The Role of Macrophages in the Host’s Defense against Sporothrix schenckii
Abstract
:1. Introduction
2. Macrophages: A Brief Overview
2.1. Macrophage Subtypes
2.1.1. M1 Classical Monocytes (Inflammatory)
2.1.2. M2 Non-Classical Monocytes (Patrolling, Anti-Inflammatory)
2.2. Macrophages Recruitment
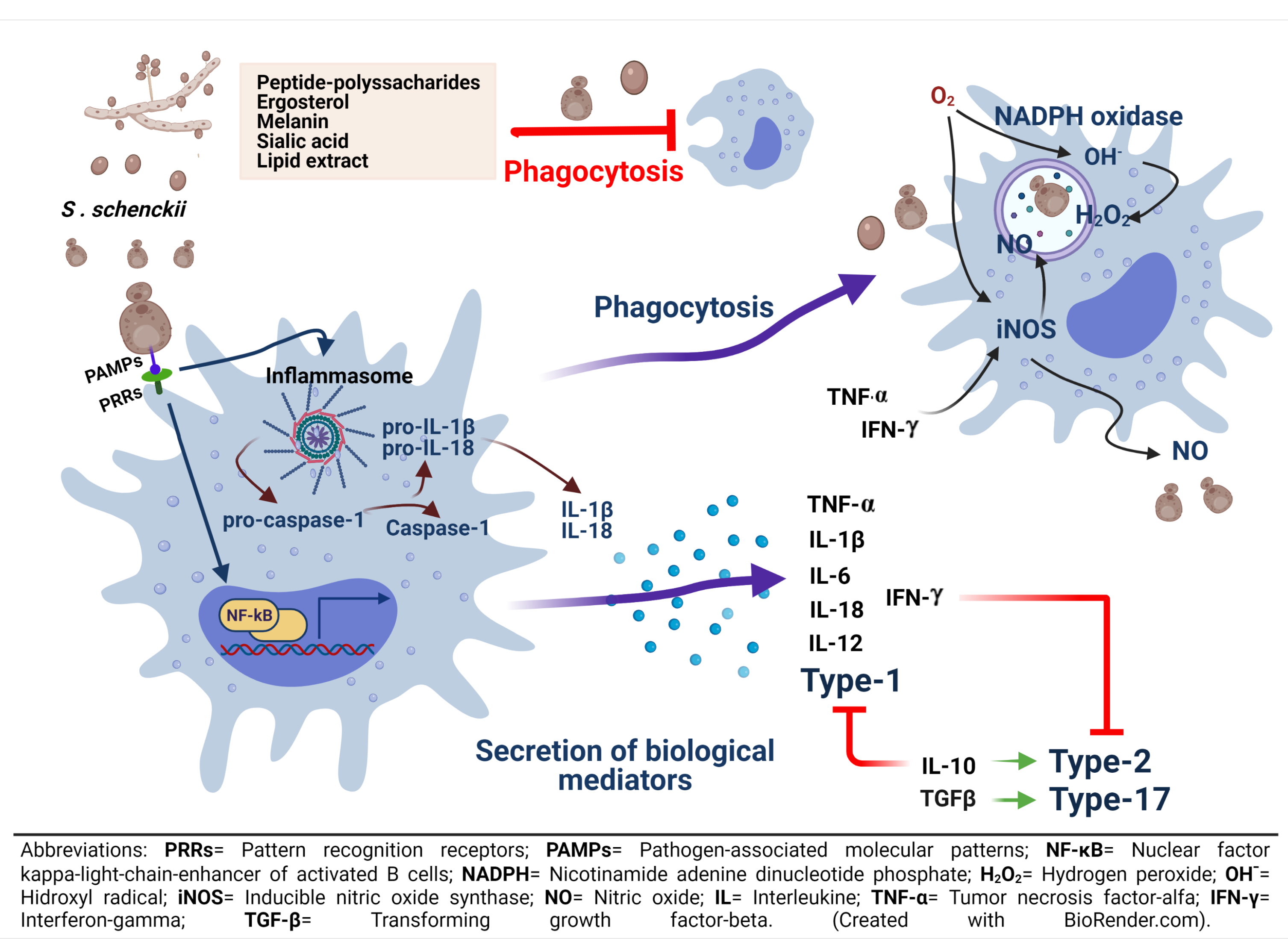
2.3. Phagocytosis
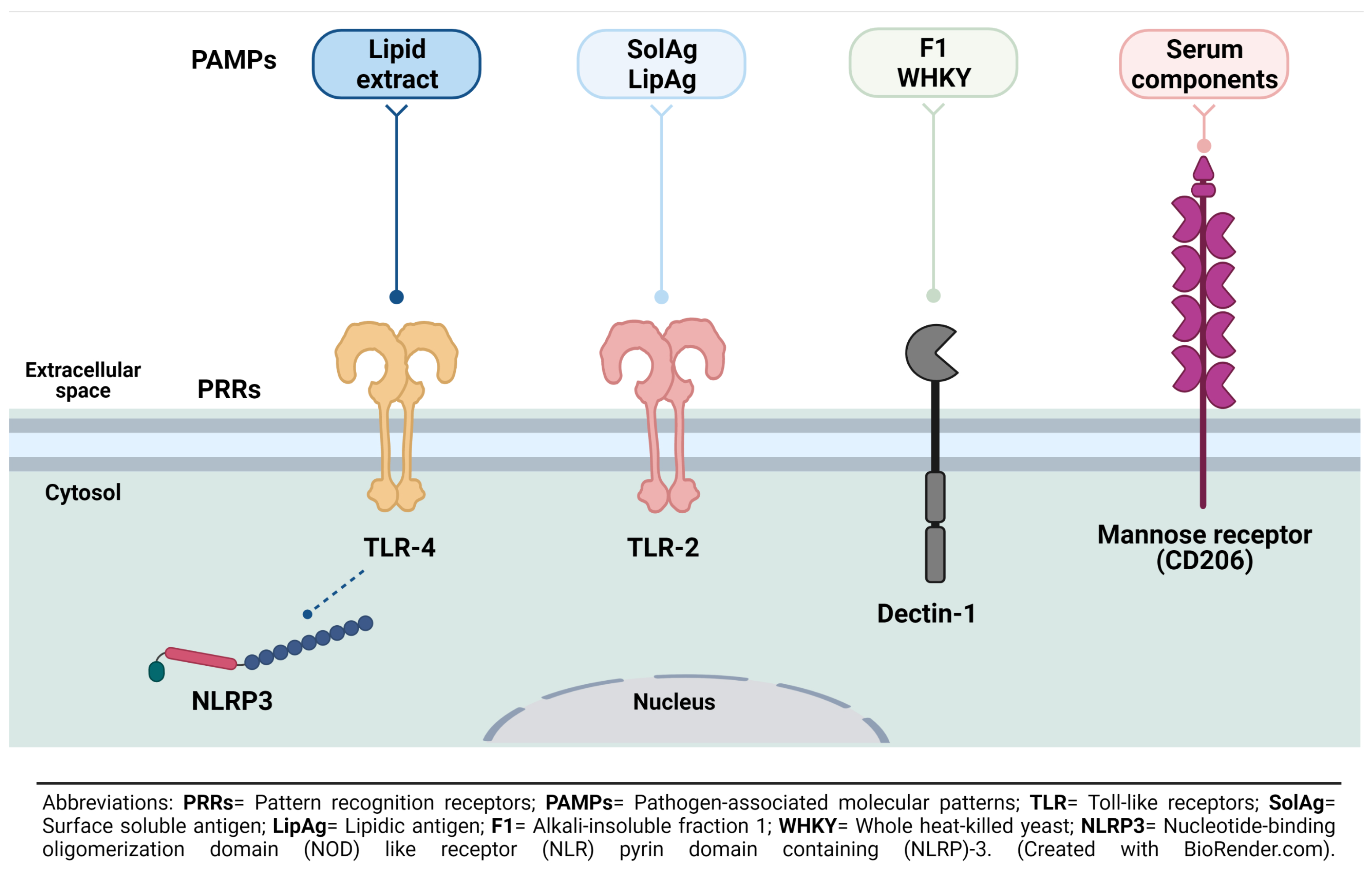
3. S. schenckii Recognition by Macrophages
4. Macrophages and the Immune Responses in Sporotrichosis
5. Macrophage-S. schenckii Interaction: Some New Aspects
A New Population of MR/Arg-1 and iNOS Negative Dermal Resident Macrophages in Mice
6. The Macrophage-S. schenckii Interaction: Virulence/Pathogenicity or Survival Factor?
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chakrabarti, A.; Bonifaz, A.; Gutierrez-Galhardo, M.C.; Mochizuki, T.; Li, S. Global epidemiology of sporotrichosis. Med. Mycol. 2015, 53, 3–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Queiroz-Telles, F.; Buccheri, R.; Benard, G. Sporotrichosis in immunocompromised hosts. J. Fungi 2019, 5, 8. [Google Scholar] [CrossRef] [Green Version]
- Toriello, C.; Brunner-Mendoza, C.; Ruiz-Baca, E.; Duarte-Escalante, E.; Pérez-Mejía, A.; del Rocío Reyes-Montes, M. Sporotrichosis in Mexico. Braz. J. Microbiol. 2020, 52, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Orofino-Costa, R.; de Macedo, P.M.; Rodrigues, A.M.; Bernardes-Engemann, A.R. Sporotrichosis: An update on epidemiology, etiopathogenesis, laboratory and clinical therapeutics. An. Bras. Dermatol. 2017, 92, 606–620. [Google Scholar] [CrossRef] [PubMed]
- Rossow, J.A.; Queiroz-Telles, F.; Caceres, D.H.; Beer, K.D.; Jackson, B.R.; Pereira, J.G.; Ferreira Gremião, I.D.; Pereira, S.A. A one health approach to combatting Sporothrix brasiliensis: Narrative review of an emerging zoonotic fungal pathogen in South America. J. Fungi 2020, 6, 247. [Google Scholar] [CrossRef]
- Barros, M.B.D.L.; de Almeida Paes, R.; Schubach, A.O. Sporothrix schenkii and Sporotrichosis. Clin. Microbiol. Rev. 2011, 24, 633–654. [Google Scholar] [CrossRef] [Green Version]
- Bonifaz, A.; Tirado-Sánchez, A. Cutaneous disseminated and extracutaneous sporotrichosis: Current status of a complex disease. J. Fungi 2017, 3, 6. [Google Scholar] [CrossRef] [Green Version]
- Mahajan, V.K. Sporotrichosis: An overview and therapeutic options. Dermatol. Res. Pract. 2014, 2014, e272376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chai, L.Y.A.; Netea, M.G.; Vonk, A.G.; Kullberg, B.J. Fungal strategies for overcoming host innate immune response. Med. Mycol. 2009, 47, 227–236. [Google Scholar] [CrossRef] [Green Version]
- Aderem, A.; Underhill, D.M. Mechanisms of phagocytosis in macrophages. Annu. Rev. Immunol. 1999, 17, 593–623. [Google Scholar] [CrossRef]
- Lee, W.L.; Harrison, R.E.; Grinstein, S. Phagocytosis by neutrophils. Microbes Infect. 2003, 5, 1299–1306. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.; Moeller, J.; Vogel, V. Mechanobiology of macrophages: How physical factors coregulate macrophage plasticity and phagocytosis. Annu. Rev. Biomed. Eng. 2019, 21, 267–297. [Google Scholar] [CrossRef]
- Hernández-Chávez, M.J.; Pérez-García, L.A.; Niño-Vega, G.A.; Mora-Montes, H.M. Fungal strategies to evade the host immune recognition. J. Fungi 2017, 3, 51. [Google Scholar] [CrossRef] [Green Version]
- Alba-Fierro, C.A.; Pérez-Torres, A.; López-Romero, E.; Cuéllar-Cruz, M.; Ruiz-Baca, E. Cell wall proteins of Sporothrix schenckii as immunoprotective agents. Rev. Iberoam. Micol. 2014, 31, 86–89. [Google Scholar] [CrossRef]
- Lopes-Bezerra, L.M.; Walker, L.A.; Niño-Vega, G.; Mora-Montes, H.M.; Neves, G.W.P.; Villalobos-Duno, H.; Barreto, L.; Garcia, K.; Franco, B.; Martínez-Álvarez, J.A.; et al. Cell walls of the dimorphic fungal pathogens Sporothrix schenckii and Sporothrix brasiliensis exhibit bilaminate structures and sloughing of extensive and intact layers. PLoS Neglected Trop. Dis. 2018, 12, e0006169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, L.; Zhang, J.; Du, W.; Liang, Z.; Li, M.; Wu, R.; Chen, S.; Hu, X.; Huang, H. Chitin-rich heteroglycan from Sporothrix schenckii sensu stricto potentiates fungal clearance in a mouse model of sporotrichosis and promotes macrophages phagocytosis. BMC Microbiol. 2021, 21, 190. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Álvarez, J.A.; Pérez-García, L.A.; Mellado-Mojica, E.; López, M.G.; Martínez-Duncker, I.; Lópes-Bezerra, L.M.; Mora-Montes, H.M. Sporothrix schenckii sensu stricto and Sporothrix brasiliensis are differentially recognized by human peripheral blood mononuclear cells. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef]
- Soudi, S.; Zavaran-Hosseini, A.; Muhammad Hassan, Z.; Soleimani, M.; Jamshidi Adegani, F.; Hashemi, S.M. Comparative Study of The Effect of LPS on The Function of BALB/c and C57BL/6 Peritoneal Macrophages. Cell J. (Yakhteh) 2013, 15, 45–54. [Google Scholar]
- Alba-Fierro, C.A.; Pérez-Torres, A.; Toriello, C.; Romo-Lozano, Y.; López-Romero, E.; Ruiz-Baca, E. Molecular components of the Sporothrix schenckii Complex that induce immune response. Curr. Microbiol. 2016, 73, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Rieger, A.M.; Hall, B.E.; Barreda, D.R. Macrophage activation differentially modulates particle binding, phagocytosis and downstream antimicrobial mechanisms. Dev. Comp. Immunol. 2010, 34, 1144–1159. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S.; Plüddemann, A.; Estrada, F.M. Macrophage heterogeneity in tissues: Phenotypic diversity and functions. Immunol. Rev. 2014, 262, 36–55. [Google Scholar] [CrossRef] [Green Version]
- Gordon, S.; Plüddemann, A. Macrophage clearance of apoptotic cells: A critical assessment. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Birbrair, A.; Frenette, P.S. Niche heterogeneity in the bone marrow. Ann. N. Y. Acad. Sci. 2016, 1370, 82–96. [Google Scholar] [CrossRef] [PubMed]
- Dzierzak, E.; de Pater, E. Chapter One—Regulation of blood stem cell development. In Current Topics in Developmental Biology; Bresnick, E.H., Ed.; Academic Press: Cambridge, MA, USA, 2016; Volume 118, pp. 1–20. [Google Scholar] [CrossRef]
- Nagasawa, T.; Omatsu, Y.; Sugiyama, T. Control of hematopoietic stem cells by the bone marrow stromal niche: The role of reticular cells. Trends Immunol. 2011, 32, 315–320. [Google Scholar] [CrossRef]
- Hoeffel, G.; Ginhoux, F. Ontogeny of tissue-resident macrophages. Front. Immunol. 2015, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geissmann, F.; Jung, S.; Littman, D.R. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 2003, 19, 71–82. [Google Scholar] [CrossRef] [Green Version]
- Ginhoux, F.; Guilliams, M. Tissue-resident macrophage ontogeny and homeostasis. Immunity 2016, 44, 439–449. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, D.; Chow, A.; Noizat, C.; Teo, P.; Beasley, M.; Leboeuf, M.; Becker, C.; See, P.; Price, J.; Lucas, D.; et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity 2013, 38, 792–804. [Google Scholar] [CrossRef] [Green Version]
- Gordon, S.; Plüddemann, A. Tissue macrophages: Heterogeneity and functions. BMC Biol. 2017, 15, 53. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S. Phagocytosis: An immunobiologic process. Immunity 2016, 44, 463–475. [Google Scholar] [CrossRef] [Green Version]
- Martinez, F.O.; Gordon, S. The M1 and M2 paradigm of macrophage activation: Time for reassessment. F1000prime Rep. 2014, 6. [Google Scholar] [CrossRef] [Green Version]
- Bonnardel, J.; Guilliams, M. Developmental control of macrophage function. Curr. Opin. Immunol. 2018, 50, 64–74. [Google Scholar] [CrossRef]
- Sheng, J.; Ruedl, C.; Karjalainen, K. Most tissue-resident macrophages except microglia are derived from fetal hematopoietic stem cells. Immunity 2015, 43, 382–393. [Google Scholar] [CrossRef] [Green Version]
- Ziegler-Heitbrock, L.; Ancuta, P.; Crowe, S.; Dalod, M.; Grau, V.; Hart, D.N.; Leenen, P.J.M.; Liu, Y.J.; MacPherson, G.; Randolph, G.J.; et al. Nomenclature of monocytes and dendritic cells in blood. Blood 2010, 116, e74–e80. [Google Scholar] [CrossRef]
- Ingersoll, M.A.; Spanbroek, R.; Lottaz, C.; Gautier, E.L.; Frankenberger, M.; Hoffmann, R.; Lang, R.; Haniffa, M.; Collin, M.; Tacke, F.; et al. Comparison of gene expression profiles between human and mouse monocyte subsets. Blood 2010, 115, e10–e19. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, L.; Yu, C.; Yang, X.F.; Wang, H. Monocyte and macrophage differentiation: Circulation inflammatory monocyte as biomarker for inflammatory diseases. Biomark. Res. 2014, 2, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mosser, D.M.; Edwards, J.P. Exploring the full spectrum of macrophage activation. Nat. Rev. Immunol. 2008, 8, 958–969. [Google Scholar] [CrossRef] [PubMed]
- Chow, A.; Brown, B.D.; Merad, M. Studying the mononuclear phagocyte system in the molecular age. Nat. Rev. Immunol. 2011, 11, 788–798. [Google Scholar] [CrossRef]
- Reese, T.A.; Liang, H.E.; Tager, A.M.; Luster, A.D.; Van Rooijen, N.; Voehringer, D.; Locksley, R.M. Chitin induces accumulation in tissue of innate immune cells associated with allergy. Nature 2007, 447, 92–96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheldon, K.E.; Shandilya, H.; Kepka-Lenhart, D.; Poljakovic, M.; Ghosh, A.; Morris, S.M. Shaping the murine mMacrophage phenotype: IL-4 and cyclic AMP synergistically activate the arginase I Promoter. J. Immunol. 2013, 191, 2290–2298. [Google Scholar] [CrossRef]
- Lv, R.; Bao, Q.; Li, Y. Regulation of M1-type and M2-type macrophage polarization in RAW264.7 cells by galectin-9. Mol. Med. Rep. 2017, 16, 9111–9119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Kubes, P. A reservoir of mature cavity macrophages that can rapidly invade visceral Organs to affect tissue repair. Cell 2016, 165, 668–678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hesketh, M.; Sahin, K.B.; West, Z.E.; Murray, R.Z. Macrophage phenotypes regulate scar formation and chronic wound healing. Int. J. Mol. Sci. 2017, 18, 1545. [Google Scholar] [CrossRef] [Green Version]
- Guilliams, M.; Mildner, A.; Yona, S. Developmental and functional heterogeneity of monocytes. Immunity 2018, 49, 595–613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, C.; Pamer, E.G. Monocyte recruitment during infection and inflammation. Nat. Rev. Immunol. 2011, 11, 762–774. [Google Scholar] [CrossRef] [Green Version]
- Flannagan, R.S.; Jaumouillé, V.; Grinstein, S. The cell biology of phagocytosis. Annu. Rev. Pathol. Mech. Dis. 2012, 7, 61–98. [Google Scholar] [CrossRef]
- Plüddemann, A.; Mukhopadhyay, S.; Gordon, S. Innate immunity to intracellular pathogens: Macrophage receptors and responses to microbial entry. Immunol. Rev. 2011, 240, 11–24. [Google Scholar] [CrossRef]
- Rosales, C.; Uribe-Querol, E. Phagocytosis: A fundamental process in immunity. Biomed Res. Int. 2017, 2017, e9042851. [Google Scholar] [CrossRef] [Green Version]
- Taylor, P.; Martinez-Pomares, L.; Stacey, M.; Lin, H.H.; Brown, G.; Gordon, S. Macrophage receptors and immune recognition. Annu. Rev. Immunol. 2004, 23, 901–944. [Google Scholar] [CrossRef]
- Licona-Limón, I.; Garay-Canales, C.A.; Muñoz-Paleta, O.; Ortega, E. CD13 mediates phagocytosis in human monocytic cells. J. Leukoc. Biol. 2015, 98, 85–98. [Google Scholar] [CrossRef]
- Kubelkova, K.; Macela, A. Innate immune recognition: An issue more complex than expected. Front. Cell. Infect. Microbiol. 2019, 9. [Google Scholar] [CrossRef] [Green Version]
- Negrini, T.d.C.; Ferreira, L.S.; Alegranci, P.; Arthur, R.A.; Sundfeld, P.P.; Maia, D.C.G.; Spolidorio, L.C.; Carlos, I.Z. Role of TLR-2 and fungal surface antigens on innate immune response against Sporothrix schenckii. Immunol. Investig. 2013, 42, 36–48. [Google Scholar] [CrossRef]
- Barreto-Bergter, E.; Figueiredo, R.T. Fungal glycans and the innate immune recognition. Front. Cell. Infect. Microbiol. 2014, 4. [Google Scholar] [CrossRef] [Green Version]
- Patin, E.C.; Thompson, A.; Orr, S.J. Pattern recognition receptors in fungal immunity. Semin. Cell Dev. Biol. 2019, 89, 24–33. [Google Scholar] [CrossRef]
- Negrini, T.d.C.; Arthur, R.A.; Carlos, I.Z. Recent advances on the role of Toll-like receptors in sporotrichosis—An overview. Microbiol. Res. J. Int. 2016, 1–10. [Google Scholar] [CrossRef]
- Mukherjee, S.; Karmakar, S.; Babu, S.P.S. TLR2 and TLR4 mediated host immune responses in major infectious diseases: A review. Braz. J. Infect. Dis. 2016, 20, 193–204. [Google Scholar] [CrossRef] [Green Version]
- Sassá, M.F.; Saturi, A.E.T.; Souza, L.F.; Ribeiro, L.C.D.A.; Sgarbi, D.B.D.G.; Carlos, I.Z. Response of macrophage Toll-like receptor 4 to a Sporothrix schenckii lipid extract during experimental sporotrichosis. Immunology 2009, 128, 301–309. [Google Scholar] [CrossRef]
- Rossato, L.; Santos, S.S.d.; Ferreira, L.G.; de Almeida, S.R. The importance of Toll-like receptor 4 during experimental Sporothrix brasiliensis infection. Med Mycol. 2019, 57, 489–495. [Google Scholar] [CrossRef]
- Sassá, M.F.; Ferreira, L.S.; de Abreu Ribeiro, L.C.; Carlos, I.Z. Immune response against Sporothrix schenckii in TLR-4-deficient mice. Mycopathologia 2012, 174, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Negrini, T.d.C.; Ferreira, L.S.; Arthur, R.A.; Alegranci, P.; Placeres, M.C.P.; Spolidorio, L.C.; Carlos, I.Z. Influence of TLR-2 in the immune response in the infection induced by fungus Sporothrix schenckii. Immunol. Investig. 2014, 43, 370–390. [Google Scholar] [CrossRef] [PubMed]
- Flores-García, A.; Velarde-Félix, J.S.; Garibaldi-Becerra, V.; Rangel-Villalobos, H.; Torres-Bugarín, O.; Zepeda-Carrillo, E.A.; Ruíz-Bernés, S.; Ochoa-Ramírez, L.A. Recombinant murine IL-12 promotes a protective Th1/cellular response in Mongolian gerbils infected with Sporothrix schenckii. J. Chemother. 2015, 27, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Maia, D.C.G.; Sassá, M.F.; Placeres, M.C.P.; Carlos, I.Z. Influence of Th1/Th2 cytokines and nitric oxide in murine systemic infection induced by Sporothrix schenckii. Mycopathologia 2006, 161, 11–19. [Google Scholar] [CrossRef]
- Carlos, I.Z.; Sassá, M.F.; da Graça Sgarbi, D.B.; Placeres, M.C.P.; Maia, D.C.G. Current research on the immune response to experimental sporotrichosis. Mycopathologia 2009, 168, 1–10. [Google Scholar] [CrossRef]
- Sollberger, G.; Strittmatter, G.E.; Garstkiewicz, M.; Sand, J.; Beer, H.D. Caspase-1: The inflammasome and beyond. Innate Immun. 2014, 20, 115–125. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Wang, H.; Kouadir, M.; Song, H.; Shi, F. Recent advances in the mechanisms of NLRP3 inflammasome activation and its inhibitors. Cell Death Dis. 2019, 10, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Gonçalves, A.C.; Ferreira, L.S.; Manente, F.A.; Faria, C.M.Q.G.D.; Polesi, M.C.; Andrade, C.R.d.; Zamboni, D.S.; Carlos, I.Z. The NLRP3 inflammasome contributes to host protection during Sporothrix schenckii infection. Immunology 2017, 151, 154–166. [Google Scholar] [CrossRef] [Green Version]
- Brown, G.D.; Taylor, P.R.; Reid, D.M.; Willment, J.A.; Williams, D.L.; Martinez-Pomares, L.; Wong, S.Y.; Gordon, S. Dectin-1 is a major β-Glucan receptor on macrophages. J. Exp. Med. 2002, 196, 407–412. [Google Scholar] [CrossRef] [Green Version]
- Plato, A.; Hardison, S.E.; Brown, G.D. Pattern recognition receptors in antifungal immunity. Semin. Immunopathol. 2015, 37, 97–106. [Google Scholar] [CrossRef] [Green Version]
- Jellmayer, J.A.; Ferreira, L.S.; Manente, F.A.; Gonçalves, A.C.; Polesi, M.C.; Batista-Duharte, A.; Carlos, I.Z. Dectin-1 expression by macrophages and related antifungal mechanisms in a murine model of Sporothrix schenckii sensu stricto systemic infection. Microb. Pathog. 2017, 110, 78–84. [Google Scholar] [CrossRef] [Green Version]
- Pavón Romero, L.; Jiménez Martínez, M.d.C.; Garcés Álvarez, M.E. Inmunología Molecular, Celular y Traslacional; Wolters Kluwer: Barcelona, España, 2020. [Google Scholar]
- Koenderman, L.; Buurman, W.; Daha, M.R. The innate immune response. Immunol. Lett. 2014, 162, 95–102. [Google Scholar] [CrossRef]
- Kajiwara, H.; Saito, M.; Ohga, S.; Uenotsuchi, T.; Yoshida, S.i. Impaired host defense against Sporothrix schenckii in mice with chronic granulomatous disease. Infect. Immun. 2004, 72, 5073–5079. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shiraishi, A.; Nakagaki, K.; Arai, T. Experimental sporotrichosis in congenitally athymic (nude) mice. J. Reticuloendothel. Soc. 1979, 26, 333–336. [Google Scholar] [PubMed]
- Dickerson, C.L.; Taylor, R.L.; Drutz, D.J. Susceptibility of congenitally athymic (nude) mice to sporotrichosis. Infect. Immun. 1983, 40, 417–420. [Google Scholar] [CrossRef] [Green Version]
- Shiraishi, A.; Nakagaki, K.; Arai, T. Role of cell-mediated immunity in the resistance to experimental sporotrichosis in mice. Mycopathologia 1992, 120, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Tachibana, T.; Matsuyama, T.; Mitsuyama, M. Involvement of CD4+ T cells and macrophages in acquired protection against infection with Sporothrix schenckii in mice. Med Mycol. 1999, 37, 397–404. [Google Scholar] [CrossRef] [Green Version]
- Carlos, I.Z.; da Graca Sgarbi, D.B.; Angluster, J.; Alviano, C.S.; Silva, C.L. Detection of cellular immunity with the soluble antigen of the fungus Sporothrix schenckii in the systemic form of the disease. Mycopathologia 1992, 117, 139–144. [Google Scholar] [CrossRef]
- Carlos, I.Z.; Sgarbi, D.B.d.g.; Placeres, M.C.P. Host organism defense by a peptide-polysaccharide extracted from the fungus Sporothrix schenckii. Mycopathologia 1998, 144, 9–14. [Google Scholar] [CrossRef]
- Verdan, F.F.; Faleiros, J.C.; Ferreira, L.S.; Monnazzi, L.G.S.; Maia, D.C.G.; Tansine, A.; Placeres, M.C.P.; Carlos, I.Z.; Santos-Junior, R.R. Dendritic cell are able to differentially recognize Sporothrix schenckii antigens and promote Th1/Th17 response in vitro. Immunobiology 2012, 217, 788–794. [Google Scholar] [CrossRef]
- Ferreira, L.S.; Gonçalves, A.C.; Portuondo, D.L.; Maia, D.C.G.; Placeres, M.C.P.; Batista-Duharte, A.; Carlos, I.Z. Optimal clearance of Sporothrix schenckii requires an intact Th17 response in a mouse model of systemic infection. Immunobiology 2015, 220, 985–992. [Google Scholar] [CrossRef]
- Maia, D.C.G.; Gonçalves, A.C.; Ferreira, L.S.; Manente, F.A.; Portuondo, D.L.; Vellosa, J.C.R.; Polesi, M.C.; Batista-Duharte, A.; Carlos, I.Z. Response of cytokines and hydrogen peroxide to Sporothrix schenkii exoantigen in systemic experimental infection. Mycopathologia 2016, 181, 207–215. [Google Scholar] [CrossRef] [Green Version]
- Alegranci, P.; de Abreu Ribeiro, L.C.; Ferreira, L.S.; Negrini, T.d.C.; Maia, D.C.G.; Tansini, A.; Gonçalves, A.C.; Placeres, M.C.P.; Carlos, I.Z. The predominance of alternatively activated macrophages following challenge with cell wall peptide-polysaccharide after prior infection with Sporothrix schenckii. Mycopathologia 2013, 176, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Garcia, B.M.; Bond, A.R.; Barry, A.K.; Steen, A.J.; LeBoit, P.E.; Ashbaugh, C.; Shinkai, K. Disseminated-cutaneous sporotrichosis in an immunocompetent adult. JAAD Case Rep. 2021, 11, 102–104. [Google Scholar] [CrossRef] [PubMed]
- Conceição-Silva, F.; Morgado, F.N. Immunopathogenesis of human Sporotrichosis: What we already know. J. Fungi 2018, 4, 89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandes, K.S.S.; Coelho, A.L.J.; Bezerra, L.M.L.; Barja-Fidalgo, C. Virulence of Sporothrix schenckii conidia and yeast cells, and their susceptibility to nitric oxide. Immunology 2000, 101, 563–569. [Google Scholar] [CrossRef]
- Fernandes, K.S.S.; Neto, E.H.; Brito, M.M.S.; Silva, J.S.; Cunha, F.Q.; Barja-Fidalgo, C. Detrimental role of endogenous nitric oxide in host defence against Sporothrix schenckii. Immunology 2008, 123, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Morgado, F.N.; Schubach, A.O.; Barros, M.B.L.; Conceição-Silva, F. The in situ inflammatory profile of lymphocutaneous and fixed forms of human sporotrichosis. Med Mycol. 2011, 49, 612–620. [Google Scholar] [CrossRef] [Green Version]
- Brito, M.M.S.; Conceição-Silva, F.; Morgado, F.N.; Raibolt, P.S.; Schubach, A.; Schubach, T.P.; Schäffer, G.M.V.; Borba, C.M. Comparison of virulence of different Sporothrix schnckii clinical isolates using experimental murine model. Med Mycol. 2007, 45, 721–729. [Google Scholar] [CrossRef] [Green Version]
- Uenotsuchi, T.; Takeuchi, S.; Matsuda, T.; Urabe, K.; Koga, T.; Uchi, H.; Nakahara, T.; Fukagawa, S.; Kawasaki, M.; Kajiwara, H.; et al. Differential induction of Th1-prone immunity by human dendritic cells activated with Sporothrix schenkii of cutaneous and visceral origins to determine their different virulence. Int. Immunol. 2006, 18, 1637–1646. [Google Scholar] [CrossRef] [Green Version]
- Morgado, F.N.; de Carvalho, L.M.V.; Leite-Silva, J.; Seba, A.J.; Pimentel, M.I.F.; Fagundes, A.; Madeira, M.F.; Lyra, M.R.; Oliveira, M.M.; Schubach, A.O.; et al. Unbalanced inflammatory reaction could increase tissue destruction and worsen skin infectious diseases—A comparative study of leishmaniasis and sporotrichosis. Sci. Rep. 2018, 8, 2898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wynn, T.A.; Barron, L. Macrophages: Master regulators of inflammation and fibrosis. Semin. Liver Dis. 2010, 30, 245–257. [Google Scholar] [CrossRef] [Green Version]
- Quintella, L.P.; Passos, S.R.L.; Vale, A.C.F.D.; Galhardo, M.C.G.; Barros, M.B.D.L.; Cuzzi, T.; Reis, R.D.S.; Carvalho, M.H.G.F.D.; Zappa, M.B.; Schubach, A.D.O. Histopathology of cutaneous sporotrichosis in Rio de Janeiro: A series of 119 consecutive cases. J. Cutan. Pathol. 2011, 38, 25–32. [Google Scholar] [CrossRef]
- Guzman-Beltran, S.; Perez-Torres, A.; Coronel-Cruz, C.; Torres-Guerrero, H. Phagocytic receptors on macrophages distinguish between different Sporothrix schenckii morphotypes. Microbes Infect. 2012, 14, 1093–1101. [Google Scholar] [CrossRef]
- Guzman Beltrán, S.; Sanchez Morales, J.; González Canto, A.; Escalona Montaño, A.; Torres Guerrero, H. Human serum proteins bind to Sporothrix schenckii conidia with differential effects on phagocytosis. Braz. J. Microbiol. 2021, 52, 33–39. [Google Scholar] [CrossRef]
- Hind, C.R.K.; Collins, P.M.; Baltz, M.L.; Pepys, M.B. Human serum amyloid P component, a circulating lectin with specificity for the cyclic 4,6-pyruvate acetal of galactose. Interactions with various bacteria. Biochem. J. 1985, 225, 107–111. [Google Scholar] [CrossRef] [Green Version]
- Yuste, J.; Botto, M.; Bottoms, S.E.; Brown, J.S. Serum amyloid P aids complement-mediated immunity to Streptococcus pneumoniae. PLoS Pathog. 2007, 3, e120. [Google Scholar] [CrossRef] [Green Version]
- Noursadeghi, M.; Bickerstaff, M.C.M.; Gallimore, J.R.; Herbert, J.; Cohen, J.; Pepys, M.B. Role of serum amyloid P component in bacterial infection: Protection of the host or protection of the pathogen. Proc. Natl. Acad. Sci. USA 2000, 97, 14584–14589. [Google Scholar] [CrossRef] [Green Version]
- Haas, C.J.C.d.; Leeuwen, E.M.M.V.; Bommel, T.V.; Verhoef, J.; Kessel, K.P.M.V.; Strijp, J.A.G.V. Serum amyloid P component bound to Gram-negative bacteria prevents lipopolysaccharide-mediated classical pathway complement activation. Infect. Immun. 2000, 68, 1753–1759. [Google Scholar] [CrossRef] [Green Version]
- Klotz, S.A.; Sobonya, R.E.; Lipke, P.N.; Garcia-Sherman, M.C. Serum amyloid P component and systemic fungal infection: Does it protect the host or is it a trojan Horse? Open Forum Infect. Dis. 2016, 3. [Google Scholar] [CrossRef] [Green Version]
- Gilchrist, K.B.; Garcia, M.C.; Sobonya, R.; Lipke, P.N.; Klotz, S.A. New features of invasive candidiasis in humans: Amyloid formation by fungi and deposition of serum amyloid P component by the host. J. Infect. Dis. 2012, 206, 1473–1478. [Google Scholar] [CrossRef]
- Garcia-Sherman, M.C.; Lundberg, T.; Sobonya, R.E.; Lipke, P.N.; Klotz, S.A. A unique biofilm in human deep mycoses: Fungal amyloid is bound by host serum amyloid P component. Npj Biofilms Microbiomes 2015, 1, 1–4. [Google Scholar] [CrossRef] [Green Version]
- Behrens, N.E.; Lipke, P.N.; Pilling, D.; Gomer, R.H.; Klotz, S.A. Serum amyloid P component binds fungal surface amyloid and decreases human macrophage phagocytosis and secretion of inflammatory cytokines. mBio 2019, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bergin, D.A.; Hurley, K.; McElvaney, N.G.; Reeves, E.P. Alpha-1 antitrypsin: A potent anti-inflammatory and potential novel therapeutic agent. Arch. Immunol. Ther. Exp. 2012, 60, 81–97. [Google Scholar] [CrossRef] [PubMed]
- Bergin, D.A.; Reeves, E.P.; Meleady, P.; Henry, M.; McElvaney, O.J.; Carroll, T.P.; Condron, C.; Chotirmall, S.H.; Clynes, M.; O’Neill, S.J.; et al. α-1 Antitrypsin regulates human neutrophil chemotaxis induced by soluble immune complexes and IL-8. J. Clin. Investig. 2010, 120, 4236–4250. [Google Scholar] [CrossRef] [Green Version]
- Agné, A.; Richter, K.; Padberg, W.; Janciauskiene, S.; Grau, V. Commercial α1-antitrypsin preparations markedly differ in their potential to inhibit the ATP-induced release of monocytic interleukin-1β. Pulm. Pharmacol. Ther. 2021, 68, 102020. [Google Scholar] [CrossRef]
- Zhou, Z.; Xu, M.J.; Gao, B. Hepatocytes: A key cell type for innate immunity. Cell. Mol. Immunol. 2016, 13, 301–315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valente, M.D.F.; Diogo, A.B.; Merlo, V.F.C.; Pegas, J.R.P.; Valente, M.d.F.; Diogo, A.B.; Merlo, V.F.C.; Pegas, J.R.P. Disseminated cutaneous sporotrichosis: Unusual presentation in an alcoholic patient. Rev. Inst. Med. Trop. SãO Paulo 2020, 62. [Google Scholar] [CrossRef]
- Benvegnú, A.M.; Stramari, J.; Dallazem, L.N.D.; Chemello, R.M.L.; Beber, A.A.C.; Benvegnú, A.M.; Stramari, J.; Dallazem, L.N.D.; Chemello, R.M.L.; Beber, A.A.C. Disseminated cutaneous sporotrichosis in patient with alcoholism. Rev. Soc. Bras. Med. Trop. 2017, 50, 871–873. [Google Scholar] [CrossRef] [Green Version]
- Nassif, P.W.; Granado, I.R.M.d.O.; Ferraz, J.S.; Souza, R.; Nassif, A.E. Esporotricose cutânea de apresentação atípica em paciente etilista. Dermatol. Online J. 2012, 18, 12. [Google Scholar] [CrossRef]
- Yang, C.Y.; Chen, J.B.; Tsai, T.F.; Tsai, Y.C.; Tsai, C.Y.; Liang, P.H.; Hsu, T.L.; Wu, C.Y.; Netea, M.G.; Wong, C.H.; et al. CLEC4F is an iducible C-type lectin in F4/80-positive cells and is involved in alpha-galactosylceramide presentation in liver. PLoS ONE 2013, 8, e65070. [Google Scholar] [CrossRef] [Green Version]
- Martinez-Pomares, L. The mannose receptor. J. Leukoc. Biol. 2012, 92, 1177–1186. [Google Scholar] [CrossRef]
- Józefowski, S.; Yang, Z.; Marcinkiewicz, J.; Kobzik, L. Scavenger receptors and β-glucan receptors participate in the recognition of yeasts by murine macrophages. Inflamm. Res. 2012, 61, 113–126. [Google Scholar] [CrossRef] [Green Version]
- Dantzer, R.; O’Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–56. [Google Scholar] [CrossRef] [Green Version]
- Øverland, G.; Stuestøl, J.F.; Dahle, M.K.; Myhre, A.E.; Netea, M.G.; Verweij, P.; Yndestad, A.; Aukrust, P.; Kullberg, B.J.; Warris, A.; et al. Cytokine responses to fungal pathogens in Kupffer cells are Toll-like receptor 4 independent and mediated by tyrosine kinases. Scand. J. Immunol. 2005, 62, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Okabe, Y.; Medzhitov, R. Tissue biology perspective on macrophages. Nat. Immunol. 2016, 17, 9–17. [Google Scholar] [CrossRef]
- Rosas, M.; Davies, L.C.; Giles, P.J.; Liao, C.T.; Kharfan, B.; Stone, T.C.; O’Donnell, V.B.; Fraser, D.J.; Jones, S.A.; Taylor, P.R. The transcription factor Gata6 links tissue macrophage phenotype and proliferative renewal. Science 2014, 344, 645–648. [Google Scholar] [CrossRef] [Green Version]
- A-Gonzalez, N.; Quintana, J.A.; García-Silva, S.; Mazariegos, M.; González de la Aleja, A.; Nicolás-Ávila, J.A.; Walter, W.; Adrover, J.M.; Crainiciuc, G.; Kuchroo, V.K.; et al. Phagocytosis imprints heterogeneity in tissue-resident macrophages. J. Exp. Med. 2017, 214, 1281–1296. [Google Scholar] [CrossRef] [Green Version]
- Gautier, E.L.; Shay, T.; Miller, J.; Greter, M.; Jakubzick, C.; Ivanov, S.; Helft, J.; Chow, A.; Elpek, K.G.; Gordonov, S.; et al. Gene-expression profiles and transcriptional regulatory pathways that underlie the identity and diversity of mouse tissue macrophages. Nat. Immunol. 2012, 13, 1118–1128. [Google Scholar] [CrossRef] [Green Version]
- Lavin, Y.; Winter, D.; Blecher-Gonen, R.; David, E.; Keren-Shaul, H.; Merad, M.; Jung, S.; Amit, I. Tissue-resident macrophage enhancer landscapes are shaped by the local microenvironment. Cell 2014, 159, 1312–1326. [Google Scholar] [CrossRef] [Green Version]
- Rückerl, D.; Campbell, S.M.; Duncan, S.; Sutherland, T.E.; Jenkins, S.J.; Hewitson, J.P.; Barr, T.A.; Jackson-Jones, L.H.; Maizels, R.M.; Allen, J.E. Macrophage origin limits functional plasticity in helminth-bacterial co-infection. PLoS Pathog. 2017, 13, e1006233. [Google Scholar] [CrossRef]
- Lee, S.H.; Charmoy, M.; Romano, A.; Paun, A.; Chaves, M.M.; Cope, F.O.; Ralph, D.A.; Sacks, D.L. Mannose receptor high, M2 dermal macrophages mediate nonhealing Leishmania major infection in a Th1 immune environment. J. Exp. Med. 2017, 215, 357–375. [Google Scholar] [CrossRef]
- Lee, S.H.; Chaves, M.M.; Kamenyeva, O.; Gazzinelli-Guimaraes, P.H.; Kang, B.; Pessenda, G.; Passelli, K.; Tacchini-Cottier, F.; Kabat, J.; Jacobsen, E.A.; et al. M2-like, dermal macrophages are maintained via IL-4/CCL24–mediated cooperative interaction with eosinophils in cutaneous leishmaniasis. Sci. Immunol. 2020, 5. [Google Scholar] [CrossRef] [PubMed]
- Murray, P.; Allen, J.; Biswas, S.; Fisher, E.; Gilroy, D.; Goerdt, S.; Gordon, S.; Hamilton, J.; Ivashkiv, L.; Lawrence, T.; et al. Macrophage activation and polarization: Nomenclature and experimental guidelines. Immunity 2014, 41, 14–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jenkins, S.J.; Ruckerl, D.; Cook, P.C.; Jones, L.H.; Finkelman, F.D.; Rooijen, N.V.; MacDonald, A.S.; Allen, J.E. Local macrophage proliferation, rather than recruitment from the blood, is a signature of TH2 inflammation. Science 2011, 332, 1284–1288. [Google Scholar] [CrossRef] [Green Version]
- Schleicher, U.; Paduch, K.; Debus, A.; Obermeyer, S.; König, T.; Kling, J.C.; Ribechini, E.; Dudziak, D.; Mougiakakos, D.; Murray, P.J.; et al. TNF-mediated restriction of arginase 1 expression in myeloid cells triggers type 2 NO synthase activity at the site of infection. Cell Rep. 2016, 15, 1062–1075. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Casadevall, A.; Pirofski, L.a. Host-pathogen interactions: Redefining the basic concepts of virulence and pathogenicity. Infect. Immun. 1999, 67, 3703–3713. [Google Scholar] [CrossRef] [Green Version]
- Casadevall, A.; Pirofski, L. Host-pathogen interactions: The attributes of virulence. J. Infect. Dis. 2001, 184, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Casadevall, A.; Pirofski, L.A. The damage-response framework of microbial pathogenesis. Nat. Rev. Microbiol. 2003, 1, 17–24. [Google Scholar] [CrossRef]
- Nemecek, J.C.; Wüthrich, M.; Klein, B.S. Global control of dimorphism and virulence in fungi. Science 2006, 312, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, G.; Klein, B.S. Insights into fungal morphogenesis and immune evasion. Microbe 2008, 3, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Sgarbi, D.B.; da Silva, A.J.R.; Carlos, I.Z.; Silva, C.L.; Angluster, J.; Alviano, C.S. Isolation of ergosterol peroxide and its reversion to ergosterol in the pathogenic fungus Sporothrix schenckii. Mycopathologia 1997, 139, 9. [Google Scholar] [CrossRef]
- Brilhante, R.S.; Pereira, V.S.; Oliveira, J.S.; Rodrigues, A.M.; de Camargo, Z.P.; Pereira-Neto, W.A.; Nascimento, N.R.; Castelo-Branco, D.S.; Cordeiro, R.A.; Sidrim, J.J.; et al. Terpinen-4-ol inhibits the growth of Sporothrix schenckii complex and exhibits synergism with antifungal agents. Future Microbiol. 2019, 14, 1221–1233. [Google Scholar] [CrossRef]
- Borba-Santos, L.P.; Visbal, G.; Gagini, T.; Rodrigues, A.M.; de Camargo, Z.P.; Lopes-Bezerra, L.M.; Ishida, K.; de Souza, W.; Rozental, S. Δ24-sterol Methyltransferase plays an important role in the growth and development of Sporothrix schenckii and Sporothrix brasiliensis. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [Green Version]
- Romero-Martinez, R.; Wheeler, M.; Guerrero-Plata, A.; Rico, G.; Torres-Guerrero, H. Biosynthesis and functions of melanin in Sporothrix schenckii. Infect. Immun. 2000, 68, 3696–3703. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Almeida-Paes, R.; Figueiredo-Carvalho, M.H.G.; Brito-Santos, F.; Almeida-Silva, F.; Oliveira, M.M.E.; Zancopé-Oliveira, R.M. Melanins protect Sporothrix brasiliensis and Sporothrix schenckii from the antifungal effects of terbinafine. PLoS ONE 2016, 11, e0152796. [Google Scholar] [CrossRef] [PubMed]
- Mario, D.A.N.; Santos, R.C.V.; Denardi, L.B.; Vaucher, R.d.A.; Santurio, J.M.; Alves, S.H. Interference of melanin in the susceptibility profile of Sporothrix species to amphotericin B. Rev. Iberoam. Micol. 2016, 33, 21–25. [Google Scholar] [CrossRef]
- Madrid, I.M.; Xavier, M.O.; Mattei, A.S.; Fernandes, C.G.; Guim, T.N.; Santin, R.; Schuch, L.F.D.; Nobre, M.D.O.; Araújo Meireles, M.C. Role of melanin in the pathogenesis of cutaneous sporotrichosis. Microbes Infect. 2010, 12, 162–165. [Google Scholar] [CrossRef] [PubMed]
- Oda, L.M.; Kubelka, C.F.; Alviano, C.S.; Travassos, L.R. Ingestion of yeast forms of Sporothrix schenckii by mouse peritoneal macrophages. Infect. Immun. 1983, 39, 497–504. [Google Scholar] [CrossRef] [Green Version]
- Alviano, C.S.; Pereira, M.E.A.; Souza, W.; Oda, L.M.; Travassos, L.R. Sialic acids are surface components of Sporothrix schenckii yeast forms. FEMS Microbiol. Lett. 1982, 15, 223–227. [Google Scholar] [CrossRef]
- Carlos, I.Z.; Sgarbi, D.B.G.; Santos, G.C.; Placeres, M.C.P. Sporothrix schenckii lipid inhibits macrophage phagocytosis: Involvement of nitric oxide and tumour necrosis factor-α. Scand. J. Immunol. 2003, 57, 214–220. [Google Scholar] [CrossRef] [Green Version]
- Gonçalves, A.C.; Maia, D.C.G.; Ferreira, L.S.; Monnazzi, L.G.S.; Alegranci, P.; Placeres, M.C.P.; Batista-Duharte, A.; Carlos, I.Z. Involvement of major components from Sporothrix schenckii cell wall in the caspase-1 activation, nitric oxide and cytokines production during experimental sporotrichosis. Mycopathologia 2015, 179, 21–30. [Google Scholar] [CrossRef]
- Garrison, R.G.; Arnold, W.N. Cytochemical localization of acid phosphatases in the dimorphic fungus Sporothrix schenckii. Curr. Microbiol. 1983, 9, 253–257. [Google Scholar] [CrossRef]
- Hogan, L.H.; Klein, B.S.; Levitz, S.M. Virulence factors of medically important fungi. Clin. Microbiol. Rev. 1996, 9, 469–488. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz-Baca, E.; Pérez-Torres, A.; Romo-Lozano, Y.; Cervantes-García, D.; Alba-Fierro, C.A.; Ventura-Juárez, J.; Torriello, C. The Role of Macrophages in the Host’s Defense against Sporothrix schenckii. Pathogens 2021, 10, 905. https://doi.org/10.3390/pathogens10070905
Ruiz-Baca E, Pérez-Torres A, Romo-Lozano Y, Cervantes-García D, Alba-Fierro CA, Ventura-Juárez J, Torriello C. The Role of Macrophages in the Host’s Defense against Sporothrix schenckii. Pathogens. 2021; 10(7):905. https://doi.org/10.3390/pathogens10070905
Chicago/Turabian StyleRuiz-Baca, Estela, Armando Pérez-Torres, Yolanda Romo-Lozano, Daniel Cervantes-García, Carlos A. Alba-Fierro, Javier Ventura-Juárez, and Conchita Torriello. 2021. "The Role of Macrophages in the Host’s Defense against Sporothrix schenckii" Pathogens 10, no. 7: 905. https://doi.org/10.3390/pathogens10070905
APA StyleRuiz-Baca, E., Pérez-Torres, A., Romo-Lozano, Y., Cervantes-García, D., Alba-Fierro, C. A., Ventura-Juárez, J., & Torriello, C. (2021). The Role of Macrophages in the Host’s Defense against Sporothrix schenckii. Pathogens, 10(7), 905. https://doi.org/10.3390/pathogens10070905







