Safety and Seroconversion of Immunotherapies against SARS-CoV-2 Infection: A Systematic Review and Meta-Analysis of Clinical Trials
Abstract
:1. Introduction
2. Results
2.1. Systematic Review of Clinical Trials
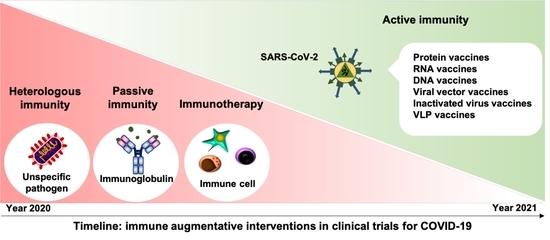
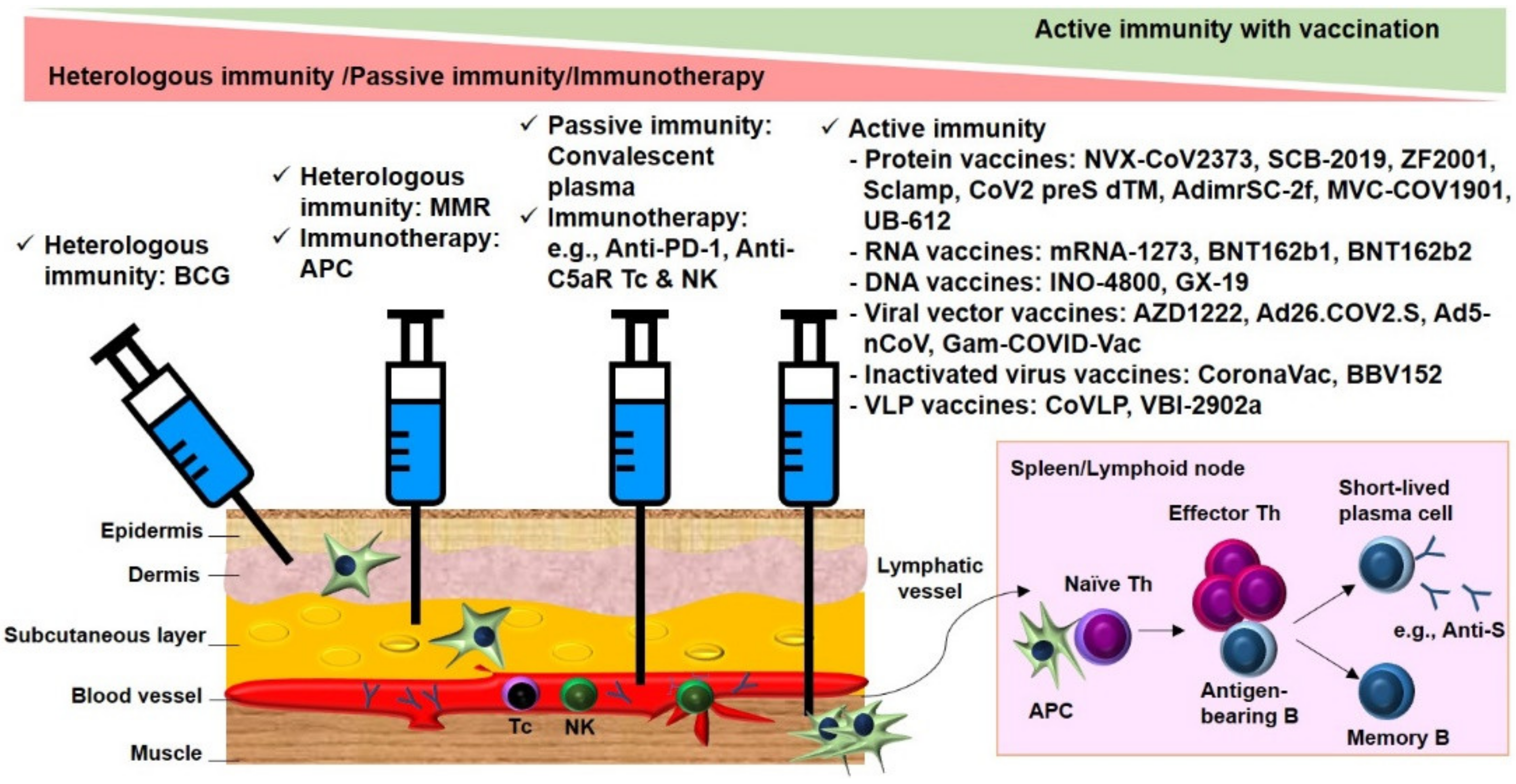
2.1.1. Cross-Protective Vaccines Realizing Heterologous Immunity
2.1.2. Inactivated and Subunit Vaccines Allowing for Active Immunity
2.1.3. Convalescent Plasma or Immunoglobulin Transfer Providing Passive Immunity
2.1.4. Immunotherapy
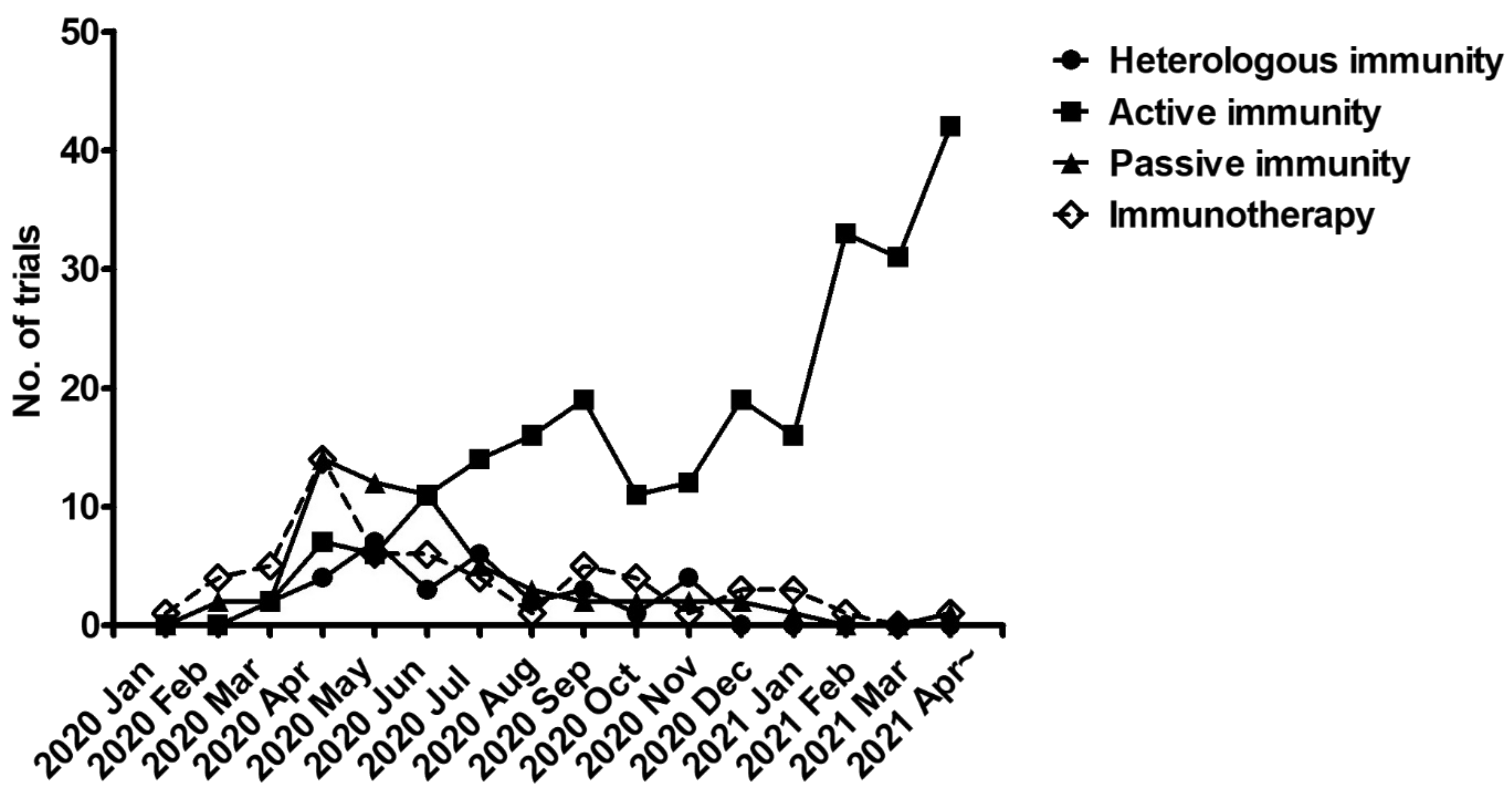
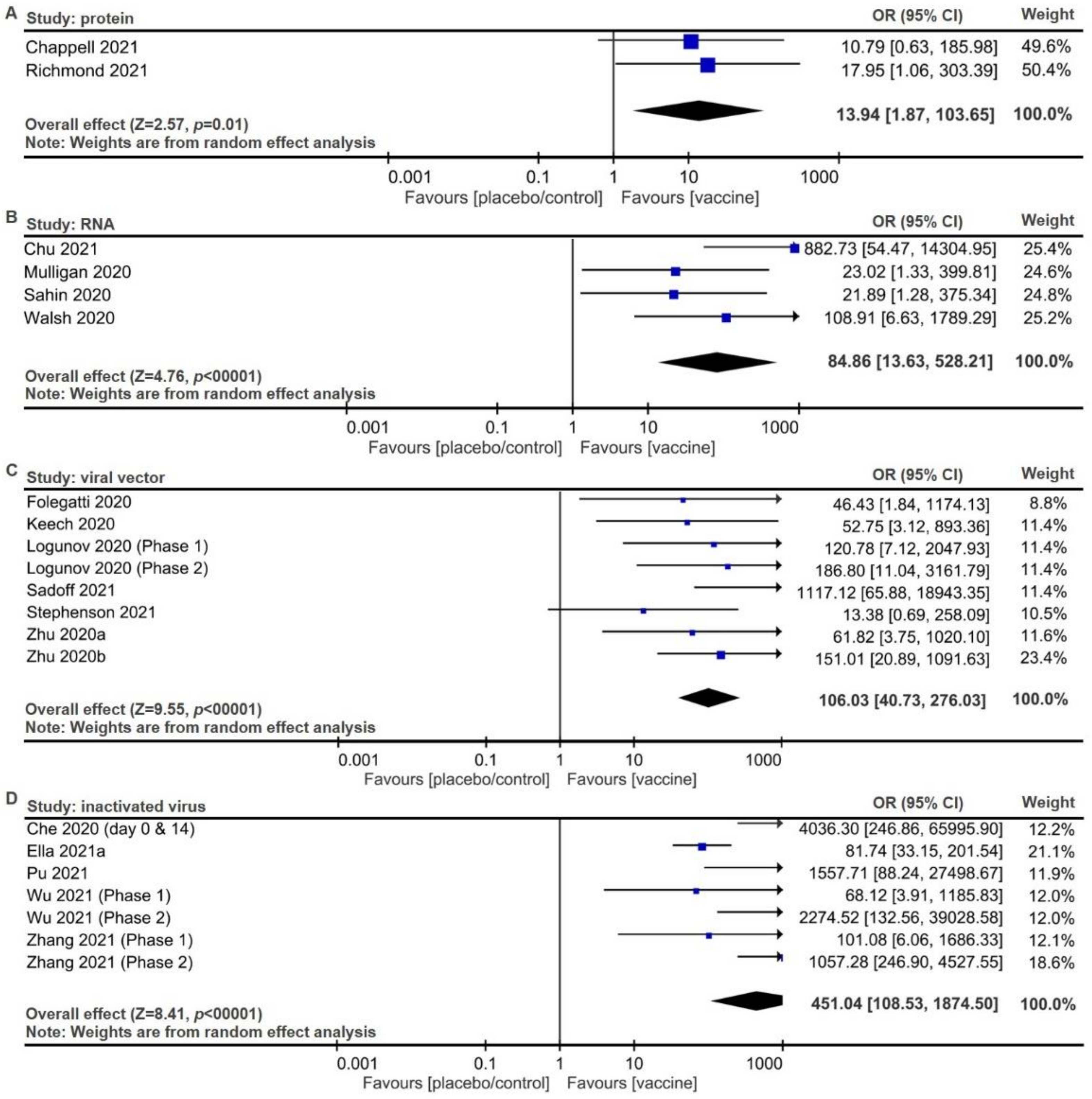
2.2. Meta-Analysis of Trial Reports
3. Discussion
4. Challenges and Perspectives
5. Conclusions
6. Materials and Methods
Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ma, K.S.; Tsai, S.Y. Big Data-driven personal protective equipment stockpiling framework under Universal Healthcare for Disease Control and Prevention in the COVID-19 Era. Int. J. Surg. 2020, 79, 290–291. [Google Scholar] [CrossRef]
- Ma, K.S. Integrating travel history via big data analytics under universal healthcare framework for disease control and prevention in the COVID-19 pandemic. J. Clin. Epidemiol. 2021, 130, 147–148. [Google Scholar] [CrossRef] [PubMed]
- Khailany, R.A.; Safdar, M.; Ozaslan, M. Genomic characterization of a novel SARS-CoV-2. Gene Rep. 2020, 19, 100682. [Google Scholar] [CrossRef]
- Dong, E.; Du, H.; Gardner, L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect. Dis. 2020, 20, 533–534. [Google Scholar] [CrossRef]
- Bonam, S.R.; Kaveri, S.V.; Sakuntabhai, A.; Gilardin, L.; Bayry, J. Adjunct Immunotherapies for the Management of Severely Ill COVID-19 Patients. Cell Rep. Med. 2020, 1, 100016. [Google Scholar] [CrossRef]
- Netea, M.G.; Joosten, L.A.; Latz, E.; Mills, K.H.; Natoli, G.; Stunnenberg, H.G.; O’Neill, L.A.; Xavier, R.J. Trained immunity: A program of innate immune memory in health and disease. Science 2016, 352, aaf1098. [Google Scholar] [CrossRef] [Green Version]
- Gerth, H.J. [Basic principles of active and passive immunizations]. Internist 1979, 20, 264–272. [Google Scholar]
- Yang, Y. Cancer immunotherapy: Harnessing the immune system to battle cancer. J. Clin. Invest. 2015, 125, 3335–3337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zwerling, A.; Behr, M.A.; Verma, A.; Brewer, T.F.; Menzies, D.; Pai, M. The BCG World Atlas: A database of global BCG vaccination policies and practices. PLoS Med. 2011, 8, e1001012. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zimmermann, P.; Perrett, K.P.; van der Klis, F.R.; Curtis, N. The immunomodulatory effects of measles-mumps-rubella vaccination on persistence of heterologous vaccine responses. Immunol. Cell Biol. 2019, 97, 577–585. [Google Scholar] [CrossRef]
- Ma, K.S.; Saeed, H.N.; Chodosh, J.; Wang, C.W.; Chung, Y.C.; Wei, L.C.; Kuo, M.T.; Liang, C.M.; Chang, J.W.; Chung, W.H.; et al. Ocular manifestations of anti-neoplastic immune checkpoint inhibitor-associated Stevens-Johnson syndrome/toxic epidermal necrolysis in cancer patients. Ocul. Surf. 2021, 22, 47–50. [Google Scholar] [CrossRef] [PubMed]
- Vabret, N.; Britton, G.J.; Gruber, C.; Hegde, S.; Kim, J.; Kuksin, M.; Levantovsky, R.; Malle, L.; Moreira, A.; Park, M.D.; et al. Immunology of COVID-19: Current State of the Science. Immunity 2020, 52, 910–941. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Gao, Y.; Wang, G.; Song, G.; Liu, S.; Sun, D.; Xu, Y.; Tian, Z. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell Mol. Immunol. 2020, 17, 533–535. [Google Scholar] [CrossRef] [Green Version]
- Risitano, A.M.; Mastellos, D.C.; Huber-Lang, M.; Yancopoulou, D.; Garlanda, C.; Ciceri, F.; Lambris, J.D. Complement as a target in COVID-19? Nat. Rev. Immunol. 2020. [Google Scholar] [CrossRef] [Green Version]
- Xu, X.; Han, M.; Li, T.; Sun, W.; Wang, D.; Fu, B.; Zhou, Y.; Zheng, X.; Yang, Y.; Li, X.; et al. Effective treatment of severe COVID-19 patients with tocilizumab. Proc. Natl. Acad. Sci. USA 2020, 117, 10970–10975. [Google Scholar] [CrossRef] [PubMed]
- Laterre, P.F.; Francois, B.; Collienne, C.; Hantson, P.; Jeannet, R.; Remy, K.E.; Hotchkiss, R.S. Association of Interleukin 7 Immunotherapy With Lymphocyte Counts Among Patients With Severe Coronavirus Disease 2019 (COVID-19). JAMA Netw. Open 2020, 3, e2016485. [Google Scholar] [CrossRef]
- Sa Ribero, M.; Jouvenet, N.; Dreux, M.; Nisole, S. Interplay between SARS-CoV-2 and the type I interferon response. PLoS Pathog. 2020, 16, e1008737. [Google Scholar] [CrossRef] [PubMed]
- Keech, C.; Albert, G.; Cho, I.; Robertson, A.; Reed, P.; Neal, S.; Plested, J.S.; Zhu, M.; Cloney-Clark, S.; Zhou, H.; et al. Phase 1-2 Trial of a SARS-CoV-2 Recombinant Spike Protein Nanoparticle Vaccine. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Richmond, P.; Hatchuel, L.; Dong, M.; Ma, B.; Hu, B.; Smolenov, I.; Li, P.; Liang, P.; Han, H.H.; Liang, J.; et al. Safety and immunogenicity of S-Trimer (SCB-2019), a protein subunit vaccine candidate for COVID-19 in healthy adults: A phase 1, randomised, double-blind, placebo-controlled trial. Lancet 2021, 397, 682–694. [Google Scholar] [CrossRef]
- Yang, S.; Li, Y.; Dai, L.; Wang, J.; He, P.; Li, C.; Fang, X.; Wang, C.; Zhao, X.; Huang, E.; et al. Safety and immunogenicity of a recombinant tandem-repeat dimeric RBD-based protein subunit vaccine (ZF2001) against COVID-19 in adults: Two randomised, double-blind, placebo-controlled, phase 1 and 2 trials. Lancet Infect. Dis. 2021, 21, 1107–1119. [Google Scholar] [CrossRef]
- Chappell, K.J.; Mordant, F.L.; Li, Z.; Wijesundara, D.K.; Ellenberg, P.; Lackenby, J.A.; Cheung, S.T.M.; Modhiran, N.; Avumegah, M.S.; Henderson, C.L.; et al. Safety and immunogenicity of an MF59-adjuvanted spike glycoprotein-clamp vaccine for SARS-CoV-2: A randomised, double-blind, placebo-controlled, phase 1 trial. Lancet Infect. Dis. 2021, 21, 1383–1394. [Google Scholar] [CrossRef]
- Goepfert, P.A.; Fu, B.; Chabanon, A.L.; Bonaparte, M.I.; Davis, M.G.; Essink, B.J.; Frank, I.; Haney, O.; Janosczyk, H.; Keefer, M.C.; et al. Safety and immunogenicity of SARS-CoV-2 recombinant protein vaccine formulations in healthy adults: Interim results of a randomised, placebo-controlled, phase 1–2, dose-ranging study. Lancet Infect. Dis. 2021. [Google Scholar] [CrossRef]
- Anderson, E.J.; Rouphael, N.G.; Widge, A.T.; Jackson, L.A.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; Pruijssers, A.J.; et al. Safety and Immunogenicity of SARS-CoV-2 mRNA-1273 Vaccine in Older Adults. N. Engl. J. Med. 2020, 383, 2427–2438. [Google Scholar] [CrossRef]
- Chu, L.; McPhee, R.; Huang, W.; Bennett, H.; Pajon, R.; Nestorova, B.; Leav, B.; on behalf of mRNA-1273 Study Group. A preliminary report of a randomized controlled phase 2 trial of the safety and immunogenicity of mRNA-1273 SARS-CoV-2 vaccine. Vaccine 2021, 39, 2791–2799. [Google Scholar] [CrossRef]
- Jackson, L.A.; Anderson, E.J.; Rouphael, N.G.; Roberts, P.C.; Makhene, M.; Coler, R.N.; McCullough, M.P.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; et al. An mRNA Vaccine against SARS-CoV-2–Preliminary Report. N. Engl. J. Med. 2020, 383, 1920–1931. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, M.J.; Lyke, K.E.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Raabe, V.; Bailey, R.; Swanson, K.A.; et al. Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults. Nature 2020, 586, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Sahin, U.; Muik, A.; Derhovanessian, E.; Vogler, I.; Kranz, L.M.; Vormehr, M.; Baum, A.; Pascal, K.; Quandt, J.; Maurus, D.; et al. COVID-19 vaccine BNT162b1 elicits human antibody and TH1 T cell responses. Nature 2020, 586, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Walsh, E.E.; Frenck, R.W., Jr.; Falsey, A.R.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Mulligan, M.J.; Bailey, R.; et al. Safety and Immunogenicity of Two RNA-Based Covid-19 Vaccine Candidates. N. Engl. J. Med. 2020, 383, 2439–2450. [Google Scholar] [CrossRef] [PubMed]
- Tebas, P.; Yang, S.; Boyer, J.D.; Reuschel, E.L.; Patel, A.; Christensen-Quick, A.; Andrade, V.M.; Morrow, M.P.; Kraynyak, K.; Agnes, J.; et al. Safety and immunogenicity of INO-4800 DNA vaccine against SARS-CoV-2: A preliminary report of an open-label, Phase 1 clinical trial. EClinicalMedicine 2021, 31, 100689. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.C.; Li, Y.H.; Guan, X.H.; Hou, L.H.; Wang, W.J.; Li, J.X.; Wu, S.P.; Wang, B.S.; Wang, Z.; Wang, L.; et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: A dose-escalation, open-label, non-randomised, first-in-human trial. Lancet 2020, 395, 1845–1854. [Google Scholar] [CrossRef]
- Folegatti, P.M.; Ewer, K.J.; Aley, P.K.; Angus, B.; Becker, S.; Belij-Rammerstorfer, S.; Bellamy, D.; Bibi, S.; Bittaye, M.; Clutterbuck, E.A.; et al. Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: A preliminary report of a phase 1/2, single-blind, randomised controlled trial. Lancet 2020, 396, 467–478. [Google Scholar] [CrossRef]
- Barrett, J.R.; Belij-Rammerstorfer, S.; Dold, C.; Ewer, K.J.; Folegatti, P.M.; Gilbride, C.; Halkerston, R.; Hill, J.; Jenkin, D.; Stockdale, L.; et al. Phase 1/2 trial of SARS-CoV-2 vaccine ChAdOx1 nCoV-19 with a booster dose induces multifunctional antibody responses. Nat. Med. 2021, 27, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.C.; Guan, X.H.; Li, Y.H.; Huang, J.Y.; Jiang, T.; Hou, L.H.; Li, J.X.; Yang, B.F.; Wang, L.; Wang, W.J.; et al. Immunogenicity and safety of a recombinant adenovirus type-5-vectored COVID-19 vaccine in healthy adults aged 18 years or older: A randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 2020, 396, 479–488. [Google Scholar] [CrossRef]
- Ramasamy, M.N.; Minassian, A.M.; Ewer, K.J.; Flaxman, A.L.; Folegatti, P.M.; Owens, D.R.; Voysey, M.; Aley, P.K.; Angus, B.; Babbage, G.; et al. Safety and immunogenicity of ChAdOx1 nCoV-19 vaccine administered in a prime-boost regimen in young and old adults (COV002): A single-blind, randomised, controlled, phase 2/3 trial. Lancet 2021, 396, 1979–1993. [Google Scholar] [CrossRef]
- Sadoff, J.; Gray, G.; Vandebosch, A.; Cardenas, V.; Shukarev, G.; Grinsztejn, B.; Goepfert, P.A.; Truyers, C.; Fennema, H.; Spiessens, B.; et al. Safety and Efficacy of Single-Dose Ad26.COV2.S Vaccine against Covid-19. N. Engl. J. Med. 2021, 384, 2187–2201. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, K.E.; Le Gars, M.; Sadoff, J.; de Groot, A.M.; Heerwegh, D.; Truyers, C.; Atyeo, C.; Loos, C.; Chandrashekar, A.; McMahan, K.; et al. Immunogenicity of the Ad26.COV2.S Vaccine for COVID-19. JAMA 2021, 325, 1535–1544. [Google Scholar] [CrossRef]
- Logunov, D.Y.; Dolzhikova, I.V.; Zubkova, O.V.; Tukhvatulin, A.I.; Shcheblyakov, D.V.; Dzharullaeva, A.S.; Grousova, D.M.; Erokhova, A.S.; Kovyrshina, A.V.; Botikov, A.G.; et al. Safety and immunogenicity of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine in two formulations: Two open, non-randomised phase 1/2 studies from Russia. Lancet 2020, 396, 887–897. [Google Scholar] [CrossRef]
- Zhang, Y.; Zeng, G.; Pan, H.; Li, C.; Hu, Y.; Chu, K.; Han, W.; Chen, Z.; Tang, R.; Yin, W.; et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18-59 years: A randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infect. Dis. 2021, 21, 181–192. [Google Scholar] [CrossRef]
- Wu, Z.; Hu, Y.; Xu, M.; Chen, Z.; Yang, W.; Jiang, Z.; Li, M.; Jin, H.; Cui, G.; Chen, P.; et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine (CoronaVac) in healthy adults aged 60 years and older: A randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infect. Dis. 2021, 21, 803–812. [Google Scholar] [CrossRef]
- Che, Y.; Liu, X.; Pu, Y.; Zhou, M.; Zhao, Z.; Jiang, R.; Yin, Z.; Xu, M.; Yin, Q.; Wang, J.; et al. Randomized, double-blinded and placebo-controlled phase II trial of an inactivated SARS-CoV-2 vaccine in healthy adults. Clin. Infect. Dis. 2020, ciaa1703. [Google Scholar] [CrossRef]
- Pu, J.; Yu, Q.; Yin, Z.; Zhang, Y.; Li, X.; Yin, Q.; Chen, H.; Long, R.; Zhao, Z.; Mou, T.; et al. The safety and immunogenicity of an inactivated SARS-CoV-2 vaccine in Chinese adults aged 18-59 years: A phase I randomized, double-blinded, controlled trial. Vaccine 2021, 39, 2746–2754. [Google Scholar] [CrossRef] [PubMed]
- Ella, R.; Vadrevu, K.M.; Jogdand, H.; Prasad, S.; Reddy, S.; Sarangi, V.; Ganneru, B.; Sapkal, G.; Yadav, P.; Abraham, P.; et al. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBV152: A double-blind, randomised, phase 1 trial. Lancet Infect. Dis. 2021, 21, 637–646. [Google Scholar] [CrossRef]
- Ella, R.; Reddy, S.; Jogdand, H.; Sarangi, V.; Ganneru, B.; Prasad, S.; Das, D.; Raju, D.; Praturi, U.; Sapkal, G.; et al. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBV152: Interim results from a double-blind, randomised, multicentre, phase 2 trial, and 3-month follow-up of a double-blind, randomised phase 1 trial. Lancet Infect. Dis. 2021, 21, 950–961. [Google Scholar] [CrossRef]
- Ward, B.J.; Gobeil, P.; Seguin, A.; Atkins, J.; Boulay, I.; Charbonneau, P.Y.; Couture, M.; D’Aoust, M.A.; Dhaliwall, J.; Finkle, C.; et al. Phase 1 randomized trial of a plant-derived virus-like particle vaccine for COVID-19. Nat. Med. 2021, 27, 1071–1078. [Google Scholar] [CrossRef]
- Piantadosi, A.; Kanjilal, S. Diagnostic Approach for Arboviral Infections in the United States. J. Clin. Microbiol. 2020, 58, e01926-19. [Google Scholar] [CrossRef]
- Aaby, P.; Benn, C.S. Saving lives by training innate immunity with bacille Calmette-Guerin vaccine. Proc. Natl. Acad. Sci. USA 2012, 109, 17317–17318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Medzhitov, R.; Janeway, C., Jr. Innate immune recognition: Mechanisms and pathways. Immunol. Rev. 2000, 173, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Rusek, P.; Wala, M.; Druszczynska, M.; Fol, M. Infectious Agents as Stimuli of Trained Innate Immunity. Int. J. Mol. Sci. 2018, 19, 456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Netea, M.G.; Dominguez-Andres, J.; Barreiro, L.B.; Chavakis, T.; Divangahi, M.; Fuchs, E.; Joosten, L.A.B.; van der Meer, J.W.M.; Mhlanga, M.M.; Mulder, W.J.M.; et al. Defining trained immunity and its role in health and disease. Nat. Rev. Immunol. 2020, 20, 375–388. [Google Scholar] [CrossRef] [Green Version]
- Kleinnijenhuis, J.; Quintin, J.; Preijers, F.; Joosten, L.A.; Ifrim, D.C.; Saeed, S.; Jacobs, C.; van Loenhout, J.; de Jong, D.; Stunnenberg, H.G.; et al. Bacille Calmette-Guerin induces NOD2-dependent nonspecific protection from reinfection via epigenetic reprogramming of monocytes. Proc. Natl. Acad. Sci. USA 2012, 109, 17537–17542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arts, R.J.W.; Moorlag, S.; Novakovic, B.; Li, Y.; Wang, S.Y.; Oosting, M.; Kumar, V.; Xavier, R.J.; Wijmenga, C.; Joosten, L.A.B.; et al. BCG Vaccination Protects against Experimental Viral Infection in Humans through the Induction of Cytokines Associated with Trained Immunity. Cell Host Microbe 2018, 23, 89–100.e105. [Google Scholar] [CrossRef] [Green Version]
- Parida, S.K.; Poiret, T.; Zhenjiang, L.; Meng, Q.; Heyckendorf, J.; Lange, C.; Ambati, A.S.; Rao, M.V.; Valentini, D.; Ferrara, G.; et al. T-Cell Therapy: Options for Infectious Diseases. Clin. Infect. Dis. 2015, 61 (Suppl. S3), S217–S224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haahr, S.; Michelsen, S.W.; Andersson, M.; Bjorn-Mortensen, K.; Soborg, B.; Wohlfahrt, J.; Melbye, M.; Koch, A. Non-specific effects of BCG vaccination on morbidity among children in Greenland: A population-based cohort study. Int. J. Epidemiol. 2016, 45, 2122–2130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Netea, M.G.; Giamarellos-Bourboulis, E.J.; Dominguez-Andres, J.; Curtis, N.; van Crevel, R.; van de Veerdonk, F.L.; Bonten, M. Trained Immunity: A Tool for Reducing Susceptibility to and the Severity of SARS-CoV-2 Infection. Cell 2020, 181, 969–977. [Google Scholar] [CrossRef] [PubMed]
- Watson, D.C.; Robbins, J.B.; Szu, S.C. Protection of mice against Salmonella typhimurium with an O-specific polysaccharide-protein conjugate vaccine. Infect. Immun. 1992, 60, 4679–4686. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghimire, T.R. The mechanisms of action of vaccines containing aluminum adjuvants: An in vitro vs in vivo paradigm. Springerplus 2015, 4, 181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baxter, D. Active and passive immunity, vaccine types, excipients and licensing. Occup. Med. 2007, 57, 552–556. [Google Scholar] [CrossRef] [Green Version]
- Al-Halifa, S.; Gauthier, L.; Arpin, D.; Bourgault, S.; Archambault, D. Nanoparticle-Based Vaccines Against Respiratory Viruses. Front. Immunol. 2019, 10, 22. [Google Scholar] [CrossRef] [Green Version]
- Hobernik, D.; Bros, M. DNA Vaccines-How Far From Clinical Use? Int. J. Mol. Sci 2018, 19, 3605. [Google Scholar] [CrossRef] [Green Version]
- Nidetz, N.F.; McGee, M.C.; Tse, L.V.; Li, C.; Cong, L.; Li, Y.; Huang, W. Adeno-associated viral vector-mediated immune responses: Understanding barriers to gene delivery. Pharmacol. Ther. 2020, 207, 107453. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.F.; Gaspar, P.D.; de Souza, H.J.L. Refrigeration of COVID-19 Vaccines: Ideal Storage Characteristics, Energy Efficiency and Environmental Impacts of Various Vaccine Options. Energies 2021, 14, 1849. [Google Scholar] [CrossRef]
- Robbins, J.B.; Schneerson, R.; Szu, S.C. Perspective: Hypothesis: Serum IgG antibody is sufficient to confer protection against infectious diseases by inactivating the inoculum. J. Infect. Dis. 1995, 171, 1387–1398. [Google Scholar] [CrossRef] [PubMed]
- Luke, T.C.; Casadevall, A.; Watowich, S.J.; Hoffman, S.L.; Beigel, J.H.; Burgess, T.H. Hark back: Passive immunotherapy for influenza and other serious infections. Crit. Care Med. 2010, 38, e66–e73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hung, I.F.; To, K.K.; Lee, C.K.; Lee, K.L.; Chan, K.; Yan, W.W.; Liu, R.; Watt, C.L.; Chan, W.M.; Lai, K.Y.; et al. Convalescent plasma treatment reduced mortality in patients with severe pandemic influenza A (H1N1) 2009 virus infection. Clin. Infect. Dis. 2011, 52, 447–456. [Google Scholar] [CrossRef]
- Van Griensven, J.; De Weiggheleire, A.; Delamou, A.; Smith, P.G.; Edwards, T.; Vandekerckhove, P.; Bah, E.I.; Colebunders, R.; Herve, I.; Lazaygues, C.; et al. The Use of Ebola Convalescent Plasma to Treat Ebola Virus Disease in Resource-Constrained Settings: A Perspective From the Field. Clin. Infect. Dis. 2016, 62, 69–74. [Google Scholar] [CrossRef]
- Rubin, R. Testing an Old Therapy Against a New Disease: Convalescent Plasma for COVID-19. JAMA 2020. [Google Scholar] [CrossRef]
- Wang, L.T.; Ma, K.S. Correspondence to ‘Normal human enthesis harbours conventional CD4+ and CD8+ T cells with regulatory features and inducible IL-17A and TNF expression’. Ann. Rheum. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Rojas, M.; Rodriguez, Y.; Monsalve, D.M.; Acosta-Ampudia, Y.; Camacho, B.; Gallo, J.E.; Rojas-Villarraga, A.; Ramirez-Santana, C.; Diaz-Coronado, J.C.; Manrique, R.; et al. Convalescent plasma in Covid-19: Possible mechanisms of action. Autoimmun. Rev. 2020, 19, 102554. [Google Scholar] [CrossRef] [PubMed]
- Coley, W.B., II. Contribution to the Knowledge of Sarcoma. Ann. Surg. 1891, 14, 199–220. [Google Scholar] [CrossRef] [PubMed]
- Riddell, S.R.; Watanabe, K.S.; Goodrich, J.M.; Li, C.R.; Agha, M.E.; Greenberg, P.D. Restoration of viral immunity in immunodeficient humans by the adoptive transfer of T cell clones. Science 1992, 257, 238–241. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.S.; Wang, L.T.; Tsai, S.Y. Correspondence to: ‘Combination of human umbilical cord mesenchymal (stromal) stem cell transplantation with IFN-gamma treatment synergistically improves the clinical outcomes of patients with rheumatoid arthritis’. Ann. Rheum. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Yen, B.L.; Yen, M.L.; Wang, L.T.; Liu, K.J.; Sytwu, H.K. Current status of mesenchymal stem cell therapy for immune/inflammatory lung disorders: Gleaning insights for possible use in COVID-19. Stem Cells Transl. Med. 2020, 9, 1163–1173. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.T.; Liu, K.J.; Sytwu, H.K.; Yen, M.L.; Yen, B.L. Advances in mesenchymal stem cell therapy for immune and inflammatory diseases: Use of cell-free products and human pluripotent stem cell-derived mesenchymal stem cells. Stem Cells Transl. Med. 2021, 10, 1288–1303. [Google Scholar] [CrossRef] [PubMed]
- Chakupurakal, G.; Onion, D.; Bonney, S.; Cobbold, M.; Mautner, V.; Moss, P. HLA-peptide multimer selection of adenovirus-specific T cells for adoptive T-cell therapy. J. Immunother. 2013, 36, 423–431. [Google Scholar] [CrossRef]
- Ma, K.S.; Chiang, C.H.; Chen, Y.W.; Wang, L.T. Correspondence to ‘Bacterial citrullinated epitopes generated by Porphyromonas gingivalis infection-a missing link for ACPA production’. Ann. Rheum. Dis. 2021. [Google Scholar] [CrossRef] [PubMed]
- Maus, M.V.; Thomas, A.K.; Leonard, D.G.; Allman, D.; Addya, K.; Schlienger, K.; Riley, J.L.; June, C.H. Ex vivo expansion of polyclonal and antigen-specific cytotoxic T lymphocytes by artificial APCs expressing ligands for the T-cell receptor, CD28 and 4-1BB. Nat. Biotechnol. 2002, 20, 143–148. [Google Scholar] [CrossRef] [PubMed]
- De Alwis, R.; Chen, S.; Gan, E.S.; Ooi, E.E. Impact of immune enhancement on Covid-19 polyclonal hyperimmune globulin therapy and vaccine development. EBioMedicine 2020, 55, 102768. [Google Scholar] [CrossRef]
- Liu, E.; Marin, D.; Banerjee, P.; Macapinlac, H.A.; Thompson, P.; Basar, R.; Nassif Kerbauy, L.; Overman, B.; Thall, P.; Kaplan, M.; et al. Use of CAR-Transduced Natural Killer Cells in CD19-Positive Lymphoid Tumors. N. Engl. J. Med. 2020, 382, 545–553. [Google Scholar] [CrossRef]
- Hu, W.; Wang, G.; Huang, D.; Sui, M.; Xu, Y. Cancer Immunotherapy Based on Natural Killer Cells: Current Progress and New Opportunities. Front. Immunol. 2019, 10, 1205. [Google Scholar] [CrossRef] [PubMed]
- Schwendener, R.A. Liposomes as vaccine delivery systems: A review of the recent advances. Ther. Adv. Vaccines 2014, 2, 159–182. [Google Scholar] [CrossRef]
- McInnes, M.D.F.; Moher, D.; Thombs, B.D.; McGrath, T.A.; Bossuyt, P.M.; PRISMA-DTA Group; Clifford, T.; Cohen, J.F.; Deeks, J.J.; Gatsonis, C.; et al. Preferred Reporting Items for a Systematic Review and Meta-analysis of Diagnostic Test Accuracy Studies: The PRISMA-DTA Statement. JAMA 2018, 319, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Gliklich, R.E.; Dreyer, N.A.; Leavy, M.B. Adverse Event Detection, Processing, and Reporting. Registries for Evaluating Patient Outcomes: A User’s Guide, 3rd ed.; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2014. [Google Scholar]
- Principi, N.; Esposito, S. Adverse events following immunization: Real causality and myths. Expert Opin. Drug Saf. 2016, 15, 825–835. [Google Scholar] [CrossRef] [PubMed]
- Hsu, W.T.; Esmaily-Fard, A.; Lai, C.C.; Zala, D.; Lee, S.H.; Chang, S.S.; Lee, C.C. Antipsychotics and the Risk of Cerebrovascular Accident: A Systematic Review and Meta-Analysis of Observational Studies. J. Am. Med. Dir. Assoc. 2017, 18, 692–699. [Google Scholar] [CrossRef] [PubMed] [Green Version]

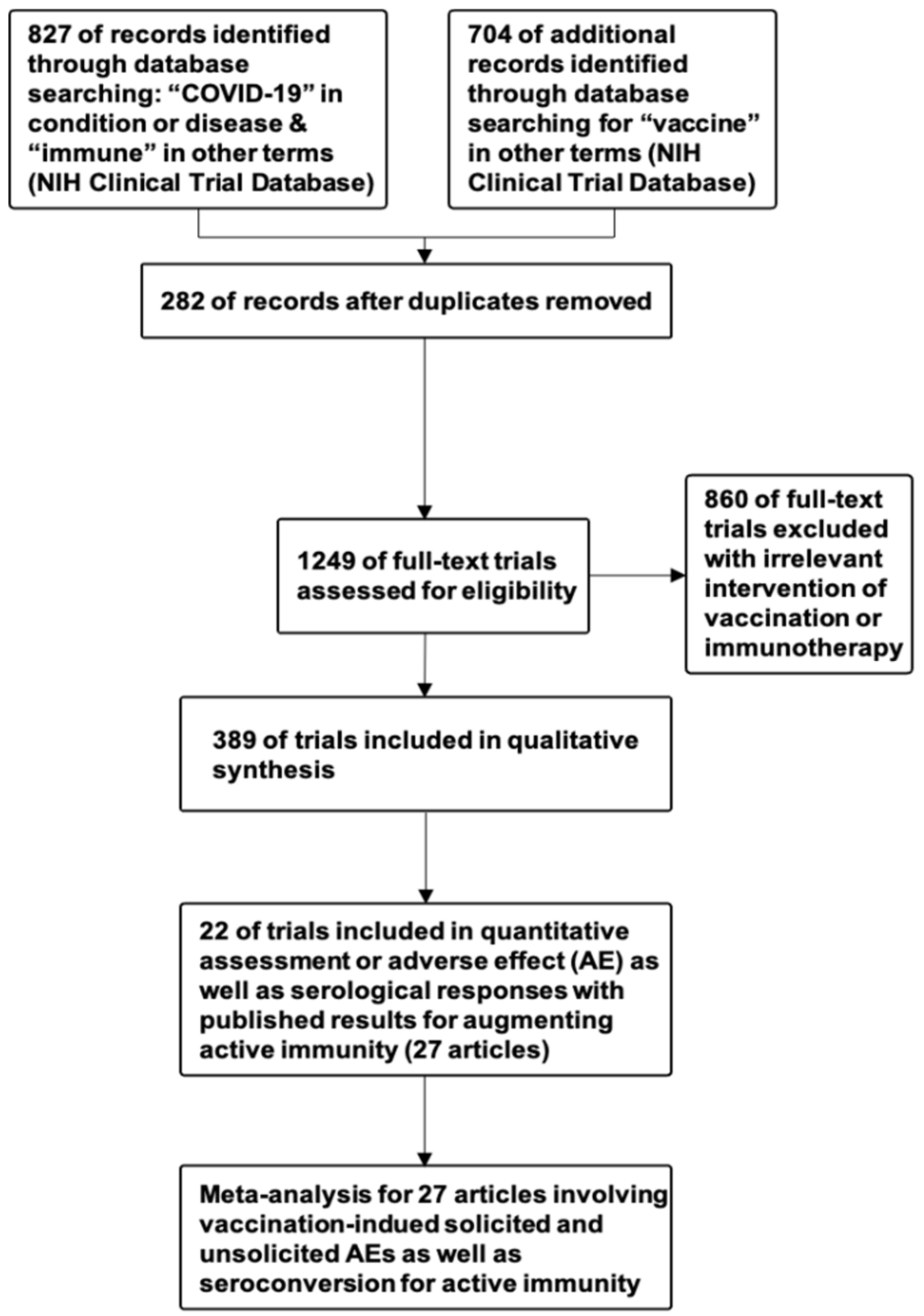
| Strategy | Treatment | Total % | Total # | # of Clinical Trial Phases | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N/A | 1 | 1&2 | 2 | 2 & 3 | 3 | 4 | ||||||
| Heterologous immunity | Mycobacterium vaccine | 8.0 | 6.5 | 32 | 26 | 0 | 0 | 0 | 1 | 0 | 19 | 6 |
| MMR vaccine | 0.5 | 2 | 0 | 0 | 0 | 0 | 0 | 2 | 0 | |||
| Polio vaccine | 0.8 | 3 | 0 | 0 | 0 | 0 | 0 | 1 | 2 | |||
| Zoster Vaccine | 0.3 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | |||
| Active immunity | Protein | 62.4 | 12.3 | 249 | 49 | 0 | 19 | 11 | 10 | 3 | 5 | 1 g |
| RNA | 17.0 | 68 | 15 a,b | 8 | 11 | 13 c,d | 5 e | 10 | 6 h | |||
| DNA | 3.3 | 13 | 0 | 4 | 7 | 0 | 2 | 0 | 0 | |||
| Viral vector | 17.5 | 70 | 5 a | 23 | 17 | 6 c,d | 3 e | 13 f | 3 g,h,i | |||
| Bacterial vector | 0.3 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | |||
| Inactivated | 10.5 | 42 | 5 a,b | 4 | 8 | 3 | 0 | 14 f | 8 i | |||
| Virus-like particle | 1.3 | 5 | 0 | 3 | 1 | 0 | 1 | 0 | 0 | |||
| Live attenuated | 0.3 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | |||
| Passive immunity | Immunoglobulin | 14.8 | 4.5 | 59 | 18 | 0 | 3 | 4 | 3 | 2 | 5 | 1 |
| Convalescent plasma | 10.3 | 41 | 8 | 4 | 1 | 19 | 3 | 6 | 0 | |||
| Immunotherapy | Neutralized antibody/ inhibitor | 14.8 | 7.3 | 59 | 30 | 3 | 1 | 1 | 18 | 3 | 3 | 0 |
| Cytokine | 3.5 | 14 | 0 | 2 | 1 | 10 | 0 | 1 | 0 | |||
| Immune cell | 4.0 | 16 | 0 | 7 | 9 | 0 | 0 | 0 | 0 | |||
| Total # of clinical trial phases | 389 | 33 | 81 | 71 | 81 | 21 | 78 | 24 | ||||
| Total % of clinical trial phases | 100.0 | 8.5 | 20.8 | 18.3 | 20.8 | 5.4 | 20.1 | 6.2 | ||||
| Strategy | Treatment | Total # | # of Participants in Clinical Trial Phases | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| N/A | 1 | 1&2 | 2 | 2 & 3 | 3 | 4 | ||||
| Heterologous immunity | Mycobacterium vaccine | 48,601 | 41,066 | 0 | 0 | 0 | 1000 | 0 | 29,202 | 10,864 |
| MMR vaccine | 260 | 0 | 0 | 0 | 0 | 260 | 0 | |||
| Polio vaccine | 7025 | 0 | 0 | 0 | 0 | 0 | 3600 | 3425 | ||
| Zoster Vaccine | 250 | 0 | 250 | 0 | 0 | 0 | 0 | 0 | ||
| Active immunity | Protein | 878,370 | 172,672 | 0 | 1616 | 6600 | 18,016 | 29,320 | 117,000 | 120 g |
| RNA | 162,052 | 9502 a,b | 981 | 13,961 | 6708 c,d | 54,550 e | 65,500 | 10,850 h | ||
| DNA | 8481 | 0 | 298 | 1105 | 0 | 7078 | 0 | 0 | ||
| Viral vector | 271,524 | 3770 a | 2688 | 9025 | 3691 c,d | 17,930 e | 224,000 f | 10,420 g,h,i | ||
| Bacterial vector | 84 | 0 | 84 | 0 | 0 | 0 | 0 | 0 | ||
| Inactivated/LVP | 263,949 | 2461 a,b | 570 | 5300 | 1750 | 30,612 | 175,790 f | 47,466 i | ||
| Live attenuated | 48 | 0 | 48 | 0 | 0 | 0 | 0 | 0 | ||
| Passive immunity | Immunoglobulin | 16,620 | 2756 | 0 | 74 | 239 | 226 | 390 | 1787 | 40 |
| Convalescent plasma | 13,864 | 6424 | 150 | 15 | 2527 | 688 | 4060 | 0 | ||
| Immunotherapy | Neutralized antibody/ inhibitor | 6988 | 3547 | 204 | 50 | 18 | 2409 | 320 | 546 | 0 |
| Cytokine | 2329 | 0 | 82 | 80 | 1129 | 0 | 1038 | 0 | ||
| Immune cell | 1112 | 0 | 280 | 832 | 0 | 0 | 0 | 0 | ||
| Total participants in clinical trial phases | 929,359 | 22,761 | 7171 | 37,175 | 34,816 | 136,888 | 618,783 | 72,765 | ||
| Type | Authors (Journal & Year) | NCT Number | Phase | Participants | Vaccination Procedures | Outcome Measures on Safety and Immunogenicity | Storage |
|---|---|---|---|---|---|---|---|
| Protein | Keech et al. (N Engl J Med 2020) [18] | 04368988 | 1 | 131 | Intramuscular injections of NVX-CoV2373 (5, 25 μg) at day 0 or/and 21 | Local, systemic, and unsolicited AEs; anti-S antibody; SARS-CoV-2-neutralizing antibody | 2–8 °C |
| Richmond et al. (Lancet 2021) [19] | 04405908 | 1 | 151 | Intramuscular injections of SCB-2019 (3, 9, or 30 μg) at days 0 and 21 | Local and systemic AEs; anti-SCB-2019 antibody; SARS-CoV-2-neutralizing antibody | 2–8 °C | |
| Yang et al. (Lancet Infect Dis 2021) [20] | 04445194 & 04466085 | 1 & 2 | 950 | Intramuscular injections of ZF2001 at day 0, 30, 60 for phase 1 trial, and at day 0, 30 or day 0, 30, 60 for phase 2 trial | Local, systemic, and unsolicited AEs; anti-RBD antibody; SARS-CoV-2-neutralizing antibody | 2–8 °C | |
| Chappell (Lancet Infect Dis 2021) [21] | 04495933 | 1 | 120 | Intramuscular injections of S-clamp vaccine (5, 15, or 45 μg) at days 0 and 28 | Local, systemic, and unsolicited AEs; Anti-clamp antibody; SARS-CoV-2-neutralizing antibody | 2–8 °C | |
| Goepfert et al. (Lancet Infect Dis 2021) [22] | 04537208 | 1 & 2 | 441 | Intramuscular injections of CoV2 preS dTM (1.3 or 2.6 μg) at day 1 for one dose or day 1 and 28 for two doses | Local, systemic, and unsolicited AEs; SARS-CoV-2-neutralizing antibody | 2–8 °C | |
| RNA | Anderson et al. (N Engl J Med 2020) [23] | 04283461 | 1 | 40 | Intramuscular injections of mRNA-1273 (25 or 100 μg) at days 1 and 29 | Local and systemic AEs; anti-S-2P antibody; anti-RBD antibody; SARS-CoV-2 neutralizing antibody | −20 °C or 2–8 °C |
| Chu et al. (Vaccine 2021) [24] | 04405076 | 2 | 600 | Intramuscular injections of mRNA-1273 (50 or 100 μg) at days 1 and 29 | Local and systemic AEs; Anti-S antibody; SARS-CoV-2-neutralizing antibody | −20 °C or 2–8 °C | |
| Jackson et al. (N Engl J Med 2020) [25] | 04283461 | 2 | 45 | Intramuscular injections of mRNA-1273 (25, 100, 250 μg) at days 1 and 29 | Local, systemic, and unsolicited AEs; anti-S-2P antibody; anti-RBD antibody; SARS-CoV-2-neutralizing antibody | −20 °C or 2–8 °C | |
| Mulligan et al. (Nature 2020) [26] | 04368728 | 1 & 2 | 45 | Intramuscular injection of BNT162b1 (10, 30 μg) at day 0 and 21 or BNT162b1 (100 μg) at day0 | Local and systemic AEs; anti-RBD antibody; SARS-CoV-2-neutralizing antibody | −80 °C | |
| Sahin et al. (Nature 2020) [27] | 04368728 | 1 & 2 | 60 | Intramuscular injections of BNT162b1 (1, 10, 30, 50 μg) at day 1 and 22 or BNT162b1 (60 μg) at day 1 | Local and systemic AEs; anti-RBD antibody; SARS-CoV-2-neutralizing antibody | −80 °C | |
| Walsh et al. (N Engl J Med 2020) [28] | 04368728 | 1 & 2 | 195 | Intramuscular injections of BNT162b1 or BNT162b2 (10, 20, 30 μg) at day 0 and 21 | Local and systemic AEs; anti-S1 antibody; SARS-CoV-2-neutralizing antibody | −80 °C | |
| DNA | Tebas et al. (EClinicalMedicine 2021) [29] | 04336410 | 1 | 40 | Intrdermal injections of INO-4800 (1, 2 mg) at weeks 0 and 4 | Local and systemic AEs; anti-S antibody; SARS-CoV-2-neutralizing antibody | 2–8 °C |
| Viral vector | Zhu et al. (Lancet 2020a) [30] | 04313127 | 1 | 108 | Intramuscular injection of adenovirus type-5 vectored COVID-19 vaccine (5 × 1010, 1 × 1011, and 1.5 × 1011 viral particles) at day 0 | Local and systemic AEs; anti-RBD antibody; SARS-CoV-2 neutralizing antibody | N/A |
| Folegatti et al. (Lancet 2020) [31] | 04324606 | 1 & 2 | 1077 | Intramuscular injections of AZD1222 (5 × 1010 viral particles) at days 0 and 28 | Local, systemic, and unsolicited AEs; anti-Spike antibody; SARS-CoV-2 neutralizing antibody | −80 °C or 2–8 K22 | |
| Barrett et al. (Nat Med 2021) [32] | 04324606 | 1 & 2 | 52 | Intramuscular injections of AZD1222 (5 × 109 or 2.5 × 1010 viral particles) at days 0 and 28 | Local and systemic AEs; anti-S antibody; anti-RBD antibody; SARS-CoV-2-neutralizing antibody | −80 °C or 2–8 °C | |
| Zhu et al. (Lancet 2020b) [33] | 04341389 | 2 | 508 | Intramuscular injection of adenovirus type-5 vectored COVID-19 vaccine (5 × 1010 or 1 × 1011 viral particles) at day 0 | Local, systemic, and unsolicited AEs; anti-RBD antibody; SARS-CoV-2-neutralizing antibody | N/A | |
| Ramasamy et al. (Lancet 2021) [34] | 04400838 | 2 & 3 | 560 | Intramuscular injections of AZD1222 (2.2 × 1010 or 3.5–6.5 × 1010 viral particles) at days 0 and 28 | Local and systemic AEs; anti-S antibody; anti-RBD antibody; SARS-CoV-2-neutralizing antibody | −80 °C or 2–8 °C | |
| Sadoff et al. (N Engl J Med 2021) [35] | 04436276 | 1 & 2 | 805 | Intramuscular injections of Ad26.COV2.S (5 × 1010 or 1 × 1011 viral particles) at day 1 or/and day 57 | Local, systemic, and severe unsolicited AEs; Anti-S antibody; SARS-CoV-2-neutralizing antibody | 2–8 °C | |
| Stephenson et al. (JAMA 2021) [36] | 04436276 | 1 | 25 | Intramuscular injections of Ad26.COV2.S (5 × 1010 or 1 × 1011 viral particles) at day 1 or/and day 57 | Local, systemic, and unsolicited AEs; Anti-S antibody; Anti-RBD antibody SARS-CoV-2-neutralizing antibody | 2–8 °C | |
| Logunov et al. (Lancet 2020) [37] | 04436471 & 04437875 | 1 & 2 | 76 | Intramuscular injections of rAd26-S and rAd5-S at day 0 for phase 1 trial, and at day 0 and 21 for phase 2 trial | Local and systemic AEs; anti-RBD antibody; SARS-CoV-2-neutralizing antibody | Frozen: −18 °C & lyophilized: 2–8 °C | |
| Inactivated virus | Zhang et al. (Lancet Infect Dis 2021) [38] | 04352608 | 1 & 2 | 744 | Intramuscular injections of CoronaVac (3 or 6 μg) at day 0 and 14 or 28 for phase 1or phase 2 | Local, systemic, and unsolicited AEs; Anti-RBD antibody; SARS-CoV-2-neutralizing antibody | 2–8 °C |
| Wu et al. (Lancet Infect Dis 2021) [39] | 04383574 | 1 & 2 | 422 | Intramuscular injections of inactivated CN02 strain at day 0 and 28 for phase 1 trial (3, 6 μg), and at day 0 for phase 2 trial (1.5, 3, 6 μg) | Local and systemic AEs; SARS-CoV-2 neutralizing antibody | 2–8 °C | |
| Che et al. (Clin Infect Dis 2020) [40] | 04412538 | 2 | 750 | Injections of inactivated virus (100 EU or 150 EU viral antigen) at day 0 and boost at day 14 or 28 | Local, systemic, and unsolicited AEs; anti-SARS-CoV-2 antibody; SARS-CoV-2-neutralizing antibody | N/A | |
| Pu et al. (Vaccine 2021) [41] | 04412538 | 1 | 192 | Intramuscular injections of inactivated virus with a D614G mutation in the S protein (50, 100, or 150 EU) at days 0 and 14 or 28. | Local, systemic, and unsolicited AEs; Anti-S antibody; SARS-CoV-2-neutralizing antibody | N/A | |
| Ella et al. (Lancet Infect Dis 2021a) [42] | 04471519 | 1 | 375 | Intramuscular injections of BBV152 (3 or 6 μg) at days 0 and 14 | Local and systemic AEs; anti-S antibody; anti-RBD antibody; SARS-CoV-2 neutralizing antibody | 2–8 °C | |
| Ella et al. (Lancet Infect Dis 2021b) [43] | 04471519 | 2 | 380 | Intramuscular injections of BBV152 (3 or 6 μg) at days 0 and 28 | Local and systemic AEs; anti-S antibody; anti-RBD antibody; SARS-CoV-2-neutralizing antibody | 2–8 °C | |
| Virus-like particle | Ward et al. (Nat Med 2021) [44] | 04450004 | 1 | 180 | Intramuscular injections of CoVLP (3.75, 7.5, or 15 μg) at days 0 and 21 | Local, systemic, and unsolicited AEs; Anti-S antibody; SARS-CoV-2-neutralizing antibody | 2–8 °C |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, K.S.-K.; Lee, C.-C.; Liu, K.-J.; Wei, J.C.-C.; Lee, Y.-T.; Wang, L.-T. Safety and Seroconversion of Immunotherapies against SARS-CoV-2 Infection: A Systematic Review and Meta-Analysis of Clinical Trials. Pathogens 2021, 10, 1537. https://doi.org/10.3390/pathogens10121537
Ma KS-K, Lee C-C, Liu K-J, Wei JC-C, Lee Y-T, Wang L-T. Safety and Seroconversion of Immunotherapies against SARS-CoV-2 Infection: A Systematic Review and Meta-Analysis of Clinical Trials. Pathogens. 2021; 10(12):1537. https://doi.org/10.3390/pathogens10121537
Chicago/Turabian StyleMa, Kevin Sheng-Kai, Chien-Chang Lee, Ko-Jiunn Liu, James Cheng-Chung Wei, Yuan-Ti Lee, and Li-Tzu Wang. 2021. "Safety and Seroconversion of Immunotherapies against SARS-CoV-2 Infection: A Systematic Review and Meta-Analysis of Clinical Trials" Pathogens 10, no. 12: 1537. https://doi.org/10.3390/pathogens10121537