Chronic Stress Detrimentally Affects In Vivo Maturation in Rat Oocytes and Oocyte Viability at All Phases of the Estrous Cycle
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Animals
2.3. Experimental Design
2.4. Stress Procedure
2.5. Vaginal Cytology and Estrous Cyclicity
2.6. Biological Samples
2.7. Hormonal Analysis
2.8. Ovarian Oocyte Recovery
2.9. Evaluation of Oocyte Viability
2.10. Evaluation of Oocyte Maturation
2.11. Statistical Analysis
3. Results
3.1. Estrous Cyclicity
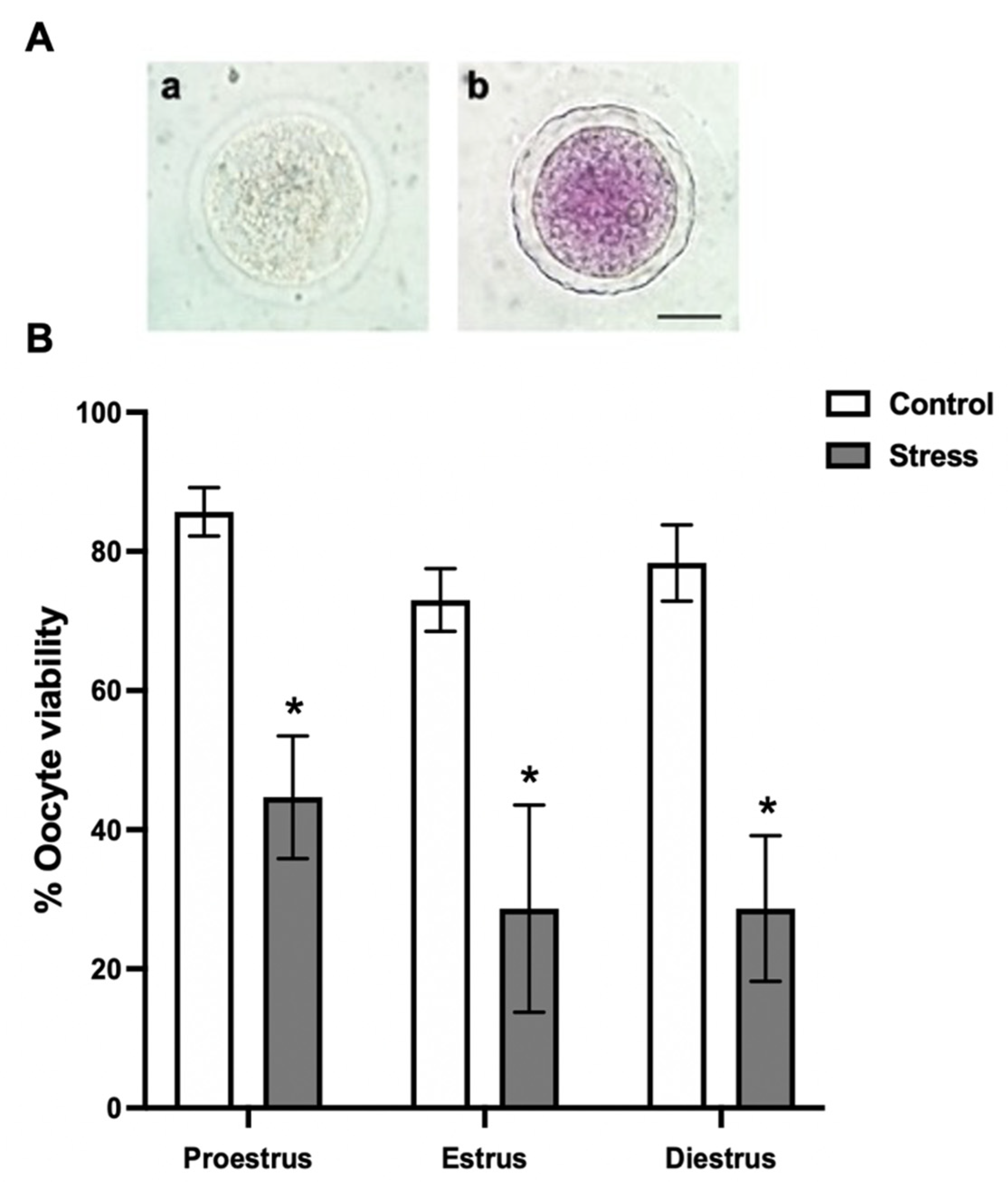
3.2. Oocyte Viability
3.3. In Vivo Oocyte Maturation
3.4. Abnormal Oocytes after Chronic Stress
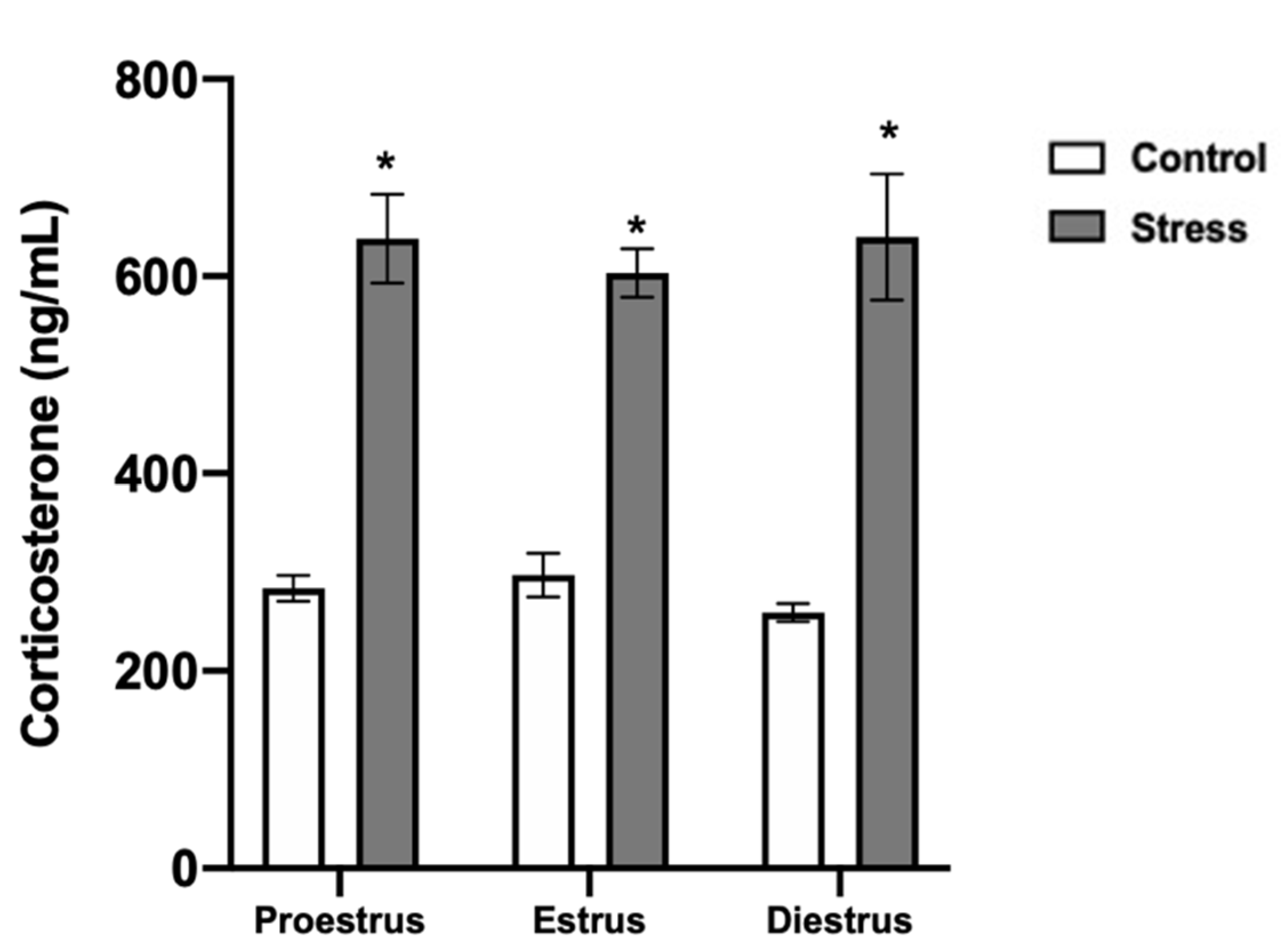
3.5. Corticosterone Serum Levels and Oocyte Viability after Chronic Stress
4. Discussion
4.1. Estrous Cyclicity
4.2. Oocyte Viability and Maturation after Chronic Stress
4.3. Abnormal Oocytes after Chronic Stress
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boivin, J.; Griffiths, E.; Venetis, C.A. Emotional distress in infertile woman and failure of assisted reproductive technologies: Meta-analysis of prospective psychosocial studies. BMJ 2011, 342, d223. [Google Scholar] [CrossRef]
- Chehreh, R.; Ozgoli, G.; Abolmaali, K.; Nasiri, M.; Mazaheri, E. Comparison of the infertility-related stress among couples and its relationship with infertility factors. Int. J. Women’s Health Reprod. Sci. 2019, 7, 313–318. [Google Scholar] [CrossRef]
- Valsamakis, G.; Chrousos, G.; Mastorakos, G. Stress, female reproduction and pregnancy. Psychoneuroendocrinology 2019, 100, 48–57. [Google Scholar] [CrossRef]
- Ebbesen, S.M.S.; Zacharie, R.; Mehlsen, M.Y.; Thomsen, D.; Hojgaard, A.; Ottosen, L.; Petersen, T.; Ingerslev, H.J. Stressful life events are associated with a poor in-vitro fertilization (IVF) outcome: A prospective study. Hum. Reprod. 2009, 2, 2173–2182. [Google Scholar] [CrossRef]
- Prasad, S.; Tiwari, M.; Pandey, A.N.; Shrivastav, T.G.; Chaube, S. Impact of stress on oocyte quality and reproductive outcome. J. Biomed. Sci. 2016, 29, 36–40. [Google Scholar] [CrossRef]
- Fu, X.Y.; Chen, H.H.; Zhang, N.; Ding, M.X.; Qiu, Y.E.; Pan, X.M.; Fang, Y.S.; Lin, Y.P.; Zheng, Q.; Wang, W.Q. Effects of chronic unpredictable mild stress on ovarian reserve in female rats: Feasibility analysis of a rat model of premature ovarian failure. Mol. Med. Rep. 2018, 18, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Dorfam, M.; Arancibia, S.; Fiedler, J.L.; Lara, H.E. Chronic intermittent cold stress activates ovarian sympathetic nerves and modifies ovarian follicular development in the rat. Biol. Reprod. 2003, 68, 2038–2043. [Google Scholar] [CrossRef]
- Zhang, S.Y.; Wang, J.Z.; Li, J.J.; Wei, D.L.; Sui, H.S.; Zhang, A.H.; Zhou, P.; Tan, J.H. Maternal restraint stress diminishes the developmental potential of oocytes. Biol. Reprod. 2011, 84, 672–681. [Google Scholar] [CrossRef]
- Dehdehi, L.; Novin, M.G.; Sadeghi, Y.; Abdollahifar, M.A.; Ziai, S.A.; Nazarian, H. Chronic stress diminishes the oocyte quality and in vitro embryonic development in maternally separated mice. Int. J. Women’s Health Reprod. Sci. 2020, 8, 29–36. [Google Scholar] [CrossRef]
- Yuan, H.-J.; Han, X.; He, N.; Wang, G.-L.; Gong, S.; Lin, J.; Gao, M.; Tan, J.-H. Glucocorticoids impair oocyte developmental potential by triggering apoptosis of ovarian cells via activating the Fas system. Sci. Rep. 2016, 6, 24036. [Google Scholar] [CrossRef]
- Xu, T.; Li, X.; Yang, L.; Zhang, Y.; Zhang, L.; Guo, Z.; Cheng, X.; Zheng, X.; Chen, B.; Hou, Z.; et al. Impact of cold exposure on the reproductive function in female rats. BioMed Res. Int. 2018, 2018, 3674906. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Chen, F.; Kong, Q.Q.; Ning, S.F.; Yuan, H.J.; Lian, H.Y.; Luo, M.J.; Tan, J.H. Stresses on female mice impair oocyte developmental potential: Effects of stress severity and duration on oocytes at the growing follicle stage. Reprod. Sci. 2016, 23, 1148–1157. [Google Scholar] [CrossRef]
- Ter Horst, J.P.; De Kloet, E.R.; Schachinger, H.; Oitzl, M.S. Relevance of stress and female sex hormones for emotion and cognition. Cell. Mol. Neurobiol. 2012, 32, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.F.; Yuan, H.J.; Li, H.; Gong, S.; Lin, J.; Miao, Y.L.; Wang, T.Y.; Tan, J.H. Restraint stress on female mice diminishes the developmental potential of oocytes: Roles of chromatin configuration and histone modification in germinal vesicle stage oocytes. Biol. Reprod. 2015, 92, 13. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Guo, Y.; Sun, J.; Bu, S.; Li, B.; Zhang, Q.; Wang, Q.; Lai, D. Melatonin protects against chronic stress-induced oxidative meiotic defects in mice MII oocytes by regulating SIRT1. Cell Cycle 2020, 19, 1667–1695. [Google Scholar] [CrossRef] [PubMed]
- Retana-Márquez, S.; Bonilla-Jaime, H.; Vázquez-Palacios, G.; Martínez-García, R.; Velázquez-Moctezuma, J. Naltrexone effects on male sexual behavior, corticosterone, and testosterone in stressed male rats. Physiol. Behav. 2009, 96, 333–342. [Google Scholar] [CrossRef] [PubMed]
- Woodward, C.; Emery, P.W. Determination of plasma corticosterone using high performance liquid chromatography. J. Chromatogr. 1987, 419, 280–284. [Google Scholar] [CrossRef]
- Casillas, F.; Betancourt, M.; Cuello, C.; Ducolomb, Y.; Lopez, A.; Juarez-Rojas, L.; Retana-Márquez, S. An efficiency comparison of different in vitro fertilization methods: IVF, ICSI and PICSI for embryo development to the blastocyst stage from vitrified porcine immature oocytes. Porc. Health Manag. 2018, 13, 16. [Google Scholar] [CrossRef]
- Divyashree, S.; Yajurvedi, H.N. Long-term chronic stress exposure induces PCO phenotype in rat. Reproduction 2016, 152, 765–774. [Google Scholar] [CrossRef]
- Retana-Márquez, S.; Juárez-Rojas, L.; Ávila-Quintero, A.; Rojas-Maya, S.; Perera, G.; Casillas, F.; Betancourt, M.; Gómez-Quiroz, L. Neuroendocrine disruption is associated to infertility in chronically stressed female rats. Reprod. Biol. 2020, 20, 474–483. [Google Scholar] [CrossRef]
- Acevedo-Rodríguez, A.; Kauffman, A.S.; Cherrington, B.D.; Borges, C.S.; Roepke, T.A.; Laconi, M. Emerging insights into hypothalamic-pituitary-gonadal axis regulation and interaction with stress signaling. J. Neuroendocrinol. 2018, 30, e12590. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, A.S. Coming of age in the kisspeptin era: Sex differences, development, and puberty. Mol. Cell. Endocrinol. 2010, 324, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.L.; Chang, H.M.; Li, R.; Yu, Y.; Quiao, J. Regulation of LH secretion by RFRP-3 -From the hypothalamus to the pituitary. Front. Neuroendocrinol. 2019, 52, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Pineda, R.; García-Galiano, D.; Sánchez-Garrido, M.A.; Romero, M.; Ruíz-Pino, F.; Aguilar, E.; Dijcks, F.A.; Blomenrohr, M.; Pinilla, L.; Van Noort, P.I. Characterization of the inhibitory roles of RFRP3, the mammalian ortholog of GnIH, in the control of gonadotropin secretion in the rat: In vivo and in vitro studies. Am. J. Physiol. Endocrinol. Metab. 2010, 299, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Tetsuka, M.; Milne, M.; Simpson, G.E.; Hillier, S.G. Expression of 11beta- hydroxysteroid dehydrogenase, glucocorticoid receptor, and mineralocorticoid receptor genes in rat ovary. Biol. Reprod. 1990, 60, 330–335. [Google Scholar] [CrossRef] [PubMed]
- Kirby, E.D.; Geraghty, A.C.; Ubuka, T.; Bentley, G.E.; Kaufer, D. Stress increases putative gonadotropin inhibitory hormone and decreases luteinizing hormone in male rats. Proc. Natl. Acad. Sci. USA 2009, 106, 11324–11329. [Google Scholar] [CrossRef]
- Byri, P.; Gangineni, A.; Reddy, R.K.; Raghavender, K.B.P. Effect of kisspeptin on in vitro maturation of sheep oocytes. Vet. World 2017, 10, 276–280. [Google Scholar] [CrossRef]
- Liu, H.; Xu, G.; Yuan, Z.; Dong, Y.; Wang, J.; Lu, W. Effect of kisspeptin on the proliferation and apoptosis of bovine granulosa cells. Anim. Reprod. Sci. 2017, 185, 1–7. [Google Scholar] [CrossRef]
- Squicciarini, V.; Riquelme, R.; Willsterman, K.; Bentley, G.E.; Lara, H.E. Role of RFRP-3 in the development of cold stress-induced polycystic ovary phenotype in rats. J. Endocrinol. 2018, 239, 81–91. [Google Scholar] [CrossRef]
- Wilsterman, K.; Bentley, G.E.; Comizzoli, P. RFRP3 influences basal lamina degradation, cellular death, and progesterone secretion in cultured preantral ovarian follicles from domestic cat. PeerJ 2019, 7, e7540. [Google Scholar] [CrossRef]
- Wang, X.; Li, X.; Hu, C. RFRP-3, the mammalian ortholog of GnIH, induces cell cycle arrest at G2/M in porcine ovarian granulosa cells. Peptides 2018, 101, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Paccola, C.C.; Resende, C.G.; Stumpp, T.; Miraglia, S.M.; Cipriano, I. The rat estrous cycle revisited: A quantitative and qualitative analysis. Anim. Reprod. 2013, 10, 677–683. [Google Scholar]
- Khamsi, F.; Roberge, S. Granulosa cells of the cumulus oophorus are different from mural granulosa cells in their response to gonadotrophins and IGF-I. J. Endocrinol. 2001, 170, 565–573. [Google Scholar] [CrossRef]
- Pan, B.; Li, J. The art of oocyte meiotic arrest regulation. Reprod. Biol. Endocrinol. 2019, 17, 8–20. [Google Scholar] [CrossRef]
- Turner, K.; Reynolds-May, M.F.; Zitek, E.M.; Tisdale, R.L.; Carlisle, A.B.; Westphal, L.M. Stress and anxiety scores in first and repeat IVF cycles: A pilot study. PLoS ONE 2013, 8, e63743. [Google Scholar] [CrossRef]
- González, R.; Ruiz-León, Y.; Gomendio, M.; Roldan, E.R.S. The effect of glucocorticoids on mouse oocyte in vitro maturation and subsequent fertilization and embryo development. Toxicol. In Vitro 2010, 24, 108–115. [Google Scholar] [CrossRef]
- Ahmed, J.A.; Nasiruddullah, N.; Dutta, D.; Biswas, R.K.; Borah, P. Cumulus cell expansion and ultrastructural changes in in vitro matured bovine oocytes under heat stress. Iran. J. Vet. Res. 2017, 18, 203–207. [Google Scholar]
- Wang, D.; Cheng, X.; Fang, H.; Ren, Y.; Li, X.; Ren, W.; Xue, B.; Yang, C. Effect of cold stress on ovarian & uterine microcirculation in rats and the role of endothelin system. Reprod. Biol. Endocrinol. 2020, 18, 29. [Google Scholar] [CrossRef]
- Liang, B.; Wei, D.L.; Cheng, Y.N.; Yuan, H.J.; Lin, J.; Cui, X.Z.; Luo, M.J.; Tan, J.H. Restraint stress impairs oocyte developmental potential in mice: Role of CRH-induced apoptosis of ovarian cells. Biol. Reprod. 2013, 89, 1–12. [Google Scholar] [CrossRef]
- Bellone, M.; Zuccotti, M.; Redi, C.; Garagna, S. The position of the germinal vesicle and the chromatin organization together provide a marker of the developmental competence of mouse antral oocytes. Reproduction 2009, 138, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Khazaei, M.; Aghaz, F. Reactive oxygen species generation and use of antioxidants during in vitro maturation of oocytes. Int. J. Fertil. Steril. 2017, 11, 63–70. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casillas, F.; Betancourt, M.; Juárez-Rojas, L.; Ducolomb, Y.; López, A.; Ávila-Quintero, A.; Zamora, J.; Ommati, M.M.; Retana-Márquez, S. Chronic Stress Detrimentally Affects In Vivo Maturation in Rat Oocytes and Oocyte Viability at All Phases of the Estrous Cycle. Animals 2021, 11, 2478. https://doi.org/10.3390/ani11092478
Casillas F, Betancourt M, Juárez-Rojas L, Ducolomb Y, López A, Ávila-Quintero A, Zamora J, Ommati MM, Retana-Márquez S. Chronic Stress Detrimentally Affects In Vivo Maturation in Rat Oocytes and Oocyte Viability at All Phases of the Estrous Cycle. Animals. 2021; 11(9):2478. https://doi.org/10.3390/ani11092478
Chicago/Turabian StyleCasillas, Fahiel, Miguel Betancourt, Lizbeth Juárez-Rojas, Yvonne Ducolomb, Alma López, Alejandra Ávila-Quintero, Jimena Zamora, Mohammad Mehdi Ommati, and Socorro Retana-Márquez. 2021. "Chronic Stress Detrimentally Affects In Vivo Maturation in Rat Oocytes and Oocyte Viability at All Phases of the Estrous Cycle" Animals 11, no. 9: 2478. https://doi.org/10.3390/ani11092478
APA StyleCasillas, F., Betancourt, M., Juárez-Rojas, L., Ducolomb, Y., López, A., Ávila-Quintero, A., Zamora, J., Ommati, M. M., & Retana-Márquez, S. (2021). Chronic Stress Detrimentally Affects In Vivo Maturation in Rat Oocytes and Oocyte Viability at All Phases of the Estrous Cycle. Animals, 11(9), 2478. https://doi.org/10.3390/ani11092478






