Zn Extraction from Zinc-Containing Sludge Using Ultrasonic Treatment Leaching with ChCl-MA DES
Abstract
1. Introduction
2. Materials and Methods
2.1. Analysis of Raw Material Composition
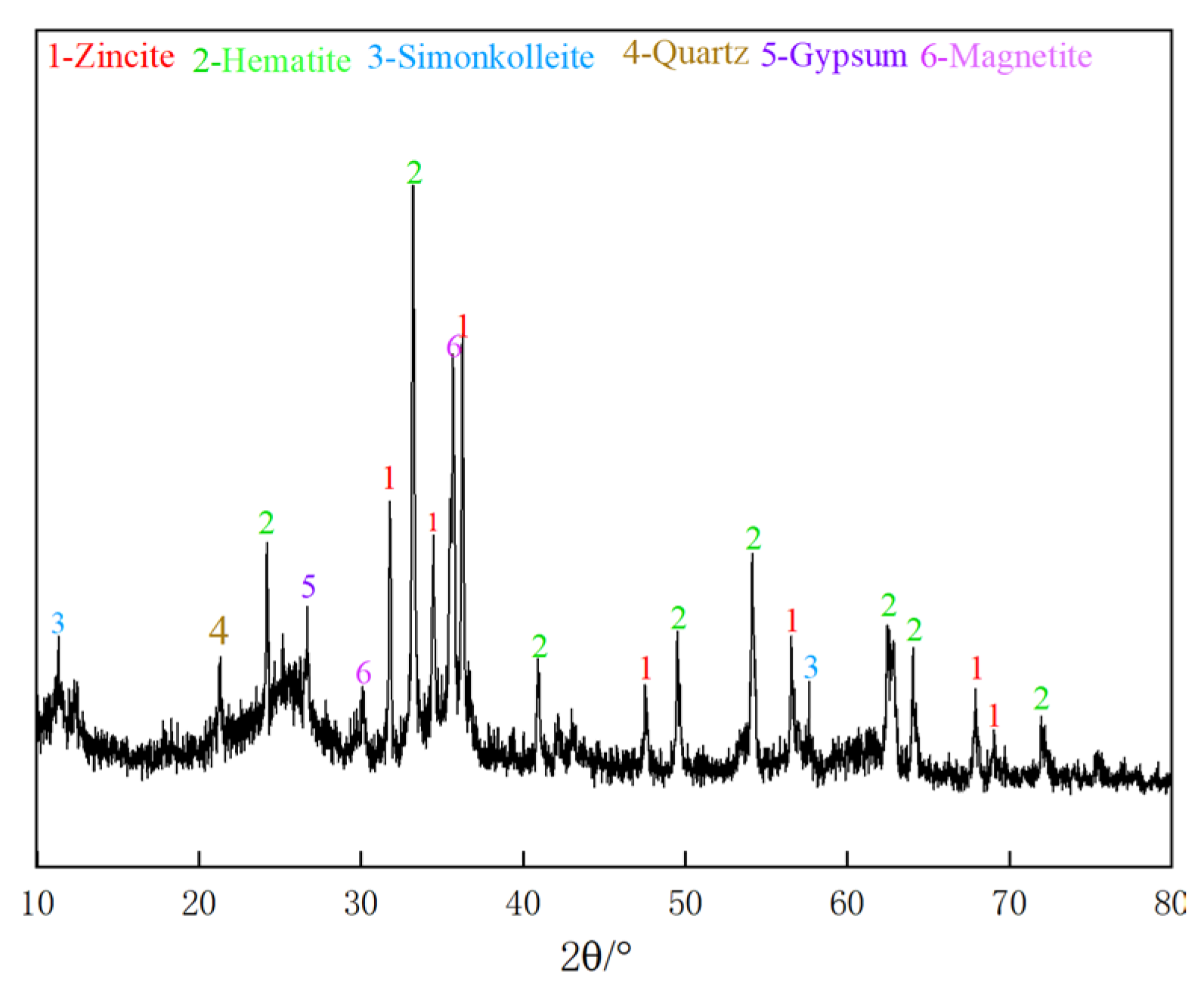
2.2. XRD Analysis
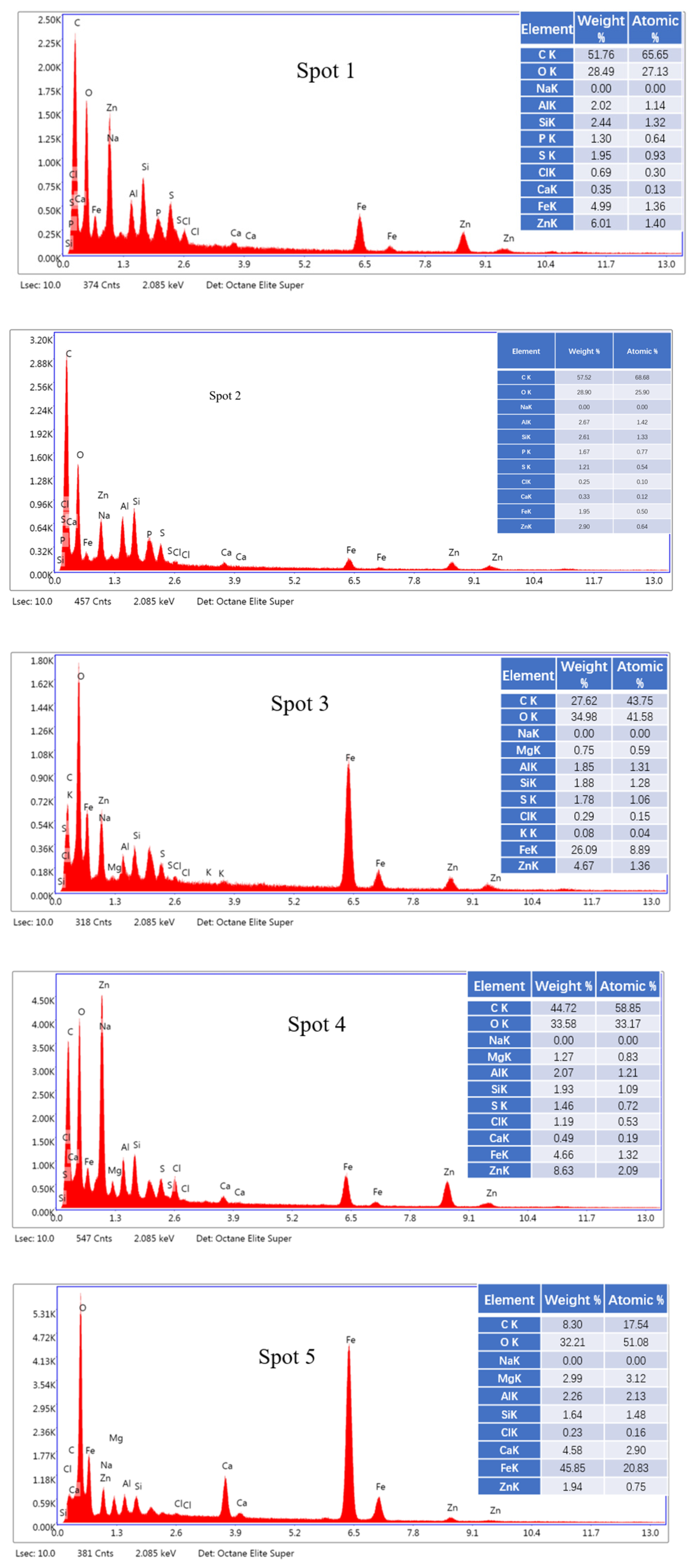
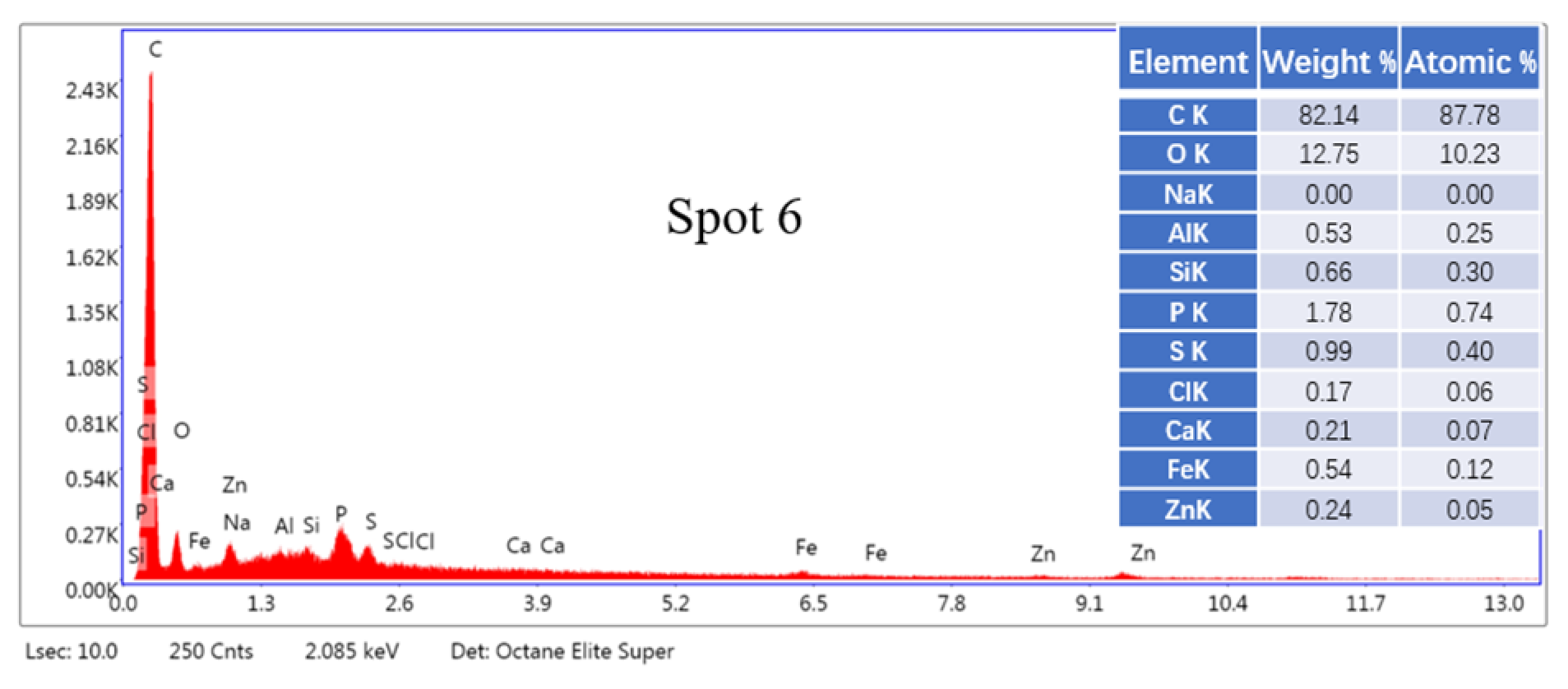
2.3. SEM-EDS Analysis
2.4. Particle Size Composition Analysis
2.5. Leaching Experiments
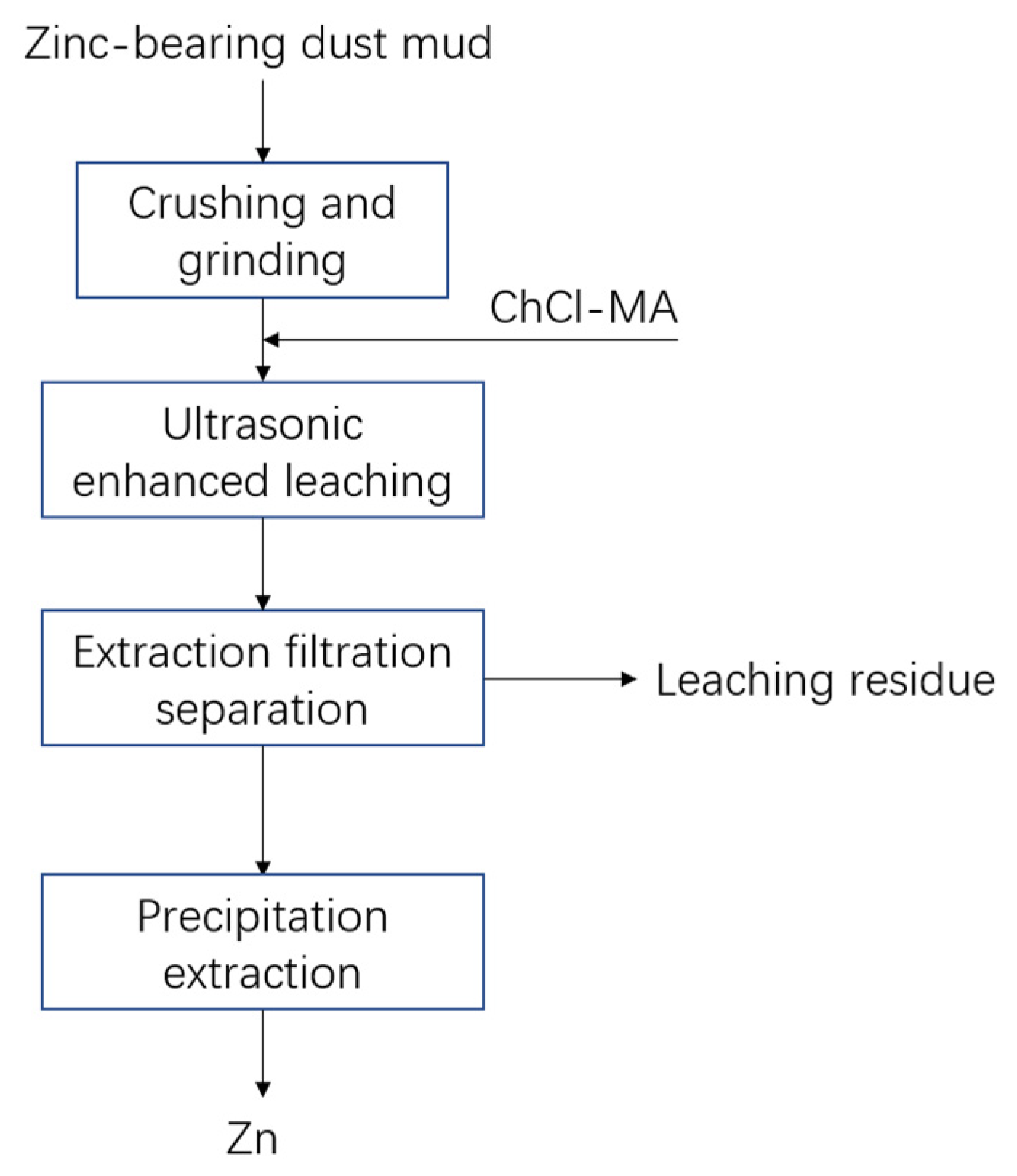
2.5.1. Principle of Leaching Experiment
2.5.2. Leaching Experiment Process
- where m0—Mass of the zinc-containing dust sludge sample, g;
- x0—Zinc content of Zn-containing dust samples, %;
- m1—Mass of the leached residue sample, g;
- x1—Zinc content of the residue sample, %.
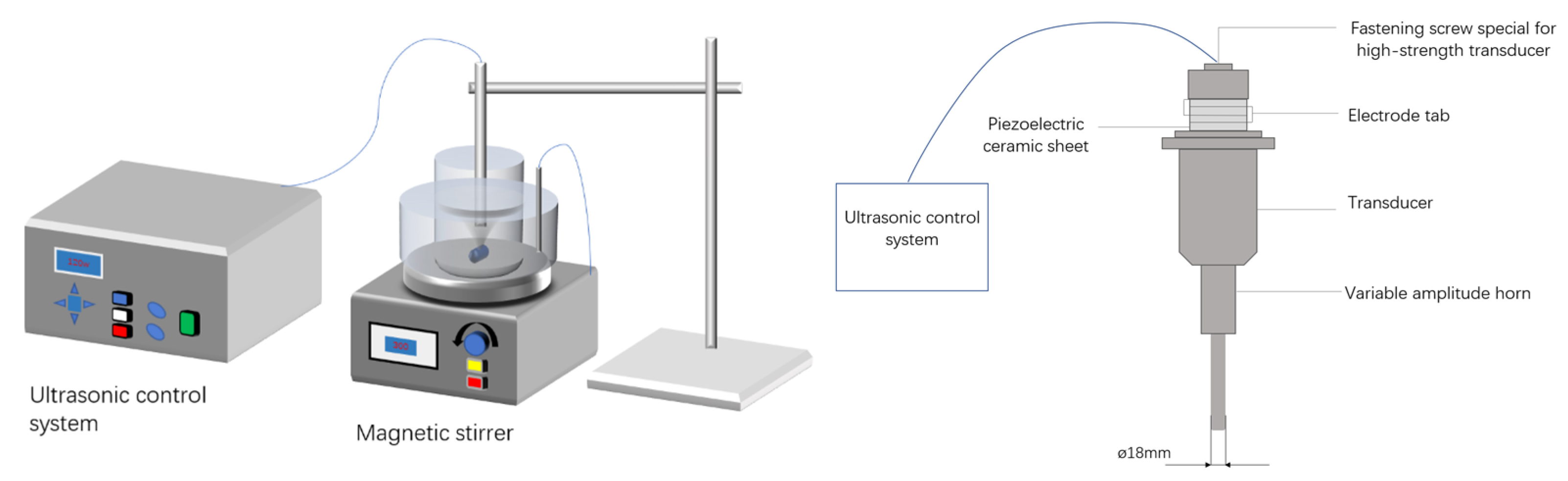
| Equipment Type | FS-600 |
|---|---|
| Power | 600 W |
| Working frequency | 20 kHz |
| Power adjustable range | 0–100% |
| Handling capacity | 500 μL–500 mL |
| Standard emitter diameter | 18 mm |
2.5.3. Full-Factorial Experimental Design
3. Results and Analysis
3.1. Design of Regression Model
3.2. Variance Analysis of Regression Model
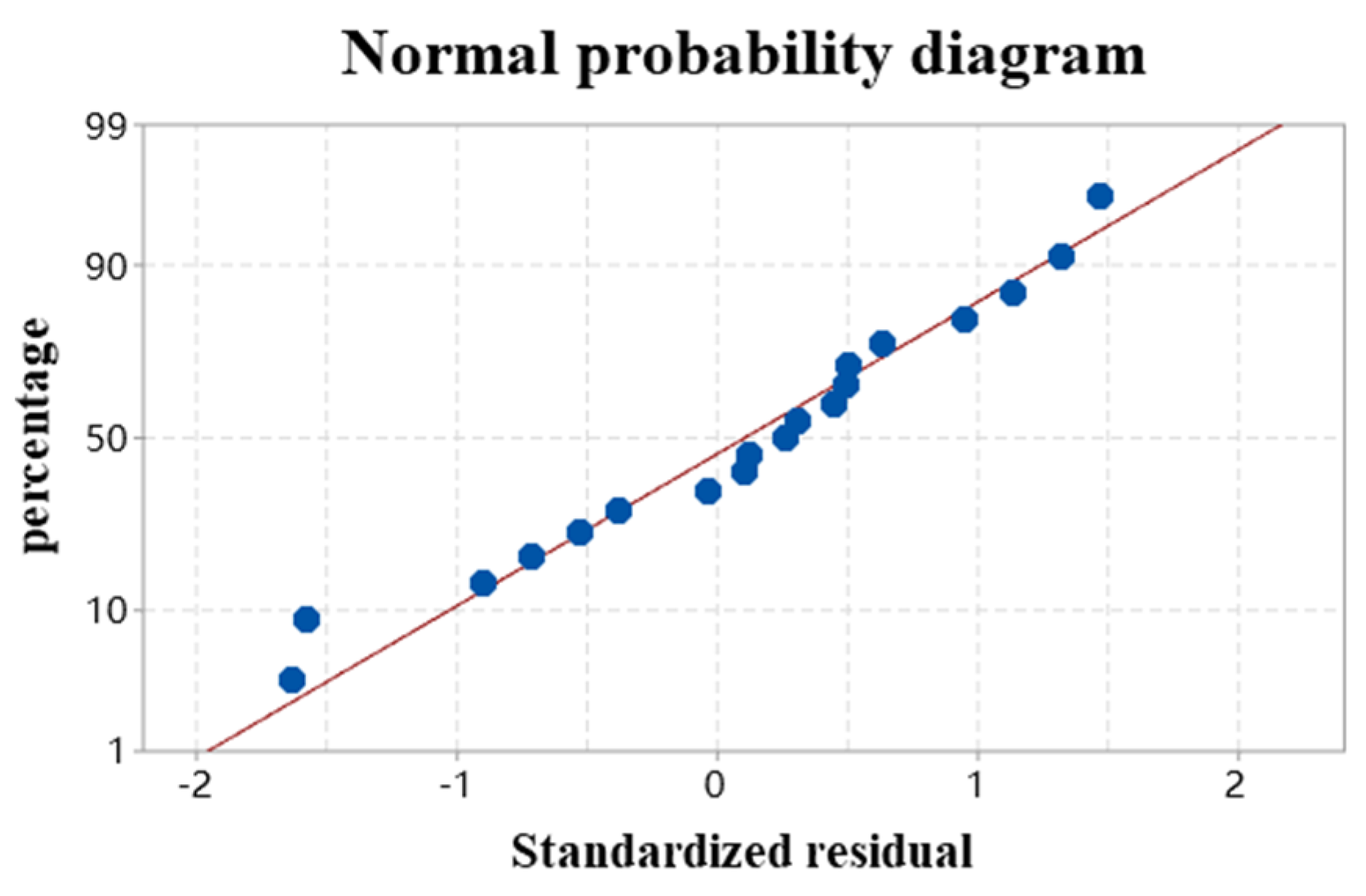
3.3. Residual Analysis
3.4. Variance Analysis of Regression Model
3.4.1. Standardized Effect Diagram Analysis
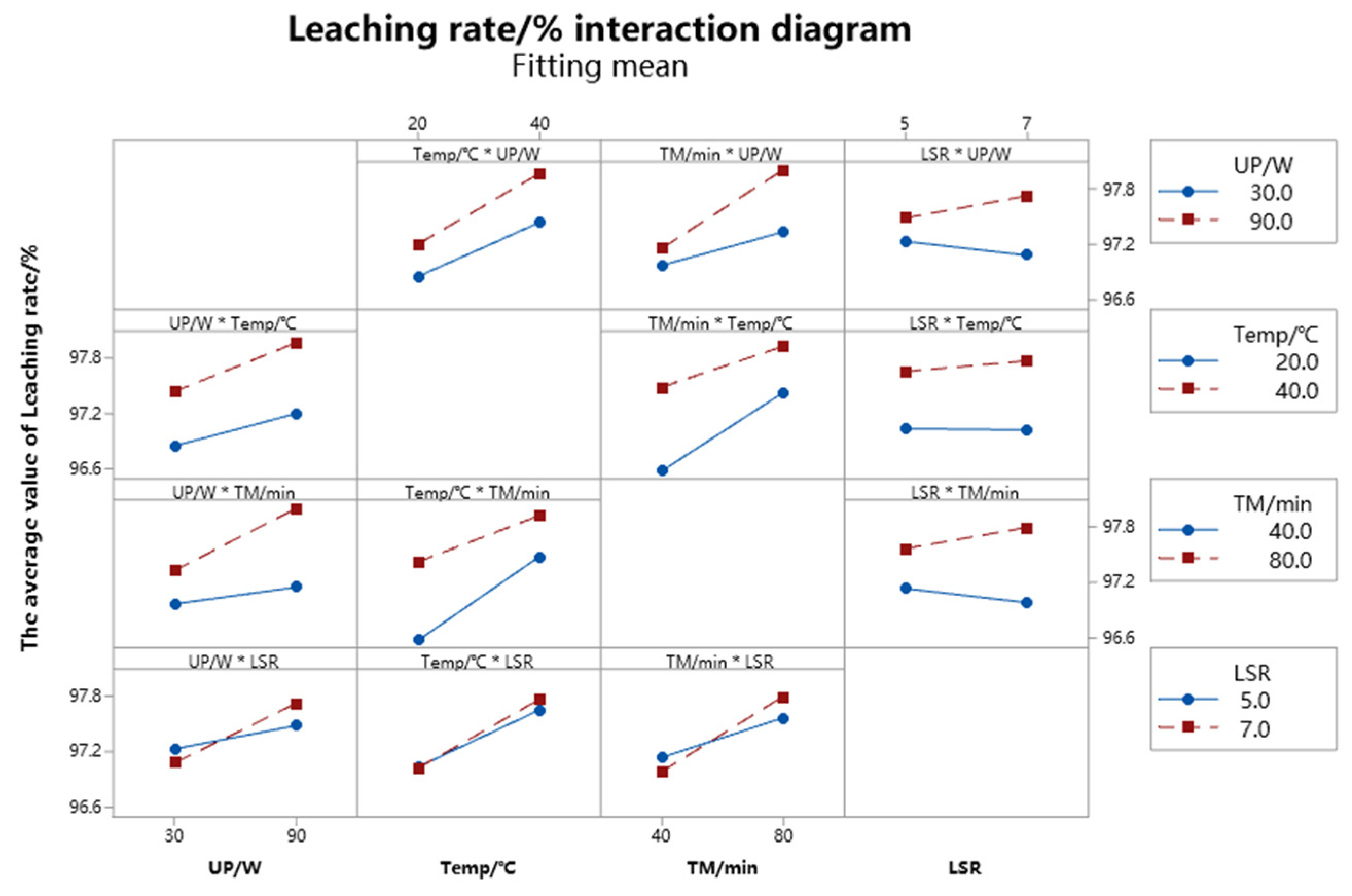
3.4.2. Factor Plot Analysis
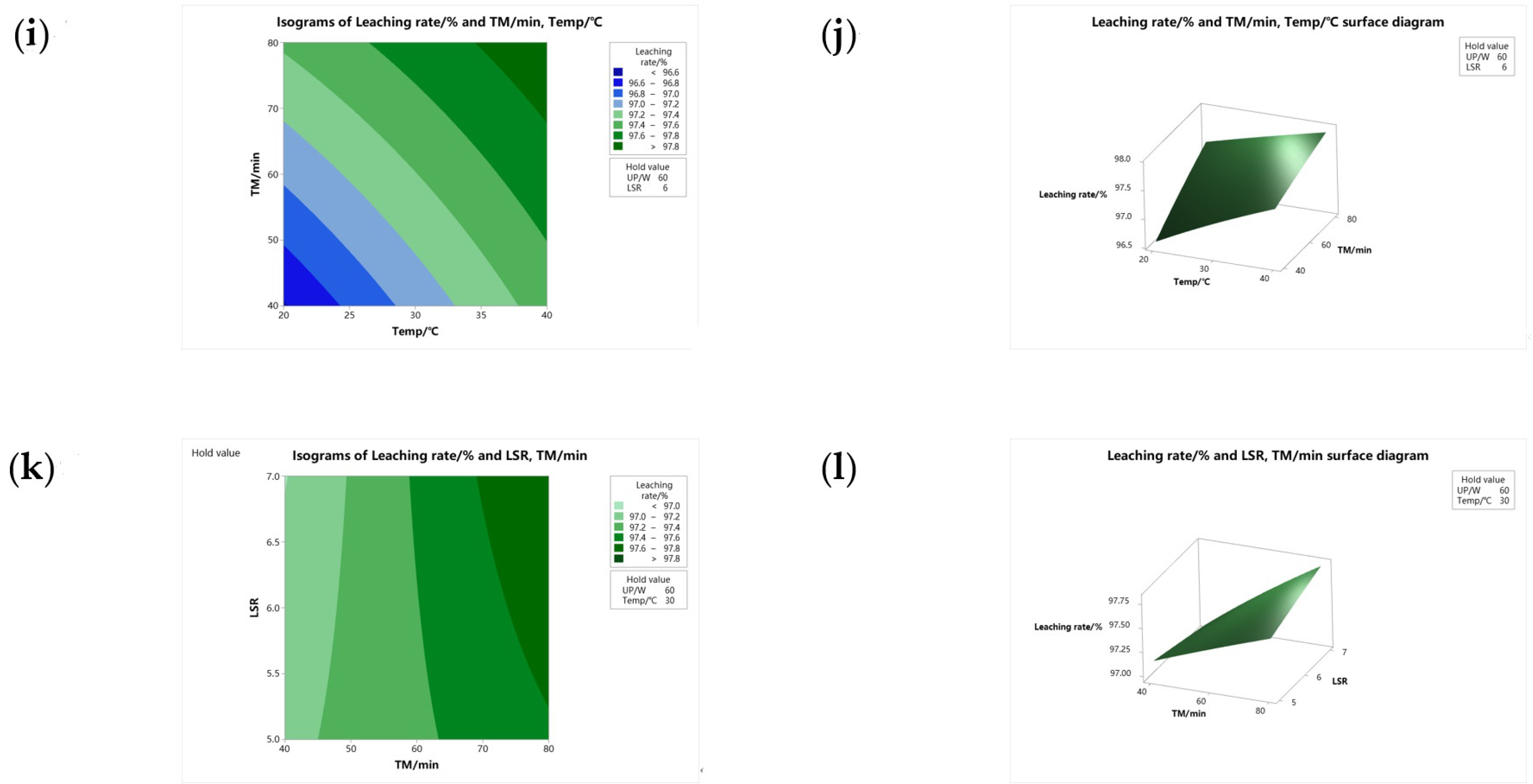
3.4.3. Contour Plot and Surface Diagram Analysis
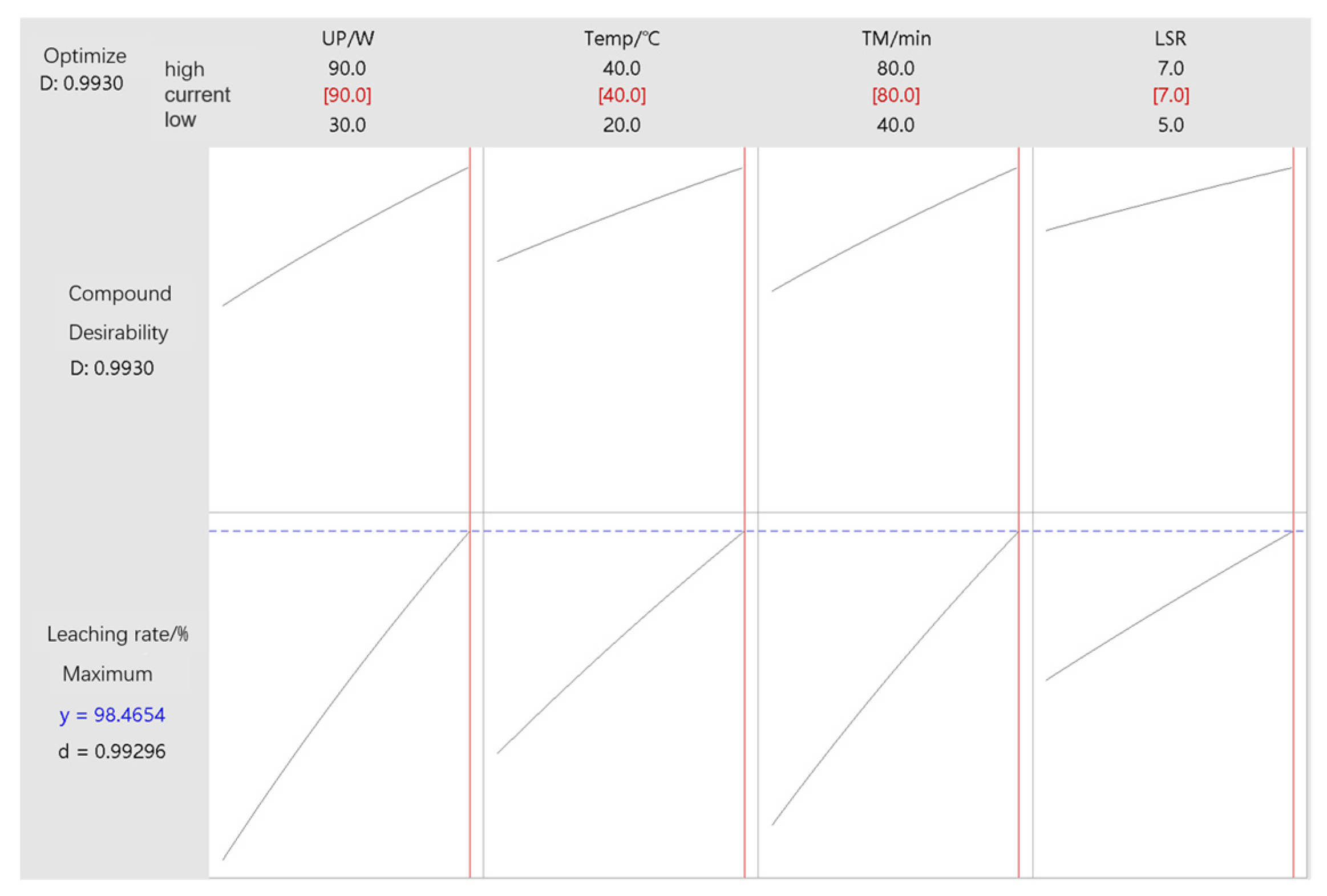
3.5. Factor Response Optimization and Experimental Validation
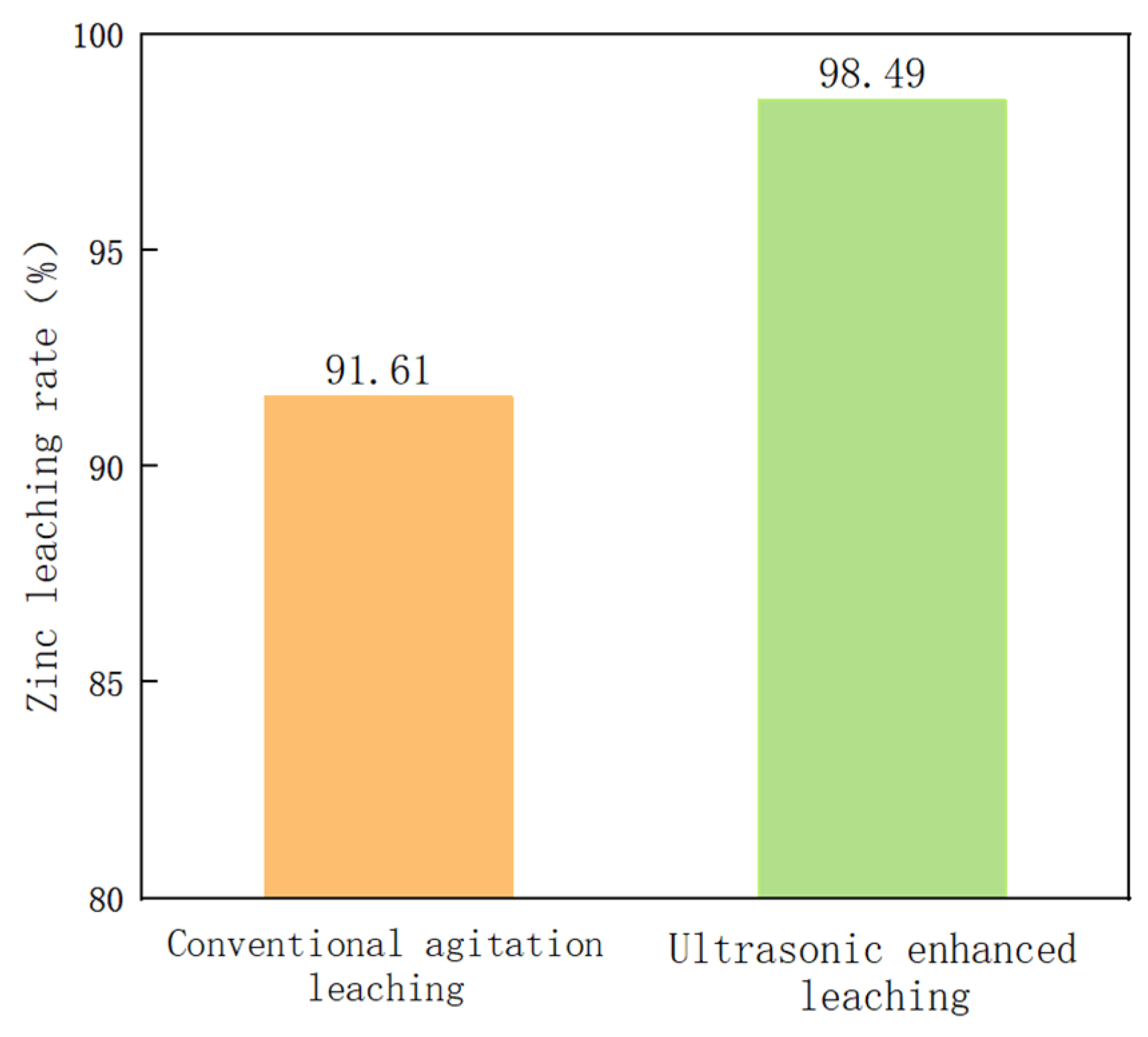
3.6. Comparative Experiment of Conventional–Ultrasonic Leaching
3.7. Leaching Residue Analysis
3.7.1. Chemical Multi-Element Analysis of Leaching Residue
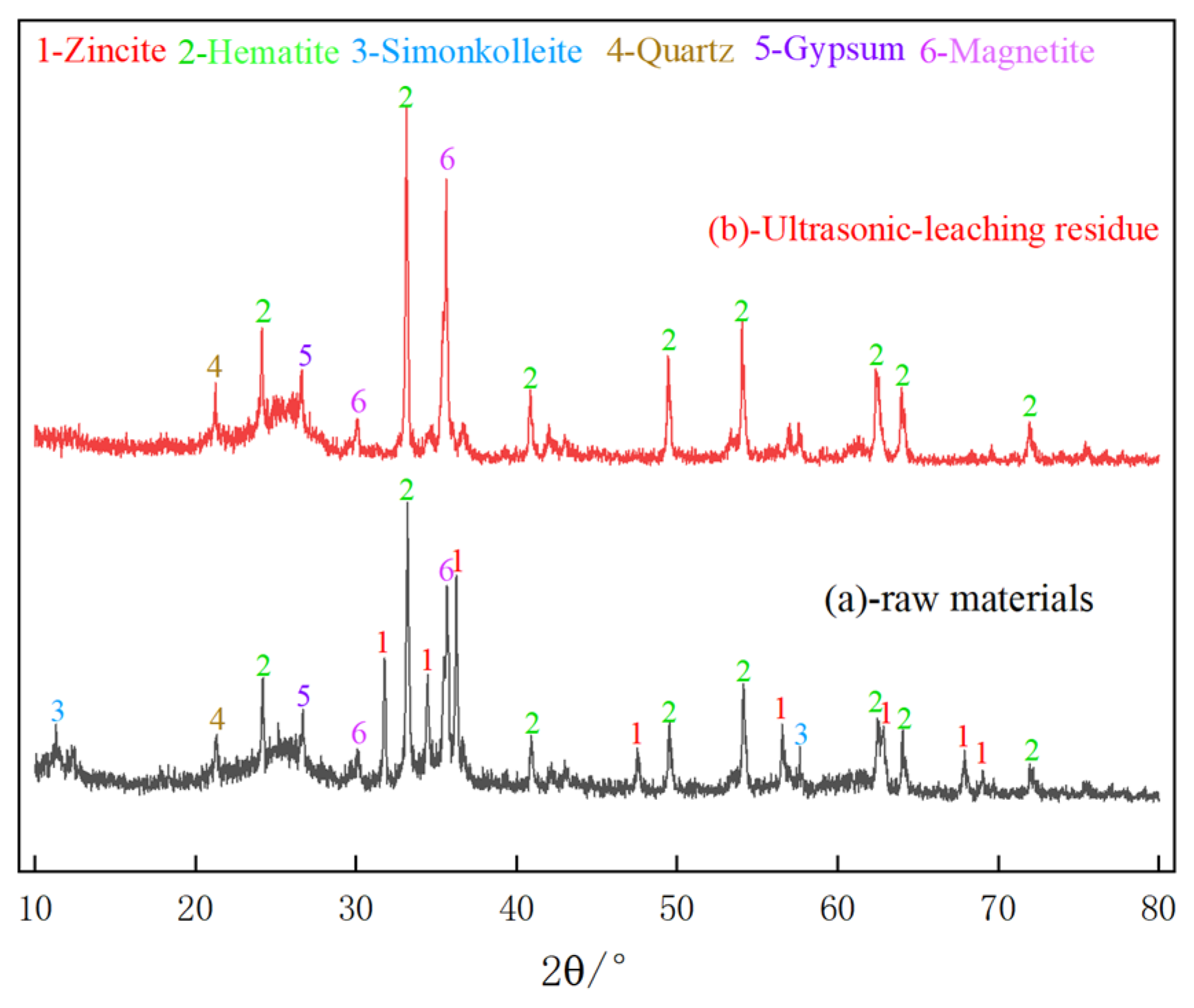
3.7.2. XRD Analysis of Leaching Residue
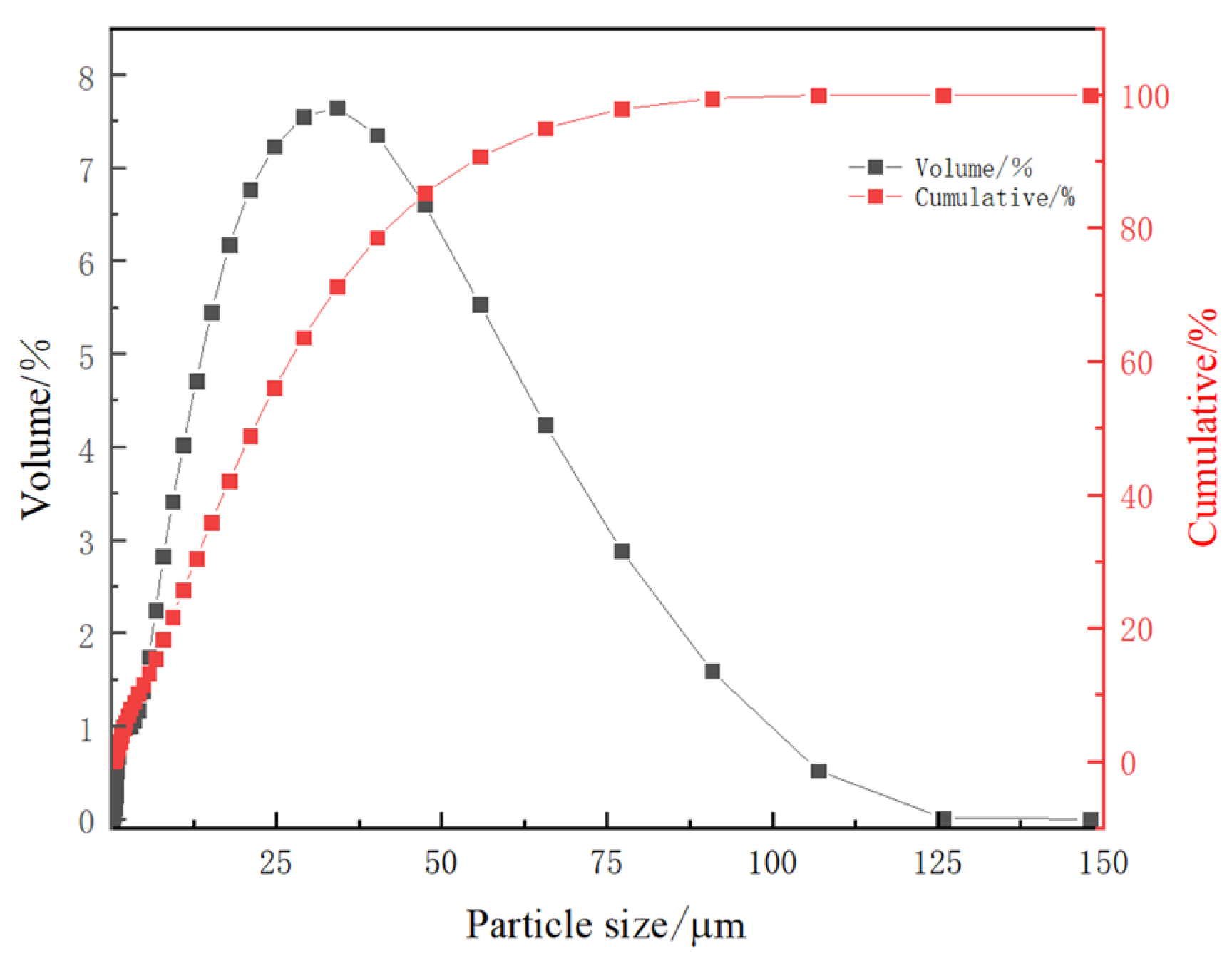
3.7.3. Analysis of Leaching Residue Particle Size Composition
3.7.4. SEM-EDS Analysis of Leaching Residue
4. Conclusions
- (1)
- In this paper, the collaborative enhanced leaching method of low eutectic solvent–ultrasonic was adopted, and the full-factorial experimental design was used to study the high-efficiency leaching of zinc in the zinc-containing dust mud. The leaching rate (𝜂) was taken as the response value, and Ultrasonic power (A, Ultrasonic power), Temperature (B, Temperature), and leaching time (C, ultrasonic power) were selected. Four conditions (Time) and liquid–solid ratios (D, liquid–solid ratio) are the investigated factors. The optimal leaching conditions obtained in this experiment are as follows: Under the optimal conditions, the ratio of liquid to solid was 7:1, the leaching temperature was 40 °C, the ultrasonic power was 90 W, and the leaching time was 80 min. The predicted value of the regression model was 98.47%. The average zinc leaching rate obtained by the 3 groups of parallel verification experiments was 98.49%, and the deviation from the predicted value of the regression model was 0.02%. The verification experiment results were consistent with the results predicted by the regression model, the experimental results were reliable, and the optimization scheme was reasonable and accurate.
- (2)
- The effects of conventional stirring and ultrasonic strengthening on the leaching rate of zinc in metallurgical dust sludge were compared under the optimum process conditions. The zinc leaching rate was increased by 6.88% under ultrasonic enhancement. At the same time, the zinc leaching rate can be kept above 98%, indicating that the process conditions have a certain guiding significance for practical application.
- (3)
- The treatment of zinc-containing dust mud is a difficult problem in the iron and steel industry. The use of low eutectic solvent–ultrasonic synergism to enhance the leaching of zinc-containing dust mud can reduce industrial loss to a certain extent and has certain economic benefits. However, the separation and utilization of other valuable elements still need further research.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- National Bureau of Statistics of China. China Statistical Yearbook; China Statistics Press: Beijing, China, 2022.
- Ju, H.; Li, Y. State of The Art on Inhibitiion for Zinc and Aluminium. Corros. Sci. Prot. Technol. 2006, 18, 353–356. [Google Scholar]
- Wang, S.M. Study on Formation Mechanism of Mechanical Zinc Plating. Master’s Thesis, Kunming University of Science and Technology, Kunming, China, 2002. [Google Scholar]
- Page, M.; Weidenfeller, B.; Hartmann, S. Influence of temperature and aging on the thermal diffusivity, thermal conductivity and heat capacity of a zinc die casting alloy. J. Alloys Compd. 2019, 786, 1060–1067. [Google Scholar] [CrossRef]
- Chang, L.M.; Ling, L.; Nie, P. Aqueous zinc ion battery: Research progress on zinc metal anode 2021. J. Jilin Norm. Univ. Nat. Sci. Ed. 2021, 42, 8–15. [Google Scholar]
- Lan, B.X. Research on Vanadium-Based Cathode Material (Ag0.33V2O5 and FeVO4•nH2O@rGO) for Aqueous Zinc Ion Batteries. Master’s Thesis, Hubei University of Technology, Wuhan, China, 2020. [Google Scholar]
- Yao, Z.Y. Lithium-Rich Li-Zn Alloy as a Novel Anode Material for Lithium Secondary Batteries. Master’s Thesis, University of Electronic Science and Technology of China, Chengdu, China, 2020. [Google Scholar]
- Liu, Q.F.; Liao, Y.L.; Wu, Y.; Xi, J.J.; Ji, G.X. Research progress on enhancing leaching efficiency of chalcopyrite. Chem. Ind. Eng. Prog. 2022, 41, 6099–6110. [Google Scholar]
- Guo, P.; Wang, S.X.; Zhang, L.B. Selective removal of antimony from refractory gold ores by ultrasound. Hydrometallurgy 2019, 190, 105161. [Google Scholar] [CrossRef]
- Zhang, J.; Wu, A.X.; Wang, Y.M.; Chen, X.S. Experimental research in leaching of copper-bearing tailings enhanced by ultrasonic treatment. J. China Univ. Min. Technol. 2008, 18, 98–102. [Google Scholar] [CrossRef]
- Zuzana, H.T.; Frantisek, K.; Zita, T.; Dusan, O.M.L.; Andrea, M.; Tomas, H. Acidic leaching both of zinc and iron from basic oxygen furnace sludge. J. Hazard. Mater. 2011, 192, 1100–1107. [Google Scholar]
- Kukurugya, F.; Vindt, T.; Havlík, T. Behavior of zinc, iron and calcium from electric arc furnace (EAF) dust in hydrometallurgical processing in sulfuric acid solutions: Thermodynamic and kinetic aspects. Hydrometallurgy 2015, 154, 20–32. [Google Scholar] [CrossRef]
- Lisarb, O.D.; Thais, L.G.; Paola, A.M.; Edson, I.M.; Fabio, A.D.; Erico, M.M.F. Ultrasound-assisted extraction of rare-earth elements from carbonatite rocks. Ultrason. Sonochem. 2018, 40 Pt B, 24–29. [Google Scholar]
- Rahimi, G.; Rastegar, S.O.; Rahmani, C.F.; Gu, T. Ultrasound-assisted leaching of vanadium from fly ash using lemon juice organic acids. RSC Adv. 2020, 10, 1685–1696. [Google Scholar] [CrossRef]
- Yu, B.Q.; Kou, J.; Sun, C.B.; Xing, Y. Extraction of copper from copper-bearing biotite by ultrasonic-assisted leaching. Int. J. Miner. Metall. Mater. 2021, 29, 212–217. [Google Scholar] [CrossRef]
- Ding, W.; Bao, S.X.; Zhang, Y.M.; Xiao, J.H. Mechanism and kinetics study on ultrasound assisted leaching of gallium and zinc from corundum flue dust. Miner. Eng. 2022, 183, 107624. [Google Scholar] [CrossRef]
- Wang, F.; Yu, J.X.; Xiong, W.L.; Xu, Y.L.; Chi, R.A. A two-step leaching method designed based on chemical fraction distribution of the heavy metals for selective leaching of Cd, Zn, Cu, and Pb from metallurgical sludge. Environ. Sci. Pollut. Res. Int. 2018, 25, 1752–1765. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.G.; Gao, J.M.; Liu, W.W.; Zhang, M.; Guo, M. Recovery of metal-doped zinc ferrite from zinc-containing electric arc furnace dust: Process development and examination of elemental migration. Hydrometallurgy 2016, 166, 1–8. [Google Scholar] [CrossRef]
- Ma, A.Y.; Zheng, X.M.; Gao, L.; Li, K.Q.; Omran, M.; Chen, G. Enhanced Leaching of Zinc from Zinc-Containing Metallurgical Residues via Microwave Calcium Activation Pretreatment. Metals 2021, 11, 1922. [Google Scholar] [CrossRef]
- Wei, L.; Fan, Y.J. Progress of Deep Eutectic Solventsand Their Applications. Chemistry 2011, 74, 333–339. [Google Scholar]
- Hu, P.C.; Jiang, W.; Zhong, L.J. Study on Properties of Deep Eutectic Solvents and Their Applications. Mod. Chem. Ind. 2018, 38, 53–57. [Google Scholar]
- Xiong, X.Q.; Han, Q.; Shi, L.; Xiao, S.Y.; Bi, C. Application of Deep-Eutectic Solvents in Green Organic Synthesis. Chin. J. Org. Chem. 2016, 36, 480–489. [Google Scholar] [CrossRef]
- Bai, F.; Li, J.; Hua, C. Research Progresses of Deep Eutectic Solvents and Its Application in Separationand Catalysis. Mater. Sci. Forum. 2018, 921, 312. [Google Scholar] [CrossRef]
- Daniel, C.; Maria, C.; Gutierrez, M.; Luisa, F. Resorcinol-Based Deep Eutectic Solvents as Both Carbonaceous Precursors and Templating Agents in the Synthesis of Hierarchical Porous Carbon Monoliths. Chem. Mater. 2010, 22, 6146–6152. [Google Scholar]
- Wang, S.X.; Xu, C.Y.; Lei, Z.; Li, J.R.; Lu, J.L.; Xiang, Q.Q.; Chen, X.; Hua, Y.X.; Li, Y. Recycling of zinc oxide dust using ChCl-urea deep eutectic solvent with nitrilotriacetic acid as complexing agents. Miner. Eng. 2022, 175, 107295. [Google Scholar] [CrossRef]
- He, X.H.; Wen, Y.P.; Wang, X.Y.; Cui, Y.R.; Li, L.B.; Ma, H.Z. Leaching NCM cathode materials of spent lithium-ion batteries with phosphate acid-based deep eutectic solvent. Waste Manag. 2023, 157, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Niu, F.S.; He, S.T.; Zhang, J.X.; Chen, W. Study on Ultrasonically-Enhanced Deep Eutectic Solvents Leaching of Zinc from Zinc-Containing Metallurgical Dust Sludge. Metals 2022, 12, 1856. [Google Scholar] [CrossRef]
- Liu, Z.X.; Wang, L. Experimental Design and Data Processing; Chemical Industry Press: Beijing, China, 2015; p. 4144. [Google Scholar]
- Zhu, M.; Dong, L.F.; Qi, H.G.; Zhao, S.Y.; Wang, L.S. Application of DOE total factor experimental design method in reducing the rate of picking off the stem line by air separation. Silicon Val. 2014, 7, 129+132. [Google Scholar]
- Dong, J.J. Study on Leaching Behaviors of Zinc-Bearing Dust Sludge Based on Deep Eutectic Solvents. Master’s Thesis, North China Institute of Technology, Tangshan, China, 2022; p. 000193. [Google Scholar]
- He, Z.; Zhang, J.K.; Wang, L.L. Six Sigma Management, 1st ed.; China Economic Publishing House: Beijing, China, 2013. [Google Scholar]
- Gürkan, E.H.; Tibet, Y.; Çoruh, S. Application of Full Factorial Design Method for Optimization of Heavy Metal Release from Lead Smelting Slag. Sustainability 2021, 13, 4890. [Google Scholar] [CrossRef]
- Li, W.Y.; Jiao, F.; Chen, C.; Qin, W.Q.; Liu, W. Full-factorial Experimental Design for Leaching of Aluminum and Silver from Waste Photovoltaic Modules. Chin. J. Nonferrous Met. 2023, 33, 898–911. [Google Scholar]
- Liu, W.; Liu, Z.L.; Liu, C. Optimization of bioleaching low-grade copper-molybdenum ore by response surface methodology. J. Cent. South Univ. Sci. Technol. 2021, 52, 3111–3120. [Google Scholar]







| Element | Fe2O3 | ZnO | SiO2 | CaO | SO3 | Al2O3 | MgO | Cl | PbO |
|---|---|---|---|---|---|---|---|---|---|
| Content | 49.754 | 14.79 | 8.702 | 6.976 | 2.872 | 5.847 | 3.246 | 5.764 | 1.638 |
| K2O | MnO | P2O5 | SnO2 | SrO | CuO | I | V2O5 | Br | |
| 0.936 | 0.377 | 0.568 | 0.178 | 0.022 | 0.053 | 0.062 | 0.011 | 0.14 |
| Fixed Condition | Stirring Speed/rpm 250 | ||||
|---|---|---|---|---|---|
| Variable | Level | Ultrasonic power (UP)/W | Temperature (TEMP)/°C | Time (TM) /min | Liquid–solid ratio (LSR)/g/L |
| Code | A | B | C | D | |
| Low | 30 | 20 | 40 | 5 | |
| Cur | 60 | 30 | 60 | 6 | |
| High | 90 | 40 | 80 | 7 | |
| Std Order | Run Order | Center Pt | Block | UP /W | Temp /°C | TM /h | LSR /g/L |
|---|---|---|---|---|---|---|---|
| 19 | 1 | 0 | 1 | 60 | 30 | 60 | 6 |
| 13 | 2 | 1 | 1 | 30 | 20 | 80 | 7 |
| 12 | 3 | 1 | 1 | 90 | 40 | 40 | 7 |
| 17 | 4 | 0 | 1 | 60 | 30 | 60 | 6 |
| 8 | 5 | 1 | 1 | 90 | 40 | 80 | 5 |
| 6 | 6 | 1 | 1 | 90 | 20 | 80 | 5 |
| 16 | 7 | 1 | 1 | 90 | 40 | 80 | 7 |
| 2 | 8 | 1 | 1 | 90 | 20 | 40 | 5 |
| 9 | 9 | 1 | 1 | 30 | 20 | 40 | 7 |
| 5 | 10 | 1 | 1 | 30 | 20 | 80 | 5 |
| 15 | 11 | 1 | 1 | 30 | 40 | 80 | 7 |
| 3 | 12 | 1 | 1 | 30 | 40 | 40 | 5 |
| 18 | 13 | 0 | 1 | 60 | 30 | 60 | 6 |
| 10 | 14 | 1 | 1 | 90 | 20 | 40 | 7 |
| 11 | 15 | 1 | 1 | 30 | 40 | 40 | 7 |
| 7 | 16 | 1 | 1 | 30 | 40 | 80 | 5 |
| 4 | 17 | 1 | 1 | 90 | 40 | 40 | 5 |
| 1 | 18 | 1 | 1 | 30 | 20 | 40 | 5 |
| 14 | 19 | 1 | 1 | 90 | 20 | 80 | 7 |
| Std Order | Run Order | UP /W | Temp /°C | TM /h | LSR /g/L | Leaching Rate/% |
|---|---|---|---|---|---|---|
| 19 | 1 | 60 | 30 | 60 | 6 | 96.78 |
| 13 | 2 | 30 | 20 | 80 | 7 | 96.95 |
| 12 | 3 | 90 | 40 | 40 | 7 | 97.62 |
| 17 | 4 | 60 | 30 | 60 | 6 | 96.85 |
| 8 | 5 | 90 | 40 | 80 | 5 | 98.15 |
| 6 | 6 | 90 | 20 | 80 | 5 | 97.58 |
| 16 | 7 | 90 | 40 | 80 | 7 | 98.52 |
| 2 | 8 | 90 | 20 | 40 | 5 | 96.46 |
| 9 | 9 | 30 | 20 | 40 | 7 | 96.41 |
| 5 | 10 | 30 | 20 | 80 | 5 | 97.4 |
| 15 | 11 | 30 | 40 | 80 | 7 | 97.86 |
| 3 | 12 | 30 | 40 | 40 | 5 | 97.59 |
| 18 | 13 | 60 | 30 | 60 | 6 | 96.8 |
| 10 | 14 | 90 | 20 | 40 | 7 | 96.87 |
| 11 | 15 | 30 | 40 | 40 | 7 | 97.19 |
| 7 | 16 | 30 | 40 | 80 | 5 | 97.3 |
| 4 | 17 | 90 | 40 | 40 | 5 | 97.74 |
| 1 | 18 | 30 | 20 | 40 | 5 | 96.85 |
| 14 | 19 | 90 | 20 | 80 | 7 | 97.93 |
| Source | Freedom | Adj SS | Adj MS | F Value | p Value |
|---|---|---|---|---|---|
| model | 10 | 5.17660 | 0.51766 | 4.09 | 0.029 |
| linear | 4 | 4.34650 | 1.08663 | 8.58 | 0.005 |
| UP/W | 1 | 0.83141 | 0.83141 | 6.56 | 0.034 |
| Temp/°C | 1 | 1.90881 | 1.90881 | 15.07 | 0.005 |
| TM/min | 1 | 1.59223 | 1.59223 | 12.57 | 0.008 |
| LSR | 1 | 0.01406 | 0.01406 | 0.11 | 0.748 |
| 2-Factor interaction | 6 | 0.83010 | 0.13835 | 1.09 | 0.441 |
| UP/W* Temp/°C | 1 | 0.07953 | 0.07953 | 0.63 | 0.451 |
| UP/W* TM/min | 1 | 0.33831 | 0.33831 | 2.67 | 0.141 |
| UP/W* LSR | 1 | 0.16484 | 0.16484 | 1.30 | 0.287 |
| Temp/°C*TM/min | 1 | 0.06831 | 0.06831 | 0.54 | 0.484 |
| Temp/°C*LSR | 1 | 0.01994 | 0.01994 | 0.16 | 0.702 |
| TM/min*LSR | 1 | 0.15917 | 0.15917 | 1.26 | 0.295 |
| Error | 8 | 1.01324 | 0.12665 | ||
| Bend | 1 | 0.35059 | 0.35059 | 3.70 | 0.096 |
| Misfitting | 5 | 0.30645 | 0.06129 | 0.34 | 0.855 |
| Pure error | 2 | 0.35620 | 0.17810 | ||
| Total | 18 | 6.18984 |
| LSR/g/L | Temp/°C | UP/W | TM/min | Actual Value/% | Expected Value/% |
|---|---|---|---|---|---|
| 7:1 | 40 | 90 | 80 | 98.52 | 98.47 |
| 7:1 | 40 | 90 | 80 | 98.46 | 98.47 |
| 7:1 | 40 | 90 | 80 | 98.49 | 98.47 |
| Element | TFe | Zn | SO3 | CaO | SiO2 |
|---|---|---|---|---|---|
| Content | 36.1 | 0.25 | 0.72 | 1.29 | 5.02 |
| Cl | K2O | CuO | PbO | ||
| 0.18 | 0.13 | 0.022 | 0.084 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niu, F.; Bu, Z.; Zhang, J.; He, S.; Chang, Z. Zn Extraction from Zinc-Containing Sludge Using Ultrasonic Treatment Leaching with ChCl-MA DES. Metals 2023, 13, 1192. https://doi.org/10.3390/met13071192
Niu F, Bu Z, Zhang J, He S, Chang Z. Zn Extraction from Zinc-Containing Sludge Using Ultrasonic Treatment Leaching with ChCl-MA DES. Metals. 2023; 13(7):1192. https://doi.org/10.3390/met13071192
Chicago/Turabian StyleNiu, Fusheng, Ziheng Bu, Jinxia Zhang, Shengtao He, and Zhenjia Chang. 2023. "Zn Extraction from Zinc-Containing Sludge Using Ultrasonic Treatment Leaching with ChCl-MA DES" Metals 13, no. 7: 1192. https://doi.org/10.3390/met13071192
APA StyleNiu, F., Bu, Z., Zhang, J., He, S., & Chang, Z. (2023). Zn Extraction from Zinc-Containing Sludge Using Ultrasonic Treatment Leaching with ChCl-MA DES. Metals, 13(7), 1192. https://doi.org/10.3390/met13071192







