Development and Evaluation of a Nested PCR for Improved Diagnosis and Genetic Analysis of Peste des Petits Ruminants Virus (PPRV) for Future Use in Nascent PPR Eradication Programme
Abstract
:Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Viruses and Cells
2.2. Primer Design
2.3. RNA Extraction and Real-Time Reverse-Transcription-Polymerase Chain Reaction (RT-qPCR)
2.4. Reverse Transcription (RT), Polymerase Chain Reaction (PCR), Nucleotide (nt) Sequencing
2.5. RNA Standards and Dilution Series Used to Determine the Limit of Detection by RT-PCR
3. Results and Discussion
3.1. Initial Testing
3.2. Lineage I
3.3. Lineage II
3.4. Lineage III
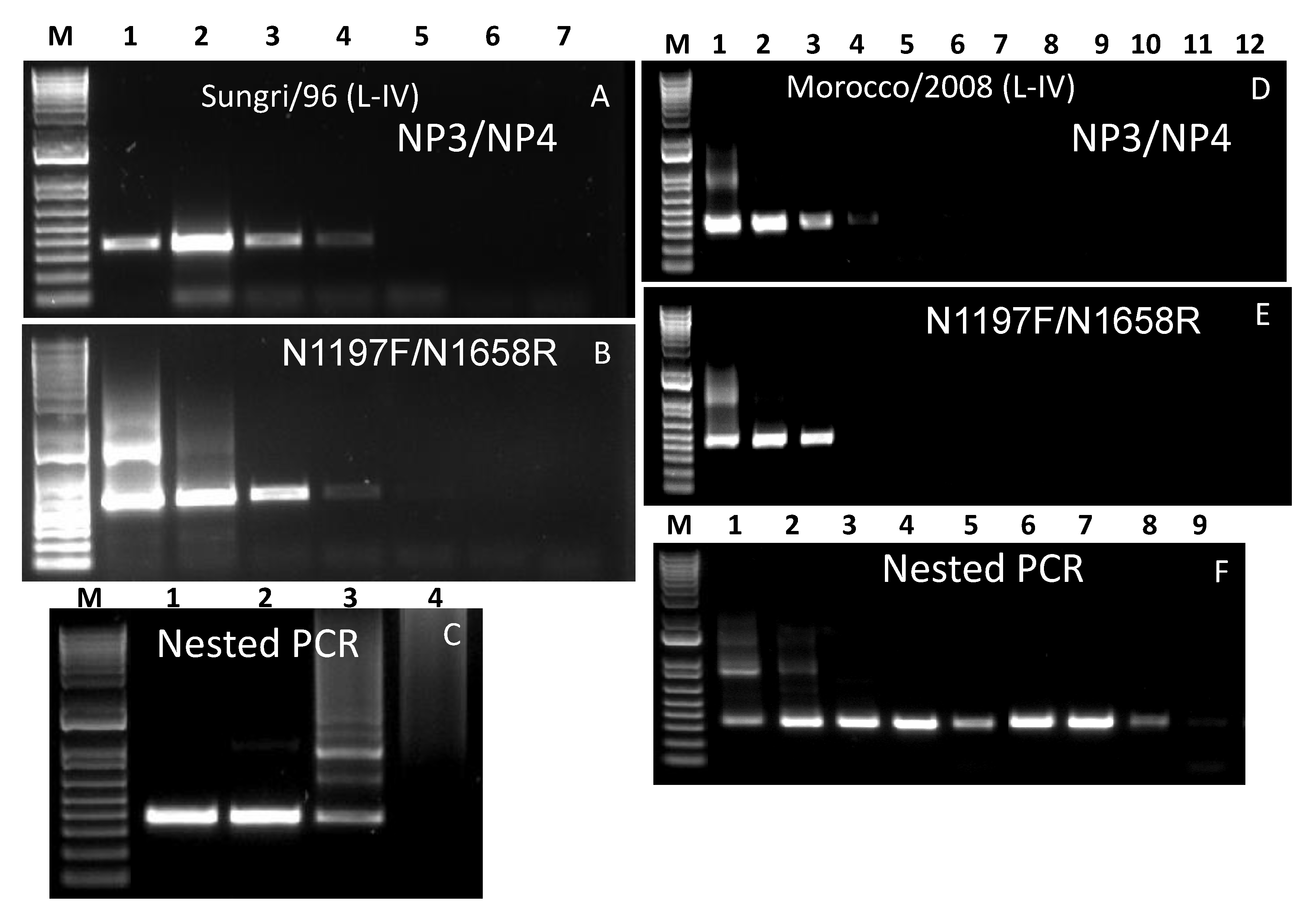
3.5. Lineage IV
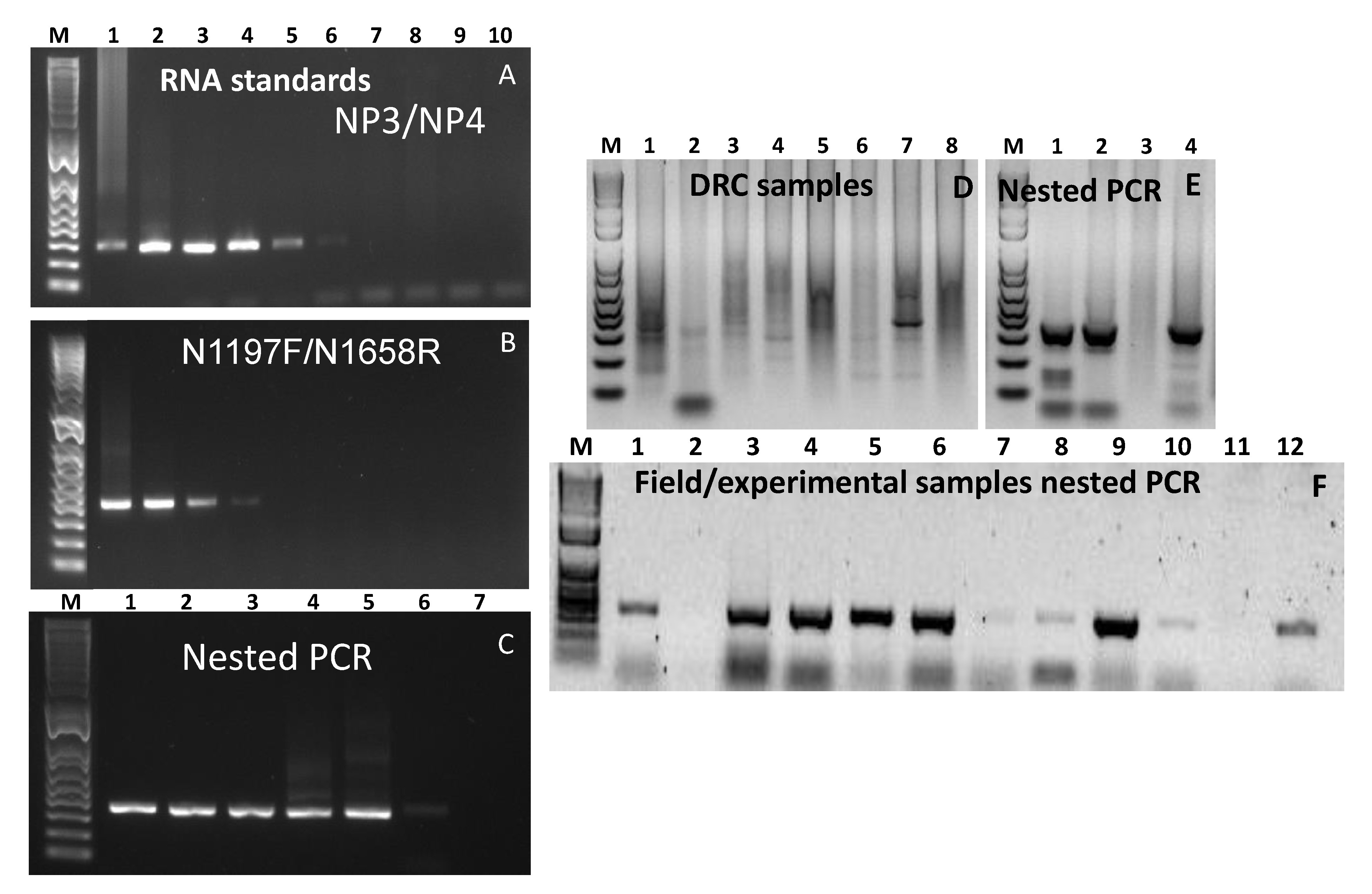
3.6. RNA Standards and Dilution Series Used to Determine Analytical Sensitivity of the Nested PCR Assay
3.7. Testing Clinical Samples from the Field and Animal Experiments
3.8. Testing Environmental Samples
3.9. Specificity Test
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Parida, S.; Muniraju, M.; Mahapatra, M.; Muthuchelvan, D.; Buczkowski, H.; Banyard, A.C. Peste des petits ruminants. Vet. Microbiol. 2015, 181, 90–106. [Google Scholar] [CrossRef] [Green Version]
- Intisar, K.S.; Ali, Y.H.; Haj, M.A.; Sahar, M.A.; Shaza, M.M.; Baraa, A.M.; Ishag, O.M.; Nouri, Y.M.; Taha, K.M.; Nada, E.M.; et al. Peste des petits ruminants infection in domestic ruminants in Sudan. Trop. Anim. Health Prod. 2017, 49, 747–754. [Google Scholar] [CrossRef]
- Schulz, C.; Fast, C.; Schlottau, K.; Hoffmann, B.; Beer, M. Neglected Hosts of Small Ruminant Morbillivirus. Emerg. Infect. Dis. 2018, 24, 2334–2337. [Google Scholar] [CrossRef] [PubMed]
- Pruvot, M.; Fine, A.E.; Hollinger, C.; Strindberg, S.; Damdinjav, B.; Buuveibaatar, B.; Chimeddorj, B.; Bayandonoi, G.; Khishgee, B.; Sandag, B.; et al. Outbreak of Peste des Petits Ruminants among Critically Endangered Mongolian Saiga and Other Wild Ungulates, Mongolia, 2016–2017. Emerg. Infect. Dis. 2020, 26, 51–62. [Google Scholar] [CrossRef]
- Abubakar, M.; Mahapatra, M.; Muniraju, M.; Arshed, M.J.; Khan, E.H.; Banyard, A.C.; Ali, Q.; Parida, S. Serological Detection of Antibodies to Peste des Petits Ruminants Virus in Large Ruminants. Transbound Emerg. Dis. 2017, 64, 513–519. [Google Scholar] [CrossRef] [Green Version]
- Dhar, P.; Sreenivasa, B.P.; Barrett, T.; Corteyn, M.; Singh, R.P.; Bandyopadhyay, S.K. Recent epidemiology of peste des petits ruminants virus (PPRV). Vet. Microbiol. 2002, 88, 153–159. [Google Scholar] [CrossRef]
- Banyard, A.C.; Parida, S.; Batten, C.; Oura, C.; Kwiatek, O.; Libeau, G. Global distribution of peste des petits ruminants virus and prospects for improved diagnosis and control. J. Gen. Virol. 2010, 91, 2885–2897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- FAO OIE. Peste Des Petits Ruminants Global Eradication Programme: Contributing to Food Security, Poverty Alleviation and Resilience; FAO, World Organization for Animal Health: Rome, Italy, 2016. [Google Scholar]
- Jones, B.A.; Mahapatra, M.; Chubwa, C.; Clarke, B.; Batten, C.; Hicks, H.; Henstock, M.; Keyyu, J.; Kock, R.; Parida, S. Characterisation of Peste Des Petits Ruminants Disease in Pastoralist Flocks in Ngorongoro District of Northern Tanzania and Bluetongue Virus Co-Infection. Viruses 2020, 12, 389. [Google Scholar] [CrossRef] [Green Version]
- Njeumi, F.; Bailey, D.; Soula, J.J.; Diop, B.; Tekola, B.G. Eradicating the Scourge of Peste Des Petits Ruminants from the World. Viruses 2020, 12, 313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parida, S.; Muniraju, M.; Altan, E.; Baazizi, R.; Raj, G.D.; Mahapatra, M. Emergence of PPR and its threat to Europe. Small Rumin. Res. 2016, 142, 16–21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niyokwishimira, A.; de D Baziki, J.; Dundon, W.G.; Nwankpa, N.; Njoroge, C.; Boussini, H.; Wamwayi, H.; Jaw, B.; Cattoli, G.; Nkundwanayo, C.; et al. Detection and molecular characterization of Peste des Petits Ruminants virus from outbreaks in Burundi, December 2017–January 2018. Transbound. Emerg. Dis. 2019, 66, 2067–2073. [Google Scholar] [CrossRef]
- Mahapatra, M.; Parida, S.; Egziabher, B.G.; Diallo, A.; Barrett, T. Sequence analysis of the phosphoprotein gene of peste des petits ruminants (PPR) virus: Editing of the gene transcript. Virus Res. 2003, 96, 85–98. [Google Scholar] [CrossRef]
- Kumar, K.S.; Babu, A.; Sundarapandian, G.; Roy, P.; Thangavelu, A.; Kumar, K.S.; Arumugam, R.; Chandran, N.D.; Muniraju, M.; Mahapatra, M.; et al. Molecular characterisation of lineage IV peste des petits ruminants virus using multi gene sequence data. Vet. Microbiol. 2014, 174, 39–49. [Google Scholar] [CrossRef] [Green Version]
- Woma, T.Y.; Adombi, C.M.; Yu, D.; Qasim, A.M.; Sabi, A.A.; Maurice, N.A.; Olaiya, O.D.; Loitsch, A.; Bailey, D.; Shamaki, D.; et al. Co-circulation of Peste-des-Petits-Ruminants Virus Asian lineage IV with Lineage II in Nigeria. Transbound. Emerg. Dis. 2016, 63, 235–242. [Google Scholar] [CrossRef] [Green Version]
- Kgotlele, T.; Macha, E.S.; Kasanga, C.J.; Kusiluka, L.J.; Karimuribo, E.D.; Van Doorsselaere, J.; Wensman, J.J.; Munir, M.; Misinzo, G. Partial genetic characterization of peste des petits ruminants virus from goats in northern and eastern Tanzania. Transbound. Emerg. Dis. 2014, 61 (Suppl. S1), 56–62. [Google Scholar] [CrossRef]
- Mahapatra, M.; Sayalel, K.; Muniraju, M.; Eblate, E.; Fyumagwa, R.; Shilinde, L.; Mdaki, M.; Keyyu, J.; Parida, S.; Kock, R. Spillover of Peste des Petits Ruminants Virus from Domestic to Wild Ruminants in the Serengeti Ecosystem, Tanzania. Emerg. Infect. Dis. 2015, 21, 2230–2234. [Google Scholar] [CrossRef] [Green Version]
- Fernandez Aguilar, X.; Mahapatra, M.; Begovoeva, M.; Kalema-Zikusoka, G.; Driciru, M.; Ayebazibwe, C.; Adwok, D.S.; Kock, M.; Lukusa, J.K.; Muro, J.; et al. Peste des Petits Ruminants at the Wildlife-Livestock Interface in the Northern Albertine Rift and Nile Basin, East Africa. Viruses 2020, 12, 293. [Google Scholar] [CrossRef] [Green Version]
- Kinimi, E.; Mahapatra, M.; Kgotlele, T.; Makange, M.R.; Tennakoon, C.; Njeumi, F.; Odongo, S.; Muyldermans, S.; Kock, R.; Parida, S.; et al. Complete Genome Sequencing of Field Isolates of Peste des Petits Ruminants Virus from Tanzania Revealed a High Nucleotide Identity with Lineage III PPR Viruses. Animals 2021, 11, 2976. [Google Scholar] [CrossRef]
- Mahapatra, M.; Pachauri, R.; Subramaniam, S.; Banyard, A.C.; ChandraSekar, S.; Ramakrishnan, M.A.; Njeumi, F.; Muthuchelvan, D.; Parida, S. Ongoing Assessment of the Molecular Evolution of Peste Des Petits Ruminants Virus Continues to Question Viral Origins. Viruses 2021, 13, 2144. [Google Scholar] [CrossRef]
- Forsyth, M.A.; Barrett, T. Evaluation of polymerase chain reaction for the detection and characterisation of rinderpest and peste des petits ruminants viruses for epidemiological studies. Virus Res. 1995, 39, 151–163. [Google Scholar] [CrossRef]
- Couacy-Hymann, E.; Roger, F.; Hurard, C.; Guillou, J.P.; Libeau, G.; Diallo, A. Rapid and sensitive detection of peste des petits ruminants virus by a polymerase chain reaction assay. J. Virol. Methods 2002, 100, 17–25. [Google Scholar] [CrossRef]
- Bao, J.; Wang, Q.; Li, L.; Liu, C.; Zhang, Z.; Li, J.; Wang, S.; Wu, X.; Wang, Z. Evolutionary dynamics of recent peste des petits ruminants virus epidemic in China during 2013–2014. Virology 2017, 510, 156–164. [Google Scholar] [CrossRef]
- Kwiatek, O.; Keita, D.; Gil, P.; Fernandez-Pinero, J.; Clavero, M.A.; Albina, E.; Libeau, G. Quantitative one-step real-time RT-PCR for the fast detection of the four genotypes of PPRV. J. Virol. Methods 2010, 165, 168–177. [Google Scholar] [CrossRef]
- Batten, C.A.; Banyard, A.C.; King, D.P.; Henstock, M.R.; Edwards, L.; Sanders, A.; Buczkowski, H.; Oura, C.C.; Barrett, T. A real time RT-PCR assay for the specific detection of Peste des petits ruminants virus. J. Virol. Methods 2011, 171, 401–404. [Google Scholar] [CrossRef]
- Li, L.; Bao, J.; Wu, X.; Wang, Z.; Wang, J.; Gong, M.; Liu, C.; Li, J. Rapid detection of peste des petits ruminants virus by a reverse transcription loop-mediated isothermal amplification assay. J. Virol. Methods 2010, 170, 37–41. [Google Scholar] [CrossRef]
- Mahapatra, M.; Howson, E.; Fowler, V.; Batten, C.; Flannery, J.; Selvaraj, M.; Parida, S. Rapid Detection of Peste des Petits Ruminants Virus (PPRV) Nucleic Acid Using a Novel Low-Cost Reverse Transcription Loop-Mediated Isothermal Amplification (RT-LAMP) Assay for Future Use in Nascent PPR Eradication Programme. Viruses 2019, 11, 699. [Google Scholar] [CrossRef] [Green Version]
- Rajko-Nenow, P.; Flannery, J.; Arnold, H.; Howson, E.L.A.; Darpel, K.; Stedman, A.; Corla, A.; Batten, C. A rapid RT-LAMP assay for the detection of all four lineages of Peste des Petits Ruminants Virus. J. Virol. Methods 2019, 274, 113730. [Google Scholar] [CrossRef]
- Thompson, J.D.; Gibson, T.; Higgins, D.G. Multiple sequence alignment using ClustalW and ClustalX. Curr. Protoc. Bioinform. 2002, 1, 2–3. [Google Scholar] [CrossRef]
- Baazizi, R.; Mahapatra, M.; Clarke, B.D.; Ait-Oudhia, K.; Khelef, D.; Parida, S. Peste des petits ruminants (PPR): A neglected tropical disease in Maghreb region of North Africa and its threat to Europe. PLoS ONE 2017, 12, e0175461. [Google Scholar] [CrossRef]
- Clarke, B.; Mahapatra, M.; Friedgut, O.; Bumbarov, V.; Parida, S. Persistence of Lineage IV Peste-des-petits ruminants virus within Israel since 1993: An evolutionary perspective. PLoS ONE 2017, 12, e0177028. [Google Scholar] [CrossRef]
- Clarke, B.D.; Islam, M.R.; Yusuf, M.A.; Mahapatra, M.; Parida, S. Molecular detection, isolation and characterization of Peste-des-petits ruminants virus from goat milk from outbreaks in Bangladesh and its implication for eradication strategy. Transbound. Emerg. Dis. 2018, 65, 1597–1604. [Google Scholar] [CrossRef]
- Parida, S.; Selvaraj, M.; Gubbins, S.; Pope, R.; Banyard, A.; Mahapatra, M. Quantifying Levels of Peste Des Petits Ruminants (PPR) Virus in Excretions from Experimentally Infected Goats and Its Importance for Nascent PPR Eradication Programme. Viruses 2019, 11, 249. [Google Scholar] [CrossRef] [Green Version]
- Colenutt, C.; Brown, E.; Paton, D.J.; Mahapatra, M.; Parida, S.; Nelson, N.; Maud, J.; Motta, P.; Sumption, K.; Adhikari, B.; et al. Environmental sampling for the detection of foot-and-mouth disease virus and peste des petits ruminants virus in a live goat market, Nepal. Transbound. Emerg. Dis. 2021. [Google Scholar] [CrossRef]
- Baron, M.D.; Parida, S.; Oura, C.A. Peste des petits ruminants: A suitable candidate for eradication? Vet. Rec. 2011, 169, 16–21. [Google Scholar] [CrossRef]
- Mariner, J.C.; Jones, B.A.; Rich, K.M.; Thevasagayam, S.; Anderson, J.; Jeggo, M.; Cai, Y.; Peters, A.R.; Roeder, P.L. The Opportunity to Eradicate Peste des Petits Ruminants. J. Immunol. 2016, 196, 3499–3506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, H.; Njeumi, F.; Parida, S.; Benfield, C.T.O. Progress towards Eradication of Peste des Petits Ruminants through Vaccination. Viruses 2021, 13, 59. [Google Scholar] [CrossRef] [PubMed]



| Serial No. | Sample Details | Mean CT # | NP3/NP4 | N1197F/N1658R | Nested PCR |
|---|---|---|---|---|---|
| 1 | DRC nasal swab-goat-5 (L-III) | 24.79 | - | - | +++ |
| 2 | DRC nasal swab-sheep-10 (L-III) | 25.09 | - | - | +++ |
| 3 | DRC nasal swab-goat-3 | 32.79 | - | - | - |
| 4 | DRC nasal swab-goat- 13 (L-III) | 22.17 | - | - | +++ |
| 5 | Tanzania occular swab-goat-16 | 26.37 | - | + | ++ |
| 6 | Tanzania occular swab-goat-11 | 36.96 | - | - | - |
| 7 | Tanzania occular swab-goat-10 (L-II) | 22.96 | + | + | +++ |
| 8 | Algeria Farm 1-blood 2 (L-IV) | 22.05 | + | + | +++ |
| 9 | Israel Field lung tissue-1 (L-IV) | 20.62 | + | + | +++ |
| 10 | Faecal sample-5 * (L-IV) | 22.45 | + | + | +++ |
| 11 | Nasal swab-2 * | 32.65 | - | - | - |
| 12 | Tanzania nasal swab-sheep-5 | 31.04 | - | - | + |
| 13 | Bangladesh Milk-B19/Nihkanchari/2015 (L-IV) | 23.54 | + | + | +++ |
| 14 | Bangladesh Milk-B53/Savara/2015 | 30.1 | - | - | + |
| 15 | Israel Field lung tissue-3 | no CT | - | - | - |
| 16 | Nasal swab-5 * | 28.35 | - | - | ++ |
| 17 | Algeria Farm 1-blood 1 (L-IV) | 25.23 | + | + | +++ |
| 18 | Algeria Farm 1-blood 5 (L-IV) | 26.34 | + | + | +++ |
| 19 | Nasal swab-4 * (L-IV) | 25.18 | + | ++ | +++ |
| 20 | Tanzania nasal swab-goat-2 | 32 | - | - | - |
| 21 | Nasal swab-3 * | 31.22 | - | - | - |
| 22 | Trb/Nepal/goatmarket/pen1/metalwall | 27.55 | - | - | - |
| 23 | Trb/Nepal/goatmarket/pen2/woodenpost/b | 31.29 | - | - | - |
| 24 | Trb/Nepal/goatmarket/pen1/metalpole/c (L-IV) | 24.61 | + | - | +++ |
| 25 | Trb/Nepal/goatmarket/pen2/metalpole/a | 29.68 | - | - | - |
| 26 | Trb/Nepal/goatmarket/pen1/woodenpost/b | 26.05 | - | - | + |
| 27 | PPRV/Ivory coast (L-I) | 20.30 | +++ | +++ | +++ |
| 28 | PPRV/Nigeria/75/1 (L-II) | 19.24 | +++ | +++ | +++ |
| 29 | PPRV/Nigeria/76/1 (L-II) | 23.68 | +++ | +++ | +++ |
| 30 | PPRV/IBRI-Oman/82 (L-III) | 19.84 | +++ | +++ | +++ |
| 31 | PPRV/Sudan/Sinar/72 (L-III) | 12.03 | +++ | +++ | +++ |
| 32 | PPRV/Sungri/96 (L-IV) | 13.45 | +++ | +++ | +++ |
| 33 | PPRV/Morocco/2008 (L-IV) | 16.85 | +++ | +++ | +++ |
| 34 | Dolphin Morbillivirus (DMV) | No CT | - | - | - |
| 35 | Measles virus (MV) | No CT | - | - | - |
| 36 | Bovine RSV (bRSV) | No CT | - | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahapatra, M.; Neto, M.M.; Khunti, A.; Njeumi, F.; Parida, S. Development and Evaluation of a Nested PCR for Improved Diagnosis and Genetic Analysis of Peste des Petits Ruminants Virus (PPRV) for Future Use in Nascent PPR Eradication Programme. Animals 2021, 11, 3170. https://doi.org/10.3390/ani11113170
Mahapatra M, Neto MM, Khunti A, Njeumi F, Parida S. Development and Evaluation of a Nested PCR for Improved Diagnosis and Genetic Analysis of Peste des Petits Ruminants Virus (PPRV) for Future Use in Nascent PPR Eradication Programme. Animals. 2021; 11(11):3170. https://doi.org/10.3390/ani11113170
Chicago/Turabian StyleMahapatra, Mana, Martin Mayora Neto, Asha Khunti, Felix Njeumi, and Satya Parida. 2021. "Development and Evaluation of a Nested PCR for Improved Diagnosis and Genetic Analysis of Peste des Petits Ruminants Virus (PPRV) for Future Use in Nascent PPR Eradication Programme" Animals 11, no. 11: 3170. https://doi.org/10.3390/ani11113170







