Impacts of Time-Fed Concentrate-Based Diets on Plasma Metabolites, Rumen Histology, and mRNA Expression of Hepatic Enzymes of Wethers
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal and Diet Management
2.2. Sample Collection and mRNA Expression
2.3. Statistical Analysis
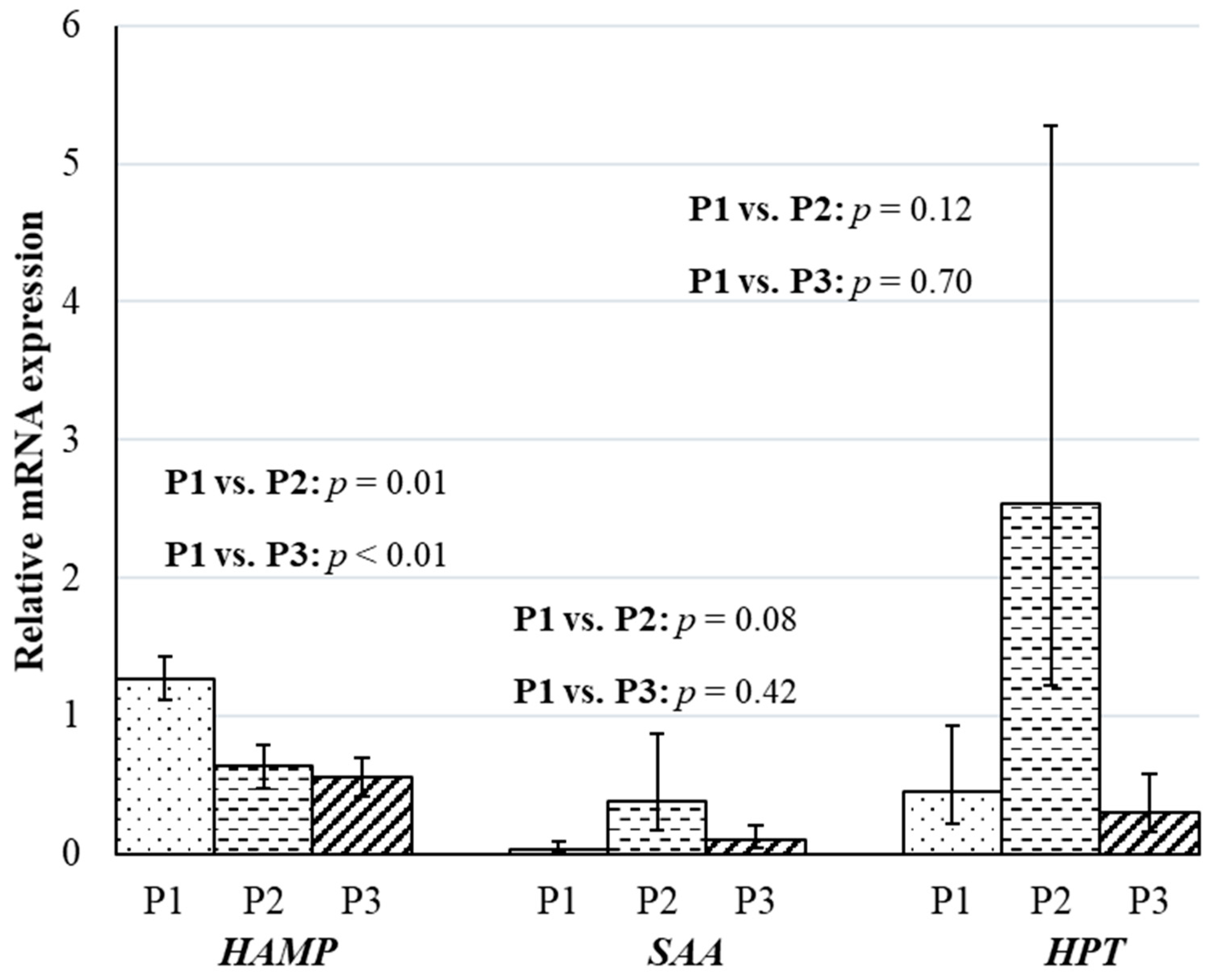
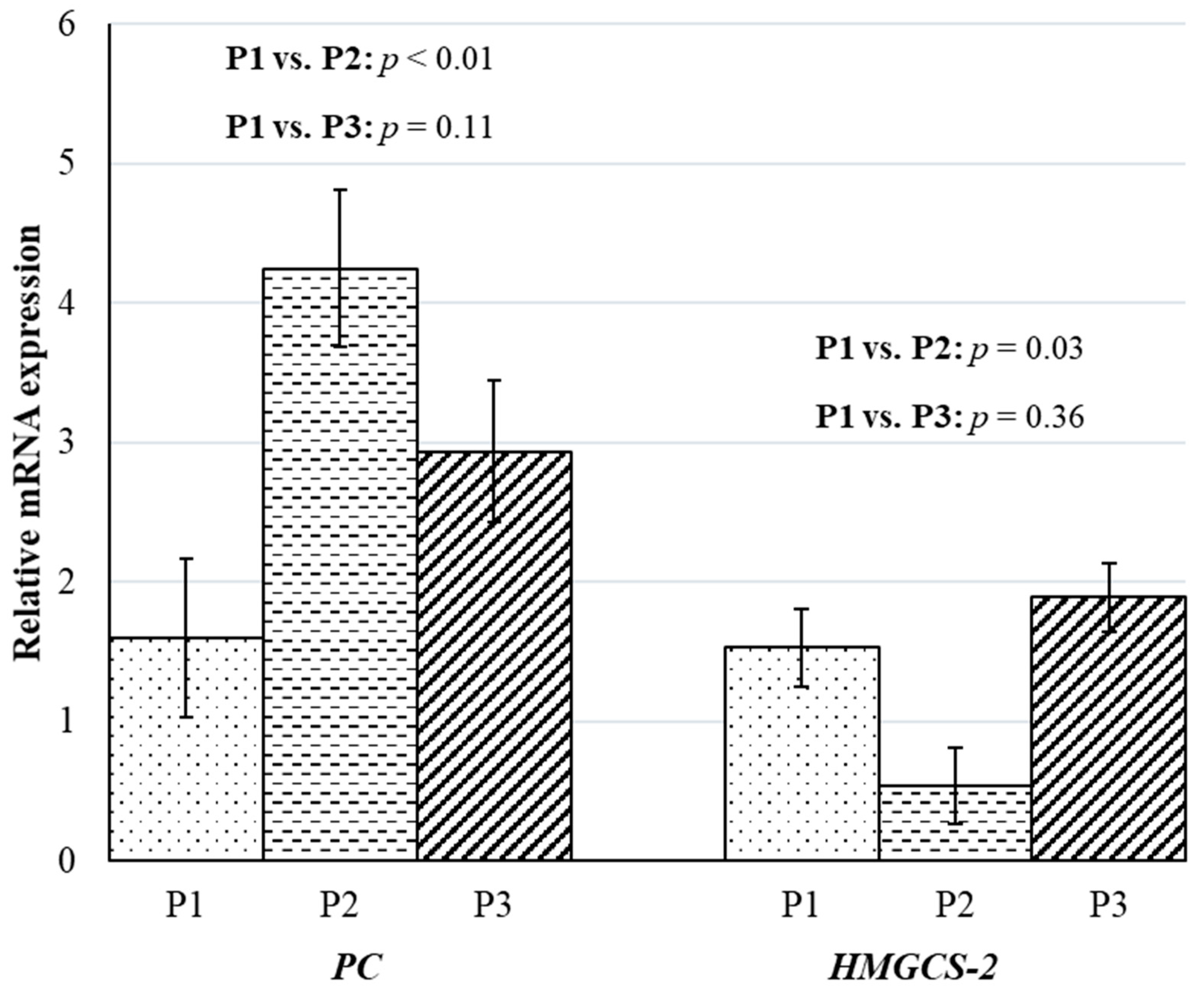
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Owens, F.N.; Secrist, D.S.; Hill, W.J.; Gill, D.R. Acidosis in cattle: A review. J. Anim Sci. 1998, 76, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Ahrens, F.A. Histamine, lactic acid, and hypertonicity as factors in the development of rumenitis in cattle. Am. J. Vet. Res. 1967, 28, 1335–1342. [Google Scholar] [PubMed]
- Steele, M.A.; AlZahal, O.; Hook, S.E.; Croom, J.; McBride, B.W. Ruminal acidosis and the rapid onset of ruminal parakeratosis in a mature dairy cow: A case report. Acta Vet. Scand. 2009, 51, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, J.; Chang, G.; Zhang, K.; Xu, L.; Jin, D.; Bilal, M.S.; Shen, X. Rumen-derived lipopolysaccharide provoked inflammatory injury in the liver of dairy cows fed a high-concentrate diet. Oncotarget 2017, 8, 46769–46780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baumann, H.; Gauldie, J. The acute phase response. Immunol. Today 1994, 15, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Murata, H.; Shimada, N.; Yoshioka, M. Current research on acute phase proteins in veterinary diagnosis: An overview. Vet. J. 2004, 168, 28–40. [Google Scholar] [CrossRef]
- Memon, R.A.; Feingold, K.R.; Moser, A.H.; Doerrler, W.; Adi, S.; Dinarello, C.A.; Grunfeld, C. Differential effects of interleukin-1 and tumor necrosis factor on ketogenesis. Am. J. Physiol. Endocrinol. Metab. 1992, 263, E301–E309. [Google Scholar] [CrossRef]
- Dong, H.; Wang, S.; Jia, Y.; Ni, Y.; Zhang, Y.; Zhuang, S.; Shen, X.; Zhao, R. Long-term effects of subacute ruminal acidosis (SARA) on milk quality and hepatic gene expression in lactating goats fed a high-concentrate diet. PLoS ONE 2013, 8, e82850. [Google Scholar] [CrossRef] [Green Version]
- National Research Council (NRC). Nutrient Requirements of Small Ruminants: Sheep, Goats, Cervids, and New World Camelids; The National Academy Press: Washington, DC, USA, 2007. [Google Scholar] [CrossRef]
- Hall, M.B. Determination of starch, including maltooligosaccharides, in animal feeds: Comparison of methods and a method recommended for AOAC collaborative study. J. AOAC Int. 2009, 92, 42–49. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Bionaz, M.; Sloboda, D.M.; Ehrlich, L.; Li, S.; Newnham, J.P.; Dudenhausen, J.W.; Henrich, W.; Plagemann, A.; Challis, J.R. The dilution effect and the importance of selecting the right internal control genes for RT-qPCR: A paradigmatic approach in fetal sheep. BMC Res. Notes 2015, 8, 58. [Google Scholar] [CrossRef] [Green Version]
- Janovick-Guretzky, N.A.; Dann, H.M.; Carlson, D.B.; Murphy, M.R.; Loor, J.J.; Drackley, J.K. Housekeeping Gene Expression in Bovine Liver is Affected by Physiological State, Feed Intake, and Dietary Treatment. J. Dairy Sci. 2007, 90, 2246–2252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chishti, G.A.; Salfer, I.J.; Suarez-Mena, F.X.; Harvatine, K.J.; Heinrichs, A.J. Relationships between physical form of oats in starter, rumen pH, and volatile fatty acids on hepatic expression of genes involved in metabolism and inflammation in dairy calves. J. Dairy Sci. 2019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xie, F.; Xiao, P.; Chen, D.; Xu, L.; Zhang, B. miRDeepFinder: A miRNA analysis tool for deep sequencing of plant small RNAs. Plant Mol. Biol. 2012, 80, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Steele, M.A.; Greenwood, S.L.; Croom, J.; McBride, B.W. An increase in dietary non-structural carbohydrates alters the structure and metabolism of the rumen epithelium in lambs. Can. J. Anim. Sci. 2012, 92, 123–130. [Google Scholar] [CrossRef]
- Odongo, N.; AlZahal, O.; Lindinger, M.; Duffield, T.; Valdes, E.; Terrell, S.; McBride, B. Effects of mild heat stress and grain challenge on acid-base balance and rumen tissue histology in lambs. J. Anim. Sci. 2006, 84, 447–455. [Google Scholar] [CrossRef]
- Gebretsadkan, T.K.; Ambachew, G.; Birhaneselassie, H. The comparison between microhematocrit and automated methods for hematocrit determination. Int. J. Blood Res. Disord 2015, 2, 1–3. [Google Scholar] [CrossRef]
- Naylor, J.M.; Kronfeld, D.S. Refractometry as a measure of the immunoglobulin status of the newborn dairy calf: Comparison with the zinc sulfate turbidity test and single radial immunodiffusion. Am. J. Vet. Res. 1977, 38, 1331–1334. [Google Scholar]
- McCarthy, M.M.; Yasui, T.; Felippe, M.J.B.; Overton, T.R. Associations between the degree of early lactation inflammation and performance, metabolism, and immune function in dairy cows. J. Dairy Sci. 2016, 99, 680–700. [Google Scholar] [CrossRef] [Green Version]
- Osorio, J.S.; Trevisi, E.; Li, C.; Drackley, J.K.; Socha, M.T.; Loor, J.J. Supplementing Zn, Mn, and Cu from amino acid complexes and Co from cobalt glucoheptonate during the peripartal period benefits postpartal cow performance and blood neutrophil function. J. Dairy Sci. 2016, 99, 1868–1883. [Google Scholar] [CrossRef] [Green Version]
- Maxie, G. Jubb, Kennedy & Palmer’s Pathology of Domestic Animals-E-Book (Volume 2); Elsevier Health Sciences: St. Louis, MI, USA, 2015; p. 41. [Google Scholar]
- Hinders, R.G.; Owen, F.G. Relation of ruminal parakeratosis development to volatile fatty acid absorption. J. Dairy Sci. 1965, 48, 1069–1073. [Google Scholar] [CrossRef]
- Lorenz, I. Ruminal Parakeratosis. In Merck Veterinary Manual; Merck & Co., Inc.: Kenilworth, NJ, USA, 2019; Available online: https://www.merckvetmanual.com/digestive-system/diseases-of-the-ruminant-forestomach/ruminal-parakeratosis (accessed on 3 December 2019).
- Penner, G.B.; Taniguchi, M.; Guan, L.L.; Beauchemin, K.A.; Oba, M. Effect of dietary forage to concentrate ratio on volatile fatty acid absorption and the expression of genes related to volatile fatty acid absorption and metabolism in ruminal tissue. J. Dairy Sci. 2009, 92, 2767–2781. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Sun, D.; Mao, S.; Zhu, W.; Liu, J. Infusion of sodium butyrate promotes rumen papillae growth and enhances expression of genes related to rumen epithelial VFA uptake and metabolism in neonatal twin lambs. J. Anim Sci. 2019, 97, 909–921. [Google Scholar] [CrossRef] [PubMed]
- Sander, E.G.; Warner, R.G.; Harrison, H.N.; Loosli, J.K. The stimulatory effect of sodium butyrate and sodium propionate on the development of rumen Mmcosa in the young calf. J. Dairy Sci. 1959, 42, 1600–1605. [Google Scholar] [CrossRef]
- Minuti, A.; Ahmed, S.; Trevisi, E.; Piccioli-Cappelli, F.; Bertoni, G.; Jahan, N.; Bani, P. Experimental acute rumen acidosis in sheep: Consequences on clinical, rumen, and gastrointestinal permeability conditions and blood chemistry1. J. Anim Sci. 2014, 92, 3966–3977. [Google Scholar] [CrossRef] [Green Version]
- Bevans, D.W.; Beauchemin, K.A.; Schwartzkopf-Genswein, K.S.; McKinnon, J.J.; McAllister, T.A. Effect of rapid or gradual grain adaptation on subacute acidosis and feed intake by feedlot cattle. J. Anim Sci. 2005, 83, 1116–1132. [Google Scholar] [CrossRef]
- Chang, G.; Zhang, K.; Xu, T.; Jin, D.; Seyfert, H.-M.; Shen, X.; Zhuang, S. Feeding a high-grain diet reduces the percentage of LPS clearance and enhances immune gene expression in goat liver. BMC Vet. Res. 2015, 11, 67. [Google Scholar] [CrossRef] [Green Version]
- Rossi, E. Hepcidin—the iron regulatory hormone. Clin. Biochemist. Rev. 2005, 26, 47–49. [Google Scholar]
- Nemeth, E.; Ganz, T. The role of hepcidin in iron metabolism. Acta Haematol. 2009, 122, 78–86. [Google Scholar] [CrossRef] [Green Version]
- Nicolas, G.; Chauvet, C.; Viatte, L.; Danan, J.L.; Bigard, X.; Devaux, I.; Beaumont, C.; Kahn, A.; Vaulont, S. The gene encoding the iron regulatory peptide hepcidin is regulated by anemia, hypoxia, and inflammation. J. Clin. Invest. 2002, 110, 1037–1044. [Google Scholar] [CrossRef]
- da Silva, C.B.; Wolkmer, P.; Paim, F.C.; da Silva, A.S.; Siqueira, L.C.; de Souza, C.L.; França, R.T.; Dornelles, G.L.; Duarte, M.M.M.F.; Monteiro, S.G.; et al. Iron metabolism and its relationship to anemia and immune system in Trypanosoma evansi infected rats. Exp. Parasitol. 2013, 133, 357–364. [Google Scholar] [CrossRef]
- Zebeli, Q.; Dunn, S.M.; Ametaj, B.N. Perturbations of plasma metabolites correlated with the rise of rumen endotoxin in dairy cows fed diets rich in easily degradable carbohydrates. J. Dairy Sci. 2011, 94, 2374–2382. [Google Scholar] [CrossRef] [PubMed]
- Steele, M.A.; Vandervoort, G.; AlZahal, O.; Hook, S.E.; Matthews, J.C.; McBride, B.W. Rumen epithelial adaptation to high-grain diets involves the coordinated regulation of genes involved in cholesterol homeostasis. Physiol. Genom. 2011, 43, 308–316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodríguez-Lecompte, J.C.; Kroeker, A.D.; Ceballos-Márquez, A.; Li, S.; Plaizier, J.C.; Gomez, D.E. Evaluation of the systemic innate immune response and metabolic alterations of nonlactating cows with diet-induced subacute ruminal acidosis. J. Dairy Sci. 2014, 97, 7777–7787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Velez, J.; Donkin, S. Feed restriction induces pyruvate carboxylase but not phosphoenolpyruvate carboxykinase in dairy cows. J. Dairy Sci. 2005, 88, 2938–2948. [Google Scholar] [CrossRef] [Green Version]
- Overton, T.R.; Drackley, J.K.; Ottemann-Abbamonte, C.J.; Beaulieu, A.D.; Emmert, L.S.; Clark, J.H. Substrate utilization for hepatic gluconeogenesis is altered by increased glucose demand in ruminants. J. Anim Sci. 1999, 77, 1940–1951. [Google Scholar] [CrossRef]
- Aschenbach, J.R.; Kristensen, N.B.; Donkin, S.S.; Hammon, H.M.; Penner, G.B. Gluconeogenesis in dairy cows: The secret of making sweet milk from sour dough. IUBMB Life 2010, 62, 869–877. [Google Scholar] [CrossRef]
- Penner, G.B.; Aschenbach, J.R.; Gäbel, G.; Rackwitz, R.; Oba, M. Epithelial capacity for apical uptake of short chain fatty acids is a key determinant for intraruminal pH and the susceptibility to subacute ruminal acidosis in sheep. J. Nutr. 2009, 139, 1714–1720. [Google Scholar] [CrossRef] [Green Version]
- Greenfield, R.; Cecava, M.; Donkin, S. Changes in mRNA Expression for Gluconeogenic Enzymes in liver of dairy cattle during the transition to lactation 1. J. Dairy Sci. 2000, 83, 1228–1236. [Google Scholar] [CrossRef]

| Item, % DM Basis | Forage | Concentrate |
|---|---|---|
| Ingredients | ||
| Grass hay 1 | 80.00 | 20.00 |
| Dry rolled corn | 8.10 | 69.80 |
| Soybean meal | 10.15 | 8.45 |
| Mineral and Vitamin Supplement 2 | 0.75 | 0.75 |
| Urea | 0.50 | 0.50 |
| NH4Cl | 0.50 | 0.50 |
| Analyzed nutrient composition | ||
| DM | 85.7 | 85.7 |
| NDF | 56.8 | 20.7 |
| ADF | 29.9 | 9.3 |
| Starch | 7.1 | 47.8 |
| CP | 14.1 | 11.9 |
| Gene | Primer | Access Code | Base Pair Length | R2 | Efficiency |
|---|---|---|---|---|---|
| ACTB | F’-CATCGGCAATGAGCGGTTCC R’-CCGTGTTGGCGTAGAGGT | NM_001009784 | 146 | 1 | 108.1 |
| PPIA | F’-TGTGCCAGGGTGGTGACTTCA R’-TGCTTGCCATCCAACCACTCAG | AY251270 | 196 | 0.999 | 100.3 |
| SAA | F’-TGATCAGTGATGCCAGAGAAAAT R’-TCTGAAGTGGTTGGGGTCTTT | XM_004019477.3 | 146 | 0.997 | 100.3 |
| HPT | F’-TGAGGCAGTGTGCGGAAAG R’-ATCCAGCGACCCACCTATGA | XM_012110686.2 | 90 | 0.998 | 102.3 |
| ALB | F’-TAGCTCGCCTGAGCCAGAAA R’-GCAAGATCTGCCCTGTCGTC | NM_001009376.1 | 136 | 0.996 | 98.0 |
| PC | F’-GAGGAGATTACCGATGTGGACC R’-GGAACACCTCGATGCGGC | XM_027959895.1 | 184 | 0.999 | 106.0 |
| HMGCS | F’-GTCCTCGAGAGAGGGCTTAGA R’-ATTTGTCCAGGGCCCGTAAG | XM_004002390.4 | 133 | 0.986 | 100.6 |
| HMGCR | F’-TGCAGATGGGATGACTCGTG R’-TGTAGACGTGCAAACCTGCT | XM_004010192.4 | 144 | 0.999 | 105.3 |
| HAMP | F’-AGACACGACAGCTCACAGAC R’-ATGGGAAAGTGGGTGTCTCG | NM_001195312.1 | 103 | 0.995 | 105.3 |
| Stability Values 1 | ACTB | PPIA |
|---|---|---|
| Delta CT method | 0.36 | 0.36 |
| BestKeeper | 0.59 | 0.66 |
| Genorm | 0.36 * | |
| Comprehensive ranking value | 1.19 | 1.41 |
| Parameter | Phases 1 | SEM 2 | p-Value | |||
|---|---|---|---|---|---|---|
| P1 | P2 | P3 | P1 vs. P2 | P1 vs. P3 | ||
| n, wethers | 4 | 4 | 5 | - | - | - |
| Dense keratin 3, μm | 19.7 | 21.8 | 32.6 | 6.32 | 0.95 | 0.16 |
| Translucent keratin 4, μm | 22.6 | 54.7 | 98.7 | 8.48 | 0.02 | 0.01 |
| Papillae width, mm | 0.350 | 0.465 | 0.512 | 0.044 | 0.09 | 0.02 |
| Papillae length, mm | 1.16 | 1.15 | 1.36 | 0.165 | 0.98 | 0.37 |
| Papillae area, mm2 | 0.410 | 0.530 | 0.696 | 0.0900 | 0.36 | 0.04 |
| Parameter | Phase 1 | SEM 2 | p-Value | |||
|---|---|---|---|---|---|---|
| P1 | P2 | P3 | P1 vs. P2 | P1 vs. P3 | ||
| n, wethers | 4 | 4 | 5 | - | - | - |
| BHBA 3, mg/dL | 2.88 | 4.16 | 6.18 | 0.543 | 0.03 | <0.01 |
| Glucose, mg/dL | 62.3 | 86.8 | 87.9 | 2.98 | <0.01 | <0.01 |
| Hematocrit, % | 36.8 | 33.7 | 34.2 | 1.02 | <0.01 | <0.01 |
| Cholesterol, mg/dL | 50.0 | 41.2 | 73.6 | 4.77 | 0.06 | <0.01 |
| Total Protein, g/dL | 6.23 | 5.76 | 6.14 | 0.127 | <0.01 | 0.47 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chishti, G.A.; Salfer, I.J.; Nedelkov, K.V.; Felix, T.L. Impacts of Time-Fed Concentrate-Based Diets on Plasma Metabolites, Rumen Histology, and mRNA Expression of Hepatic Enzymes of Wethers. Animals 2020, 10, 686. https://doi.org/10.3390/ani10040686
Chishti GA, Salfer IJ, Nedelkov KV, Felix TL. Impacts of Time-Fed Concentrate-Based Diets on Plasma Metabolites, Rumen Histology, and mRNA Expression of Hepatic Enzymes of Wethers. Animals. 2020; 10(4):686. https://doi.org/10.3390/ani10040686
Chicago/Turabian StyleChishti, Ghazanfar A., Isaac J. Salfer, Krum V. Nedelkov, and Tara L. Felix. 2020. "Impacts of Time-Fed Concentrate-Based Diets on Plasma Metabolites, Rumen Histology, and mRNA Expression of Hepatic Enzymes of Wethers" Animals 10, no. 4: 686. https://doi.org/10.3390/ani10040686
APA StyleChishti, G. A., Salfer, I. J., Nedelkov, K. V., & Felix, T. L. (2020). Impacts of Time-Fed Concentrate-Based Diets on Plasma Metabolites, Rumen Histology, and mRNA Expression of Hepatic Enzymes of Wethers. Animals, 10(4), 686. https://doi.org/10.3390/ani10040686





