A Retrospective Study of Clinical and Histopathological Features of 81 Cases of Canine Apocrine Gland Adenocarcinoma of the Anal Sac: Independent Clinical and Histopathological Risk Factors Associated with Outcome
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection Criteria
2.2. Data Collection
2.3. Histological Analysis
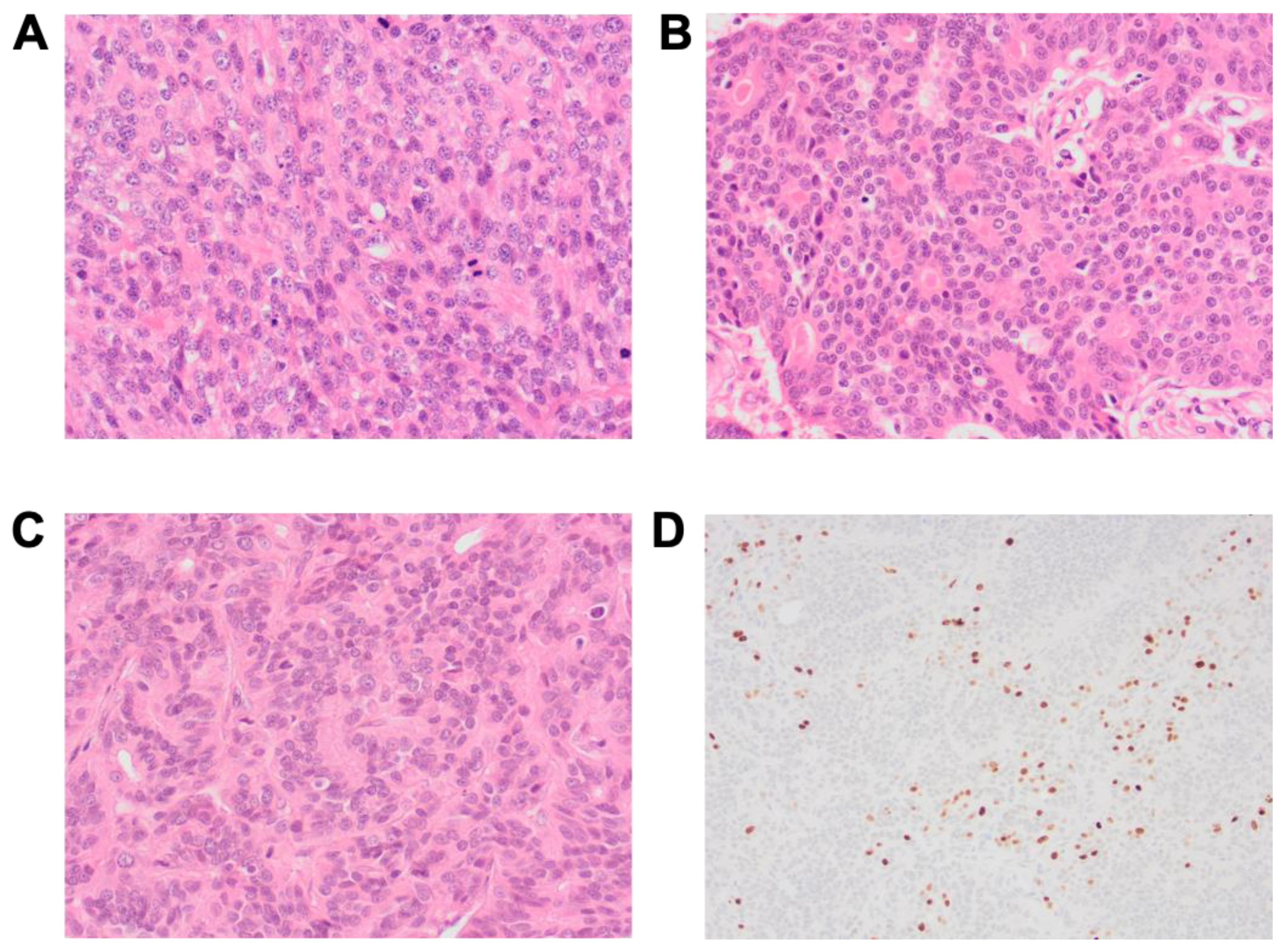
2.4. Immunohistochemistry
2.5. Statistics
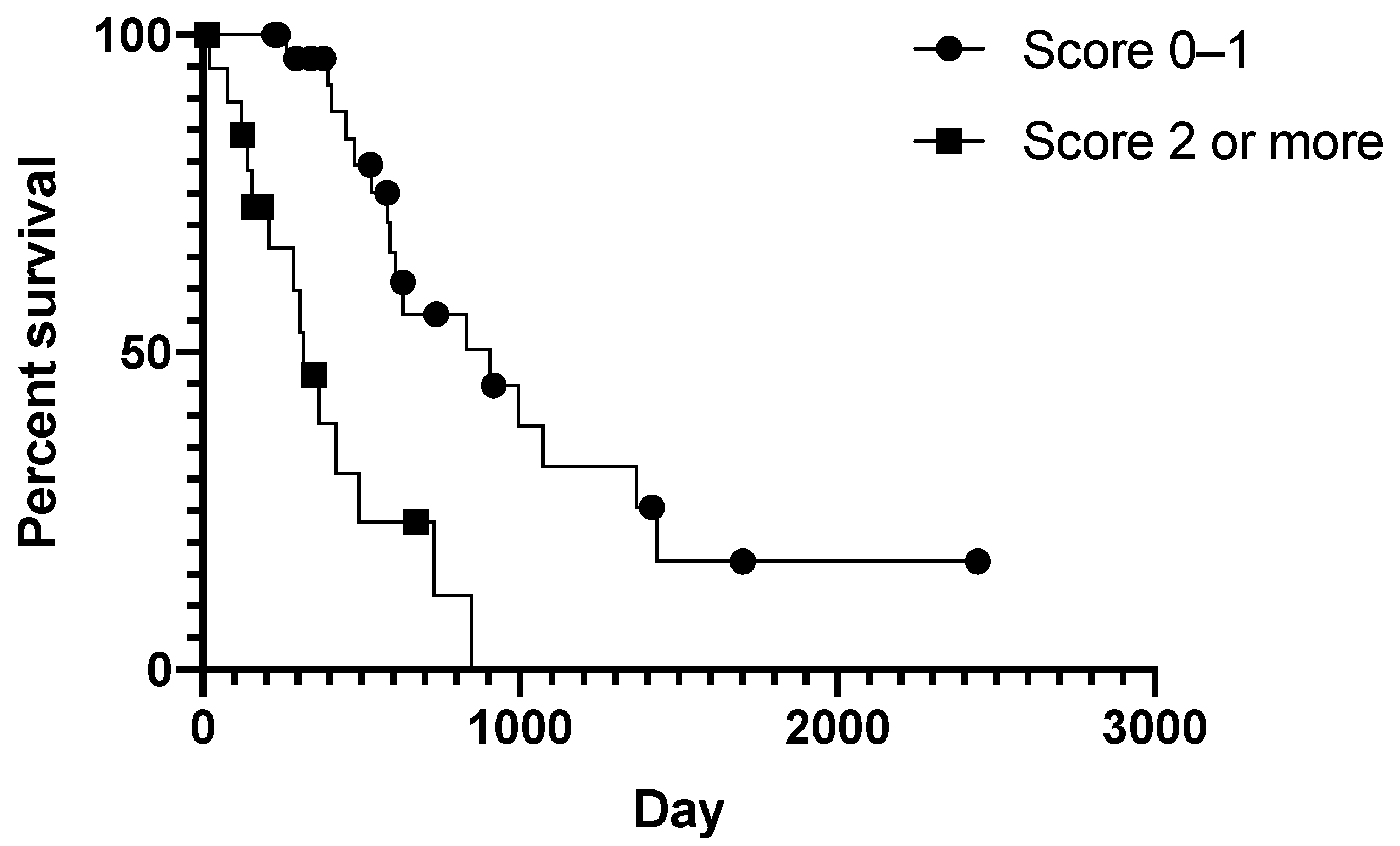
3. Results
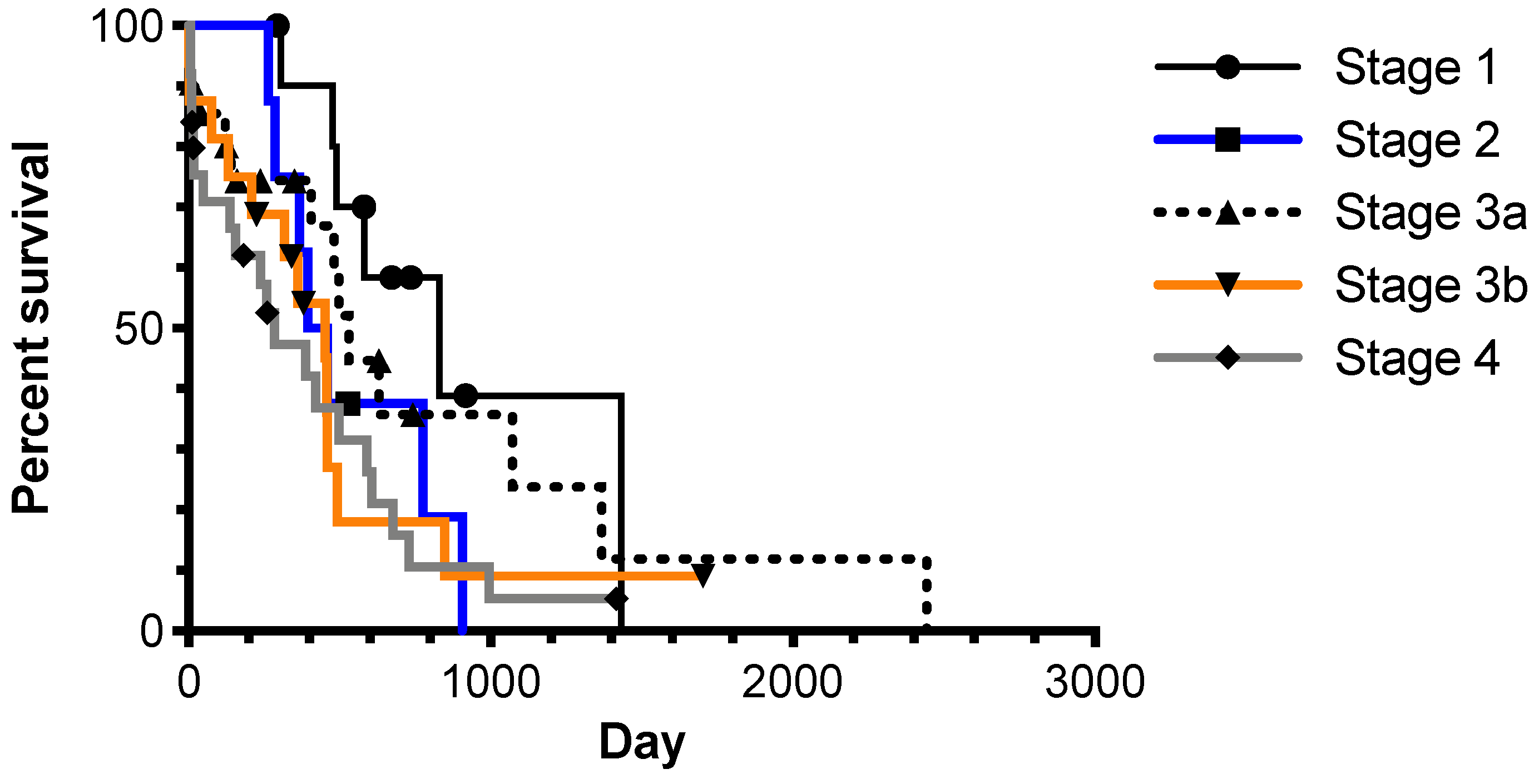
3.1. Signalment, Clinical Signs, and Clinical Staging
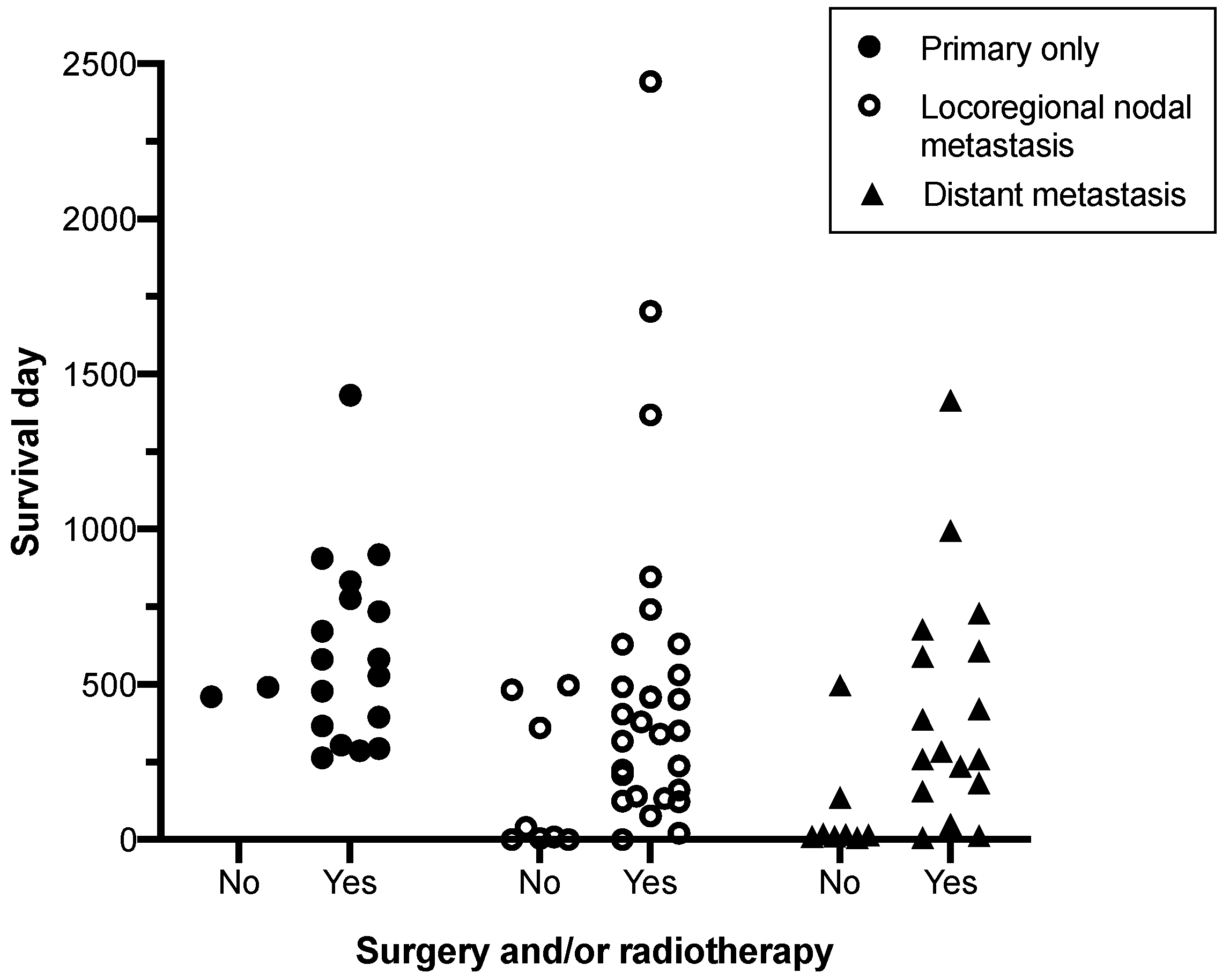
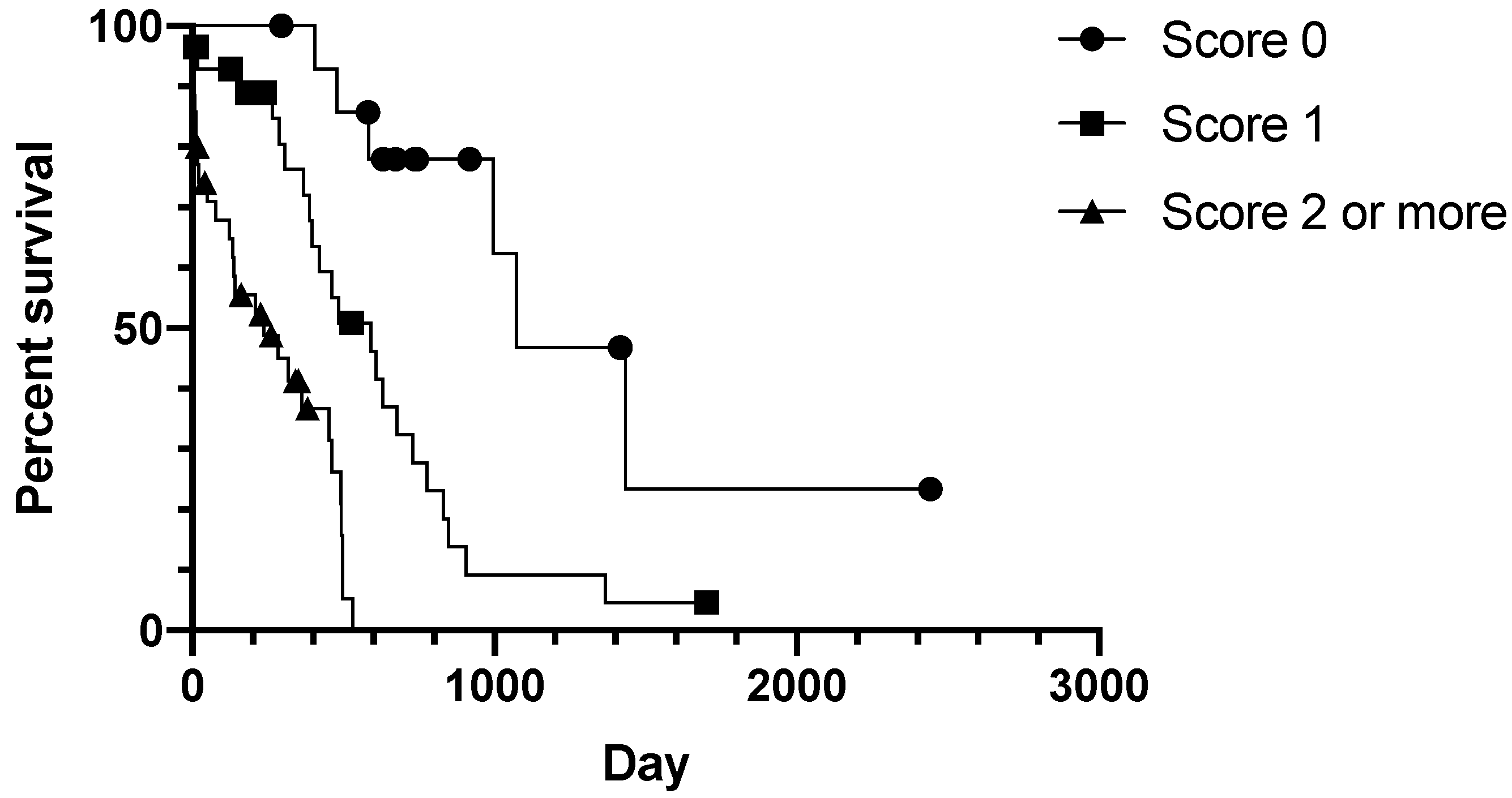
3.2. Prognostic Clinical Variables and Patient Survival Time
3.3. Prognostic Histopathological Parameters
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Appendix B
Appendix C
References
- Berrocal, A.; Vos, J.H.; Van Den Ingh, T.S.; Molenbeek, R.F.; van Sluijs, F.J. Canine Perineal Tumours. J. Vet. Med. Ser. A 1989, 36, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.E.; Gliatto, J.M.; Dodge, R.K.; Johnson, J.L.; Gamblin, R.M.; Thamm, D.; Lana, S.E.; Szymkowski, M.; Moore, A.; Veterinary Cooperative Oncology Group. Carcinoma of the apocrine glands of the anal sac in dogs: 113 cases (1985–1995). J. Am. Vet. Med Assoc. 2003, 223, 825–831. [Google Scholar] [CrossRef]
- Polton, G.A.; Mowat, V.; Lee, H.C.; McKee, K.A.; Scase, T.J. Breed, gender and neutering status of British dogs with anal sac gland carcinoma. Vet. Comp. Oncol. 2006, 4, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Aguirre-Hernández, J.; Polton, G.; Kennedy, L.J.; Sargan, D.R. Association between anal sac gland carcinoma and dog leukocyte antigen-DQB1 in the English Cocker Spaniel. Tissue Antigens 2010, 76, 476–481. [Google Scholar] [CrossRef] [PubMed]
- Bennett, P.F.; DeNicola, D.B.; Bonney, P.L.; Glickman, N.W.; Knapp, D.W. Clinical Anal Sac Adenocarcinomas: Clinical Presentation and Response to Therapy. J. Vet. Intern. Med. 2002, 16, 100–104. [Google Scholar] [CrossRef]
- Brisson, B.A.; Whiteside, D.P.; Holmberg, D.L. Metastatic Anal Sac Adenocarcinoma in a Dog Presenting for Acute Paralysis. Can. Vet. J. 2004, 45, 678–681. [Google Scholar] [PubMed]
- Polton, G.A.; Brearley, M.J. Clinical Stage, Therapy, and Prognosis in Canine Anal Sac Gland Carcinoma. J. Vet. Intern. Med. 2007, 21, 274–280. [Google Scholar] [CrossRef]
- Meuten, D.J.; Cooper, B.J.; Capen, C.C.; Chew, D.J.; Kociba, G.J. Hypercalcemia Associated with an Adenocarcinoma Derived from the Apocrine Glands of the Anal Sac. Vet. Pathol. 1981, 18, 454–471. [Google Scholar] [CrossRef]
- Rosol, T.J.; Capen, C.C.; Danks, J.A.; Suva, L.J.; Steinmeyer, C.L.; Hayman, J.; Ebeling, P.R.; Martin, T.J. Identification of Parathyroid Hormone-Related Protein in Canine Apocrine Adenocarcinoma of the Anal Sac. Vet. Pathol. 1990, 27, 89–95. [Google Scholar] [CrossRef]
- Messinger, J.S.; Windham, W.R.; Ward, C.R. Ionized Hypercalcemia in Dogs: A Retrospective Study of 109 Cases (1998–2003). J. Vet. Intern. Med. 2009, 23, 514–519. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, K.; Morita, R.; Hojo, Y.; Nomura, K.; Shibutani, M.; Mitsumori, K. Immunohistochemical Characterization of Neuroendocrine Differentiation of Canine Anal Sac Glandular Tumours. J. Comp. Pathol. 2013, 149, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Bowlt, K.; Friend, E.; Delisser, P.; Murphy, S.; Polton, G. Temporally Separated Bilateral Anal Sac Gland Carcinomas in Four Dogs. J. Small Anim. Pr. 2013, 54, 432–436. [Google Scholar] [CrossRef] [PubMed]
- Tripp, C.D.; Fidel, J.; Anderson, C.L.; Patrick, M.; Pratt, C.; Sellon, R.; Bryan, J.N. Tolerability of Metronomic Administration of Lomustine in Dogs with Cancer. J. Vet. Intern. Med. 2011, 25, 278–284. [Google Scholar] [CrossRef]
- Urie, B.K.; Russell, D.S.; Kisseberth, W.C.; London, C.A. Evaluation of Expression and Function of Vascular Endothelial Growth Factor Receptor 2, Platelet Derived Growth Factor Receptors-Alpha and -Beta, KIT, and RET in Canine Apocrine Gland Anal Sac Adenocarcinoma and Thyroid Carcinoma. BMC Vet. Res. 2012, 8, 1. [Google Scholar] [CrossRef] [Green Version]
- Wouda, R.M.; Borrego, J.; Keuler, N.S.; Stein, T. Evaluation of Adjuvant Carboplatin Chemotherapy in the Management of Surgically Excised Anal Sac Apocrine Gland Adenocarcinoma in Dogs. Vet. Comp. Oncol. 2016, 14, 139–148. [Google Scholar] [CrossRef]
- Pradel, J.; Berlato, D.; Dobromylskyj, M.; Rasotto, R. Prognostic Significance of Histopathology in Canine Anal Sac Gland Adenocarcinomas: Preliminary Results in a Retrospective Study of 39 Cases. Vet. Comp. Oncol. 2018, 16, 518–528. [Google Scholar] [CrossRef]
- Skorupski, K.; Alarcón, C.; de Lorimier, L.; LaDouceur, E.; Rodriguez, C.; Rebhun, R. Outcome and Clinical, Pathological, and Immunohistochemical Factors Associated with Prognosis for Dogs with Early-Stage Anal Sac Adenocarcinoma Treated with Surgery Alone: 34 Cases (2002–2013). J. Am. Vet. Med. Assoc. 2018, 253, 84–91. [Google Scholar] [CrossRef]
- Barnes, D.C.; Demetriou, J.L. Surgical Management of Primary, Metastatic and Recurrent Anal Sac Adenocarcinoma in the Dog: 52 Cases. J. Small Anim. Pract. 2017, 58, 263–268. [Google Scholar] [CrossRef]
- Potanas, C.; Padgett, S.; Gamblin, R. Surgical Excision of Anal Sac Apocrine Gland Adenocarcinomas with and without Adjunctive Chemotherapy in Dogs: 42 Cases (2005–2011). J. Am. Vet. Med. Assoc. 2015, 246, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Emms, S. Anal Sac Tumours of the Dog and Their Response to Cytoreductive Surgery and Chemotherapy. Aust. Vet. J. 2008, 83, 340–343. [Google Scholar] [CrossRef]
- Hobson, H.; Brown, M.; Rogers, K. Surgery of Metastatic Anal Sac Adenocarcinoma in Five Dogs. Vet. Surg. 2006, 35, 267–270. [Google Scholar] [CrossRef]
- Meier, V.; Polton, G.; Cancedda, S.; Roos, M.; Laganga, P.; Emmerson, T.; Rohrer Bley, C. Outcome in Dogs with Advanced (Stage 3b) Anal Sac Gland Carcinoma Treated with Surgery or Hypofractionated Radiation Therapy. Vet. Comp. Oncol. 2017, 15, 1073–1086. [Google Scholar] [CrossRef] [PubMed]
- McQuown, B.; Keyerleber, M.; Rosen, K.; McEntee, M.; Burgess, K. Treatment of Advanced Canine Anal Sac Adenocarcinoma with Hypofractionated Radiation Therapy: 77 Cases (1999–2013). Vet. Comp. Oncol. 2017, 15, 840–851. [Google Scholar] [CrossRef]
- Hammer, A.S.; Couto, C.G.; Ayl, R.D.; Shank, K.A. Treatment of Tumor-Bearing Dogs With Actinomycin D. J. Vet. Intern. Med. 1994, 8, 236–239. [Google Scholar] [CrossRef] [PubMed]
- Poirier, V.J.; Burgess, K.E.; Adams, W.M.; Vail, D.M. Toxicity, Dosage, and Efficacy of Vinorelbine (Navelbine) in Dogs with Spontaneous Neoplasia. J. Vet. Intern. Med. 2004, 18, 536–539. [Google Scholar] [CrossRef]
- Dominguez, P.; Dervisis, N.; Cadile, C.; Sarbu, L.; Kitchell, B. Combined Gemcitabine and Carboplatin Therapy for Carcinomas in Dogs. J. Vet. Intern. Med. 2009, 23, 130–137. [Google Scholar] [CrossRef]
- London, C.; Mathie, T.; Stingle, N.; Clifford, C.; Haney, S.; Mary, K.; Beaver, L.; Vickery, K.; Vail, D.M.; Hershey, B.; et al. Preliminary Evidence for Biologic Activity of Toceranib Phosphate (Palladia®) in Solid Tumors. Vet. Comp. Oncol. 2012, 10, 194–205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leach, T.N.; Childress, M.O.; Greene, S.N.; Mohamed, A.S.; Moore, G.E.; Schrempp, D.R.; Lahrman, S.R.; Knapp, D.W. Prospective Trial of Metronomic Chlorambucil Chemotherapy in Dogs with Naturally Occurring Cancer. Vet. Comp. Oncol. 2012, 10, 102–112. [Google Scholar] [CrossRef]
- Heaton, C.M.; Fernandes, A.F.A.; Jark, P.C.; Pan, X. Evaluation of Toceranib for Treatment of Apocrine Gland Anal Sac Adenocarcinoma in Dogs. J. Vet. Intern. Med., 2020, 34, 873–881. [Google Scholar] [CrossRef]
- Morello, E.M.; Cino, M.; Giacobino, D.; Nicoletti, A.; Iussich, S.; Buracco, P.; Martano, M. Prognostic Value of Ki67 and Other Clinical and Histopathological Factors in Canine Apocrine Gland Anal Sac Adenocarcinoma. Animals 2021, 11, 1–17. [Google Scholar] [CrossRef]
- Schlag, A.; Johnson, T.; Vinayak, A.; Kuvaldina, A.; Skinner, O.; Wustefeld-Janssens, B. Comparison of Methods to Determine Primary Tumour Size in Canine Apocrine Gland Anal Sac Adenocarcinoma. J. Small Anim. Pr. 2020, 61, 185–189. [Google Scholar] [CrossRef]
- Nguyen, S.; Thamm, D.; Vail, D.; London, C. Response Evaluation Criteria for Solid Tumours in Dogs (v1.0): A Veterinary Cooperative Oncology Group (VCOG) Consensus Document. Vet. Comp. Oncol. 2015, 13, 176–183. [Google Scholar] [CrossRef]
- Heinze, G.; Wallisch, C.; Dunkler, D. Variable Selection—A Review and Recommendations for the Practicing Statistician. Biom. J. 2018, 60, 431–449. [Google Scholar] [CrossRef] [Green Version]
- Youden, W.J. Index for Rating Diagnostic Tests. Cancer 1950, 3, 32–35. [Google Scholar] [CrossRef]
- Akaike, H. New Look at the Statistical Model Identification. In Selected Papers of Hirotugu Akaike; Parazen, E., Tanabe, K., Kitagawa, G., Eds.; Springer: New York, NY, USA, 1974. [Google Scholar]
- Goldschmidt, M.H.; Goldschmidt, K.H. Epithelial and Melanocytic Tumors of the Skin. In Tumors in Domestic Animals, 5th ed.; Meuten, D.J., Ed.; Wiley-Blackwell: Ames, IA, USA, 2017; pp. 120–122. [Google Scholar]
- Moik, F.; Zöchbauer-Müller, S.; Posch, F.; Pabinger, I.; Ay, C. Systemic Inflammation and Activation of Haemostasis Predict Poor Prognosis and Response to Chemotherapy in Patients with Advanced Lung Cancer. Cancers 2020, 12, 1–17. [Google Scholar] [CrossRef]
- Heinze, G.; Dunkler, D. Five Myths about Variable Selection. Transpl. Int. 2017, 30, 6–10. [Google Scholar] [CrossRef]
- Nadeau, M.E.; Kitchell, B.E. Evaluation of the Use of Chemotherapy and Other Prognostic Variables for Surgically Excised Canine Thyroid Carcinoma with and without Metastasis. Can. Vet. J. 2011, 52, 994–998. [Google Scholar] [PubMed]
- Campos, M.; Ducatelle, R.; Rutteman, G.; Kooistra, H.S.; Duchateau, L.; de Rooster, H.; Peremans, K.; Daminet, S. Clinical, Pathologic, and Immunohistochemical Prognostic Factors in Dogs with Thyroid Carcinoma. J. Vet. Intern. Med. 2014, 28, 1805–1813. [Google Scholar] [CrossRef] [PubMed]
- Bundred, N.J.; Ratcliffe, W.A.; Walker, R.A.; Coley, S.; Morrison, J.M.; Ratcliffe, J.G. Parathyroid Hormone Related Protein and Hypercalcaemia in Breast Cancer. Br. Med. J. 1991, 303, 1506–1509. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Powell, G.J.; Southby, J.; Danks, J.A.; Stillwell, R.G.; Hayman, J.A.; Henderson, M.A.; Bennett, R.C.; Martin, T.J. Localization of Parathyroid Hormone-Related Protein in Breast Cancer Metastases: Increased Incidence in Bone Compared with Other Sites. Cancer Res. 1991, 51, 3059–3061. [Google Scholar]

| Parameter | Number |
|---|---|
| Sex | 54 males |
| 27 females | |
| 2:1 male:female | |
| Age | 9.7 years mean |
| 5−15 years interquartile range | |
| Breed most affected | Cocker spaniel n = 30 |
| Labrador retriever n = 12 | |
| Cross-breed n = 11 | |
| Primary clinical sign | Owner-detected perianal mass n = 40 |
| Incidental finding of perianal mass n = 17 | |
| Dysuria/tenesmus n = 13 | |
| Clinical signs of hypercalcaemia | |
| Mass location | Right anal sac n = 41 |
| Left anal sac n = 34 | |
| Bilateral n = 6 | |
| Sublumbar lymphadenopathy on rectal palpation | Present n = 30 |
| Absent n = 51 |
| Parameter | Groups | No. | MST 1 | Hazard Ratio | p 2 |
|---|---|---|---|---|---|
| Tumour diameter | ≤2 cm | 35 | 678 | 1.983 | 0.048 * |
| >2 cm | 42 | 360 | |||
| Lymph node diameter | ≤1.6 cm | 39 | 631 | 3.053 | 0.001 ** |
| 1.6 < x ≤ 5 cm | 24 | 493 | |||
| >5 cm | 18 | 135 | |||
| Surgery | No | 18 | 137 | 0.332 | 0.004 ** |
| Yes | 63 | 531 | |||
| Radiotherapy | No | 73 | 452 | 0.307 | 0.014 * |
| Yes | 8 | 995 | |||
| Medical therapy | No | 49 | 452 | Eliminated | |
| Yes | 32 | 461 | |||
| Calcium status | Normal | 58 | 497 | Eliminated | |
| Increased | 19 | 286 |
| Clinical Stage | Median Survival Time (days) | |
|---|---|---|
| No Surgery or Radiotherapy | Surgery and/or Radiotherapy | |
| Primary disease only | 476 | 776 |
| Locoregional nodal metastasis | 360 1 | 493 1 |
| Distant metastasis | 15 1 | 421 1 |
| Parameter | Group | Score |
|---|---|---|
| Tumour diameter cm | ≤2 | 0 |
| >2 | 1 | |
| Lymph node diameter cm | ≤1.6 | 0 |
| 1.6 < x ≤ 5 | 1 | |
| >5 | 2 | |
| Treatment | Surgery and/or radiotherapy | 0 |
| Neither surgery nor radiotherapy | 1 |
| Parameter | Groups | No. | MST 1 | Hazard Ratio | p 2 |
|---|---|---|---|---|---|
| Tumour diameter | ≤1.3 cm | 9 | 445 | 5.948 | 0.006 ** |
| >1.3 cm | 26 | 230 | |||
| Distant metastasis | No | 29 | 252 | 5.708 | 0.001 ** |
| Yes | 8 | 133 | |||
| Lymph node size | ≤3.8 cm | 27 | 230 | 0.385 | 0.053 |
| >3.8 cm | 10 | 258 | |||
| Calcium status | Normal | 26 | 239 | Eliminated | |
| Increased | 8 | 275 |
| Parameter | Groups | No. | MST 1 | Hazard Ratio | p 2 |
|---|---|---|---|---|---|
| Necrosis | None | 29 | 831 | 2.984 | 0.008 ** |
| Present (any amount) | 21 | 421 | |||
| Predominant histological pattern | Non-solid | 26 | 631 | 2.63 | 0.04 * |
| Solid | 23 | 452 | |||
| Vascular invasion | None | 31 | 831 | 2.794 | 0.02 * |
| Present | 18 | 367 | |||
| Ki67% | 6.1 | 21 | 607 | Eliminated | |
| >6.1 | 25 | 590 |
| Parameter | Group | Score |
|---|---|---|
| Tumour necrosis | None | 0 |
| Present | 1 | |
| Predominant histological pattern | Rosette or tubular | 0 |
| Solid | 1 | |
| Vascular invasion | None | 0 |
| Present | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wong, H.; Byrne, S.; Rasotto, R.; Drees, R.; Taylor, A.; Priestnall, S.L.; Leo, C. A Retrospective Study of Clinical and Histopathological Features of 81 Cases of Canine Apocrine Gland Adenocarcinoma of the Anal Sac: Independent Clinical and Histopathological Risk Factors Associated with Outcome. Animals 2021, 11, 3327. https://doi.org/10.3390/ani11113327
Wong H, Byrne S, Rasotto R, Drees R, Taylor A, Priestnall SL, Leo C. A Retrospective Study of Clinical and Histopathological Features of 81 Cases of Canine Apocrine Gland Adenocarcinoma of the Anal Sac: Independent Clinical and Histopathological Risk Factors Associated with Outcome. Animals. 2021; 11(11):3327. https://doi.org/10.3390/ani11113327
Chicago/Turabian StyleWong, Hannah, Stephanie Byrne, Roberta Rasotto, Randi Drees, Angela Taylor, Simon L. Priestnall, and Chiara Leo. 2021. "A Retrospective Study of Clinical and Histopathological Features of 81 Cases of Canine Apocrine Gland Adenocarcinoma of the Anal Sac: Independent Clinical and Histopathological Risk Factors Associated with Outcome" Animals 11, no. 11: 3327. https://doi.org/10.3390/ani11113327
APA StyleWong, H., Byrne, S., Rasotto, R., Drees, R., Taylor, A., Priestnall, S. L., & Leo, C. (2021). A Retrospective Study of Clinical and Histopathological Features of 81 Cases of Canine Apocrine Gland Adenocarcinoma of the Anal Sac: Independent Clinical and Histopathological Risk Factors Associated with Outcome. Animals, 11(11), 3327. https://doi.org/10.3390/ani11113327






