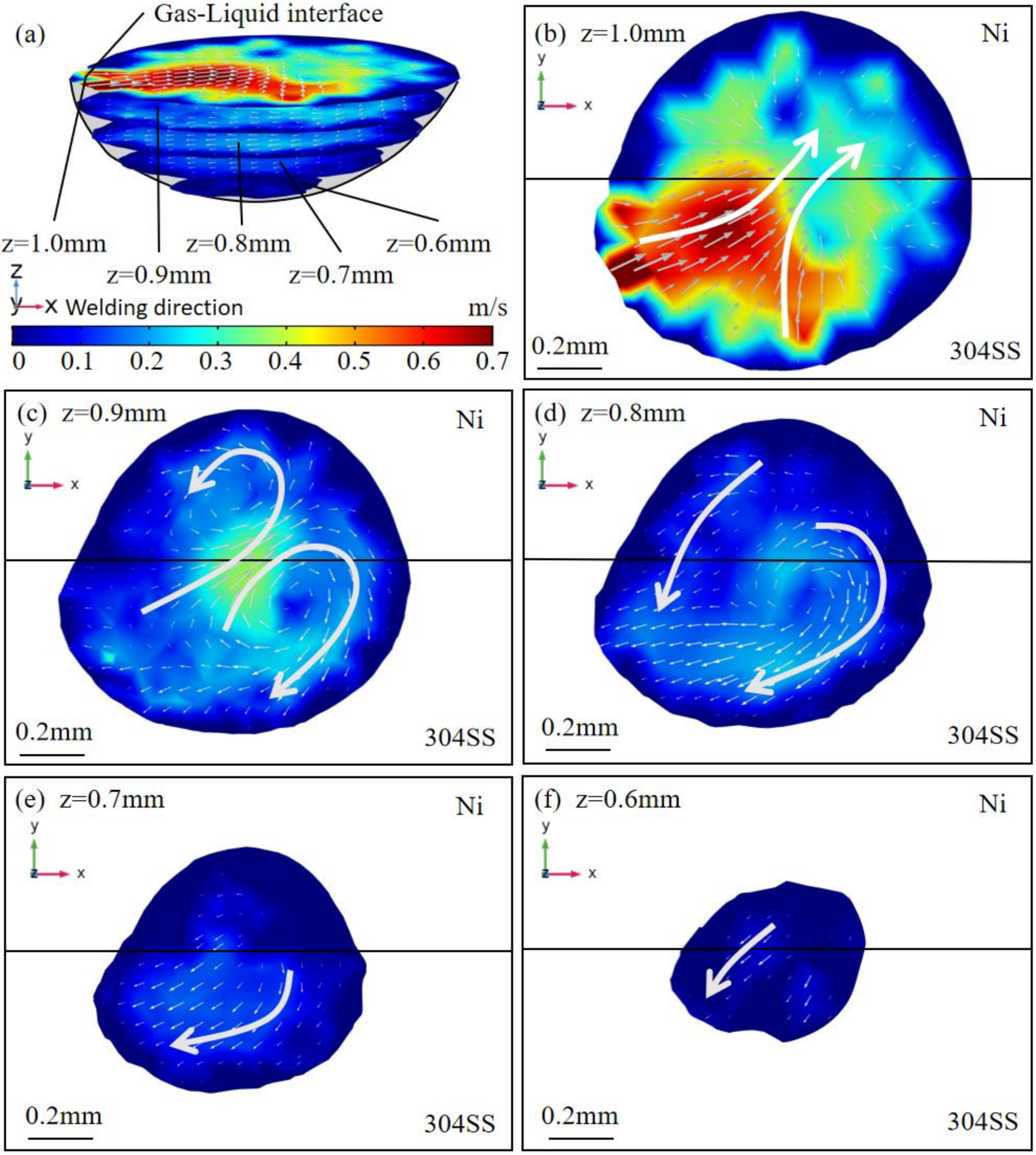
4.1. Thermal Behavior
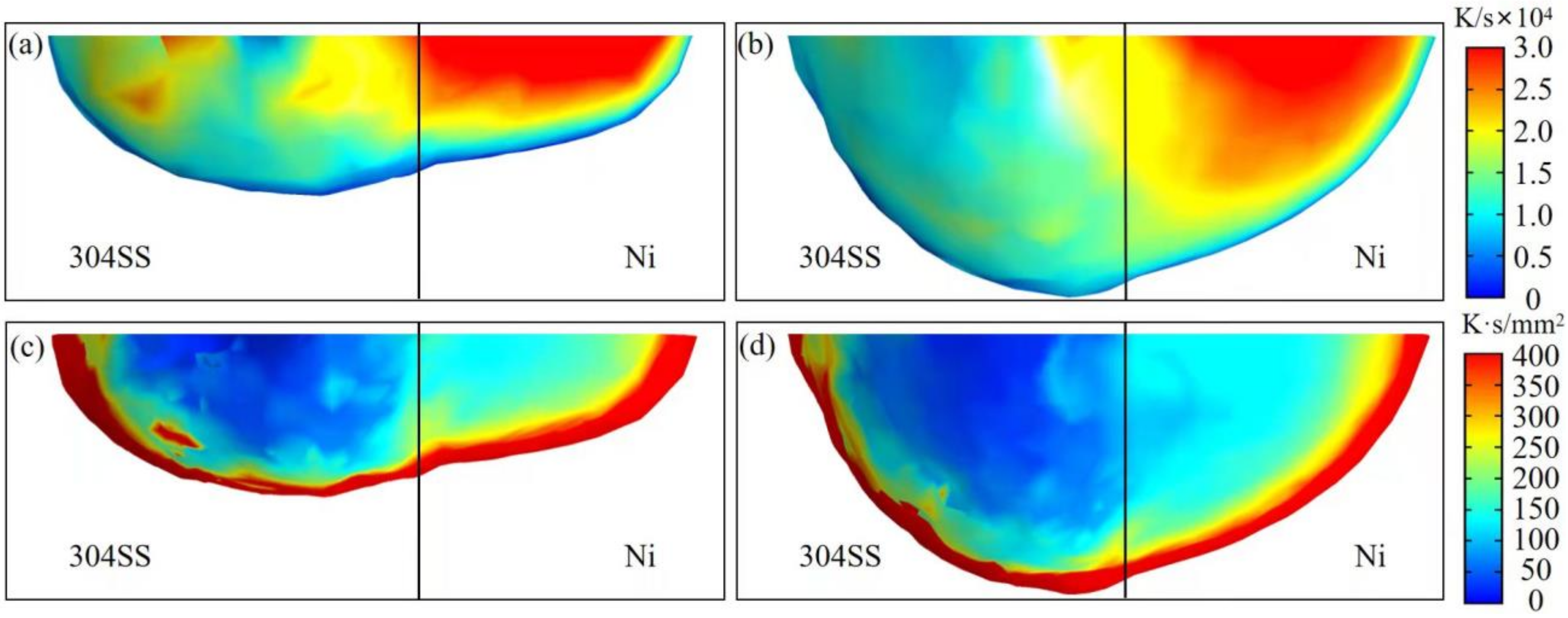
4.2. Solidification Characteristics
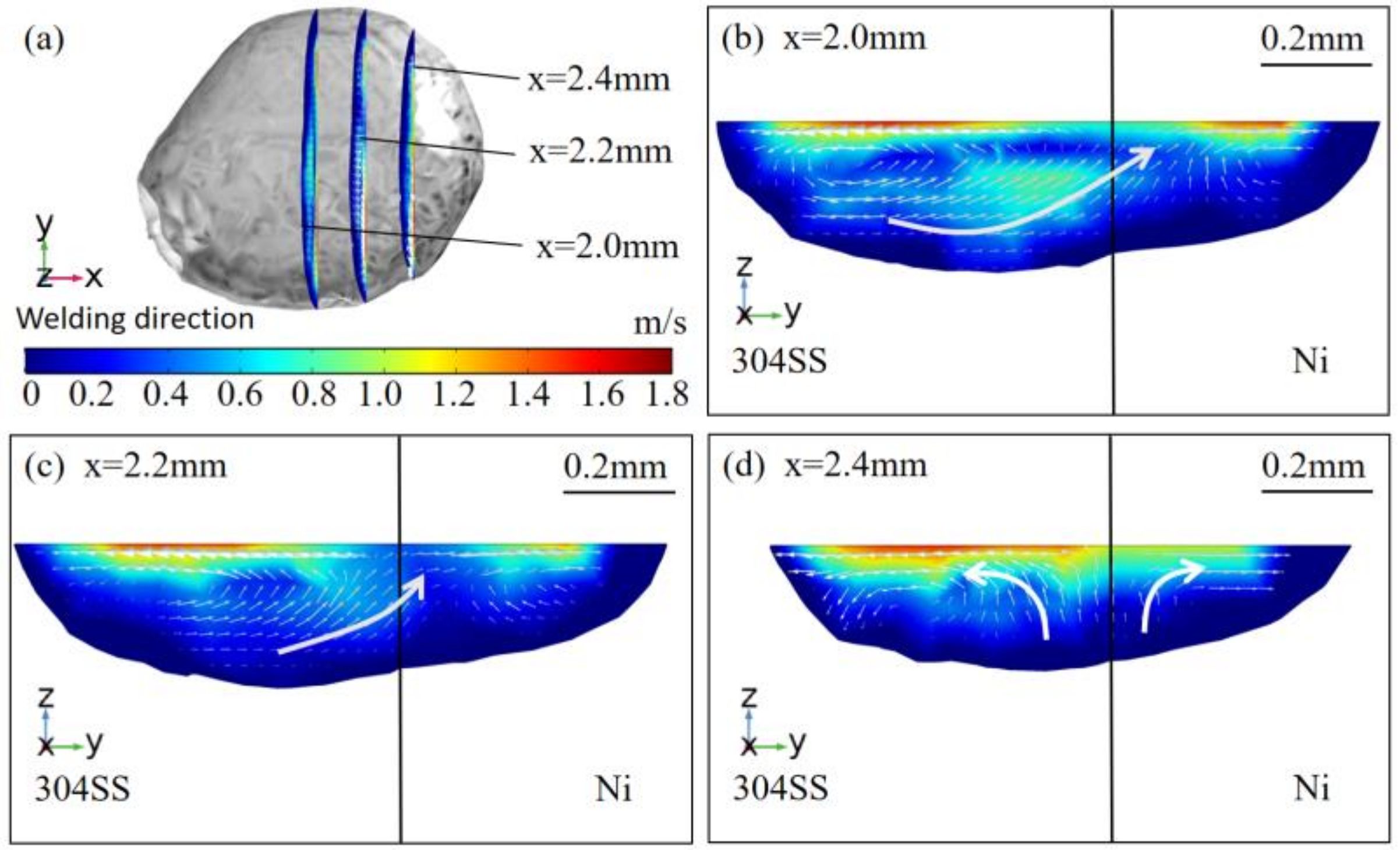
4.3. Correlation between Dilution and Fluid Flow
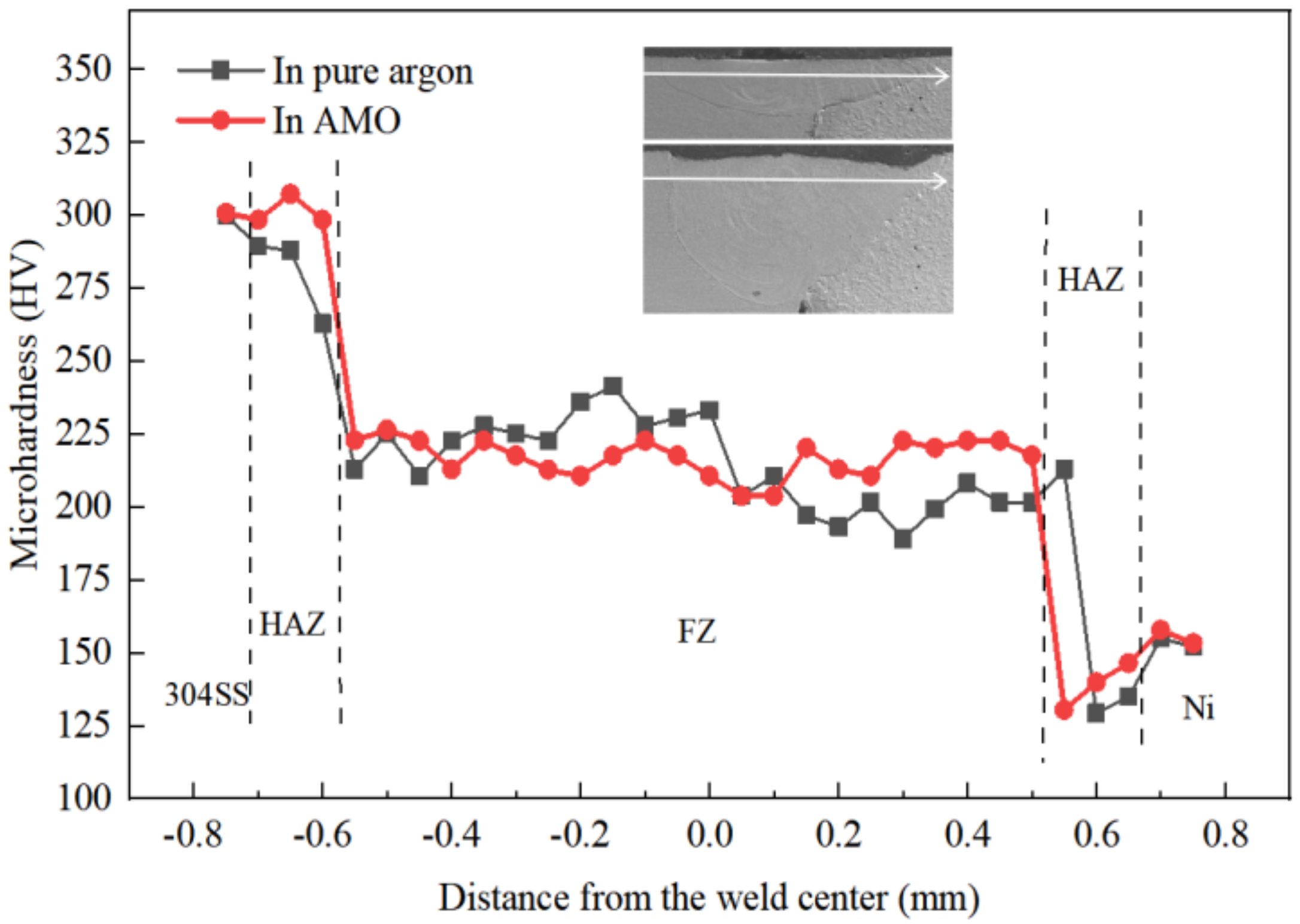
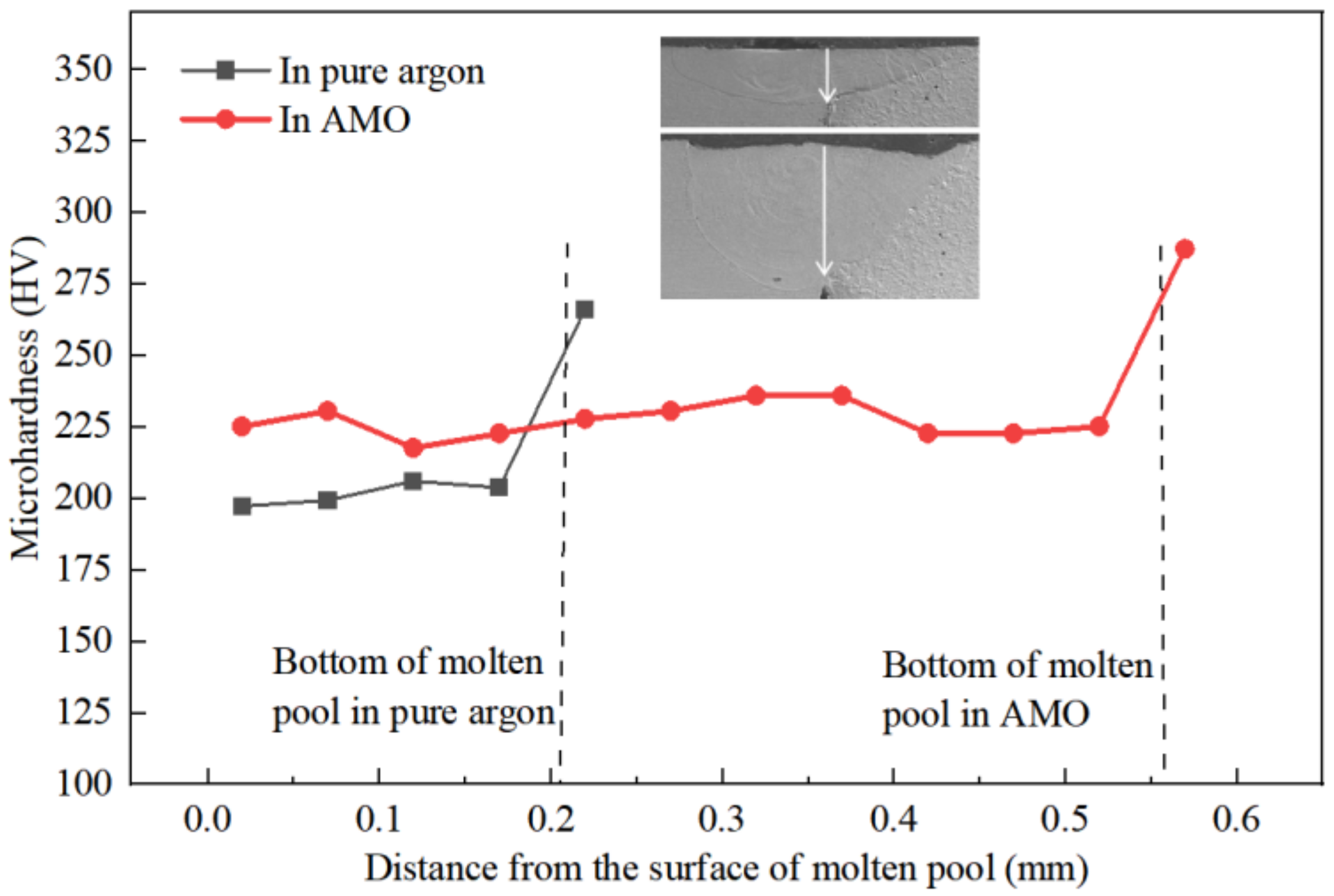
4.4. Effect of Oxygen on Microhardness of the Dissimilar Joint
5. Conclusions
- (1)
- The oxygen from the gas atmosphere changed the temperature coefficient of surface tension from negative to positive, resulting in the transition of flow mode inside the molten pool from outward convection to inward convection. As a result, compared with the welding in pure argon, the molten pool was deeper, the maximum velocity was smaller, and the peak temperature was larger when oxygen was added. Additionally, the tail at the end of the molten pool was smaller in the AMO because of the convective heat transfer by the inward convection;
- (2)
- In both situations (AMO and pure argon), the morphology parameter G/R for solidification was smaller on the 304SS side; thus, equiaxed dendrites were observed on the 304SS side, and columnar dendrites were found on the nickel side in the solidified molten pool. The G/R at the substrate interface was smaller in the AMO, and the solidified microstructures were equiaxed dendrites for the AMO and columnar for pure Argon in this area. The cooling rate GR changed little between pure argon and the AMO, and, thus, the microstructure size was similar;
- (3)
- The distribution of the Fe, Cr, and Ni elements in the molten pool in the AMO was more uniform compared with that in pure argon. In the molten pool in pure argon, Fe and Ni were enriched at the edge of the 304SS side of the molten pool, where the content of Ni was small, and the three elements were relatively evenly distributed on the nickel side. There were strong flows across the substrate interface for planes at different depths of the molten pool in the AMO, which promoted the mixing of elements in the molten pool. In the molten pool in pure argon, the outward flow at the top of the molten pool caused almost no movement, while at the bottom, only the flows deriving from the metals on both sides near the substrate interface intersected and moved to the nickel side. Therefore, the elements on the nickel side of the molten pool were evenly distributed;
- (4)
- The microhardness distribution was more uniform in the AMO, and this partly resulted from the homogenous dilution of alloy elements.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Sun, Z.; Ion, J.C. Laser welding of dissimilar metal combinations. J. Mater. Sci. 1995, 30, 4205–4214. [Google Scholar] [CrossRef]
- Maurya, A.K.; Pandey, C.; Chhibber, R. Dissimilar welding of duplex stainless steel with Ni alloys: A review. Int. J. Press. Vessel. Pip. 2021, 192, 104439. [Google Scholar] [CrossRef]
- Sun, Z. Feasibility of producing ferritic/austenitic dissimilar metal joints by high energy density laser beam process. Int. J. Press. Vessel. Pip. 1996, 68, 153–160. [Google Scholar] [CrossRef]
- Yu, G.; He, X.L.; Li, S.X. Laser Manufacturing and Its Application; National Defense Industry Press: Beijing, China, 2017; pp. 93–97. ISBN 978-7-118-10984-9. [Google Scholar]
- Hu, Y.; He, X.; Yu, G.; Ge, Z.; Zheng, C.; Ning, W. Heat and mass transfer in laser dissimilar welding of stainless steel and nickel. Appl. Surf. Sci. 2012, 258, 5914–5922. [Google Scholar] [CrossRef] [Green Version]
- Fang, Y.; Jiang, X.; Mo, D.; Zhu, D.; Luo, Z. A review on dissimilar metals’ welding methods and mechanisms with interlayer. Int. J. Adv. Manuf. Technol. 2019, 102, 2845–2863. [Google Scholar] [CrossRef]
- Sirohi, S.; Taraphdar, P.K.; Dak, G.; Pandey, C.; Sharma, S.K.; Goyal, A. Study on evaluation of through-thickness residual stresses and microstructure-mechanical property relation for dissimilar welded joint of modified 9cr–1mo and ss304h steel. Int. J. Press. Vessel. Pip. 2021, 194, 104557. [Google Scholar] [CrossRef]
- Bunaziv, I.; Akselsen, O.M.; Ren, X.; Nyhus, B.; Eriksson, M.; Gulbrandsen-Dahl, S. A Review on Laser-Assisted Joining of Aluminium Alloys to Other Metals. Metals 2021, 11, 1680. [Google Scholar] [CrossRef]
- Yuan, R.; Deng, S.; Cui, H.; Chen, Y.; Lu, F. Interface characterization and mechanical properties of dual beam laser welding-brazing Al/steel dissimilar metals. J. Manuf. Process. 2019, 40, 37–45. [Google Scholar] [CrossRef]
- Zhang, L.J.; Lu, G.F.; Ning, J.; Zhu, Q.; Zhang, J.X.; Na, S.J. Effects of minor Zr addition on the microstructure and mechanical properties of laser welded dissimilar joint of titanium and molybdenum. Mater. Sci. Eng. A 2019, 742, 788–797. [Google Scholar] [CrossRef]
- Cao, L.; Liu, D.; Jiang, P.; Shao, X.; Zhou, Q.; Wang, Y. Multi-physics simulation of dendritic growth in magnetic field assisted solidification. Int. J. Heat Mass Transf. 2019, 144, 118673. [Google Scholar] [CrossRef]
- Li, Z.; Yu, G.; He, X.; Li, S.; Li, Z. Fluid flow and solute dilution in laser linear butt joining of 304SS and Ni. Int. J. Heat Mass Transf. 2020, 161, 120233. [Google Scholar] [CrossRef]
- Li, Z.; Yu, G.; He, X.; Li, S.; Zhao, Y. Numerical and experimental investigations of solidification parameters and mechanical property during laser dissimilar welding. Metals 2018, 8, 799. [Google Scholar] [CrossRef] [Green Version]
- He, X.; DebRoy, T.; Fuerschbach, P.W. Alloying element vaporization during laser spot welding of stainless steel. J. Phys. D 2003, 36, 3079. [Google Scholar] [CrossRef]
- Landowski, M.; Świerczyńska, A.; Rogalski, G.; Fydrych, D. Autogenous fiber laser welding of 316L austenitic and 2304 lean duplex stainless steels. Materials 2020, 13, 2930. [Google Scholar] [CrossRef]
- Kumar, A.; Pandey, C. Autogenous laser-welded dissimilar joint of ferritic/martensitic P92 steel and Inconel 617 alloy: Mechanism, microstructure, and mechanical properties. Arch. Civ. Mech. Eng. 2022, 22, 1–20. [Google Scholar] [CrossRef]
- Chakraborty, N. The effects of turbulence on molten pool transport during melting and solidification processes in continuous conduction mode laser welding of copper–nickel dissimilar couple. Appl. Therm. Eng. 2009, 29, 3618–3631. [Google Scholar] [CrossRef] [Green Version]
- Zargari, H.H.; Ito, K.; Miwa, T.; Parchuri, P.K.; Sharma, A. Metallurgical characterization of penetration shape change in workpiece vibration-assisted tandem-pulsed gas metal arc welding. Materials 2020, 13, 3096. [Google Scholar] [CrossRef]
- Lu, S.; Fujii, H.; Nogi, K. Sensitivity of Marangoni convection and weld shape variations to welding parameters in O2–Ar shielded GTA welding. Scr. Mater. 2004, 51, 271–277. [Google Scholar] [CrossRef]
- Zhao, C.X.; Kwakernaak, C.; Pan, Y.; Richardson, I.M.; Saldi, Z.; Kenjeres, S.; Kleijn, C.R. The effect of oxygen on transitional Marangoni flow in laser spot welding. Acta Mater. 2010, 58, 6345–6357. [Google Scholar] [CrossRef]
- Kidess, A.; Kenjereš, S.; Kleijn, C.R. The influence of surfactants on thermocapillary flow instabilities in low Prandtl melting pools. Phys. Fluids 2016, 28, 062106. [Google Scholar] [CrossRef] [Green Version]
- Heiple, C.R.; Roper, J.R.; Stagner, R.T.; Aden, R.J. Surface active element effects on the shape of GTA, laser and electron beam welds. Weld J. 1983, 62, 72. [Google Scholar]
- Sahoo, P.; Collur, M.M.; DebRoy, T. Effects of oxygen and sulfur on alloying element vaporization rates during laser welding. Metall. Trans. B 1988, 19, 967–972. [Google Scholar] [CrossRef]
- Li, S.; Deng, Z.; Deng, H.; Xu, W. Microstructure and properties of weld joint during 10 kW laser welding with surface-active element sulfur. Appl. Surf. Sci. 2017, 426, 704–713. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, Y.; Xiong, Z.; Zhu, M.; Zhang, Z.; Li, Z. Mechanism and optimization of activating fluxes for process stability and weldability of hybrid laser-arc welded HSLA steel. Weld World 2021, 65, 753–766. [Google Scholar] [CrossRef]
- Gan, Z.; Yu, G.; He, X.; Li, S. Surface-active element transport and its effect on liquid metal flow in laser-assisted additive manufacturing. Int. Commun. Heat Mass Transf. 2017, 86, 206–214. [Google Scholar] [CrossRef] [Green Version]
- Hu, Y.; He, X.; Yu, G.; Li, S.; Zheng, C.; Ning, W. Experimental and numerical study on laser keyhole welding of 42CrMo under air and argon atmosphere. Int. J. Adv. Manuf. Technol. 2017, 90, 3555–3565. [Google Scholar] [CrossRef] [Green Version]
- Unni, A.K.; Muthukumaran, V. Numerical simulation of the influence of oxygen content on the weld pool depth during activated TIG welding. Int. J. Adv. Manuf. Technol. 2021, 112, 467–489. [Google Scholar] [CrossRef]
- Zargari, H.H.; Ito, K.; Sharma, A.; Kumar, M. Visualizing the vibration effect on the tandem-pulsed gas metal arc welding in the presence of surface tension active elements. Int. J. Heat Mass Transf. 2020, 161, 120310. [Google Scholar] [CrossRef]
- Voller, V.R.; Prakash, C. A fixed grid numerical modelling methodology for convection-diffusion mushy region phase-change problems. Int. J. Heat Mass Transf. 1987, 30, 1709–1719. [Google Scholar] [CrossRef]
- Li, Z.; Yu, G.; He, X.; Tian, C.; Li, S.; Li, H. Probing thermocapillary convection and multisolute dilution in laser welding of dissimilar miscible metals. Int. J. Therm. Sci. 2022, 172, 107242. [Google Scholar] [CrossRef]
- Bahrami, A.; Helenbrook, B.T.; Valentine, D.T.; Aidun, D.K. Fluid flow and mixing in linear GTA welding of dissimilar ferrous alloys. Int. J. Heat Mass Transf. 2016, 93, 729–741. [Google Scholar] [CrossRef]
- Bahrami, A.; Valentine, D.T.; Helenbrook, B.T.; Aidun, D.K. Study of mass transport in autogenous GTA welding of dissimilar metals. Int. J. Heat Mass Transf. 2015, 85, 41–53. [Google Scholar] [CrossRef]
- Li, Z.; Yu, G.; He, X.; Li, S.; Tian, C.; Dong, B. Analysis of surface tension driven flow and solidification behavior in laser linear welding of stainless steel. Opt. Laser Technol. 2020, 123, 105914. [Google Scholar] [CrossRef]
- Folkhard, E. Welding Metallurgy of Stainless Steels; John Wiley & Sons: Hoboken, NJ, USA, 2005; pp. 46–50. ISBN 0-471-47379-0. [Google Scholar]
- Kou, S. Welding Metallurgy; John Wiley & Sons: Hoboken, NJ, USA, 2003; pp. 17–20. ISBN 0-471-434919-4. [Google Scholar]
- Gan, Z.; Li, H.; Wolff, S.J.; Bennett, J.L.; Hyatt, G.; Wagner, G.J.; Liu, W.K. Data-driven microstructure and microhardness design in additive manufacturing using a self-organizing map. Engineering 2019, 5, 730–735. [Google Scholar] [CrossRef]
- Ridha Mohammed, G.; Ishak, M.; Ahmad, S.N.A.S.; Abdulhadi, H.A. Fiber laser welding of dissimilar 2205/304 stainless steel plates. Metals 2017, 7, 546. [Google Scholar] [CrossRef] [Green Version]
- Lehto, P.; Remes, H.; Saukkonen, T.; Hänninen, H.; Romanoff, J. Influence of grain size distribution on the Hall–Petch relationship of welded structural steel. Mater. Sci. Eng. C 2014, 592, 28–39. [Google Scholar] [CrossRef] [Green Version]







| Elements | Si | Mn | Cr | Ni | Fe |
|---|---|---|---|---|---|
| 304SS | 0.83 | 1.32 | 18.43 | 8.45 | Bal. |
| Laser Power | Laser Spot Diameter | Welding Speed | Gas Flow |
|---|---|---|---|
| 800 W | 1.16 mm | 20 mm/s | 15 L/min |
| Parameter | Value |
|---|---|
| Power distribution factor | 2 |
| Laser absorption efficiency | 0.3 |
| Ambient temperature (K) | 298.15 |
| Convection coefficient (W/m2·K) | 100 |
| Emissivity | 0.2 |
| Stefan–Boltzmann constant (W/m2·K4) | 5.67 × 10−8 |
| Property | 304SS | Nickel |
|---|---|---|
| Liquidus temperature (K) | 1720 | 1733 |
| Solidus temperature (K) | 1637 | 1723 |
| Heat of fusion (kJ/kg) | 261 | 298 |
| Specific heat of liquid (J/kg·K) | 800 | 734 |
| Specific heat of solid (J/kg·K) | 645 | 617 |
| Thermal conductivity of liquid (W/m·K) | 29 | 69 |
| Thermal conductivity of solid (W/m·K) | 33 | 86 |
| Density of solid metal (kg/m3) | 7450 | 8200 |
| Density of liquid metal (kg/m3) | 6910 | 7700 |
| Dynamic viscosity (kg/m·s) | 7.20 × 10−3 | 6.5 × 10−3 |
| Surface tension (N/m) | 1.872 | 1.778 |
| Temperature coefficient of surface tension (N/m·K) | −4.30 × 10−4 | −3.40 × 10−4 |
| Liquid volume thermal expansion (K−1) | 1.15 × 10−5 | 1.52 × 10−5 |
| Liquid volume concentration expansion | 0.078 | 0.078 |
| Effective mass diffusivity (m2/s) | 7.00 × 10−7 | 7.00 × 10−7 |
| Position | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| Fe in pure argon | 61.56 | 46.92 | 26.10 | 30.09 |
| Cr in pure argon | 18.14 | 13.93 | 7.6 | 8.44 |
| Ni in pure argon | 20.29 | 39.15 | 66.3 | 61.47 |
| Fe in the AMO | 53.08 | 52.40 | 50.44 | 47.27 |
| Cr in the AMO | 14.60 | 14.93 | 14.10 | 13.52 |
| Ni in the AMO | 32.33 | 32.66 | 35.55 | 39.22 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dong, B.; Li, Z.; Yu, G.; Li, S.; Tian, C.; Bian, Y.; Shu, Z.; He, X. Effect of Surface-Active Element Oxygen on Heat and Mass Transfer in Laser Welding of Dissimilar Metals: Numerical and Experimental Study. Metals 2022, 12, 556. https://doi.org/10.3390/met12040556
Dong B, Li Z, Yu G, Li S, Tian C, Bian Y, Shu Z, He X. Effect of Surface-Active Element Oxygen on Heat and Mass Transfer in Laser Welding of Dissimilar Metals: Numerical and Experimental Study. Metals. 2022; 12(4):556. https://doi.org/10.3390/met12040556
Chicago/Turabian StyleDong, Binxin, Zhiyong Li, Gang Yu, Shaoxia Li, Chongxin Tian, Yanhua Bian, Zhuang Shu, and Xiuli He. 2022. "Effect of Surface-Active Element Oxygen on Heat and Mass Transfer in Laser Welding of Dissimilar Metals: Numerical and Experimental Study" Metals 12, no. 4: 556. https://doi.org/10.3390/met12040556
APA StyleDong, B., Li, Z., Yu, G., Li, S., Tian, C., Bian, Y., Shu, Z., & He, X. (2022). Effect of Surface-Active Element Oxygen on Heat and Mass Transfer in Laser Welding of Dissimilar Metals: Numerical and Experimental Study. Metals, 12(4), 556. https://doi.org/10.3390/met12040556







