Risk Factors for Antimicrobial Use on Irish Pig Farms
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Farm Selection
2.2. Farm Production and Performance
2.3. Antimicrobial Use
2.4. Biosecurity Assessment and Farm Management Practices
2.5. Respiratory Disease Status and Farm Prevalence
2.6. Data Processing and Statistical Analysis
3. Results
4. Discussion
4.1. Farm Characteristics
4.2. Farm Performance Indicators
4.3. Biosecurity Practices
4.4. Respiratory Disease
4.5. Disease Management
4.6. Implications for Irish Pig Sector
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organisation. Global Action Plan on Antimicrobial Resistance. 2015. Available online: https://apps.who.int/iris/bitstream/handle/10665/193736/9789241509763_eng.pdf?sequence=1 (accessed on 2 April 2021).
- Department of Health. Ireland’s National Action Plan on Antimicrobial Resistance 2017–2020. (iNAP). 2017. Available online: https://www.agriculture.gov.ie/media/migration/animalhealthwelfare/amr/inap/IrelandNationalActionPlanAntimicrobialResistance030818.pdf (accessed on 2 December 2020).
- European Commission. A European One Health Action Plan against Antimicrobial Resistance (AMR). 2017. Available online: https://ec.europa.eu/health/amr/sites/health/files/antimicrobial_resistance/docs/amr_2017_action-plan.pdf (accessed on 2 April 2021).
- Tiseo, K.; Huber, L.; Gilbert, M.; Robinson, T.P.; van Boeckel, T.P. Global Trends in Antimicrobial Use in Food Animals from 2017 to 2030. Antibiotics 2020, 9, 918. [Google Scholar] [CrossRef]
- Aarestrup, F.M. The Livestock Reservoir for Antimicrobial Resistance: A Personal View on Changing Patterns of Risks, Effects of Interventions and the Way Forward. Phil. Trans. R. Soc. B 2015, 370, 20140085. [Google Scholar] [CrossRef] [Green Version]
- Tang, K.L.; Caffrey, N.P.; Nóbrega, D.B.; Cork, S.C.; Ronksley, P.E.; Barkema, H.W.; Polachek, A.J.; Ganshorn, H.; Sharma, N.; Kellner, J.D.; et al. Restricting the Use of Antibiotics in Food-Producing Animals and Its Associations with Antibiotic Resistance in Food-Producing Animals and Human Beings: A Systematic Review and Meta-Analysis. Lancet Planet. Health 2017, 1, e316–e327. [Google Scholar] [CrossRef]
- DANMAP (Danish Integrated Antimicrobial Resistance Monitoring and Research Programme). DANMAP 2019—Use of Antimicrobial Agents and Occurrence of Antimicrobial Resistance in Bacteria From Food Animals, Food and Humans in Denmark. 2020. Available online: https://www.danmap.org/reports/2019 (accessed on 23 January 2021).
- The Netherlands Veterinary Medicines Institute. Usage of Antibiotics in Agricultural Livestock in The Netherlands in 2018. Trends and Benchmarking of Livestock Farms and Veterinarians. 2020. Available online: https://cdn.i-pulse.nl/autoriteitdiergeneesmiddelen/userfiles/EN/SDa-rapporten/sda-rapport-usage-of-antibiotics-in-agricultural-livestock-in-2019-corr-fig5b.(1).pdf (accessed on 23 January 2021).
- Belvet-SAC. Belgian Veterinary Surveillance of Antibacterial Consumption National Consumption Report 2019. 2020. Available online: https://belvetsac.ugent.be/BelvetSac_report_2019.pdf (accessed on 23 January 2021).
- Sjölund, M.; Postma, M.; Collineau, L.; Lösken, S.; Backhans, A.; Belloc, C.; Emanuelson, U.; Beilage, E.G.; Stärk, K.; Dewulf, J. Quantitative and Qualitative Antimicrobial Usage Patterns in Farrow-to-Finish Pig Herds in Belgium, France, Germany and Sweden. Prev. Vet. Med. 2016, 130, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Sarrazin, S.; Joosten, P.; Gompel, L.V.; Luiken, R.E.C.E.; Mevius, D.J.; Wagenaar, J.A.; Heederik, D.J.J.J.; Dewulf, J. Quantitative and Qualitative Analysis of Antimicrobial Usage Patterns in 180 Selected Farrow-to-Finish Pig Farms from Nine European Countries Based on Single Batch and Purchase Data. J. Antimicrob. Chemother. 2019, 74, 807–816. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, S.; Kehrenberg, C.; Walsh, T.R. Use of Antimicrobial Agents in Veterinary Medicine and Food Animal Production. Int. J. Antimicrob. Agents 2001, 17, 431–437. [Google Scholar] [CrossRef]
- Prescott, J.F. Antimicrobial Use in Food and Companion Animals. Anim. Health Res. Rev. 2008, 9, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Burch, D.G. Antimicrobial drug use in swine. In Antimicrobial Therapy in Veterinary Medicine; Giguère, S., Prescott, J.F., Dowling, P.M., Eds.; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 2013; pp. 553–568. ISBN 9780470963029. [Google Scholar]
- Jensen, V.; Eorg, H.; Aarestrup, F. Indications and Patterns of Therapeutic Use of Antimicrobial Agents in the Danish Pig Production from 2002 to 2008. J. Vet. Pharmacol. Ther. 2012, 35, 33–46. [Google Scholar] [CrossRef]
- Van Rennings, L.; von Münchhausen, C.; Ottilie, H.; Hartmann, M.; Merle, R.; Honscha, W.; Käsbohrer, A.; Kreienbrock, L. Cross-Sectional Study on Antibiotic Usage in Pigs in Germany. PLoS ONE 2015, 10, e0119114. [Google Scholar] [CrossRef] [Green Version]
- Collineau, L.; Bougeard, S.; Backhans, A.; Dewulf, J.; Emanuelson, U.; Beilage, G.E.; Lehébel, A.; Lösken, S.; Postma, M.; Sjölund, M.; et al. Application of Multiblock Modelling to Identify Key Drivers for Antimicrobial Use in Pig Production in Four European Countries. Epidemiol. Infect. 2018, 146, 1003–1014. [Google Scholar] [CrossRef] [Green Version]
- Kruse, A.; Kristensen, C.; Lavlund, U.; Stege, H. Antimicrobial Prescription Data in Danish National Database Validated against Treatment Records in Organic Pig Farms and Analysed for Associations with Lesions Found at Slaughter. BMC Vet. Res. 2019, 15, 218. [Google Scholar] [CrossRef]
- Vieira, A.R.; Pires, S.M.; Houe, H.; Emborg, H.-D. Trends in Slaughter Pig Production and Antimicrobial Consumption in Danish Slaughter Pig Herds, 2002–2008. Epidemiol. Infect. 2010, 139, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Van der Fels-Klerx, H.; Puister-Jansen, L.; van Asselt, E.; Burgers, S. Farm Factors Associated with the Use of Antibiotics in Pig Production. J. Anim. Sci. 2011, 89, 1922–1929. [Google Scholar] [CrossRef] [Green Version]
- Hemme, M.; Ruddat, I.; Hartmann, M.; Werner, N.; van Rennings, L.; Käsbohrer, A.; Kreienbrock, L. Antibiotic Use on German Pig Farms—A Longitudinal Analysis for 2011, 2013 and 2014. PLoS ONE 2018, 13, e0199592. [Google Scholar] [CrossRef] [PubMed]
- Arnold, C.; Schüpbach-Regula, G.; Hirsiger, P.; Malik, J.; Scheer, P.; Sidler, X.; Spring, P.; Peter-Egli, J.; Harisberger, M. Risk Factors for Oral Antimicrobial Consumption in Swiss Fattening Pig Farms—A Case–Control Study. Porc. Health Manag. 2016, 2, 5. [Google Scholar] [CrossRef] [Green Version]
- Raasch, S.; Postma, M.; Dewulf, J.; Stärk, K.; Beilage, E.G. Association between Antimicrobial Usage, Biosecurity Measures as Well as Farm Performance in German Farrow-to-Finish Farms. Porc. Health Manag. 2018, 4, 30. [Google Scholar] [CrossRef] [PubMed]
- Postma, M.; Backhans, A.; Collineau, L.; Loesken, S.; Sjölund, M.; Belloc, C.; Emanuelson, U.; Beilage, E.G.; Nielsen, E.; Stärk, K.D.; et al. Evaluation of the Relationship between the Biosecurity Status, Production Parameters, Herd Characteristics and Antimicrobial Usage in Farrow-to-Finish Pig Production in Four EU Countries. Porc. Health Manag. 2016, 2, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laanen, M.; Persoons, D.; Ribbens, S.; de Jong, E.; Callens, B.; Strubbe, M.; Maes, D.; Dewulf, J. Relationship between Biosecurity and Production/Antimicrobial Treatment Characteristics in Pig Herds. Vet. J. 2013, 198, 508–512. [Google Scholar] [CrossRef]
- Casal, J.; Mateu, E.; Mejía, W.; Martín, M. Factors Associated with Routine Mass Antimicrobial Usage in Fattening Pig Units in a High Pig-Density Area. Vet. Res. 2007, 38, 481–492. [Google Scholar] [CrossRef] [Green Version]
- EMA (European Medicines Agency); EFSA (European Food Safety Authority). EMA and EFSA Joint Scientific Opinion on measures to reduce the need to use antimicrobial agents in animal husbandry in the European Union, and the resulting impacts on food safety (RONAFA). EFSA J. 2017, 15, 4666. [Google Scholar] [CrossRef]
- Postma, M.; Stärk, K.; Sjölund, M.; Backhans, A.; Beilage, E.; Lösken, S.; Belloc, C.; Collineau, L.; Iten, D.; Visschers, V.; et al. Alternatives to the Use of Antimicrobial Agents in Pig Production: A Multi-Country Expert-Ranking of Perceived Effectiveness, Feasibility and Return on Investment. Prev. Vet. Med. 2015, 118, 457–466. [Google Scholar] [CrossRef]
- Dupont, N.; Diness, L.H.; Fertner, M.; Kristensen, C.S.; Stege, H. Antimicrobial Reduction Measures Applied in Danish Pig Herds Following the Introduction of the “Yellow Card” Antimicrobial Scheme. Prev. Vet. Med. 2017, 138, 9–16. [Google Scholar] [CrossRef]
- Coyne, L.A.; Latham, S.M.; Dawson, S.; Donald, I.J.; Pearson, R.B.; Smith, R.F.; Williams, N.J.; Pinchbeck, G.L. Exploring Perspectives on Antimicrobial Use in Livestock: A Mixed-Methods Study of UK Pig Farmers. Front. Vet. Sci. 2019, 6, 257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bak, H.; Rathkjen, P.H. Reduced Use of Antimicrobials after Vaccination of Pigs against Porcine Proliferative Enteropathy in a Danish SPF Herd. Acta Vet. Scand. 2009, 51, 1. [Google Scholar] [CrossRef] [Green Version]
- Kristensen, C.; Baadsgaard, N.; Toft, N. A Meta-Analysis Comparing the Effect of PCV2 Vaccines on Average Daily Weight Gain and Mortality Rate in Pigs from Weaning to Slaughter. Prev. Vet. Med. 2011, 98, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Sacristán, R.D.P.; Michiels, A.; Martens, M.; Haesebrouck, F.; Maes, D. Efficacy of Vaccination against Actinobacillus Pleuropneumoniae in Two Belgian Farrow-to-Finish Pig Herds with a History of Chronic Pleurisy. Vet. Rec. 2014, 174, 302. [Google Scholar] [CrossRef] [PubMed]
- Temtem, C.; Kruse, A.; Nielsen, L.; Pedersen, K.; Alban, L. Comparison of the Antimicrobial Consumption in Weaning Pigs in Danish Sow Herds with Different Vaccine Purchase Patterns during 2013. Porc. Health Manag. 2016, 2, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kruse, A.; Kristensen, C.; Nielsen, L.; Alban, L. A Register-Based Study on Associations between Vaccination, Antimicrobial Use and Productivity in Conventional Danish Finisher Pig Herds during 2011 to 2014. Prev. Vet. Med. 2019, 164, 33–40. [Google Scholar] [CrossRef]
- Backhans, A.; Sjölund, M.; Lindberg, A.; Emanuelson, U. Antimicrobial Use in Swedish Farrow-to-Finish Pig Herds Is Related to Farmer Characteristics. Porc. Health Manag. 2016, 2, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Visschers, V.; Postma, M.; Sjölund, M.; Backhans, A.; Collineau, L.; Loesken, S.; Belloc, C.; Dewulf, J.; Emanuelson, U.; Beilage, G.E.; et al. Higher Perceived Risk of Antimicrobials Is Related to Lower Antimicrobial Usage among Pig Farmers in Four European Countries. Vet. Rec. 2016, 179, 490. [Google Scholar] [CrossRef]
- Moreno, M.A. Opinions of Spanish Pig Producers on the Role, the Level and the Risk to Public Health of Antimicrobial Use in Pigs. Res. Vet. Sci. 2014, 97, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Visschers, V.H.M.; Backhans, A.; Collineau, L.; Iten, D.; Loesken, S.; Postma, M.; Belloc, C.; Dewulf, J.; Emanuelson, U.; Grosee Beilage, E.; et al. Perceptions of Antimicrobial Usage, Antimicrobial Resistance and Policy Measures to Reduce Antimicrobial Usage in Convenient Samples of Belgian, French, German, Swedish and Swiss Pig Farmers. Prev. Vet. Med. 2015, 119, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Diana, A.; Snijders, S.; Rieple, A.; Boyle, L. Why Do Irish Pig Farmers Use Medications? Barriers for Effective Reduction of Antimicrobials in Irish Pig Production. Ir. Vet. J. 2021, 74, 12. [Google Scholar] [CrossRef]
- Ström, G.; Boqvist, S.; Albihn, A.; Fernström, L.-L.; Djurfeldt, A.A.; Sokerya, S.; Sothyra, T.; Magnusson, U. Antimicrobials in Small-Scale Urban Pig Farming in a Lower Middle-Income Country—Arbitrary Use and High Resistance Levels. Antimicrob. Resist. Infect. Control 2018, 7, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Isomura, R.; Matsuda, M.; Sugiura, K. An Epidemiological Analysis of the Level of Biosecurity and Animal Welfare on Pig Farms in Japan and Their Effect on the Use of Veterinary Antimicrobials. J. Vet. Med. Sci. 2018, 80, 18–0287. [Google Scholar] [CrossRef] [Green Version]
- Danish Veterinary and Food Administration. Guideline for Prescribing Antimicrobial for Pigs. 2018. Available online: https://www.foedevarestyrelsen.dk/english/Animal/AnimalHealth/Veterinary_medicine/Documents/Antimicrobial%20guideline%20for%20pigs%20Denmark%202018.pdf (accessed on 23 August 2021).
- AMCRA. Vade-Mecum Pour Un Usage Responsable Des Produits Antibacteriens Chez Les Porcs. 2021. Available online: https://formularium.amcra.be/a/3 (accessed on 23 August 2021).
- Jensen, V.F.; Jorsal, S.-E.L.; Toft, N. A Cross-Sectional Study of Oral Antibacterial Treatment Patterns in Relation to Specific Diarrhoeal Pathogens in Weaner Pigs. Vet. Microbiol. 2017, 203, 18–27. [Google Scholar] [CrossRef] [Green Version]
- Sarrazin, S. Analysis of herd level antimicrobial usage in pigs and poultry in 9 European countries based on EFFORT data. Presented to Quantification, Benchmarking and Stewardship of Veterinary Antimicrobial Usage. In Proceedings of the First International Conference, Ghent University, Ghent, Belgium, 27 February 2018; Available online: https://aacting.org/first-aacting-conference/ (accessed on 4 July 2021).
- O’Neill, L.; da Costa, M.; Leonard, F.C.; Gibbons, J.; Díaz, J.; McCutcheon, G.; Manzanilla, E. Quantification, Description and International Comparison of Antimicrobial Use on Irish Pig Farms. Porc. Heal Manag. 2020, 6, 30. [Google Scholar] [CrossRef]
- Central Statistics Office (CSO). Pig Survey June 2017. 2017. Available online: https://www.cso.ie/en/releasesandpublications/er/ps/pigsurveyjune2017/ (accessed on 2 December 2020).
- Timmerman, T.; Dewulf, J.; Catry, B.; Feyen, B.; Opsomer, G.; de Kruif, A.; Maes, D. Quantification and Evaluation of Antimicrobial Drug Use in Group Treatments for Fattening Pigs in Belgium. Prev. Vet. Med. 2006, 74, 251–263. [Google Scholar] [CrossRef]
- European Medicines Agency. Defined Daily Doses for Animals (DDDvet) and Defined Course Doses for Animals (DCDvet) European Surveillance of Veterinary Antimicrobial Consumption (ESVAC). 2016. Available online: https://www.ema.europa.eu/en/documents/other/defined-daily-doses-animalsdddvet-defined-course-doses-animals-dcdvet-european-surveillance_en.pdf (accessed on 20 March 2020).
- European Medicine Agency. Revised ESVAC Reflection Paper on Collecting Data on Consumption of Antimicrobial Agents per Animal Species, on Technical Units of Measurement and Indicators for Reporting Consumption of Antimicrobial Agents in Animals. 2013. Available online: https://www.ema.europa.eu/documents/scientific-guideline/revised-europeansurveillance-veterinary-antimicrobial-consumption-esvac-reflection-papercollecting_en.pdf (accessed on 2 August 2019).
- Rodrigues da Costa, M.; Gasa, J.; Díaz, J.A.C.; Postma, M.; Dewulf, J.; McCutcheon, G.; Manzanilla, E.G. Using the Biocheck.UGentTM Scoring Tool in Irish Farrow-to-Finish Pig Farms: Assessing Biosecurity and Its Relation to Productive Performance. Porc. Health Manag. 2019, 5, 4. [Google Scholar] [CrossRef] [Green Version]
- Rodrigues da Costa, M.; Fitzgerald, R.; Manzanilla, E.; O’Shea, H.; Moriarty, J.; McElroy, M.C.; Leonard, F. A Cross-Sectional Survey on Respiratory Disease in a Cohort of Irish Pig Farms. Ir. Vet. J. 2020, 73, 24. [Google Scholar] [CrossRef]
- Madec, F.; Derrien, H. Fréquence, intensité et localisation des lésions pulmonaires chez le porc charcutier: Résultats d’une premiére série d’observations en abattoir. Journées Rech. Porc. Fr. 1981, 13, 231–236. [Google Scholar]
- Dottori, M.; Nigrelli, A.D.; Bonilauri, P.; Merialdi, G.; Gozio, S.; Cominotti, F. Proposta Di Un Nuovo Sistema Di Punteggiatura Delle Pleuriti Suine in Sede Di Macellazione. La Griglia, S.P.E.S. (Slaughterhouse Pleuritis Evaluation System) O. Large Anim. Rev. 2007, 13, 161–165. [Google Scholar]
- Merialdi, G.; Dottori, M.; Bonilauri, P.; Luppi, A.; Gozio, S.; Pozzi, P.; Spaggiari, B.; Martelli, P. Survey of Pleuritis and Pulmonary Lesions in Pigs at Abattoir with a Focus on the Extent of the Condition and Herd Risk Factors. Vet. J. 2012, 193, 234–239. [Google Scholar] [CrossRef]
- Robinson, N.A.; Loynachan, A.T. Cardiovascular and Hematopoietic Systems. In Diseases of Swine, 11th ed.; Zimmerman, J.J., Karriker, L.A., Ramirez, A., Schwartz, K.J., Stevenson, G.W., Zhang, J., Eds.; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 2019; pp. 221–233. ISBN 9781119350927. [Google Scholar] [CrossRef]
- Thomson, J.R.; Friendship, R.M. Digestive system. In Diseases of Swine, 11th ed.; Zimmerman, J.J., Karriker, L.A., Ramirez, A., Schwartz, K.J., Stevenson, G.W., Zhang, J., Eds.; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 2019; pp. 234–263. ISBN 9781119350927. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: https://www.R-project.org/ (accessed on 20 November 2020).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; Available online: https://ggplot2.tidyverse.org (accessed on 4 March 2021).
- Hanbo Chen. VennDiagram: Generate High-Resolution Venn and Euler Plots. R Package Version 1.6.20. 2018. Available online: https://CRAN.R-project.org/package=VennDiagram (accessed on 4 March 2021).
- Marquer, P.; Rabade, T.; Forti, R. Pig farming in the European Union: Considerable variations from one member state to another. Eurostat Statis Focus. 2014, 15, 1–13. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php?title=Archive:Pig_farming_sector_-_statistical_portrait_2014 (accessed on 26 September 2021).
- World Health Organization (WHO). Critically Important Antimicrobials for Human Medicine, 6th Revision 2018. World Health Organization: Geneva, Switzerland, 2019. Available online: https://apps.who.int/iris/bitstream/handle/10665/312266/9789241515528-eng.pdf?ua=1 (accessed on 1 April 2020).
- European Medicines Agency. Categorisation of Antibiotics for Use in Animals for Prudent and Responsible Use. 2019. Available online: https://www.ema.europa.eu/en/documents/report/categorisation-antibiotics-use-animalsprudent-responsible-use_en.pdf (accessed on 2 February 2020).
- Regulation (EU) 2019/4 of the European Parliament and of the Council of 11 December 2018 on the Manufacture, Placing on the Market and Use of Medicated Feed, Amending Regulation (EC) No 183/2005 of the European Parliament and of the Council and Repealing Council Directive 90/167/EEC (Text with EEA Relevance). 2019, pp. 1–23. Available online: http://data.europa.eu/eli/reg/2019/4/oj (accessed on 2 July 2021).
- Rodrigues da Costa, M. Evaluation of Current Feeding and Management Practices in Irish Pig Production and Future Strategies for Improvement. Ph.D. Thesis, Universitat Autònoma de Barcelona, Barcelona, Spain, 2018. Available online: http://hdl.handle.net/10803/666618 (accessed on 3 March 2021).
- Stege, H.; Jensen, T.K.; Møller, K.; Bækbo, P.; Jorsal, S.E. Risk Factors for Intestinal Pathogens in Danish Finishing Pig Herds. Prev. Vet. Med. 2001, 50, 153–164. [Google Scholar] [CrossRef]
- Campbell, J.M.; Crenshaw, J.D.; Polo, J. The Biological Stress of Early Weaned Piglets. J Anim. Sci. Biotechnol. 2013, 4, 19. [Google Scholar] [CrossRef] [Green Version]
- Moeser, A.J.; Pohl, C.S.; Rajput, M. Weaning Stress and Gastrointestinal Barrier Development: Implications for Lifelong Gut Health in Pigs. Anim. Nutr. 2017, 3, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Gebhardt, J.T.; Tokach, M.D.; Dritz, S.S.; DeRouchey, J.M.; Woodworth, J.C.; Goodband, R.D.; Henry, S.C. Postweaning Mortality in Commercial Swine Production II: Review of Infectious Contributing Factors. Transl. Anim. Sci. 2020, 4, txaa052. [Google Scholar] [CrossRef] [PubMed]
- Gebhardt, J.T.; Tokach, M.D.; Dritz, S.S.; DeRouchey, J.M.; Woodworth, J.C.; Goodband, R.D.; Henry, S.C. Postweaning Mortality in Commercial Swine Production. I: Review of Non-Infectious Contributing Factors. Transl. Anim. Sci. 2020, 4, txaa068. [Google Scholar] [CrossRef] [PubMed]
- Stevens, K.; Gilbert, J.; Strachan, W.; Robertson, J.; Johnston, A.; Pfeiffer, D. Characteristics of Commercial Pig Farms in Great Britain and Their Use of Antimicrobials. Vet. Rec. 2007, 161, 45. [Google Scholar] [CrossRef] [PubMed]
- Scali, F.; Santucci, G.; Maisano, A.M.; Giudici, F.; Guadagno, F.; Tonni, M.; Amicabile, A.; Formenti, N.; Giacomini, E.; Lazzaro, M.; et al. The Use of Antimicrobials in Italian Heavy Pig Fattening Farms. Antibiotics 2020, 9, 892. [Google Scholar] [CrossRef]
- Filippitzi, M.; Kruse, B.A.; Postma, M.; Sarrazin, S.; Maes, D.; Alban, L.; Nielsen, L.; Dewulf, J. Review of Transmission Routes of 24 Infectious Diseases Preventable by Biosecurity Measures and Comparison of the Implementation of These Measures in Pig Herds in Six European Countries. Transbound. Emerg. Dis. 2018, 65, 381–398. [Google Scholar] [CrossRef]
- Collineau, L.; Rojo-Gimeno, C.; Léger, A.; Backhans, A.; Loesken, S.; Nielsen, E.O.; Postma, M.; Emanuelson, U.; Beilage, E.; Sjölund, M.; et al. Herd-Specific Interventions to Reduce Antimicrobial Usage in Pig Production without Jeopardising Technical and Economic Performance. Prev. Vet. Med. 2017, 144, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Raasch, S.; Collineau, L.; Postma, M.; Backhans, A.; Sjölund, M.; Belloc, C.; Emanuelson, U.; Beilage, E.; Stärk, K.; Dewulf, J.; et al. Effectiveness of Alternative Measures to Reduce Antimicrobial Usage in Pig Production in Four European Countries. Porc. Heal. Manag. 2020, 6, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- USDA. Swine 2012: Part I: Baseline Reference of Swine Health and Management in the United States, 2012. USDA-APHIS-VS, CEAH, Fort Collins, CO. 2015; #663.0814. Available online: https://www.aphis.usda.gov/animal_health/nahms/swine/downloads/swine2012/Swine2012_dr_PartI.pdf (accessed on 16 March 2021).
- Yaeger, M.J.; Alstine, W.G. Respiratory system. In Diseases of Swine, 11th ed.; Zimmerman, J.J., Karriker, L.A., Ramirez, A., Schwartz, K.J., Stevenson, G.W., Zhang, J., Eds.; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 2019; pp. 393–407. ISBN 9781119350927. [Google Scholar] [CrossRef]
- Gottschalk, M.; Segura, M. Streptococcosis. In Diseases of Swine, 11th ed.; Zimmerman, J.J., Karriker, L.A., Ramirez, A., Schwartz, K.J., Stevenson, G.W., Zhang, J., Eds.; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 2019; pp. 934–950. ISBN 9781119350927. [Google Scholar] [CrossRef]
- Aragon, V.; Segales, J.; Tucker, A.W. Glässer’s disease. In Diseases of Swine, 11th ed.; Zimmerman, J.J., Karriker, L.A., Ramirez, A., Schwartz, K.J., Stevenson, G.W., Zhang, J., Eds.; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 2019; pp. 844–853. ISBN 9781119350927. [Google Scholar] [CrossRef]
- Steenhard, N.R.; Jungersen, G.; Kokotovic, B.; Beshah, E.; Dawson, H.D.; Urban, J.F.; Roepstorff, A.; Thamsborg, S.M. Ascaris suum Infection Negatively Affects the Response to a Mycoplasma Hyopneumoniae Vaccination and Subsequent Challenge Infection in Pigs. Vaccine 2009, 27, 5161–5169. [Google Scholar] [CrossRef] [PubMed]
- Callens, B.; Persoons, D.; Maes, D.; Laanen, M.; Postma, M.; Boyen, F.; Haesebrouck, F.; Butaye, P.; Catry, B.; Dewulf, J. Prophylactic and Metaphylactic Antimicrobial Use in Belgian Fattening Pig Herds. Prev. Vet. Med. 2012, 106, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Cameron-Veas, K.; Solà-Ginés, M.; Moreno, M.A.; Fraile, L.; Migura-Garcia, L. Impact of the Use of β-Lactam Antimicrobials on the Emergence of Escherichia Coli Isolates Resistant to Cephalosporins under Standard Pig-Rearing Conditions. Appl. Environ. Microbiol. 2015, 81, 1782–1787. [Google Scholar] [CrossRef] [Green Version]
- Kruse, A.B.; Knegt, L.V.; de Nielsen, L.R.; Alban, L. No Clear Effect of Initiating Vaccination against Common Endemic Infections on the Amounts of Prescribed Antimicrobials for Danish Weaner and Finishing Pigs during 2007–2013. Front. Vet. Sci. 2017, 3, 120. [Google Scholar] [CrossRef] [Green Version]
- Holt, H.R.; Alarcon, P.; Velasova, M.; Pfeiffer, D.U.; Wieland, B. BPEX Pig Health Scheme: A Useful Monitoring System for Respiratory Disease Control in Pig Farms? BMC Vet. Res. 2011, 7, 82. [Google Scholar] [CrossRef] [Green Version]
- Opriessnig, T.; Giménez-Lirola, L.; Halbur, P. Polymicrobial Respiratory Disease in Pigs. Anim. Health Res. Rev. 2011, 12, 133–148. [Google Scholar] [CrossRef]
- Díaz, J.A.C.; Fitzgerald, R.M.; Shalloo, L.; da Costa, M.R.; Niemi, J.; Leonard, F.C.; Kyriazakis, I.; Manzanilla, E.G. Financial Analysis of Herd Status and Vaccination Practices for Porcine Reproductive and Respiratory Syndrome Virus, Swine Influenza Virus, and Mycoplasma Hyopneumoniae in Farrow-to-Finish Pig Farms Using a Bio-Economic Simulation Model. Front. Vet. Sci. 2020, 7, 556674. [Google Scholar] [CrossRef]
- Regulation (EU). 2019/6 of the European Parliament and of the Council of 11 December 2018 on Veterinary Medicinal Products and Repealing Directive 2001/82/EC (Text with EEA Relevance). 2019, pp. 43–167. Available online: http://data.europa.eu/eli/reg/2019/6/oj (accessed on 4 April 2020).
- UK-VARSS. UK Veterinary Antibiotic Resistance and Sales Surveillance Report (UK-VARSS 2019). Veterinary Medicines Directorate. 2020. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/950126/UK-VARSS_2019_Report__2020-TPaccessible.pdf (accessed on 20 December 2020).
- Aarestrup, F. Sustainable Farming: Get Pigs off Antibiotics. Nature 2012, 486, 465–466. [Google Scholar] [CrossRef]
- Speksnijder, D.; Mevius, D.; Bruschke, C.; Wagenaar, J. Reduction of Veterinary Antimicrobial Use in the Netherlands. The Dutch Success Model. Zoonoses Public Health 2015, 62, 79–87. [Google Scholar] [CrossRef]
- Rojo-Gimeno, C.; Postma, M.; Dewulf, J.; Hogeveen, H.; Lauwers, L.; Wauters, E. Farm-Economic Analysis of Reducing Antimicrobial Use Whilst Adopting Improved Management Strategies on Farrow-to-Finish Pig Farms. Prev. Vet. Med. 2016, 129, 74–87. [Google Scholar] [CrossRef]
- Postma, M.; Vanderhaeghen, W.; Sarrazin, S.; Maes, D.; Dewulf, J. Reducing Antimicrobial Usage in Pig Production without Jeopardizing Production Parameters. Zoonoses Public Health 2017, 64, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Diana, A.; Boyle, L.A.; Leonard, F.C.; Carroll, C.; Sheehan, E.; Murphy, D.; Manzanilla, E.G. Removing Prophylactic Antibiotics from Pig Feed: How Does It Affect Their Performance and Health? BMC Vet. Res. 2019, 15, 67. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Commission Implementing Decision of 26.6.2017 Concerning, in the Framework of Article 35 of Directive 2001/82/EC of the European Parliament and of the Council, the Marketing Authorisations for Veterinary Medicinal Products Containing “Zinc Oxide” to be Administered Orally to Food Producing Species. 2017. Available online: https://ec.europa.eu/health/documents/community-register/2017/20170626136754/dec_136754_en.pdf (accessed on 16 September 2021).
- Nielsen, C.; Kongsted, H.; Sørensen, J.; Krogh, M. Antibiotic and Medical Zinc Oxide Usage in Danish Conventional and Welfare-Label Pig Herds in 2016–2018. Prev Vet Med 2021, 189, 105283. [Google Scholar] [CrossRef] [PubMed]
- Maes, D.; Deluyker, H.; Verdonck, M.; Kruif, A.; Ducatelle, R.; Castryck, F.; Miry, C.; Vrijens, B. Non-infectious Factors Associated with Macroscopic and Microscopic Lung Lesions in Slaughter Pigs from Farrow-to-finish Herds. Vet. Rec. 2001, 148, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Agostini, P.; Fahey, A.; Manzanilla, E.; O’Doherty, J.; de Blas, C.; Gasa, J. Management Factors Affecting Mortality, Feed Intake and Feed Conversion Ratio of Grow-Finishing Pigs. Animal 2014, 8, 1312–1318. [Google Scholar] [CrossRef] [Green Version]
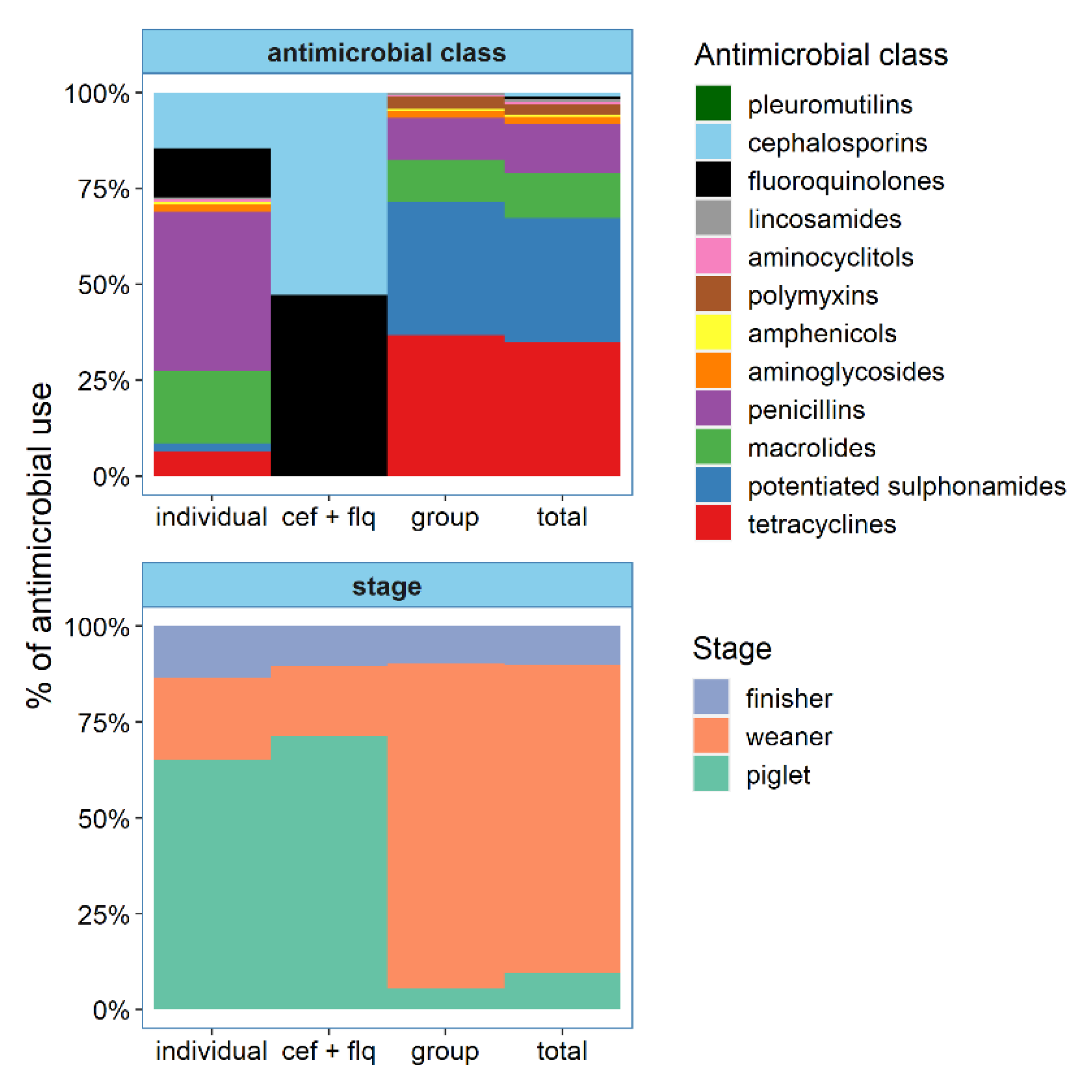
| Antimicrobial Class | Farms with Use | Mean (SD) | Median (Range) |
|---|---|---|---|
| Oral group treatments | |||
| Tetracylines | 38 (73.1%) | 7.7 (12) | 1.2 (0–49.1) |
| Potentiated sulphonamides | 17 (32.7%) | 6.6 (22.8) | 0 (0–150.1) |
| Penicillins | 36 (69.2%) | 2.7 (5.5) | 0.9 (0–29.7) |
| Macrolides | 23 (44.2%) | 2.3 (4.4) | 0 (0–23.6) |
| Lincosamides | 10 (19.2%) | 0.1 (0.5) | 0 (0–2.9) |
| Amphenicols | 7 (13.5%) | 0.1 (0.4) | 0 (0–2.5) |
| Aminoglycosides | 27 (51.9%) | 0.5 (1.3) | 0 (0–8.9) |
| Aminocyclitols * | 10 (19.2%) | 0.1 (0.3) | 0 (0–1.9) |
| Polymyxins | 13 (25%) | 0.8 (2.6) | 0 (0–13) |
| Total oral group treatments (TIgroup) | 50 (96.2%) | 21 (28.5) | 13.3 (0–167.8) |
| Individual treatments | |||
| Tetracyclines | 22 (42.3%) | 0.1 (0.2) | 0 (0–0.9) |
| Potentiated sulphonamides | 8 (15.4%) | 0.1 (0.3) | 0 (0–2.2) |
| Penicillins | 52 (100%) | 0.6 (0.6) | 0.4 (0–2) |
| Macrolides | 18 (34.6%) | 0.2 (0.6) | 0 (0–4) |
| Lincosamides | 21 (40.4%) | 0 (0) | 0 (0–0.2) |
| Amphenicols | 5 (9.6%) | 0 (0) | 0 (0–0.3) |
| Aminoglycosides | 14 (26.9%) | 0 (0.1) | 0 (0–0.3) |
| Aminocyclitols | 23 (44.2%) | 0 (0) | 0 (0–0.2) |
| Fluoroquinolones | 45 (86.5%) | 0.2 (0.3) | 0.1 (0–1.2) |
| Cephalosporins | 12 (23.1%) | 0.2 (0.8) | 0 (0–4.6) |
| Pleuromutilins | 1 (1.9%) | 0 (0) | 0 (0–0) |
| Total individual treatments (TIindividual) | 52 (100%) | 1.5 (1.6) | 1.1 (0.1–11.4) |
| Total antimicrobial use (TItotal) | 52 (100%) | 22.5 (28.6) | 14.2 (0.2–169.1) |
| Cephalopsorin and/or fluoroquinolone use (TIceflq) | 47 (90.4%) | 0.5 (0.9) | 0.2 (0–5.8) |
| Outcome Variable | Predictor Variables | Estimate a | Std. Error | Back-Transformed Estimate (95% CI) | p |
|---|---|---|---|---|---|
| LOG TItotal | Intercept | 3.28 | 0.936 | 26.48 | |
| adjusted R2 = 0.53 | Finisher mortality_2016 | 0.28 | 0.135 | 1.32 (1.01–1.74) | 0.043 |
| p < 0.001 | Home milling (ref = no) | −1.08 | 0.265 | 0.34 (0.2–0.58) | 0.000 |
| Weaner stage (days) | −0.03 | 0.013 | 0.97 (0.95–1) | 0.027 | |
| IAv vaccination (ref = no) | 1.06 | 0.272 | 2.88 (1.66–4.97) | 0.000 | |
| Lung abscesses (%) | 0.07 | 0.037 | 1.07 (0.99–1.16) | 0.070 | |
| Pericarditis (%) | 0.06 | 0.031 | 1.06 (1–1.13) | 0.059 | |
| LOG TIgroup | Intercept | 2.93 | 0.832 | 18.72 | |
| adjusted R2 = 0.55 | Finisher mortality | 0.28 | 0.120 | 1.33 (1.04–1.69) | 0.023 |
| p < 0.001 | Home milling (ref = no) | −0.97 | 0.235 | 0.38 (0.23–0.61) | 0.000 |
| Weaner stage (days) | −0.02 | 0.011 | 0.98 (0.95–1) | 0.041 | |
| IAv vaccination (ref = no) | 0.95 | 0.242 | 2.58 (1.58–4.19) | 0.000 | |
| Lung abscesses (%) | 0.07 | 0.033 | 1.07 (1–1.14) | 0.046 | |
| Pericarditis (%) | 0.06 | 0.028 | 1.06 (1–1.12) | 0.037 | |
| LOG TIindividual | Intercept | −0.58 | 0.473 | 0.56 | |
| adjusted R2 = 0.29 | Feed, water and equipment supply b | −0.01 | 0.007 | 0.99 (0.97–1) | 0.048 |
| p < 0.001 | Vermin and bird control b | 0.01 | 0.005 | 1.01 (1–1.02) | 0.018 |
| Piglet prophylaxis (ref = no) c | 0.60 | 0.192 | 1.82 (1.23–2.67) | 0.003 | |
| Liver milk spots (%) | 0.01 | 0.004 | 1.01 (1–1.02) | 0.018 | |
| LOG TIceflq | Intercept | −1.32 | 0.816 | 0.27 | |
| adjusted R2 = 0.37 | Weaner mortality | −0.41 | 0.126 | 0.66 (0.51–0.85) | 0.002 |
| p < 0.001 | Feed, water and equipment supply b | −0.03 | 0.012 | 0.97 (0.95–1) | 0.031 |
| Sow prophylaxis (ref = no) c | 0.72 | 0.378 | 2.06 (0.96–4.41) | ||
| Piglet prophylaxis (ref = no) c | 1.16 | 0.350 | 3.19 (1.58–6.45) | 0.002 | |
| Pericarditis (%) | 0.14 | 0.041 | 1.15 (1.06–1.25) | 0.001 |
| Outcome Variables: Oral Group Treatment with Antimicrobial Class | ||||||||
|---|---|---|---|---|---|---|---|---|
| Tetracyclines | Potentiated Sulphonamides | Penicillins | Macrolides | Lincosamides | Amphenicols | Aminoglycosides | Polymyxins | |
| Predictor Variable | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) |
| Herd characteristics | ||||||||
| Home milling (ref = no) | 0.16 (0.02–0.86) | - | - | - | - | - | - | - |
| Finisher mortality (%) | - | 7.7 (1.7–88.88) | - | 2.81 (1.32–7.18) | - | - | - | - |
| Age at weaning (days) | - | - | - | 1.32 (1.05–1.78) | - | - | - | - |
| Weaner stage (days) | 0.89 (0.78–0.98) | 0.69 (0.45–0.87) | - | - | 0.9 (0.8–0.98) | - | - | - |
| Finisher stage (days) | - | - | - | - | - | - | 1.07 (1–1.16) | - |
| No. of sows per employee | - | - | - | - | - | - | 0.98 (0.95–1.00) | 1.02 (1–1.04) |
| Farmer experience (years) | - | - | - | - | - | - | 1.08 (1.00–1.18) | - |
| Youngest building (years) | - | - | - | - | 1.3 (1.07–1.68) | - | - | - |
| Biosecurity scores | ||||||||
| Feed, water and equipment supply | - | 0.92 (0.82–1.00) | - | - | - | - | - | - |
| Environment and region | - | 0.95 (0.87–1.00) | - | - | - | - | - | - |
| Nursery unit management | 0.93 (0.87–0.99) | - | - | - | - | - | - | - |
| Measures between compartments and use of equipment | - | - | - | - | - | - | - | 0.94 (0.89–0.99) |
| Pluck lesions at slaughter and disease status | ||||||||
| Pleurisy (%) | - | 1.11 (1.03–1.25) | - | - | - | - | - | - |
| Moderate/severe pleurisy (%) | - | - | - | - | - | - | 0.89 (0.81–0.96) | - |
| Scars (%) | 1.13 (1.02–1.3) | - | - | - | - | - | 1.1 (1.02–1.23) | - |
| Lung abscesses (%) | - | - | - | - | - | 1.47 (1.09–2.9) | - | - |
| Pericarditis (%) | - | - | 1.35 (1.11–1.73) | - | - | - | - | 1.19 (1.01–1.44) |
| Liver milk spots (%) | - | - | - | - | - | 1.04 (1–1.11) | - | - |
| IAv status (ref = negative) | - | - | 0.11 (0.01–0.74) | - | - | - | - | - |
| Mhyo status (ref = negative) | - | - | - | - | 134.31 (4.66–>9999) | - | - | - |
| PRRSv status (ref = negative) | - | - | - | - | - | 270.67 (5.26–>9999) | - | - |
| Vaccination and prophylactic antimicrobial use practices | ||||||||
| IAv vaccination (ref = no) | 12.53 (1.72–183.73) | 106.48 (5.14–>9999) | - | 24.11 (4.8–191.38) | - | - | - | - |
| Piglet prophylaxis (ref = no) | - | - | 10.18 (2.22–64.81) | - | - | 11.3 (1.09–367.08) | - | - |
| Outcome Variables: Individual Treatment with Antimicrobial Class | ||||||||
|---|---|---|---|---|---|---|---|---|
| Tetracyclines | Potentiated Sulphonamides | Macrolides | Lincosamides | Aminoglycosides | Aminocyclitols | Fluoroquinolones | Cephalosporins | |
| Predictor variable | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) |
| Herd characteristics | ||||||||
| Piglet mortality (%) | - | - | 0.77 (0.57–0.98) | - | - | - | - | - |
| Weaner mortality (%) | 1.78 (1.04–3.66) | - | - | - | - | - | - | - |
| Finisher mortality (%) | - | - | 0.52 (0.22–1.04) | - | - | - | - | - |
| Average daily gain (g) | - | - | - | 1.02 (1–1.03) | - | - | - | 0.99 (0.97–1) |
| Feed conversion ratio | 0 (0–0.15) | - | - | - | - | - | - | - |
| Pigs per sow per year | - | - | - | - | 0.43 (0.2–0.73) | 0.61 (0.38–0.89) | - | - |
| Weaning age (days) | - | 0.72 (0.5–0.95) | - | - | 0.79 (0.6–0.97) | - | - | - |
| Total rearing period (days) | - | - | - | - | - | 0.91 (0.83–0.97) | - | - |
| No. of sows per employee | - | - | - | - | 1.04 (1.01–1.07) | - | - | 1.03 (1.01–1.06) |
| Biosecurity scores | ||||||||
| Transport of animals, removal of manure and dead animals | - | - | 1.11 (1.03–1.23) | - | - | - | - | - |
| Feed, water and equipment supply | - | - | - | - | - | - | 0.9 (0.8–0.97) | - |
| Vermin and bird control | - | 1.08 (1.02–1.19) | - | - | - | - | - | - |
| Disease management | 0.97 (0.93–1.00) | - | - | - | - | - | - | - |
| Fattening unit management | 1.06 (1.01–1.13) | 1.07 (1.01–1.16) | - | - | - | - | - | - |
| Overall internal biosecurity | - | - | - | - | - | 1.06 (1.01–1.13) | - | - |
| Pluck lesions at slaughter and disease status | ||||||||
| Pericarditis (%) | - | - | - | - | - | 0.82 (0.65–0.98) | 1.59 (1.11–2.67) | - |
| Liver milk spots (%) | 0.96 (0.93–0.99) | - | - | 1.04 (1.01–1.07) | - | - | - | - |
| IAv status (ref = negative) | - | - | - | - | 0.07 (0–0.51) | - | - | - |
| Vaccination and prophylactic antimicrobial use practices | ||||||||
| IAv vaccination (ref = no) | - | - | - | 8.18 (1.8–50.29) | - | - | - | - |
| PRRSv vaccination (ref = no) | - | - | - | 7.27 (1.54–45.87) | - | - | - | - |
| Piglet prophylaxis (ref = no) | - | 0.01 (0–0.21) | - | - | - | - | - | 4.35 (0.96–26.2) |
| Sow prophylaxis (ref = no) | - | - | - | - | - | - | 17.22 (1.15–1032.14) | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
O’Neill, L.; Calderón Díaz, J.A.; Rodrigues da Costa, M.; Oakes, S.; Leonard, F.C.; Manzanilla, E.G. Risk Factors for Antimicrobial Use on Irish Pig Farms. Animals 2021, 11, 2828. https://doi.org/10.3390/ani11102828
O’Neill L, Calderón Díaz JA, Rodrigues da Costa M, Oakes S, Leonard FC, Manzanilla EG. Risk Factors for Antimicrobial Use on Irish Pig Farms. Animals. 2021; 11(10):2828. https://doi.org/10.3390/ani11102828
Chicago/Turabian StyleO’Neill, Lorcan, Julia Adriana Calderón Díaz, Maria Rodrigues da Costa, Sinnead Oakes, Finola C. Leonard, and Edgar García Manzanilla. 2021. "Risk Factors for Antimicrobial Use on Irish Pig Farms" Animals 11, no. 10: 2828. https://doi.org/10.3390/ani11102828






