1. Introduction
A lithium galvanic battery is a primary battery with lithium metal or alloy as the negative electrode, which has the advantages of large specific energy, a high working voltage, a wide temperature range, good storage performance, and a small self-discharge rate as well as being easy to carry and use. It can form various battery packs through a series-parallel connection, which is one of the preferred power sources for military and civilian equipment [
1]. The lithium-thionyl chloride (Li\SOCl
2) cell in the lithium galvanic battery system is a novel chemical power source that has developed rapidly in the past 40 years, whose mass-specific energy is relatively high among the known ones, which can reach more than 500 Wh/kg, 4 times that of the conventional alkaline zinc-manganese battery. In recent years, it has been widely used in the field of national defense [
2].
As the negative electrode of the battery, lithium metal will release electrons when discharging, and the generated Li
+ will enter the electrolyte and react with Cl
− on the surface of the positive electrode to generate LiCl. SOCl
2 is not only the active material in the positive electrode of batteries, but it could also be a solvent [
3]. SOCl
2 is a mixture of sulfuric acid and hydrochloric acid in water, and it is easy to produce. When SOCl
2 encounters water, it is easy to generate mixed acid. This mixture is very corrosive and easily corrodes stainless steel and general metals, so it is difficult to store or handle it in the air.
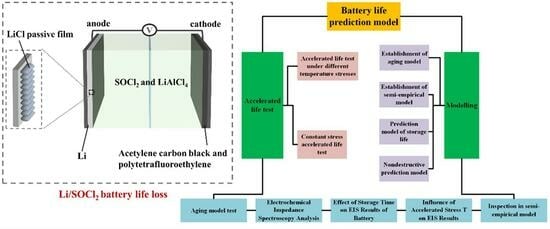
The negative electrode in the Li\SOCl
2 cell is lithium metal, and the positive electrode material is a kind of inert porous carbon material. SOCl
2 dissolved with inorganic salts such as LiClO
4 or LiAlCl
4 could be used as an electrolyte. Li\SOCl
2 cell and its working principle [
4] could be expressed as follows:
(−) Li|SOCl2−/LiAlCl4|SOCl2 (C) (+)
Anode: Li → Li+ + e− (oxidation reaction)
Cathode: 2SOCl2 + 4e−→ SO2 ↑ + S ↓ + 4Cl− (reduction reaction)
Total reaction: 4Li + 2SOCl2 → SO2 ↑ + S ↓ + 4LiCl ↓
The cathode of a Li\SOCl
2 battery is a mixture of polytetrafluoroethylene (PTFE) and acetylene black, which are carriers of discharge products S and LiCl. The reaction products sulfur and sulfur dioxide are dissolved in excess thionyl chloride electrolyte, and during discharge, there will be a certain degree of pressure due to the production of sulfur dioxide [
5]. Wang et al. [
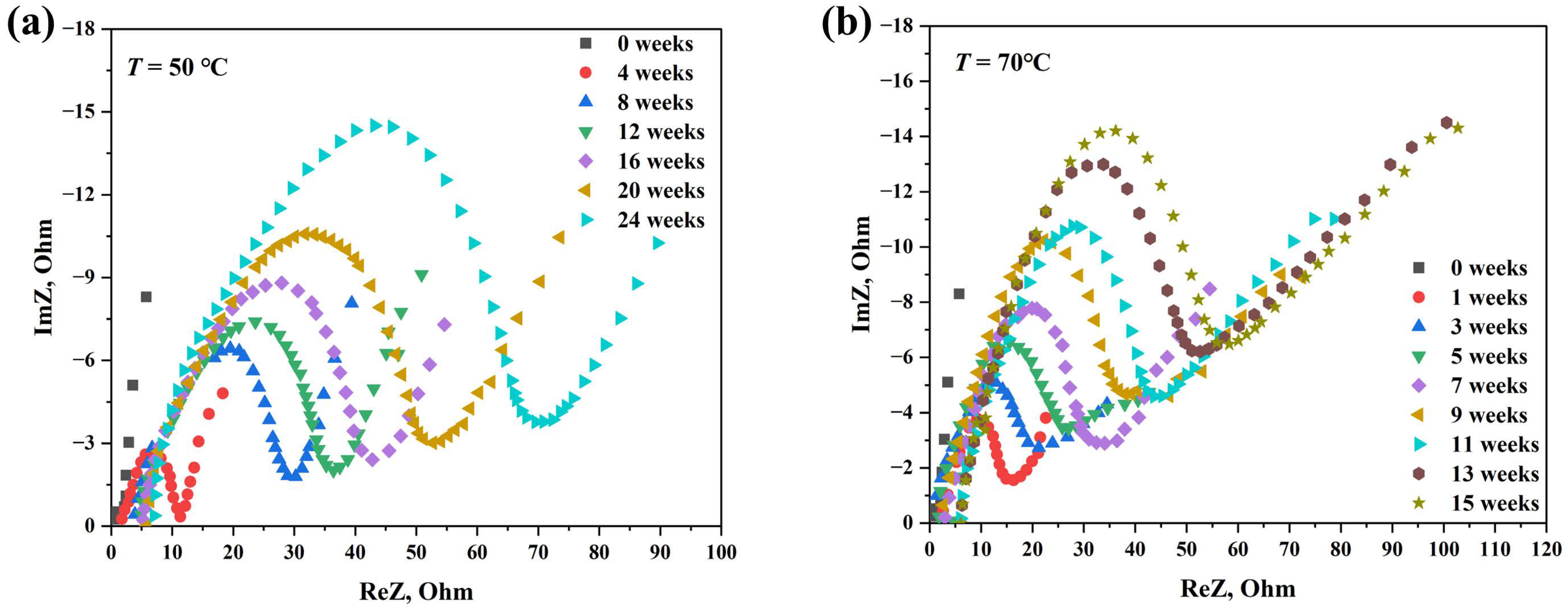
6] found that, during the storage period, the lithium negative electrode gradually reacts with the thionyl chloride electrolyte to form LiCl, and the lithium negative electrode can be protected by the LiCl film formed on it. This passivation film is beneficial to prolong the storage life of the battery, but it will cause voltage lag at the beginning of discharge. If the voltage of a lithium primary battery cannot immediately reach the required working state due to passivation or other reasons, it suffers a voltage lag. After long-term storage at high temperatures, the voltage lag phenomenon will be particularly obvious when the battery is discharged at a low temperature or with a slightly large current.
The carbon cathode of a Li\SOCl
2 battery plays an important role in the flow of electrolytes in the battery and has the function of storing the reaction product lithium chloride. The discharge time, discharge voltage platform, and energy density of the battery are closely related to the porosity and pore size distribution of the carbon cathode. As electrolyte and cathode active material, SOCl
2 needs to pass through the pores of the carbon cathode during the battery reaction. The uniform distribution of pores directly affects the reaction degree of SOCl
2. The distribution of lithium chloride particles in the carbon cathode is directly related to pore size, and the reaction product lithium chloride can enter the carbon cathode through mesopores and macropores, which will directly affect the discharge time and capacity of the battery. It is found that the carbon cathode with a large specific surface area can prolong the discharge time of the battery [
7].
In recent years, a lot of research on the long-term storage of batteries has been mostly aimed at lithium-ion secondary batteries rather than primary batteries, and the existing life prediction models are mostly developed based on lithium-ion secondary batteries. There are few studies on the life prediction model of lithium primary batteries.
At present, the research on the anode of a Li\SOCl
2 battery is mainly aimed at the voltage lag phenomenon of the battery and the lithium chloride passivation film that appears on the anode lithium, which leads to the low initial discharge voltage platform of the battery. During the storage of the battery, lithium metal will contact SOCl
2, and a redox reaction will occur, resulting in a lithium chloride passivation film insoluble in electrolytes. The transfer of electrons and the flow of electrolytes will be hindered by this passivation film [
8]. At present, the existing research is mainly divided into three solutions [
9]. The first is to change the anode lithium into lithium alloy, which increases the difficulty of forming a purified membrane. The second is to coat conductive electrolytes on a lithium sheet to improve the conductivity of the passivation film. The third is to make a lithium-thionyl chloride battery and a supercapacitor hybrid battery and destroy the passivation film of the anode by pulse current. Metal oxide electrodes can effectively solve the problem of passive-film deposition. Almessiere et al. [
10] and Zdorovet et al. [
11] developed a Dy-substituted Ni-Cu-Zn spinel ferrite material and a FeCo-Fe
2CoO
4/Co
3O
4-spinel type material. These two materials can not only fundamentally solve the problem of passive film, but also have excellent electrochemical performance. They are ideal candidates for electrode materials. In addition, many studies have shown that the addition of the second phase can significantly improve the electronic properties of the resulting composite film. The telluride glass doped with CeO
2 obtained by Kozlovskiy et al. [
12] not only has excellent anti-degradation ability but also has excellent electromagnetic shielding ability. Almessiere et al. [
13] doped Sc
3+ in Sr
0.5Ba
0.5ScxFe
12−xO
19 (SrBaSc)/NiFe
2O
4 (NiFe) hard-soft nanocomposites (NCs) to change their structure, morphology, and electromagnetic properties. Zhang and Wang [
14] used the accelerated life test method. The charging and discharging tests and analysis of lithium batteries at different temperatures are carried out, and it is considered that the environmental temperature has a very serious impact on the life of lithium batteries. The results show that when the ambient temperature is higher than the normal working temperature, the higher the charging and discharging temperature of a lithium battery, the faster the battery aging speed, and the more serious the battery life attenuation. When the ambient temperature is lower than the normal working temperature, the electrochemical reaction of ions inside the battery will intensify, which will also accelerate the decay process of battery life. Research by Abraham et al. [
15] studies the aging mechanism of batteries based on the lithium loss of negative electrodes. According to the representative accelerated aging data, the relationship between lithium loss and temperature and time was finally established, and the battery life prediction model was developed. Kassem et al. [
16], through the experimental study of lithium batteries at different temperatures such as 30 °C, 45 °C, and 60 °C, explored the influence of different temperatures on the life of lithium batteries. The test results show that with the increase of ambient temperature, the life of lithium batteries will be reduced, and the battery-capacity loss is the most serious under the most serious aging condition (
T = 60 °C).
Because the storage life of lithium primary batteries is longer than that of other lithium battery systems, it is necessary to obtain its key characteristic parameter information through an accelerated life test, which is convenient for establishing a model for prediction [
17]. Under the premise of not changing the failure mechanism of the product, the test conditions are enhanced, so that the sample leaks out its defects in a short time, and the failure effect is aggravated [
18]. The prediction of the storage life of lithium primary batteries is usually carried out at room temperature (RT), and the test time is quite long. To get the storage life of lithium primary batteries quickly, the accelerated life test method is usually adopted [
19,
20].
The basic principle of accelerated life tests is to deduce the life change law of test samples under normal strength stress by analyzing and processing the life change law of test samples under high strength stress. The relationship between different strength stress and the life change law of test samples is established, and the bridge reflecting the mathematical relationship between stress and the life of test samples is called the aging model. At present, there is a certain amount of theoretical basis for the construction of a lithium battery aging model. Through the study of the existing literature [
21,
22], it is found that the aging models of lithium primary batteries are mainly divided into life-prediction models based on physical and chemical mechanism-mechanism models, data-driven life-prediction-empirical model, and integrated life-prediction-semi-empirical model. The advantage of the semi-empirical model is that compared with the empirical model, there are not so many parameters that need to be obtained through experimental tests, and it can connect the internal electrochemical reaction of the battery with the external stress test so that the working process of the battery can be more intuitively reflected [
23,
24,
25,
26].
In the accelerated life experiment of the battery, the previous results show that the capacity attenuation of the battery is the key factor in determining whether the battery can continue to work with the increase of storage time and the change of storage temperature [
27]. Dubarry et al. [
28] found that the battery-capacity attenuation mechanism can be divided into three categories: active lithium loss, negative active material loss, and positive active material loss. For lithium primary batteries, the capacity attenuation mainly depends on the degree of lithium loss, and the main ways of lithium loss during storage are self-discharge reaction and the formation of solid electrolyte interface film (SEI) at the interface between electrode and electrolyte [
29,
30,
31,
32]. Combined with other testing methods such as battery charging and discharging performance tests, electrochemical impedance spectroscopy (EIS), capacity increment analysis (ICA), and differential voltage analysis (DVA), a lot of information about battery aging mechanisms can be obtained. In actual production processing and during normal use, the storage and shelving statuses of batteries are very common. The high-temperature storage environment will cause the battery capacity to significantly decay, and the influence degree will be aggravated with the increase in temperature [
33,
34,
35,
36,
37,
38,
39].
In this study, a lithium primary battery was taken as the object, and a lithium thionyl chloride battery was taken as an example to study its performance degradation during storage through accelerated life tests at high temperatures. By analyzing the reaction mechanism of the lithium-thionyl chloride battery and combining it with the change in the AC impedance spectrum, the main aging mechanism of the battery was determined. On this basis, the semi-empirical model between performance characteristic parameters and storage time and temperature stress is established. Through this model, the relationship between accelerated stress and battery life can be quantitatively described. By comparing the experimental data with the aging model to predict the battery life, the accuracy of the model is verified.
Table 1 lists the comparison between this work and other main references in this paper.