The Healthy and Diseased Equine Endometrium: A Review of Morphological Features and Molecular Analyses
Abstract
Simple Summary
Abstract
1. Introduction
2. Physiological Characteristics of the Equine Endometrium
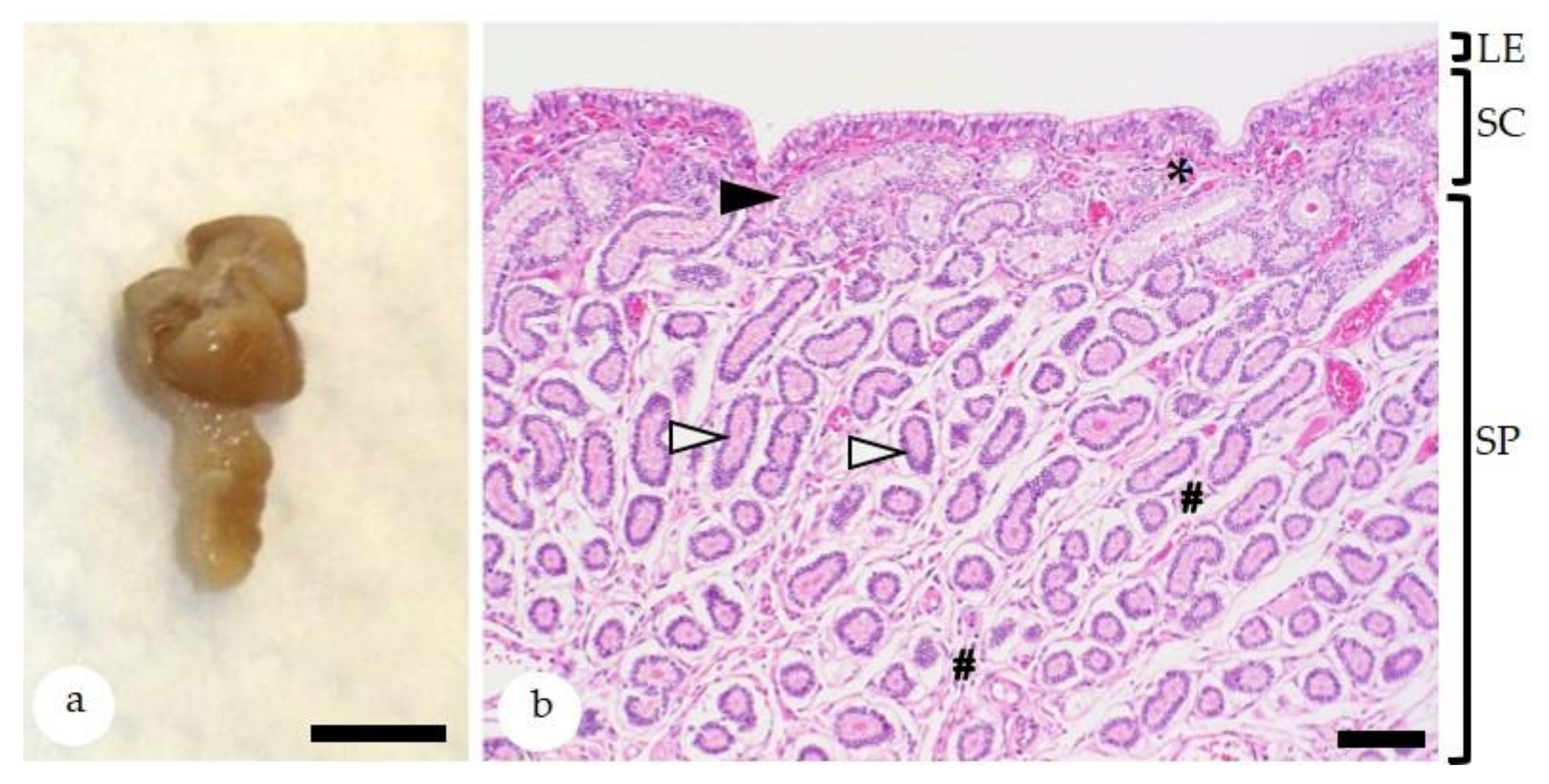
2.1. Endometrial Histology and Influencing Factors
2.2. Expression of Estrogen and Progesterone Receptors during the Estrous Cycle
2.3. Expression of Glandular Secretory Proteins
2.4. Expression of Cytoskeletal Proteins
2.5. Resident Immune Cells
3. Innate Immune Defenses
4. The Endometrial Biopsy as Diagnostic Tool
5. Endometrial Diseases
5.1. Equine Endometrosis
5.1.1. Diagnostic Features of Endometrosis
5.1.2. Molecular Characteristics of Endometrosis
5.1.3. The Pathogenesis of Endometrosis—Still to be Revealed
5.2. Endometritis—Subclassification and Disease Susceptibility
5.2.1. Suppurative Endometritis
5.2.2. Nonsuppurative Endometritis
5.2.3. Endometritis Eosinophilica
5.2.4. Granulomatous Endometritis
5.2.5. Cytology as Diagnostic Tool for Endometritis
5.2.6. The Susceptible Mare
5.3. Endometrial Maldifferentiation During the Breeding Season
5.3.1. Glandular Inactivity During the Breeding Season
5.3.2. Irregular Glandular Differentiation During the Breeding Season
5.3.3. Unequal Glandular Differentiation During the Breeding Season
5.4. Angiopathies
5.4.1. Angiosis—Diagnostic Features
5.4.2. Angiosis—Influence of Age and Parity
5.5. Endometrial Cancer: A Rare Disease in Mares
6. Discussion
6.1. Different Equine Endometrial Diseases: A Common Cause for Subfertility?
6.2. Molecular Features of Equine Endometrial Pathophysiology—an Important Research Aspect
6.2.1. Biomarkers for Equine Endometrial Health and Disease
6.2.2. Molecular Research to Improve Treatment
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kenney, R.M.; Doig, P.A. Equine endometrial biopsy. In Current Therapy in Theriogenology, 2nd ed.; Morrow, D.A., Ed.; WB Saunders: Philadelphia, PA, USA, 1986; pp. 723–729. [Google Scholar]
- Schoon, H.-A.; Schoon, D.; Klug, E. Uterusbiopsien als Hilfsmittel für Diagnose und Prognose von Fertilitätsstörungen der Stute. Pferdeheilkunde 1992, 8, 355–362. [Google Scholar] [CrossRef]
- Schoon, H.-A.; Schoon, D.; Klug, E. Die Endometriumbiopsie bei der Stute im klinisch-gynäkologischen Kontext. Pferdeheilkunde 1997, 13, 453–464. [Google Scholar] [CrossRef]
- Schoon, D.; Schoon, H.-A.; Klug, E. Angioses in the equine endometrium—Pathogenesis and clinical correlations. Pferdeheilkunde 1999, 15, 541–546. [Google Scholar] [CrossRef]
- Schoon, H.-A.; Schoon, D.; Wiegandt, I.; Bartmann, C.-P.; Aupperle, H. “Endometrial maldifferentiation”—A clinically significant diagnosis in equine reproduction? Pferdeheilkunde 1999, 15, 555–559. [Google Scholar] [CrossRef][Green Version]
- Schoon, H.-A.; Wiegandt, I.; Schoon, D.; Aupperle, H.; Bartmann, C.-P. Functional disturbances in the endometrium of barren mares: A histological and immunohistological study. J. Reprod. Fertil. Suppl. 2000, 56, 381–391. [Google Scholar]
- Schoon, H.-A.; Schoon, D. The category I mare (Kenney and Doig 1986): Expected foaling rate 80–90%—Fact or fiction? Pferdeheilkunde 2003, 19, 698–701. [Google Scholar] [CrossRef]
- Buczkowska, J.; Kozdrowski, R.; Nowak, M.; Ra, A.; Mrowiec, J. Endometrosis—Significance for horse reproduction, pathogenesis, diagnosis, and proposed therapeutic methods. Pol. J. Vet. Sci. 2014, 17, 547–554. [Google Scholar] [CrossRef]
- Killisch, R.; Böttcher, D.; Theuß, T.; Edzards, H.; Martinsson, G.; Einspanier, A.; Gottschalk, J.; Schoon, H.-A. Seasonal or pathological findings? Morphofunctional characteristics of the equine endometrium during the autumn and spring transition. Reprod. Dom. Anim. 2017, 52, 1011–1018. [Google Scholar] [CrossRef]
- Katila, T. Onset and duration of uterine inflammatory response of mares after insemination with fresh semen. Biol. Reprod. 1995, 1, 515–517. [Google Scholar] [CrossRef]
- Troedsson, M.H.T. Uterine clearance and resistance to persistent endometritis in the mare. Theriogenology 1999, 52, 461–471. [Google Scholar] [CrossRef]
- LeBlanc, M.M.; Causey, R.C. Clinical and subclinical endometritis in the mare: Both threats to fertility. Reprod. Dom. Anim. 2009, 44, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Kilgenstein, H.J.; Schöniger, S.; Schoon, D.; Schoon, H.-A. Microscopic examination of endometrial biopsies of retired sports mares: An explanation for the clinically observed subfertility? Res. Vet. Sci. 2015, 99, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Mambelli, L.I.; Mattos, R.C.; Winter, G.H.Z.; Madeiro, D.S.; Morais, B.P.; Malschitzky, E.; Miglino, M.A.; Kerkis, A.; Kerkis, I. Changes in expression pattern of selected endometrial proteins following mesenchymal stem cells infusion in mares with endometrosis. PLoS ONE 2014, 9, e97889. [Google Scholar] [CrossRef] [PubMed]
- Aupperle, H.; Steiger, K.; Reischauer, A.; Schoon, H.-A. Ultrastructural and immunohistochemical characterization of the physiological and pathological inactivity of the equine endometrium. Pferdeheilkunde 2003, 19, 629–632. [Google Scholar] [CrossRef]
- Aupperle, H.; Schoon, D.; Schoon, H.-A. Physiological and pathological expression of intermediate filaments in the equine endometrium. Res. Vet. Sci. 2004, 76, 249–255. [Google Scholar] [CrossRef]
- Schöniger, S.; Gräfe, H.; Schoon, H.-A. Beta-defensin is a component of the endometrial immune defence in the mare. Pferdeheilkunde 2013, 29, 335–346. [Google Scholar] [CrossRef]
- Huth, N.; Schöniger, S.; Sieme, H.; Bartmann, C.-P.; Schoon, H.-A. Lymphocyte subpopulations in the equine endometrium: A methodical approach with diagnostic implications. Pferdeheilkunde 2014, 30, 4–14. [Google Scholar] [CrossRef]
- Rebordão, M.R.; Galvão, A.; Szóstek, A.; Amaral, A.; Mateus, L.; Skarzynski, D.J.; Ferreira-Dias, G. Physiopathologic mechanisms involved in mare endometrosis. Reprod. Dom. Anim. 2014, 49, 82–87. [Google Scholar] [CrossRef]
- Klose, K.; Schoon, H.-A. Periglandular inflammatory cells in the endometrium of the mare—A physiological defence mechanisms which impacts on the development of endometrosis. Pferdeheilkunde 2016, 32, 15–23. [Google Scholar] [CrossRef]
- Schöniger, S.; Böttcher, D.; Theuß, T.; Gräfe, H.; Schoon, H.-A. New insights into the innate immune defences of the equine endometrium: In situ and in vitro expression pattern of beta-defensin. Pferdeheilkunde 2016, 32, 4–14. [Google Scholar] [CrossRef][Green Version]
- Rudolph, N.; Schoon, H.-A.; Schöniger, S. Immunohistochemical characterization of immune cells in fixed equine endometrial tissue: A diagnostic relevant method. Pferdeheilkunde 2017, 33, 524–537. [Google Scholar] [CrossRef]
- Schöniger, S.; Gräfe, H.; Schoon, H.-A. Expression of Toll-like receptors 2, 4 and 6 in different cell populations of the equine endometrium. Vet. Immunol. Immunopathol. 2017, 185, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Schöniger, S.; Böttcher, D.; Theuß, T.; Gräfe, H.; Schoon, H.-A. Expression of Toll-like receptors 2, 4 and 6 in equine endometrial cells: A comparative in situ and in vitro study. Res. Vet. Sci. 2017, 112, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Schöniger, S.; Gräfe, H.; Richter, F.; Schoon, H.-A. Expression of indoleamine 2,3-dioxygenase 1 as transcript and protein in the healthy and diseased equine endometrium. Res. Vet. Sci. 2018, 118, 278–287. [Google Scholar] [CrossRef]
- Schöniger, S.; Gräfe, H.; Schoon, H.-A. Innate immunity mechanisms of the equine endometrium—Benefit or harm? Pferdeheilkunde 2018, 34, 5–11. [Google Scholar] [CrossRef]
- Bischofberger, L.; Szewczyk, K.; Schoon, H.-A. Unequal glandular differentiation of the equine endometrium—A separate endometrial alteration? Pferdeheilkunde 2019, 35, 304–315. [Google Scholar] [CrossRef]
- Minkwitz, C.; Schoon, H.-A.; Zhang, Q.; Schöniger, S. Plasticity of endometrial epithelial and stromal cells—A new approach towards the pathogenesis of equine endometrosis. Reprod. Dom. Anim. 2019, 54, 835–845. [Google Scholar] [CrossRef]
- Moore, R.E.; Kirwan, J.; Doherty, M.K.; Whitfield, P.D. Biomarker discovery in animal health and disease: The application of post-genomic technologies. Biomark. Insights 2007, 2, 185–196. [Google Scholar] [CrossRef]
- Califf, R.M. Biomarker definitions and their applications. Exp. Biol. Med. 2018, 243, 213–221. [Google Scholar] [CrossRef]
- Taylor, C.R. Introduction to predictive biomarkers: Definitions and characteristics. In Predictive Biomarkers in Oncology; Badve, S., Kumar, G.L., Eds.; Springer Nature: Cham, Switzerland, 2019; pp. 3–18. [Google Scholar]
- Ricketts, S.W. The endometrial technique and clinical application of endometrial biopsy in the mare. Equine Vet. J. 1975, 7, 102–108. [Google Scholar] [CrossRef]
- Kenney, M. Cyclic and pathologic changes of the mare endometrium as detected by biopsy, with a note on early embryonic death. J. Am. Vet. Med. Assoc. 1978, 172, 241–262. [Google Scholar]
- Van Camp, S.D. Endometrial biopsy of the mare. A review and update. Vet. Clin. North Am. Equine Pract. 1988, 4, 229–245. [Google Scholar] [CrossRef]
- Aurich, C. Reproductive cycles of horses. Anim. Reprod. Sci. 2011, 124, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Brunckhorst, D.; Schoon, H.-A.; Bader, H.; Sieme, H. Morphologische, enzyme- and immunhistologische Charakteristika des endometrialen Zyklus der Stute. Fertilität 1991, 7, 44–51. [Google Scholar]
- Kilgenstein, H.J. Die Subfertilität der Sportstute: Welchen Einblick gewährt die histopathologische Untersuchung von Endometriumbioptaten? Inaugural-Dissertation, Leipzig University, Leipzig, Germany, 2014. [Google Scholar]
- Strankmeyer, O. Morphometrische Untersuchungen am Endometrium zyklischer und ovariektomierter hormonbehandelter Stuten: Entwicklung eines diagnostischen Verfahrens. Inaugural Dissertation, University of Veterinary Medicine Hannover, Hannover, Germany, 1993. [Google Scholar]
- Watson, E.D.; Skolnik, S.B.; Zanecosky, H.G. Progesterone and estrogen receptor distribution in the endometrium of the mare. Theriogenology 1992, 38, 575–580. [Google Scholar] [CrossRef]
- Aupperle, H.; Özgen, S.; Schoon, H.-A.; Schoon, D.; Hoppen, H.O.; Sieme, H.; Tannapfel, A. Cyclical endometrial steroid hormone receptor expression and proliferation intensity in the mare. Equine Vet. J. 2000, 32, 228–232. [Google Scholar] [CrossRef]
- Hartt, L.S.; Carling, S.J.; Joyce, M.M.; Johnson, G.A.; Vanderwall, D.K.; Ott, T.L. Temporal and spatial associations of oestrogen receptor alpha and progesterone receptor in the endometrium of cyclic and early pregnant mares. Reproduction 2005, 130, 241–250. [Google Scholar] [CrossRef]
- Silva, E.S.M.; Scoggin, K.E.; Canisso, I.F.; Troedsson, M.H.T.; Squires, E.L.; Ball, B.A. Expression of receptors for ovarian steroids and prostaglandin E2 in the endometrium and myometrium of mares during estrus, diestrus and early pregnancy. Anim. Reprod. Sci. 2014, 151, 169–181. [Google Scholar] [CrossRef]
- Hammond, M.E.; Hayes, D.F.; Dowsett, M.; Allred, D.C.; Hagerty, K.L.; Badve, S.; Fitzgibbons, P.L.; Francis, G.; Goldstein., N.S.; Hayes, M.; et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer (unabridged version). Arch. Pathol. Lab. Med. 2010, 134, e48–Ce72. [Google Scholar]
- Cooke, P.S.; Buchanan, D.L.; Young, P.; Setiawan, T.; Brody, J.; Korach, K.S.; Taylor, J.; Lubahn, D.B.; Cunha, G.R. Stromal estrogen receptors mediate mitogenic effects of estradiol on uterine epithelium. Proc. Natl. Acad. Sci. USA 1997, 94, 6535–6540. [Google Scholar] [CrossRef]
- Kurita, T.; Lee, K.J.; Cooke, P.S.; Lydon, J.P.; Cunha, G.R. Paracrine regulation of epithelial progesterone receptor and lactoferrin by progesterone in the mouse uterus. Biol. Reprod. 2000, 62, 831–838. [Google Scholar] [CrossRef] [PubMed]
- Cunha, G.R.; Cooke, P.S.; Kurita, T. Role of stromal-epithelial interactions in hormonal responses. Arch. Histol. Cytol. 2004, 67, 417–434. [Google Scholar] [CrossRef] [PubMed]
- Chung, D.; Gao, F.; Jegga, A.G.; Das, S.K. Estrogen mediated epithelial proliferation in the uterus is directed by stromal Fgf10 and Bmp8a. Mol. Cell. Endocrinol. 2015, 400, 48–60. [Google Scholar] [CrossRef]
- Pierro, E.; Minici, F.; Alesiani, O.; Miceli, F.; Proto, C.; Screpanti, I.; Mancuso, S.; Lanzone, A. Stromal-epithelial interactions modulate estrogen responsiveness in normal human endometrium. Biol. Reprod. 2001, 64, 831–838. [Google Scholar] [CrossRef] [PubMed]
- Crossett, B.; Suire, S.; Herrler, A.; Allen, W.R.; Stewart, F. Transfer of a uterine lipocalin from the endometrium of the mare to the developing equine conceptus. Biol. Reprod. 1998, 59, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Stewart, F.; Gerstenberg, C.; Suire, S.; Allen, W.R. Immunolocalization of a novel protein (P19) in the endometrium of fertile and subfertile mares. J. Reprod. Fertil. Suppl. 2000, 56, 593–599. [Google Scholar]
- Hoffmann, C.; Bazer, F.W.; Klug, J.; Aupperle, H.; Ellenberger, C.; Özgen, S.; Schoon, D.; Schoon, H.-A. Immunohistochemical and histochemical identification of proteins and carbohydrates in the equine endometrium: Reaction patterns in the cycling mare. Pferdeheilkunde 2009, 25, 212–219. [Google Scholar] [CrossRef]
- Suire, S.; Stewart, F.; Beauchamp, J.; Kennedy, M.W. Uterocalin, a lipocalin provisioning the preattachment equine conceptus: Fatty acid and retinol binding properties, and structural characterization. Biochem. J. 2001, 356, 369–376. [Google Scholar] [CrossRef]
- Bramer, S.A.; Macedo, A.; Klein, C. Hexokinase 2 drives glycogen accumulation in equine endometrium at day 12 of diestrus and pregnancy. Reprod. Biol. Endocrinol. 2017, 15, 4. [Google Scholar] [CrossRef]
- Grimm, A.-L.; Schoon, H.-A.; Schöniger, S. Histopathological features of endometritis eosinophilica in mares. Histol. Histopathol. 2017, 32, 1161–1173. [Google Scholar]
- Schulz, S. Untersuchungen zu Vorkommen, Nachweis und Bedeutung von Mastzellen im Endometrium der Stute. Inaugural-Dissertation, Leipzig University, Leipzig, Germany, 1997. [Google Scholar]
- Watson, E.D.; Thomson, S.R.M. Lymphocyte subsets in the endometrium of genitally normal mares and mares susceptible to endometritis. Equine Vet. J. 1996, 28, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Waelchli, R.O.; Winder, N.C. Mononuclear cell infiltration of the equine endometrium: Immunohistochemical studies. Equine Vet. J. 1991, 23, 470–474. [Google Scholar] [CrossRef] [PubMed]
- Schöniger, S.; Gräfe, H.; Richter, F.; Helmschrodt, C.; Schoon, H.-A. Expression of NOD1 and NOD2 transcripts in the healthy and diseased equine endometrium. Pferdeheilkunde 2017, 33, 216–222. [Google Scholar] [CrossRef]
- Christoffersen, M.; Woodward, E.; Bojesen, A.M.; Jacobsen, S.; Petersen, M.R.; Troedsson, M.H.T.; Lehn-Jensen, H. Inflammatory responses to induced infectious endometritis in mares resistant or susceptible to persistent endometritis. BMC Vet. Res. 2012, 8, 41. [Google Scholar] [CrossRef] [PubMed]
- Woodward, E.M.; Christoffersen, M.; Campos, J.; Betancourt, A.; Horohov, D.; Scoggin, K.E.; Squires, E.L.; Troedsson, M.H.T. Endometrial inflammatory markers of the early immune response in mares susceptible or resistant to persistent breeding-induced endometritis. Reproduction 2013, 145, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Constantine, V.S. A combined tissue stain for the selective staining of collagen, elastic fibers and acidic carbohydrates. J. Invest. Dermatol. 1969, 52, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Ebert, A.; Schoon, D.; Schoon, H.-A. Age-related endometrial alterations in mares—biopsy findings of the last 20 years. In Leipziger Blaue Hefte, 7th Leipzig Veterinary Congress, 8th International Conference on Equine Reproductive Medicine; Rackwitz, R., Pees, M., Aschenbach, J.R., Gäbel, G., Eds.; Lehmanns Media GmbH: Berlin, Germany, 2014; Volume 2, pp. 230–232. [Google Scholar]
- Alimi, Y.; Iwanaga, J.; Loukas, M.; Tubbs, R.S. The clinical anatomy of endometriosis: A review. Cureus 2018, 10, e3361. [Google Scholar] [CrossRef]
- Hoffman, C.; Ellenberger, C.; Mattos, R.C.; Aupperle, H.; Dhein, S.; Stief, B.; Schoon, H.-A. The equine endometrosis: New insights into the pathogenesis. Anim. Reprod. Sci. 2009, 111, 261–278. [Google Scholar] [CrossRef]
- Lehmann, J.; Ellenberger, C.; Hoffmann, C.; Bazer, F.W.; Klug, J.; Allen, W.R.; Sieme, H.; Schoon, H.-A. Morpho-functional studies regarding the fertility prognosis of mares suffering from equine endometrosis. Theriogenology 2011, 76, 1326–1336. [Google Scholar] [CrossRef]
- Hoffmann, C.; Bazer, F.W.; Klug, J.; Aupperle, H.; Ellenberger, C.; Schoon, H.-A. Immunohistochemical and histochemical identification of proteins and carbohydrates in the equine endometrium. Expression patterns for mares suffering from endometrosis. Theriogenology 2009, 71, 264–274. [Google Scholar] [CrossRef]
- Rebordão, M.R.; Amaral, A.; Lukasik, K.; Szóstek-Mioduchowska, A.; Pinto-Bravo, P.; Galvão, A.; Skarzynski, D.J.; Ferreira-Dias, G. Constituents of neutrophil extracellular traps induce in vitro collagen formation in mare endometrium. Theriogenology 2018, 113, 8–18. [Google Scholar]
- Szóstek-Mioduchowska, A.Z.; Lukasik, K.; Skarzynski, D.J.; Okuda, K. Effect of transforming growth factor-β1 on α-smooth muscle actin and collagen expression in equine endometrial fibroblasts. Theriogenology 2019, 124, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Rebordão, M.R.; Carneiro, C.; Alexandre-Pires, G.; Brito, P.; Pereira, C.; Nunes, T.; Galvão, A.; Leitão, A.; Vitela, C.; Ferreira-Dias, G. Neutrophil extracellular traps formation by bacteria causing endometritis in the mare. J. Reprod. Immunol. 2014, 106, 41–49. [Google Scholar]
- Woodward, E.M.; Christoffersen, M.; Campos, J.; Horohov, D.W.; Scoggin, K.E.; Squires, E.; Troedsson, M.H.T. An investigation of uterine nitric oxide production in mares susceptible and resistant to persistent breeding-induced endometritis and the effects of immunomodulation. Reprod. Dom. Anim. 2013, 48, 554–561. [Google Scholar] [CrossRef]
- Kabisch, J.; Klose, K.; Schoon, H.-A. Endometrial biopsies of old mares—What to expect?! Pferdeheilkunde 2019, 35, 211–219. [Google Scholar] [CrossRef]
- Wittenbrink, M.W.; Hoelzle, K.; Hoelzle, L.E. What’s new in the bacteriology of the mare’s genital tract. Pferdeheilkunde 2008, 24, 53–55. [Google Scholar] [CrossRef][Green Version]
- Petersen, M.R.; Skive, B.; Christoffersen, M.; Lu, K.; Nielsen, J.M.; Troedsson, M.H.; Bojesen, A.M. Activation of persistent Streptococcus equi subspecies zooepidemicus in mares with subclinical endometritis. Vet. Microbiol. 2015, 179, 119–125. [Google Scholar] [CrossRef]
- Christoffersen, M.; Troedsson, M.H.T. Inflammation and fertility in the mare. Reprod. Dom. Anim. 2017, 52, 14–20. [Google Scholar] [CrossRef]
- Tunón, A.-M.; Katila, T.; Magnusson, U.; Nummijärvi, A.; Rodriguez-Martinez, H. T-cell distribution in two different segments of the equine endometrium 6 and 48 hours after insemination. Theriogenology 2000, 54, 835–841. [Google Scholar] [CrossRef]
- Slusher, S.H.; Freeman, K.P.; Roszel, J.F. Eosinophils in equine uterine cytology and histology specimens. J. Am. Vet. Med. Assoc. 1984, 184, 665–670. [Google Scholar]
- Klug, E.; Bartmann, C.P.; Schöning, A.; Schoon, D.; Schoon, H.-A. Auswirkung einer Progestagen-Langzeit-Applikation auf Sexualzyklus und Endometrium des Pferdes—Zwei Seiten einer therapeutischen Medaille. Pferdeheilkunde 1997, 13, 490–498. [Google Scholar] [CrossRef][Green Version]
- Ellenberger, C.; Schoon, D.; Schoon, H.-A. Außergewöhnliche Befunde bei der Untersuchung von Uterusbiopsien der Stute. Pferdeheilkunde 2006, 22, 171–176. [Google Scholar] [CrossRef][Green Version]
- Fumuso, E.A.; Aguilar, J.; Giguère, S.; Rivulgo, M.; Wade, J.; Rogan, D. Immune parameters in mares resistant and susceptible to persistent post-breeding endometritis: Effects of immunomodulation. Vet. Immunol. Immunopathol. 2007, 118, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Häfner, I.; Schoon, H.-A.; Schoon, D.; Aupperle, H. Glanduläre Differenzierungsstörungen im Endometrium der Stute—Lichtmikroskopische und immunhistologische Untersuchungen. Pferdeheilkunde 2001, 17, 103–110. [Google Scholar] [CrossRef]
- Ellenberger, C.; Aupperle, H.; Bartmann, C.-P.; Hoppen, H.-O.; Schoon, D.; Schoon, H.-A. Endometrial maldifferentiation caused by ovarian disorders in the mare—morphological and immunohistochemical studies. Theriogenology 2002, 58, 499–502. [Google Scholar]
- Ellenberger, C.; Bartmann, C.-P.; Klug, E.; Hoppen, H.-O.; Hoffmann, C.; Bazer, F.W.; Klug, J.; Allen, W.R.; Schoon, D.; Schoon, H.-A. Immunohistochemical characterization of equine endometrial maldifferentiation with special emphasis on uterine secretory proteins. In Havemeyer Foundation Monograph Series No. 14, Proceedings of the 6th International Symposium on Equine Embryo Transfer, Rio de Janeiro, Brazil, 4th–6th August 2004; Alvarenga, M., Wade, J.F., Eds.; R & W Communications: Newmarket, UK, 2005; pp. 13–15. [Google Scholar]
- Grüninger, B.; Schoon, H.-A.; Schoon, D.; Menger, S.; Klug, E. Incidence and morphology of endometrial angiopathies in mares in relationship to age and parity. J Comp. Pathol. 1998, 119, 293–309. [Google Scholar] [CrossRef]
- Ludwig, S.; Schoon, D.; Aupperle, H.; von Reiswitz, A.; Schoon, H.-A. Angiopathies in the equine endometrial biopsy—A marker for extrauterine vascular lesions? Pferdeheilkunde 2001, 17, 608–614. [Google Scholar] [CrossRef]
- Aupperle, H.; Hasenbein, I.; Ellenberger, C.; Ludwig, S.; Schoon, D.; Schoon, H.-A. Histopathological findings in ovarian vessels, in comparison to alterations in extraovarian, extrauterine and uterine vessels in the mare. Pferdeheilkunde 2003, 19, 589–594. [Google Scholar] [CrossRef]
- Agnew, D.W.; MacLachlan, N.J. Tumors of the genital system. In Tumors in Domestic Animals, 5th ed.; Meuten, D.J., Ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2016; pp. 689–722. [Google Scholar]
- Gunson, D.E.; Gillette, D.M.; Beech, J.; Orsini, J. Endometrial adenocarcinoma in a mare. Vet. Pathol. 1980, 17, 776–780. [Google Scholar] [CrossRef]
- Chaffin, M.K.; Fuentealba, I.C.; Schmitz, D.G.; Read, W.K. Endometrial adenocarcinoma in a mare. Cornell. Vet. 1990, 80, 65–73. [Google Scholar]
- Thompson, R.; Armién, A.G.; Rasmussen, J.M.; Wolf, T.M. Uterine adenocarcinoma in a Przewalski‘s wild horse (Equus ferus przewalskii). J. Zoo. Wildl. Med. 2014, 45, 441–445. [Google Scholar] [CrossRef] [PubMed]
- Lopez, C.; Ciccarelli, M.; Gold, J.R.; Tibary, A. Uterine adenocarcinoma in Quarter horse mare. Equine Vet. Educ. 2017, 30, 640–644. [Google Scholar] [CrossRef]
- Govaere, J.; Maes, S.; Saey, V.; Blancke, W.; Hoogewijs, M.; Deschauwer, C.; Smits, K.; Roles, K.; Vercauteren, G.; de Kruif, A. Uterine fibrsarcoma in a Warmblood mare. Reprod. Domest. Anim. 2011, 46, 564–566. [Google Scholar] [CrossRef] [PubMed]
- Freeman, S.L.; England, G.C.; Bjornson, S.; Smith, R.K. Uterine T cell lymphoma in a mare, with multicentric involvement. Vet Rec. 1997, 141, 391–393. [Google Scholar] [CrossRef]
- Canisso, I.F.; Pinn, T.L.; Gerdin, J.A.; Ollivett, T.L.; Buckles, E.L.; Schweizer, C.M.; Ainsworth, D.M. B-cell multicentric lymphoma as a probable cause of abortion in a Quarter horse broodmare. Can Vet. J. 2013, 54, 288–291. [Google Scholar]
- Claes, a.; Ball, B.A.; Liu, I.K.M.; Vaughan, B.; Highland, M.A.; Brown, J.A. Uterine B cell lymphoma in a mare. Equine Vet. Educ. 2015, 27, e5–Ce8. [Google Scholar] [CrossRef]
- Yamini, B.; Borg, L. Endometrial polyps and endometritis in a thoroughbred filly. J. Vet. Diagn. Invest. 1994, 6, 496–498. [Google Scholar] [CrossRef]
- Hamir, A.N.; Hunt, P.R.; Kenney, R.M. Hyperplastic endometrial polyps in a two-year-old filly. Vet. Pathol. 1989, 26, 185–187. [Google Scholar] [CrossRef]
- Allen, W.R.; Stewart, F. Equine placentation. Reprod. Fertil. Dev. 2001, 13, 623–634. [Google Scholar] [CrossRef]
- Alghamdi, A.; Troedsson, M.H.T.; Laschkewitsch, T.; Xue, J.L. Uterine secretion from mares with post-breeding endometritis alters sperm motion characteristics in vitro. Theriogenology 2001, 55, 1019–1028. [Google Scholar] [CrossRef]
- Troedsson, M.H.T.; Desvousges, A.; Alghamdi, A.S.; Dahms, B.; Dow, C.A.; Hayna, J.; Valesco, R.; Collahan, P.T.; Macpherson, M.L.; Pozor, M.; et al. Components in seminal plasma regulating sperm transport and elimination. Anim. Reprod. Sci. 2005, 89, 171–186. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, R.O. The effects of endometritis on the establishment of pregnancy in cattle. Reprod. Fertil. Dev. 2012, 24, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Ferris, R.A.; Frisbie, D.D.; McCue, P.M. Use of mesenchymal stem cells or autologous conditioned serum to modulate the inflammatory response to spermatozoa in mares. Theriogenology 2014, 82, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Rink, E.B.; Amilon, K.R.; Esteves, C.L.; French, H.M.; Watson, E.; Aurich, E.; Donadeu, F.X. Isolation and characterization of equine endometrial mesenchymal stromal cells. Stem Cell Res. Ther. 2017, 8, 166. [Google Scholar] [CrossRef]






| Disease Entities | Subgroups |
|---|---|
| Endometrosis |
|
| Endometritis |
|
| Maldifferentiation during the breeding season |
|
| Angiosis |
|
| Neoplasia |
|
| Category | Microscopic Findings and Additional Factors | Estimated Foaling Rate * |
|---|---|---|
| I | No significant alterations | >80–90% |
| IIa | Mild endometritis OR Mild endometrosis OR Mild lymphatic lacunae OR Partial endometrial atrophy during the late breeding season | 50–80% |
| IIb | Moderate endometritis OR Moderate endometrosis OR Moderate lymphatic lacunae OR Barrenness ≥2 years OR Combined presence of two category IIa findings | 10–50% |
| III | Marked endometritis OR Marked endometrosis OR Marked lymphatic lacunae OR Deep endometrial atrophy during the breeding season OR Combined presence of ≥3 category IIa or ≥2 category IIb or III findings | <10% |
| Molecule | Cell(s)/Structure | Normal and Altered Functions |
|---|---|---|
| Estrogen Receptor | E, S | Hormonal Responsiveness |
| Progesterone Receptor | E, S | Hormonal Responsiveness |
| Vimentin | E, S | Cellular Differentiation |
| Desmin | S | Cellular Differentiation |
| Smooth muscle actin | S | Cellular Differentiation |
| Calponin | E, S | Cellular Differentiation |
| Laminin | BM | Interaction GE/S |
| Uteroglobin | E | Secretory activity |
| Uterocalin | E | Secretory activity |
| Uteroferrin | E | Secretory activity |
| Calbindin | E | Secretory activity |
| β-Defensin | E | Immune defense |
| Indoleamine 2,3 dioxygenase | E | Immune defense |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schöniger, S.; Schoon, H.-A. The Healthy and Diseased Equine Endometrium: A Review of Morphological Features and Molecular Analyses. Animals 2020, 10, 625. https://doi.org/10.3390/ani10040625
Schöniger S, Schoon H-A. The Healthy and Diseased Equine Endometrium: A Review of Morphological Features and Molecular Analyses. Animals. 2020; 10(4):625. https://doi.org/10.3390/ani10040625
Chicago/Turabian StyleSchöniger, Sandra, and Heinz-Adolf Schoon. 2020. "The Healthy and Diseased Equine Endometrium: A Review of Morphological Features and Molecular Analyses" Animals 10, no. 4: 625. https://doi.org/10.3390/ani10040625
APA StyleSchöniger, S., & Schoon, H.-A. (2020). The Healthy and Diseased Equine Endometrium: A Review of Morphological Features and Molecular Analyses. Animals, 10(4), 625. https://doi.org/10.3390/ani10040625





