Genome-Wide Identification and Characterization of the BRD Family in Nile Tilapia (Oreochromis niloticus)
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Fish and Experimental Conditions
2.2. Identification of BRD Genes in Representative Animals
2.3. Phylogenetic Analysis, Synteny Analysis, and Functional Domains Analysis of BRD Genes
2.4. Spatiotemporal Expression Pattern of BRDs in Tilapia
2.5. Validation of Spatiotemporal Expression Pattern of BRD Genes by qRT-PCR and Fluorescence In Situ Hybridization
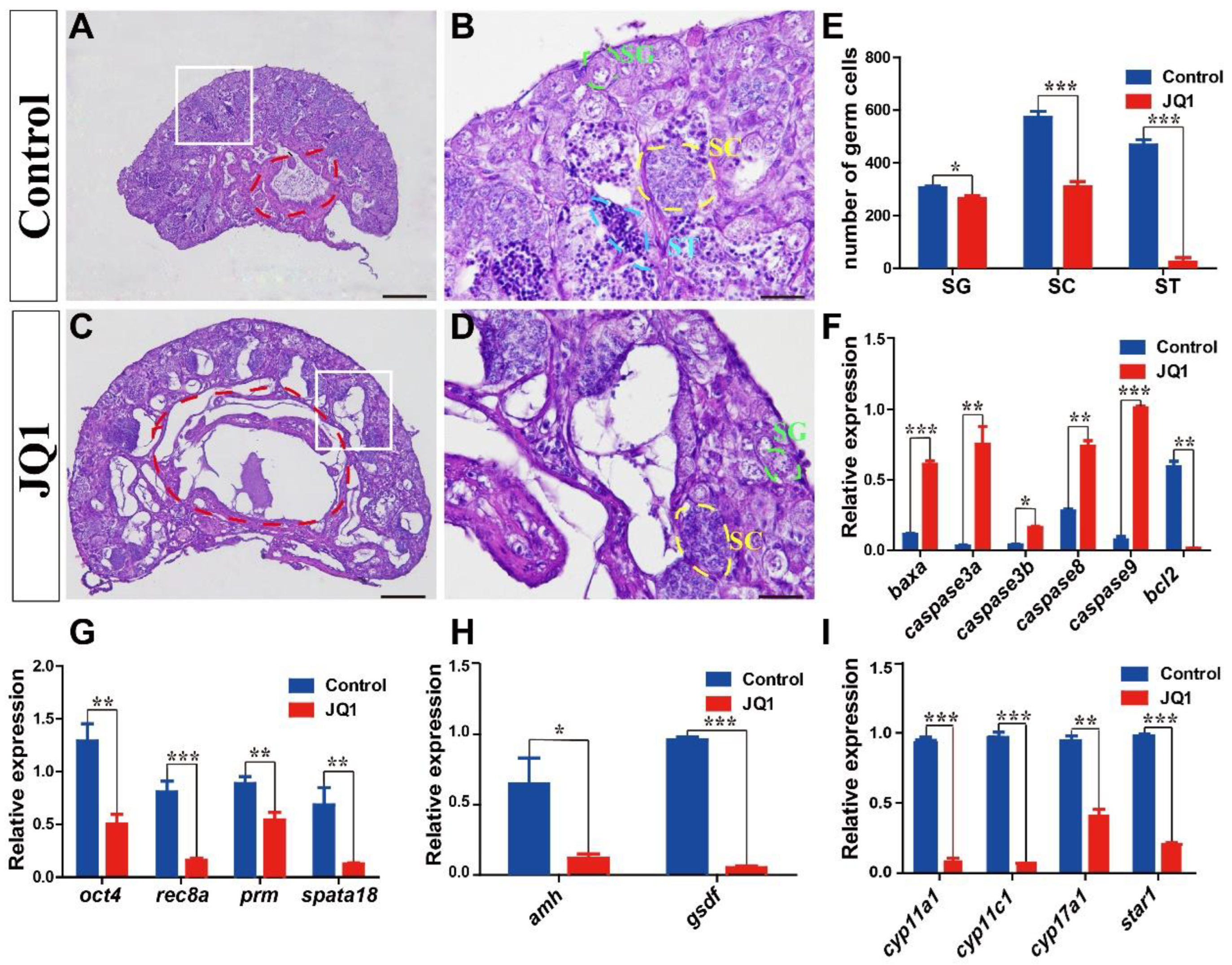
2.6. JQ1 Treatment
3. Results
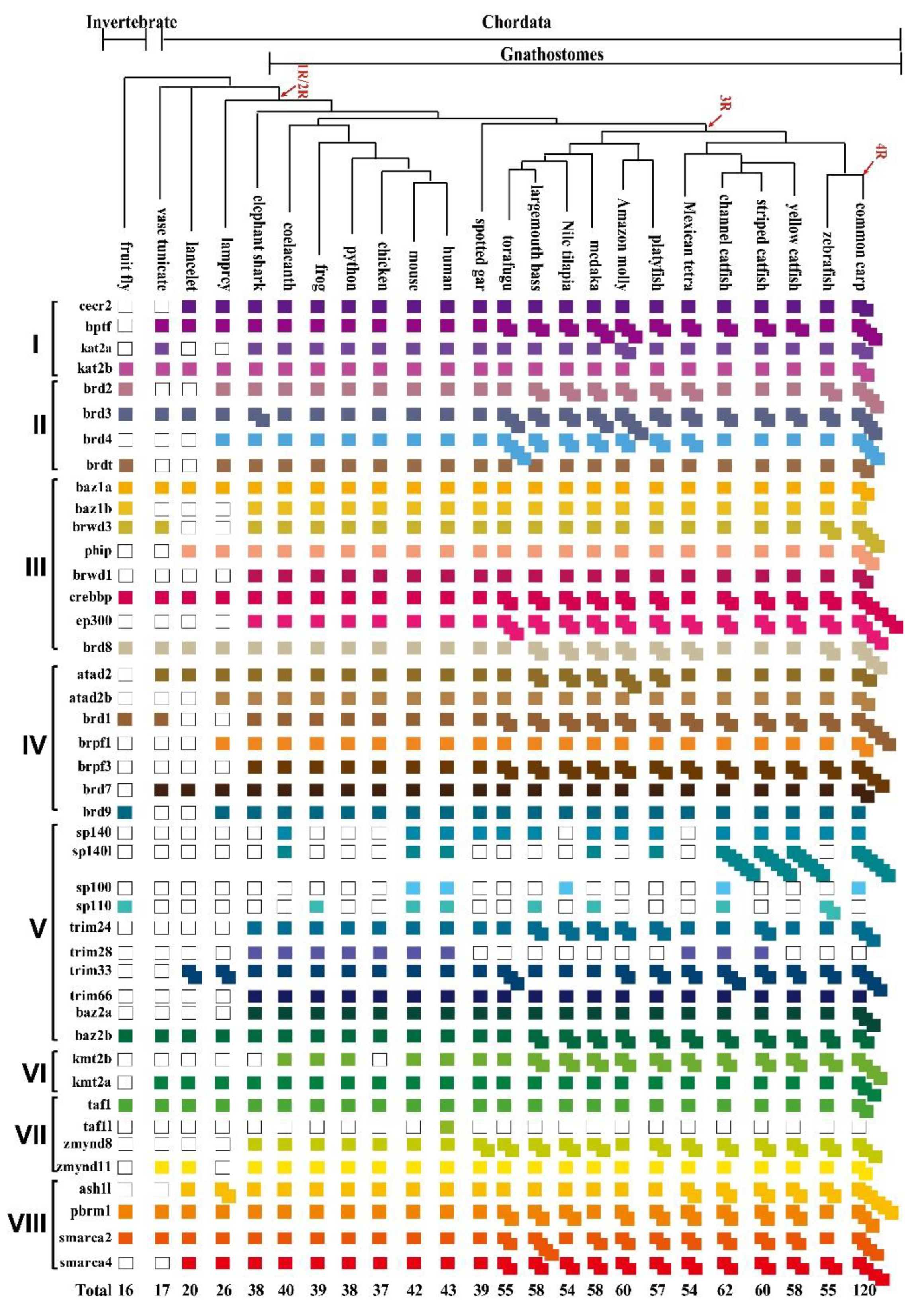
3.1. Identification of BRDs from Tilapia and Representative Species
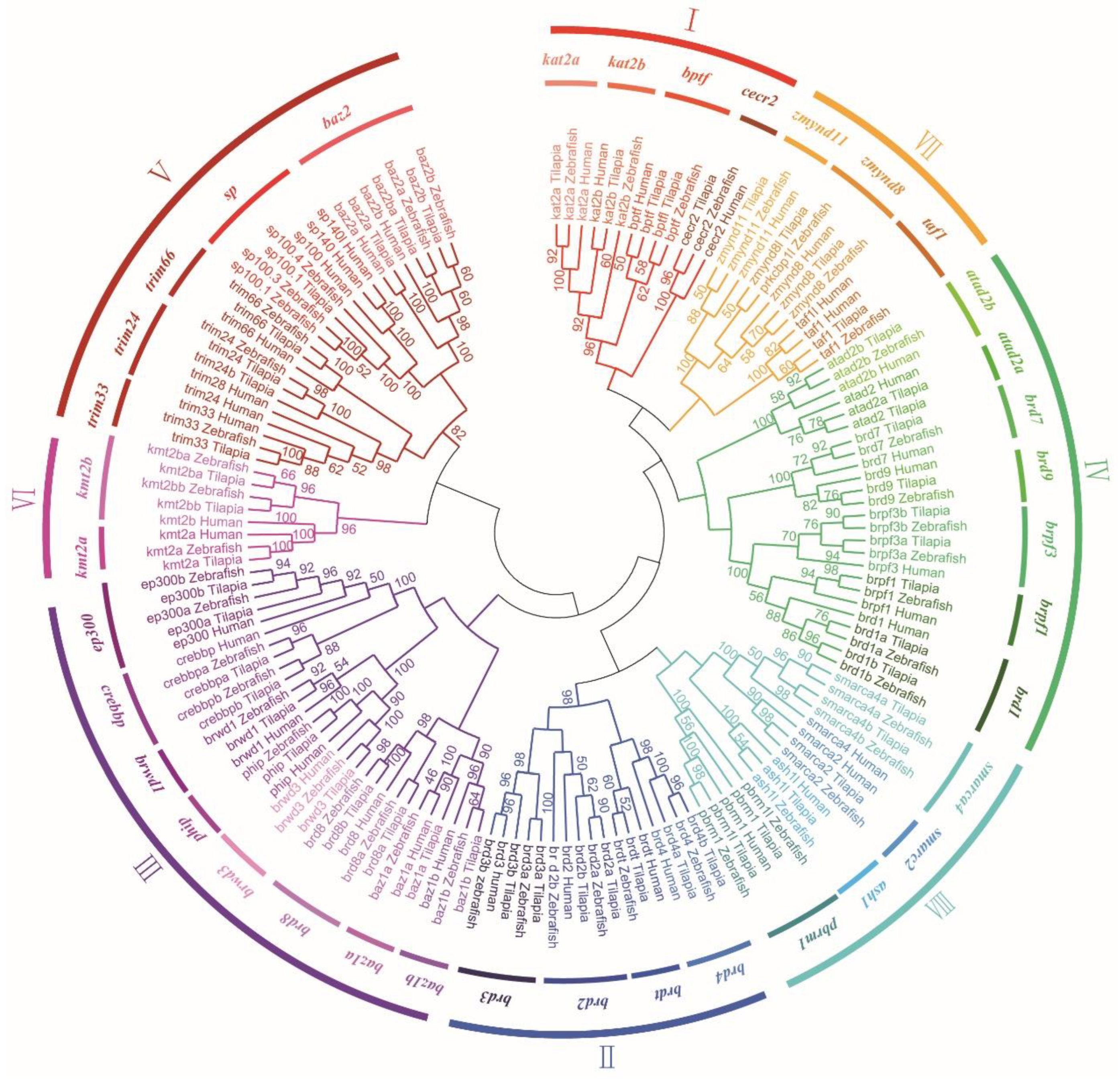
3.2. Phylogenetic and Syntenic Analyses of BRD Members
3.3. Chromosomal Localization of BRD Genes and BRD Protein Domains in Tilapia
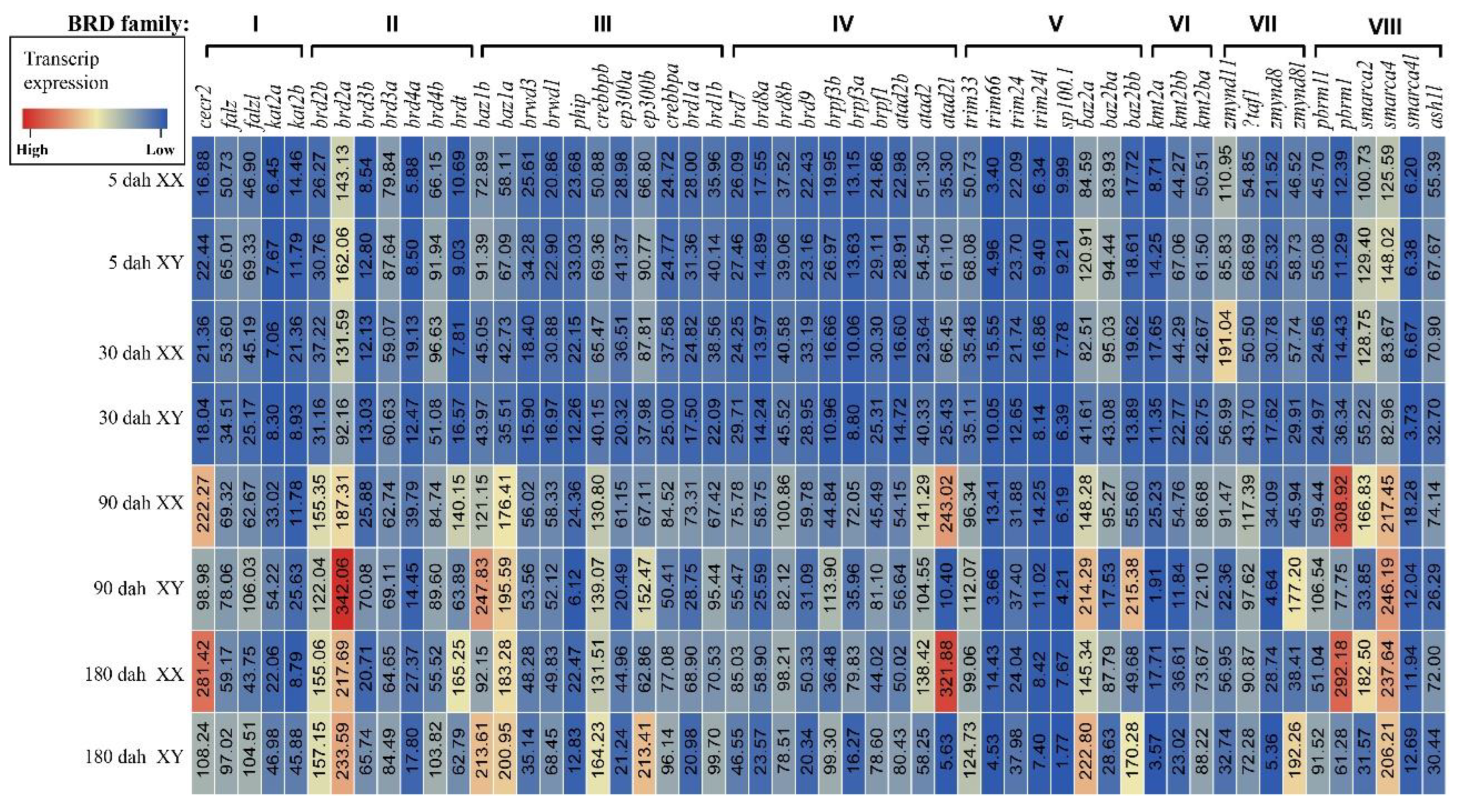
3.4. Expression and Distribution of BRDs in Eight Tilapia Tissues
3.5. Expression of BRDs in Tilapia Gonads at Different Stages
3.6. Expression Validation and Cellular Location of BRDs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Haynes, S.R.; Dollard, C.; Winston, F.; Beck, S.; Trowsdale, J.; Dawid, I.B. The bromodomain: A conserved sequence found in human, Drosophila and yeast proteins. Nucleic Acids Res. 1992, 20, 2603. [Google Scholar] [CrossRef] [PubMed]
- Jeanmougin, F.; Wurtz, J.M.; Le Douarin, B.; Chambon, P.; Losson, R. The bromodomain revisited. Trends Biochem. Sci. 1997, 22, 151–153. [Google Scholar] [CrossRef]
- Filippakopoulos, P.; Picaud, S.; Mangos, M.; Keates, T.; Lambert, J.P.; Barsyte-Lovejoy, D.; Felletar, I.; Volkmer, R.; Müller, S.; Pawson, T.; et al. Histone recognition and large-scale structural analysis of the human bromodomain family. Cell 2012, 149, 214–231. [Google Scholar] [CrossRef]
- Dhalluin, C.; Carlson, J.E.; Zeng, L.; He, C.; Aggarwal, A.K.; Zhou, M.M. Structure and ligand of a histone acetyltransferase bromodomain. Nature 1999, 399, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Fujisawa, T.; Filippakopoulos, P. Functions of bromodomain-containing proteins and their roles in homeostasis and cancer. Nat. Rev. Mol. Cell Biol. 2017, 18, 246–262. [Google Scholar] [CrossRef]
- Muller, S.; Filippakopoulos, P.; Knapp, S. Bromodomains as therapeutic targets. Expert Rev. Mol. Med. 2011, 13, e29. [Google Scholar] [CrossRef]
- Lee, S.Y.; Kim, J.J.; Miller, K.M. Bromodomain proteins: Protectors against endogenous DNA damage and facilitators of genome integrity. Exp. Mol. Med. 2021, 53, 1268–1277. [Google Scholar] [CrossRef]
- Tamkun, J.W.; Deuring, R.; Scott, M.P.; Kissinger, M.; Pattatucci, A.M.; Kaufman, T.C.; Kennison, J.A. brahma: A regulator of Drosophila homeotic genes structurally related to the yeast transcriptional activator SNF2/SWI2. Cell 1992, 68, 561–572. [Google Scholar] [CrossRef]
- Zhang, J.; Huang, L.; Zhang, P.; Huang, X.; Yang, W.; Liu, R.; Sun, Q.; Lu, Y.; Zhang, M.; Fu, Q. Genomic Identification, Evolution, and Expression Analysis of Bromodomain Genes Family in Buffalo. Genes 2022, 13, 103. [Google Scholar] [CrossRef]
- DiBenedetto, A.J.; Guinto, J.B.; Ebert, T.D.; Bee, K.J.; Schmidt, M.M.; Jackman, T.R. Zebrafish brd2a and brd2b are paralogous members of the bromodomain-ET (BET) family of transcriptional coregulators that show structural and expression divergence. BMC Dev. Biol. 2008, 8, 39. [Google Scholar] [CrossRef] [Green Version]
- Pivot-Pajot, C.; Caron, C.; Govin, J.; Vion, A.; Rousseaux, S.; Khochbin, S. Acetylation-dependent chromatin reorganization by BRDT, a testis-specific bromodomain-containing protein. Mol. Cell Biol. 2003, 23, 5354–5365. [Google Scholar] [CrossRef] [PubMed]
- Severinsen, J.; Bjarkam, C.R.; Kiar-Larsen, S.; Olsen, I.M.; Nielsen, M.M.; Blechingberg, J.; Nielsen, A.L.; Holm, I.E.; Foldagr, L.; Young, B.D. Evidence implicating BRD1 with brain development and susceptibility to both schizophrenia and bipolar affective disorder. Mol. Psychiatry 2006, 11, 1126–1138. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.A.; Lee, J.W.; Kim, H.S.; Lee, Y.Y.; Kim, T.J.; Choi, C.H.; Choi, J.J.; Jeon, H.K.; Cho, Y.J.; Ryu, J.Y. Tumor suppressive effects of bromodomain-containing protein 7 (BRD7) in epithelial ovarian carcinoma. Clin. Cancer Res. 2014, 20, 565–575. [Google Scholar] [CrossRef] [PubMed]
- Browne, J.A.; NandyMazumdar, M.; Paranjapye, A.; Leir, S.H.; Harris, A. The bromodomain containing 8 (BRD8) transcriptional network in human lung epithelial cells. Mol. Cell Endocrinol. 2021, 524, 111169. [Google Scholar] [CrossRef]
- Zaware, N.; Zhou, M.M. Bromodomain biology and drug discovery. Nat. Struct. Mol. Biol. 2019, 26, 870–879. [Google Scholar] [CrossRef] [PubMed]
- Russell, J.O.; Ko, S.; Saggi, H.S.; Singh, S.; Poddar, M.; Shin, D.; Monga, S.P. Bromodomain and Extraterminal (BET) Proteins Regulate Hepatocyte Proliferation in Hepatocyte-Driven Liver Regeneration. Am. J. Pathol. 2018, 188, 1389–1405. [Google Scholar] [CrossRef] [PubMed]
- Andricovich, J.; Perkail, S.; Kai, Y.; Casasanta, N.; Peng, W.; Tzatsos, A. Loss of KDM6A Activates Super-Enhancers to Induce Gender-Specific Squamous-like Pancreatic Cancer and Confers Sensitivity to BET Inhibitors. Cancer Cell 2018, 33, 512–526. [Google Scholar] [CrossRef]
- Piunti, A.; Hashizume, R.; Morgan, M.A.; Bartom, E.T.; Horbinski, C.M.; Marshall, S.A.; Rendleman, E.J.; Ma, Q.; Takahashi, Y.H.; Woodfin, A.R.; et al. Therapeutic targeting of polycomb and BET bromodomain proteins in diffuse intrinsic pontine gliomas. Nat. Med. 2017, 23, 493–500. [Google Scholar] [CrossRef]
- Shang, E.; Salazar, G.; Crowley, T.E.; Wang, X.; Lopez, R.A.; Wang, X.; Wolgemuth, D.J. Identification of unique, differentiation stage-specific patterns of expression of the bromodomain-containing genes Brd2, Brd3, Brd4, and Brdt in the mouse testis. Gene Expr. Patterns 2004, 4, 513–519. [Google Scholar] [CrossRef]
- Bryant, J.M.; Berger, S.L. Low-hanging fruit: Targeting Brdt in the testes. EMBO J. 2012, 31, 3788–3789. [Google Scholar] [CrossRef] [Green Version]
- Berkovits, B.D.; Wolgemuth, D.J. The Role of the Double Bromodomain-Containing BET Genes During Mammalian Spermatogenesis. Curr. Top. Dev. Biol. 2013, 102, 293–326. [Google Scholar]
- Matzuk, M.M.; McKeown, M.R.; Filippakopoulos, P.; Li, Q.; Ma, L.; Agno, J.E.; Lemieux, M.E.; Picaud, S.; Richard, N.Y.; Qi, J. Small-molecule inhibition of BRDT for male contraception. Cell 2012, 150, 673–684. [Google Scholar] [CrossRef] [PubMed]
- Úbeda-Manzanaro, M.; Ortiz-Delgado, J.B.; Sarasquete, C. The Bromodomain testis-specific gene (Brdt) characterization and expression in gilthead seabream, Sparus aurata, and European seabass, Dicentrarchus labrax. Eur. J. Histochem. 2016, 60, 2638. [Google Scholar] [CrossRef] [PubMed]
- Shang, E.; Nickerson, H.D.; Wen, D.; Wang, X.; Wolgemuth, D.J. The first bromodomain of Brdt, a testis-specific member of the BET sub-family of double-bromodomain-containing proteins, is essential for male germ cell differentiation. Development 2007, 134, 3507–3515. [Google Scholar] [CrossRef]
- Aston, K.I.; Krausz, C.; Laface, I.; Ruiz-Castane, E.; Carrell, D.T. Evaluation of 172 candidate polymorphisms for association with oligozoospermia or azoospermia in a large cohort of men of European descent. Hum. Reprod. 2010, 25, 1383–1397. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sang, M.; Gong, S.; Chen, Z.; Zhao, X.; Wang, G.; Li, Z.; Huang, Y.; Chen, S.; Xie, G. BET bromodomain inhibitor JQ1 regulates spermatid development by changing chromatin conformation in mouse spermatogenesis. Genes Dis. 2021, 9, 1062–1073. [Google Scholar] [CrossRef]
- Brawand, D.; Wagner, C.E.; Li, Y.I.; Malinsky, M.; Keller, I.; Fan, S.; Simakov, O.; Ng, A.Y.; Lim, Z.W.; Bezault, E. The genomic substrate for adaptive radiation in African cichlid fish. Nature 2014, 513, 375–381. [Google Scholar] [CrossRef]
- Tao, W.; Yuan, J.; Zhou, L.; Sun, L.; Sun, Y.; Yang, S.; Li, M.; Zeng, S.; Huang, B.; Wang, D. Characterization of gonadal transcriptomes from Nile tilapia (Oreochromis niloticus) reveals differentially expressed genes. PLoS ONE 2013, 8, 63604. [Google Scholar] [CrossRef]
- Emms, D.M.; Kelly, S. OrthoFinder: Solving fundamental biases in whole genome comparisons dramatically improves orthogroup inference accuracy. Genome Biol. 2015, 16, 157. [Google Scholar] [CrossRef]
- Tavares, D.L.M.; Canete, S.C.; Henkin, R.; Estrela, P.C.; Freitas, T.R.O.; Galante1, R.; Freitas, C.M.D.S. Taxonomy Browser: A biodiversity data management system. J. Comput. Interdiscip. Sci. 2011, 2, 37–46. [Google Scholar]
- Hall, T. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7. Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Muffato, M.; Louis, A.; Poisnel, C.E.; Roest Crollius, H. Genomicus: A database and a browser to study gene synteny in modern and ancestral genomes. Bioinformatics 2010, 26, 1119–1121. [Google Scholar] [CrossRef]
- Chen, C.; Chen, H.; Zhang, Y.; Thomas, H.R.; Frank, M.H.; He, Y.; Xia, R. TBtools: An Integrative Toolkit Developed for Interactive Analyses of Big Biological Data. Mol. Plant 2020, 13, 1194–1202. [Google Scholar] [CrossRef]
- Tao, W.; Chen, J.; Tan, D.; Yang, J.; Sun, L.; Wei, J.; Conte, M.A.; Kocher, T.D.; Wang, D. Transcriptome display during tilapia sex determination and differentiation as revealed by RNA-Seq analysis. BMC Genom. 2018, 19, 363. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.N.; Jiang, X.L.; Xie, Q.P.; Yuan, J.; Huang, B.F.; Tao, W.J.; Zhou, L.Y.; Nagahama, Y.; Wang, D.S. Transdifferentiation of differentiated ovary into functional testis by long-term treatment of aromatase inhibitor in Nile tilapia. Endocrinology 2014, 155, 1476–1488. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Wang, F.; Qin, Z.; Li, Z.; Yang, S.; Gao, T.; Sun, L.; Wang, D. Dnmt3aa but Not Dnmt3ab is Required for Maintenance of Gametogenesis in Nile Tilapia (Oreochromis niloticus). Int J. Mol. Sci. 2021, 22, 10170. [Google Scholar] [CrossRef]
- Yang, L.; Li, Y.; Wu, Y.; Sun, S.; Song, Q.; Wei, J.; Sun, L.; Li, M.; Wang, D.; Zhou, L. Rln3a is a prerequisite for spermatogenesis and fertility in male fish. J. Steroid Biochem. Mol. Biol. 2020, 197, 105517. [Google Scholar] [CrossRef]
- Wang, D.S.; Kobayashi, T.; Zhou, L.Y.; Paul-Prasanth, B.; Ijiri, S.; Sakai, F.; Okubo, K.; Morohashi, K.; Nagahama, Y. Foxl2 up-regulates aromatase gene transcription in a female-specific manner by binding to the promoter as well as interacting with ad4 binding protein/steroidogenic factor 1. Mol. Endocrinol. 2007, 21, 712–725. [Google Scholar] [CrossRef]
- Magadum, S.; Banerjee, U.; Murugan, P.; Gangapur, D.; Ravikesavan, R. Gene duplication as a major force in evolution. J. Genet. 2013, 92, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Hoegg, S.; Brinkmann, H.; Taylor, J.S.; Meyer, A. Phylogenetic timing of the fish-specific genome duplication correlates with the diversification of teleost fish. J. Mol. Evol. 2004, 59, 190–203. [Google Scholar] [CrossRef] [PubMed]
- Escriva, H.; Manzon, L.; Youson, J.; Laudet, V. Analysis of lamprey and hagfish genes reveals a complex history of gene duplications during early vertebrate evolution. Mol. Biol. Evol. 2002, 19, 1440–1450. [Google Scholar] [CrossRef] [Green Version]
- Chiu, C.H.; Dewar, K.; Wagner, G.P.; Takahashi, K.; Ruddle, F.; Ledje, C.; Bartsch, P.; Scemama, J.L.; Stellwag, E.; Fried, C. Bichir HoxA cluster sequence reveals surprising trends in ray-finned fish genomic evolution. Genome Res. 2004, 14, 11–17. [Google Scholar] [CrossRef]
- Kuraku, S.; Meyer, A.; Kuratani, S. Timing of genome duplications relative to the origin of the vertebrates: Did cyclostomes diverge before or after? Mol. Biol. Evol. 2009, 26, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Gillis, W.Q.; St John, J.; Bowerman, B.; Schneider, S.Q. Whole genome duplications and expansion of the vertebrate GATA transcription factor gene family. BMC Evol. Biol. 2009, 9, 207. [Google Scholar] [CrossRef]
- Kuang, G.; Tao, W.; Zheng, S.; Wang, X.; Wang, D. Genome-Wide Identification, Evolution and Expression of the Complete Set of Cytoplasmic Ribosomal Protein Genes in Nile Tilapia. Int. J. Mol. Sci. 2020, 21, 1230. [Google Scholar] [CrossRef]
- Liu, X.Y.; Zhang, X.-B.; Li, M.-H.; Zheng, S.Q.; Liu, Z.L.; Cheng, Y.Y.; Wang, D.S. Genome-wide identification, evolution of chromobox family genes and their expression in Nile tilapia. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2017, 203, 25–34. [Google Scholar] [CrossRef]
- Charlop-Powers, Z.; Zeng, L.; Zhang, Q.; Zhou, M.M. Structural insights into selective histone H3 recognition by the human Polybromo bromodomain 2. Cell Res. 2010, 20, 529–538. [Google Scholar] [CrossRef]
- Park, S.; Stanfield, R.L.; Martinez-Yamout, M.A.; Dyson, H.J.; Wilson, I.A.; Wright, P.E. Role of the CBP catalytic core in intramolecular SUMOylation and control of histone H3 acetylation. Proc. Natl. Acad. Sci. USA 2017, 114, E5335–E5342. [Google Scholar] [CrossRef]
- Umehara, T.; Nakamura, Y.; Jang, M.K.; Nakano, K.; Tanaka, A.; Ozato, K.; Padmanabhan, B.; Yokoyama, S. Structural basis for acetylated histone H4 recognition by the human BRD2 bromodomain. J. Biol. Chem. 2010, 285, 7610–7618. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Zhang, J.; Shen, W.; Wang, X.; Wu, J.; Wu, J.; Shi, Y. Solution structure of the second bromodomain of Brd2 and its specific interaction with acetylated histone tails. BMC Struct. Biol. 2007, 7, 57. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, J.T.; Glass, K.C. Biological function and histone recognition of family IV bromodomain-containing proteins. J. Cell Physiol. 2018, 233, 1877–1886. [Google Scholar] [CrossRef]
- Li, H.; Ilin, S.; Wang, W.; Duncan, E.M.; Wysocka, J.; Allis, C.D.; Patel, D.J. Molecular basis for site-specific read-out of histone H3K4me3 by the BPTF PHD finger of NURF. Nature 2006, 442, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Belkina, A.C.; Denis, G.V. BET domain co-regulators in obesity, inflammation and cancer. Nat. Rev. Cancer 2012, 12, 465–477. [Google Scholar] [CrossRef]
- Wang, F.; Liu, H.; Blanton, W.P.; Belkina, A.; Lebrasseur, N.K.; Denis, G.V. Brd2 disruption in mice causes severe obesity without Type 2 diabetes. Biochem. J. 2009, 425, 71–83. [Google Scholar] [CrossRef]
- Shang, E.; Wang, X.; Wen, D.; Greenberg, D.A.; Wolgemuth, D.J. Double bromodomain-containing gene Brd2 is essential for embryonic development in mouse. Dev. Dyn. 2009, 238, 908–917. [Google Scholar] [CrossRef]
- Yu, Z.; Chen, T.; Mo, H.; Guo, C.; Liu, Q. BRD8, which is negatively regulated by miR-876–3p, promotes the proliferation and apoptosis resistance of hepatocellular carcinoma cells via KAT5. Arch. Biochem. Biophys. 2020, 693, 108550. [Google Scholar] [CrossRef]
- Lu, J.; Qian, C.; Ji, Y.; Bao, Q.; Lu, B. Gene Signature Associated with Bromodomain Genes Predicts the Prognosis of Kidney Renal Clear Cell Carcinoma. Front. Genet. 2021, 12, 643935. [Google Scholar] [CrossRef]
- Jones, D.T.; Jager, N.; Kool, M.; Zichner, T.; Hutter, B.; Sultan, M.; Cho, Y.J.; Pugh, T.J.; Hovestadt, V.; Stutz, A.M.; et al. Dissecting the genomic complexity underlying medulloblastoma. Nature 2012, 488, 100–105. [Google Scholar] [CrossRef]
- Hasselblatt, M.; Thomas, C.; Federico, A.; Nemes, K.; Johann, P.D.; Bison, B.; Bens, S.; Dahlum, S.; Kordes, U.; Redlich, A.; et al. SMARCB1-deficient and SMARCA4-deficient Malignant Brain Tumors with Complex Copy Number Alterations and TP53 Mutations May Represent the First Clinical Manifestation of Li-Fraumeni Syndrome. Am. J. Surg. Pathol. 2022, 46, 1277–1283. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.; Kong, Q.; Li, C.; Mao, B. Xenopus RCOR2 (REST corepressor 2) interacts with ZMYND8, which is involved in neural differentiation. Biochem. Biophys. Res. Commun. 2010, 394, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhao, R.; Guo, C.; Jiang, S.; Yang, J.; Xu, Y.; Liu, Y.; Fan, L.; Xiong, W.; Ma, J.; et al. Knockout of BRD7 results in impaired spermatogenesis and male infertility. Sci. Rep. 2016, 6, 21776. [Google Scholar] [CrossRef] [Green Version]
- Trousdale, R.K.; Wolgemuth, D.J. Bromodomain containing 2 (Brd2) is expressed in distinct patterns during ovarian folliculogenesis independent of FSH or GDF9 action. Mol. Reprod. Dev. 2004, 68, 261–268. [Google Scholar] [CrossRef]
- Shibata, Y.; Uchida, M.; Takeshita, H.; Nishiwaki, K.; Sawa, H. Multiple functions of PBRM-1/Polybromo- and LET-526/Osa-containing chromatin remodeling complexes in C. elegans development. Dev. Biol. 2012, 361, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P.J.; Norton, K.A.; Niri, F.H.; Dawe, C.E.; McDermid, H.E. CECR2 is involved in spermatogenesis and forms a complex with SNF2H in the testis. J. Mol. Biol. 2012, 415, 793–806. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yan, J. Directional divergence of Ep300 duplicates in teleosts and its implications. BMC Evol. Biol. 2020, 20, 140. [Google Scholar] [CrossRef]
- Paillisson, A.; Levasseur, A.; Gouret, P.; Callebaut, I.; Bontoux, M.; Pontarotti, P.; Monget, P. Bromodomain testis-specific protein is expressed in mouse oocyte and evolves faster than its ubiquitously expressed paralogs BRD2, -3, and -4. Genomics 2007, 89, 215–223. [Google Scholar] [CrossRef]
- Wang, P.J.; Manterola, M.; Brown, T.M.; Oh, M.Y.; Garyn, C.; Gonzalez, B.J.; Wolgemuth, D.J. BRDT is an essential epigenetic regulator for proper chromatin organization, silencing of sex chromosomes and crossover formation in male meiosis. PLOS Genet. 2018, 14, e1007209. [Google Scholar]
- Gaucher, J.; Boussouar, F.; Montellier, E.; Curtet, S.; Buchou, T.; Bertrand, S.; Hery, P.; Jounier, S.; Depaux, A.; Vitte, A.L. Bromodomain-dependent stage-specific male genome programming by Brdt. EMBO J. 2012, 31, 3809–3820. [Google Scholar] [CrossRef]
- Berkovits, B.D.; Wolgemuth, D.J. The first bromodomain of the testis-specific double bromodomain protein Brdt is required for chromocenter organization that is modulated by genetic background. Dev. Biol. 2011, 360, 358–368. [Google Scholar] [CrossRef] [PubMed]
- Barda, S.; Paz, G.; Yogev, L.; Yavetz, H.; Lehavi, O.; Hauser, R.; Botchan, A.; Breitbart, H.; Kleiman, S.E. Expression of BET genes in testis of men with different spermatogenic impairments. Fertil. Steril. 2012, 97, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Jostes, S.; Nettersheim, D.; Fellermeyer, M.; Schneider, S.; Hafezi, F.; Honecker, F.; Schumacher, V.; Geyer, M.; Kristiansen, G.; Schorle, H. The bromodomain inhibitor JQ1 triggers growth arrest and apoptosis in testicular germ cell tumours in vitro and in vivo. J. Cell Mol. Med. 2017, 21, 1300–1314. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, C.; Yu, M.; Zhang, Q.; Ma, Z.; Du, K.; You, H.; Wei, J.; Wang, D.; Tao, W. Genome-Wide Identification and Characterization of the BRD Family in Nile Tilapia (Oreochromis niloticus). Animals 2022, 12, 2266. https://doi.org/10.3390/ani12172266
Xu C, Yu M, Zhang Q, Ma Z, Du K, You H, Wei J, Wang D, Tao W. Genome-Wide Identification and Characterization of the BRD Family in Nile Tilapia (Oreochromis niloticus). Animals. 2022; 12(17):2266. https://doi.org/10.3390/ani12172266
Chicago/Turabian StyleXu, Chunmei, Miao Yu, Qingqing Zhang, Zhisheng Ma, Kang Du, Huiqin You, Jing Wei, Deshou Wang, and Wenjing Tao. 2022. "Genome-Wide Identification and Characterization of the BRD Family in Nile Tilapia (Oreochromis niloticus)" Animals 12, no. 17: 2266. https://doi.org/10.3390/ani12172266




