Effects of Housing and Management Systems on the Growth, Immunity, Antioxidation, and Related Physiological and Biochemical Indicators of Donkeys in Cold Weather
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Experiment Design
2.2. Meteorological Observation
2.3. Measurement of Growth Performance and Nutrient Digestibility
2.4. Measurement of Basic Physiological Indexes
2.5. Preparation and Analysis of Blood Sample
2.6. Statistical Analysis
3. Results
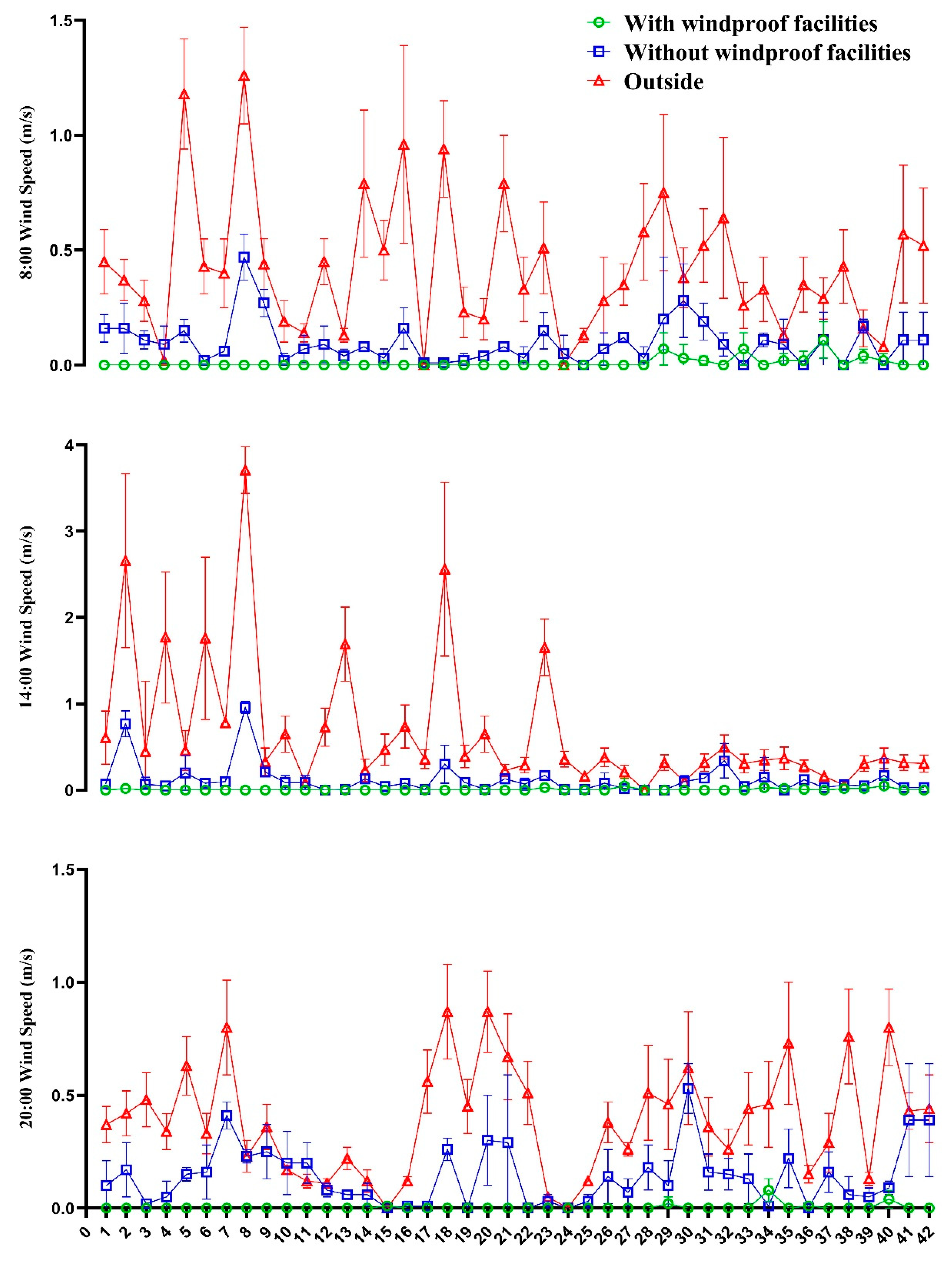
3.1. Ambient Temperature and Wind Speed
3.2. Growth Performance and Nutrient Digestibility
3.3. Basic Physiological Indexes
3.4. Blood Biochemical and Hormone Indexes
3.5. Immune and Antioxidative Indexes
4. Discussions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Simenew, K.; Gezahegne, M.; Getachew, M.; Wondyefraw, M.; Eyob, I. Reference Values of Clinically Important Physiological, Hematological and Serum Biochemical Parameters of Apparently Healthy Working Equids of Ethiopia. Glob. Vet. 2011, 7, 01–06. [Google Scholar]
- Turner, J.W. Genetic and biological aspects of Zebu adaptability. J. Anim. Sci. 1980, 50, 1201–1205. [Google Scholar] [CrossRef] [PubMed]
- Lenis Sanin, Y.; Zuluaga Cabrera, A.M.; Tarazona Morales, A.M. Adaptive responses to thermal stress in mammals. Rev. Bras. Med. 2016, 31, 121–135. [Google Scholar]
- Zhang, D.-J.; Liu, D.; Wang, L.; Wang, W.-T.; He, X.-M.; Yang, G.-W. Gene expression profile analysis of pig muscle in response to cold stress. J. Appl. Anim. Res. 2017, 45, 195–198. [Google Scholar]
- Young, B. Cold stress as it affects animal production. J. Anim. Sci. 1981, 52, 154–163. [Google Scholar] [CrossRef]
- Young, B. Ruminant cold stress: Effect on production. J. Anim. Sci. 1983, 57, 1601–1607. [Google Scholar] [CrossRef]
- Gwazdauskas, F.C. Effects of climate on reproduction in cattle. J. Dairy Sci. 1985, 68, 1568–1578. [Google Scholar] [CrossRef]
- Alves, F.M.S.; Felix, G.A.; Almeida, I.C.L.; Nääs, I.A.; Souza, G.M.; Caldara, F.R.; Garcia, R.G. Impact of exposure to cold on layer production. Revista Brasileira de Ciência Avícola 2012, 14, 223–226. [Google Scholar] [CrossRef]
- Fukuhara, K.; Kvetnansky, R.; Cizza, G.; Pacak, K.; Ohara, H.; Goldstein, D.S.; Kopin, I.J. Interrelations between sympathoadrenal system and hypothalamo-pituitary-adrenocortical/thyroid systems in rats exposed to cold stress. J. Neuroendocrinol. 1996, 8, 533–541. [Google Scholar] [CrossRef]
- He, J.; Zheng, W.; Tao, C.; Guo, H.; Xue, Y.; Zhao, R.; Yao, W. Heat stress during late gestation disrupts maternal microbial transmission with altered offspring’s gut microbial colonization and serum metabolites in a pig model. Environ. Pollut. 2020, 266, 115111. [Google Scholar] [CrossRef]
- Hangalapura, B.N.; Nieuwland, M.G.; de Vries Reilingh, G.; Heetkamp, M.J.; Van den Brand, H.; Kemp, B. Effects of cold stress on immune responses and body weight of chicken lines divergently selected for antibody responses to sheep red blood cells. Poult. Sci. 2003, 82, 1692–1700. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.; Greene, E.; Ishola, P.; Huff, G.; Donoghue, A.; Bottje, W. Chronic mild cold conditioning modulates the expression of hypothalamic neuropeptide and intermediary metabolic-related genes and improves growth performances in young chicks. PLoS ONE 2015, 10, e0142319. [Google Scholar]
- Chandra, J.; Samali, A.; Orrenius, S. Triggering and modulation of apoptosis by oxidative stress. Free Radic. Biol. Med. 2000, 3, 323–333. [Google Scholar] [CrossRef]
- Meng, X.; Liu, P.; Li, J.; Gao, B.; Chen, P. Physiological responses of swimming crab Portunus trituberculatus under cold acclimation: Antioxidant defense and heat shock proteins. Aquaculture 2014, 434, 11–17. [Google Scholar] [CrossRef]
- Miest, J.J. Apoptosis and Its Association with Immunomodulation and Disease in Common Carp (Cyprinus carpio L.); Keele University: Keele, UK, 2013. [Google Scholar]
- Escarcha, J.F.; Lassa, J.A.; Zander, K.K. Livestock under climate change: A systematic review of impacts and adaptation. Climate 2018, 6, 54. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis, 18th ed.; Association of Official Analytical Chemists: Washington, DC, USA, 2004. [Google Scholar]
- Van Keulen, J.; Young, B.A. Evaluation of acid-insoluble ash as a natural marker in ruminant digestibility studies. J. Anim. Sci. 1977, 44, 282–287. [Google Scholar] [CrossRef]
- McBride, G.E.; Christopherson, R.J.; Sauer, W. Metabolic rate and plasma thyroid hormone concentrations of mature horses in response to changes in ambient temperature. Can. J. Anim. Sci. 1985, 65, 375–382. [Google Scholar] [CrossRef]
- Chen, X.Y.; Li, R.; Wang, M.; Geng, Z.Y. Identification of differentially expressed genes in hypothalamus of chicken during cold stress. Mol. Biol. Rep. 2014, 41, 2243–2248. [Google Scholar] [CrossRef]
- Yang, X.; Luo, Y.H.; Zeng, Q.F.; Zhang, K.Y.; Ding, X.M.; Bai, S.P.; Wang, J.P. Effects of low ambient temperatures and dietary vitamin C supplement on growth performance, blood parameters, and antioxidant capacity of 21-day-old broilers. Poult. Sci. 2014, 93, 898–905. [Google Scholar] [CrossRef]
- Nonnecke, B.J.; Foote, M.R.; Miller, B.L.; Fowler, M.; Johnson, T.E.; Horst, R.L. Effects of chronic environmental cold on growth, health, and select metabolic and immunologic responses of preruminant calves. J. Dairy Sci. 2009, 92, 6134–6143. [Google Scholar] [CrossRef]
- Kennedy, P.M.; Early, R.J.; Christopherson, R.J.; Milligan, L.P. Nitrogen transformations and duodenal amino acid content in sheep given four forage diets and exposed to warm and cold ambient temperatures. Can. J. Anim. Sci. 1986, 66, 951–957. [Google Scholar] [CrossRef]
- Graham, A.D.; Christopherson, R.J. Effects of adrenaline and noradrenaline on the heat production of warm- and cold-acclimated sheep. Can. J. Physiol. Pharm. 1981, 59, 985–993. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.W.G.; Mcqueen, R.E.; Burgess, P.L. Effect of cold on digestibility of chopped or pelleted hay by sheep. Can. J. Anim. Sci. 1980, 60, 571. [Google Scholar]
- Razzaque, M.A.; Abbas, S.; Bedair, A.M. Performance of pre-weaned female calves confined in housing and open environment hutches in Kuwait. Pak. Vet. J. 2009, 29, 1–4. [Google Scholar]
- Alsarra, I.; Ahmed, M.; Alanazi, F.; ElTahir, H.; Alsheikh, M.; Neau, S. Influence of Cyclodextrin Complexation with NSAIDs on NSAID/Cold Stress-Induced Gastric Ulceration in Rats. Int. J. Med. Sci. 2010, 7, 232. [Google Scholar] [CrossRef][Green Version]
- Sykes, A.R.; Slee, J. Acclimatization of Scottish blackface sheep to cold. 2. skin temperature, heart rate, respiration rate, shivering intensity and skinfold thickness. Anim. Sci. 1968, 10, 17–35. [Google Scholar] [CrossRef]
- Diesel, D.A.; Tucker, A.; Robertshaw, D. Cold-induced changes in breathing pattern as a strategy to reduce respiratory heat loss. J. Appl. Physiol. 1990, 69, 1946–1952. [Google Scholar] [CrossRef]
- Feldman, B.F.; Zinkl, J.G.; Jain, N.C. Schalm’s Veterinary Hematology; Lippincott Williams and Wilkins: Philadelphia, PA, USA; Baltimore, MD, USA; New York, NY, USA; London, UK; Buenos Aires, Argentina; Hong Kong, China; Sydney, Australia; Tokyo, Japan, 2002. [Google Scholar]
- Otto, F.; Baggasse, P.; Bogin, E.; Harun, M.; Vilela, F. Biochemical blood profile of Angoni cattle in Mozambique. Israel Vet. Med. Assoc. 2000, 55, 95–102. [Google Scholar]
- Davis, S.L. Environmental modulation of the immune system via the endocrine system. Domest. Anim. Endocrin. 1998, 15, 283–289. [Google Scholar] [CrossRef]
- Nilaweera, K.N.; Speakman, J.R. Regulation of intestinal growth in response to variations in energy supply and demand. Obes. Rev. 2018, 19, 61–72. [Google Scholar]
- Yu, A.S.; McCarthy, K.M.; Francis, S.A.; McCormack, J.M.; Lai, J.; Rogers, R.A.; Lynch, R.D.; Schneeberger, E.E. Knockdown of occludin expression leads to diverse phenotypic alterations in epithelial cells. American Journal of Physiology. Cell Physiol. 2005, 288, C1231–C1241. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.W.; Bi, M.Y.; Yao, H.D.; Fu, J.; Li, S.; Xu, S.W. Effect of cold stress on expression of AMPKα–PPARα pathway and inflammation genes. Avian Dis. 2014, 58, 415–426. [Google Scholar] [CrossRef] [PubMed]
- Stanley, C.C.; Williams, C.C.; Jenny, B.F.; Fernandez, J.M.; Bateman, H.G.; Nipper, W.A.; Lovejoy, J.C.; Gantt, D.T.; Goodier, G.E. Effects of Feeding Milk Replacer Once Versus Twice Daily on Glucose Metabolism in Holstein and Jersey Calves 1. J. Dairy Sci. 2002, 85, 2335–2343. [Google Scholar] [CrossRef]
- Kanungo, S.; Wells, K.; Tribett, T.; El-Gharbawy, A. Glycogen metabolism and glycogen storage disorders. Ann. Transl. Med. 2018, 6, 474. [Google Scholar] [CrossRef] [PubMed]
- Cockrem, J.F.; Barrett, D.P.; Candy, E.J.; Potter, M.A. CORT responses in birds: Individual variation and repeatability in Adelie penguins (Pygoscelis adeliae) and other species, and the use of power analysis to determine sample sizes. Gen. Comp. Endocr. 2009, 163, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Goodrich, E.L.; Behling-Kelly, E. Clinical pathology of donkeys and mules. Vet. Clin. N. Am. Equine Pract. 2019, 35, 433–455. [Google Scholar] [CrossRef]
- Helmreich, D.L.; Parfitt, D.B.; Lu, X.Y. Relation between the hypothalamic-pituitary-thyroid (HPT) axis and the hypothalamic-pituitary-adrenal (HPA) axis during repeated stress. Neuroendocrinology 2005, 81, 183–192. [Google Scholar] [CrossRef]
- Hauger, R.L.; Lorang, M.; Irwin, M.; Aguilera, G. CRF receptor regulation and sensitization of ACTH responses to acute ether stress during chronic intermittent immobilization stress. Brain Res. 1990, 532, 34–40. [Google Scholar] [CrossRef]
- Young, E.A.; Akana, S.F.; Dallman, M.F. Decreased sensitivity to glucocorticoid fast feedback in chronically stressed rats. Neuroendocrinology 1990, 51, 536–542. [Google Scholar] [CrossRef]
- Kant, G.J.; Bunnell, B.N.; Mougey, E.H.; Pennington, L.L.; Meyerhoff, J.L. Effects of repeated stress on pituitary cyclic AMP, and plasma prolactin, corticosterone and growth hormone in male rats. Pharmacol. Biochem. Behav. 1983, 18, 967–971. [Google Scholar] [CrossRef]
- Boer, S.R.; Koopmans, S.J.; Slangen, J.L.; Gugten, J. Plasma catecholamine, corticosterone and glucose responses to repeated stress in rats: Effect of interstress or interval length. Physiol. Behav. 1990, 47, 117–124. [Google Scholar]
- Pitman, D.L.; Ottenweller, J.E.; Natelson, B.H. Effect of stressor intensity on habituation and sensitization of glucocorticoid responses in rats. Behav. Neurosci. 1990, 104, 28–36. [Google Scholar] [CrossRef]
- Salak-Johnson, J.L.; Mcglone, J.J. Making sense of apparently conflicting data: Stress and immunity in swine and cattle. J. Anim. Sci. 2007, 85, E81–E88. [Google Scholar] [PubMed]
- Vijayan, M.; Reddy, P.H. Stroke, vascular dementia, and Alzheimer’s disease: Molecular links. J. Alzheimers Dis. 2016, 54, 427–443. [Google Scholar] [PubMed]
- Calabrese, V.; Giord, J.; Signorile, A.; Laura, O.M.; Castorina, S.; De, P.C.; Eckert, G.; Calabrese, E.J. Major pathogenic mechanisms in vascular dementia: Roles of cellular stress response and hormesis in neuroprotection. J. Neurosci Res. 2016, 94, 1588–1603. [Google Scholar]
- Jin, X.; Li, T.; Zhang, L.; Ma, J.; Yu, L.; Li, C.; Niu, L. Environmental enrichment improves spatial learning and memory in vascular dementia rats with activation of wnt/beta-catenin signal pathway. Med. Sci. Monit. 2017, 23, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Testa, G.; Staurenghi, E.; Zerbinati, C.; Gargiulo, S.; Iuliano, L.; Giaccone, G.; Fanto, F.; Poli, G.; Leonarduzzi, G.; Gamba, P. Changes in brain oxysterols at different stages of Alzheimer’s disease: Their involvement in neuroinflammation. Redox. Biol. 2016, 10, 24–33. [Google Scholar] [CrossRef]
- Sudduth, T.L.; Weekman, E.M.; Price, B.R.; Gooch, J.L.; Woolums, A.; Norris, C.M.; Wilcock, D.M. Time-course of glial changes in the hyperhomocysteinemia model of vascular cognitive impairment and dementia (VCID). Neuroscience 2017, 341, 42–51. [Google Scholar] [CrossRef]
- Dumont, F.J. Modulation of Th1 and Th2 responses for immunotherapy. Expert Opin. Ther. Pat. 2005, 12, 341–367. [Google Scholar] [CrossRef]
- Cong, P.; Liu, Y.; Liu, N. Cold exposure induced oxidative stress and apoptosis in the myocardium by inhibiting the Nrf2-Keap1 signaling pathway. BMC Cardiovasc. Disord. 2018, 18, 36. [Google Scholar] [CrossRef]
- Litman, G.W.; Rast, J.P.; Shamblott, M.J.; Haire, R.N.; Hulst, M.; Roess, W.; Litman, R.T.; Hinds Frey, K.R.; Zilch, A.; Amemiya, C.T. Phylogenetic diversification of immunoglobulin genes and the antibody repertoire. Mol. Biol. Evol. 1993, 10, 60–72. [Google Scholar] [PubMed]
- Xu, Y.Q.; Wang, Z.Q.; Qin, Z.; Yan, S.M.; Shi, B.L. Effects of chitosan addition on growth performance, diarrhoea, anti-oxidative function and serum immune parameters of weaned piglets. S. Afr. J. Anim. Sci. 2018, 48, 142–150. [Google Scholar] [CrossRef]
- Fang, Y.Z.; Yang, S.; Wu, G. Free radicals, antioxidants, and nutrition. Nutrition 2002, 18, 872–879. [Google Scholar] [CrossRef]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [PubMed]
- Rao, G.N.; Berk, B.C. Active oxygen species stimulate vascular smooth muscle cell growth and protooncogene expression. Circ. Res. 1992, 70, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, H.; Tian, J.; Wang, J.; Khan, M.A.; Wang, Y.; Zhang, L.; Wang, T. Effects of dietary sodium selenite and selenium yeast on antioxidant enzyme activities and oxidative stability of chicken breast meat. J. Agric. Food Chem. 2012, 60, 7111–7120. [Google Scholar] [CrossRef] [PubMed]
- Gumuslu, S.; Sarikcioglu, B.; Sahin, E.; Yargiçoglu, P.; Agar, A. Influences of different stress models on the antioxidant status and lipid peroxidation in rat erythrocytes. Free Radic. Res. 2002, 36, 1277–1282. [Google Scholar] [CrossRef]
- Sahin, E.; Gumuslu, S. Cold-stress-induced modulation of antioxidant defence: Role of stressed conditions in tissue injury followed by protein oxidation and lipid peroxidation. Int. J. Biometeorol. 2004, 48, 165–171. [Google Scholar] [CrossRef]

| Items | No Windproof f. | Windproof f. | SEM | p | p | ||||
|---|---|---|---|---|---|---|---|---|---|
| Cold Water | Lukewarm Water | Cold Water | Lukewarm Water | WF | WT | WF × WT | |||
| IBW kg | 127.6 | 128.8 | 128.0 | 127.9 | 1.80 | 0.996 | - | - | - |
| ADG kg/d | |||||||||
| 1–21 d | 0.241 b | 0.244 b | 0.299 a | 0.309 a | 1.89 | <0.001 | <0.001 | 0.132 | 0.359 |
| 22–42 d | 0.254 b | 0.266 b | 0.348 a | 0.333 a | 2.61 | <0.001 | <0.001 | 0.821 | 0.021 |
| 1–42 d | 0.248 b | 0.255 b | 0.323 a | 0.321 a | 1.90 | <0.001 | <0.001 | 0.538 | 0.224 |
| ADFI g/d | |||||||||
| 1–21 d | 4029 ab | 4270 a | 3685 c | 3955 bc | 50.0 | 0.005 | 0.004 | 0.019 | 0.888 |
| 22–42 d | 4230 ab | 4453 a | 4061 b | 4042 b | 46.9 | 0.020 | 0.006 | 0.290 | 0.214 |
| 1–42 d | 4129 ab | 4361 a | 3873 b | 3998 b | 47.1 | 0.010 | 0.004 | 0.073 | 0.577 |
| F:G | |||||||||
| 1–21 d | 17.32 a | 16.90 a | 12.58 b | 12.57 b | 0.20 | <0.001 | <0.001 | 0.597 | 0.609 |
| 22–42 d | 17.43 a | 16.01 b | 11.69 c | 12.16 c | 0.17 | <0.001 | <0.001 | 0.177 | 0.011 |
| 1–42 d | 17.38 a | 16.42 a | 12.10 b | 12.36 b | 0.17 | <0.001 | <0.001 | 0.328 | 0.094 |
| Water intake L/d | |||||||||
| 1–21 d | 3.68 c | 4.93 a | 4.21 bc | 4.61 ab | 0.11 | 0.005 | 0.645 | 0.002 | 0.073 |
| 22–42 d | 3.79 b | 4.82 a | 4.06 b | 4.29 ab | 0.11 | 0.019 | 0.552 | 0.008 | 0.073 |
| 1–42 d | 3.73 c | 4.87 a | 4.12 bc | 4.44 ab | 0.10 | 0.006 | 0.923 | 0.002 | 0.059 |
| Items | No Windproof f. | Windproof f. | SEM | p | p | ||||
|---|---|---|---|---|---|---|---|---|---|
| Cold Water | Lukewarm Water | Cold Water | Lukewarm Water | WF | WT | WF × WT | |||
| 21 d | |||||||||
| DM | 72.20 b | 72.17 b | 73.88 a | 74.01 a | 0.18 | 0.004 | 0.005 | 0.064 | 0.053 |
| CF | 60.52 b | 62.26 ab | 64.38 ab | 66.91 a | 0.65 | 0.013 | 0.007 | 0.840 | 0.276 |
| CP | 63.99 b | 65.74 ab | 67.23 a | 67.44 a | 0.32 | 0.003 | 0.140 | 0.237 | 0.001 |
| Ash | 29.56 b | 27.02 b | 35.97 a | 36.53 a | 0.50 | <0.001 | 0.164 | 0.079 | 0.384 |
| Ca | 57.08 bc | 54.15 c | 59.05 ab | 61.10 a | 0.61 | 0.005 | 0.361 | 0.177 | 0.957 |
| P | 28.35 b | 26.88 b | 32.51 a | 32.94 a | 0.40 | <0.001 | <0.001 | 0.337 | 0.140 |
| NDF | 54.97 | 53.28 | 52.63 | 51.24 | 0.56 | 0.345 | 0.002 | 0.719 | 0.054 |
| ADF | 35.31 | 34.61 | 34.24 | 33.45 | 0.38 | 0.455 | <0.001 | 0.532 | 0.255 |
| 42 d | |||||||||
| DM | 66.19 b | 66.55 b | 69.26 a | 70.14 a | 0.20 | <0.001 | <0.001 | 0.132 | 0.523 |
| CF | 65.02 b | 64.56 b | 71.87 a | 71.94 a | 0.94 | 0.011 | 0.001 | 0.920 | 0.891 |
| CP | 61.72 | 61.84 | 61.62 | 61.04 | 0.31 | 0.808 | 0.479 | 0.719 | 0.578 |
| Ash | 29.82 b | 31.19 b | 36.54 a | 38.92 a | 0.52 | <0.001 | 0.700 | 0.388 | 0.841 |
| Ca | 49.45 | 52.23 | 52.96 | 52.24 | 0.66 | 0.277 | 0.901 | 0.097 | 0.994 |
| P | 27.43 b | 27.41 b | 31.81 a | 31.94 a | 0.32 | <0.001 | <0.001 | 0.084 | 0.633 |
| NDF | 49.01 | 48.20 | 47.53 | 47.28 | 0.48 | 0.807 | 0.198 | 0.442 | 0.200 |
| ADF | 34.16 | 34.03 | 32.19 | 32.04 | 0.39 | 0.406 | <0.001 | 0.928 | 0.909 |
| Items | Time | No Windproof f. | Windproof f. | SEM | p | p | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cold Water | Lukewarm Water | Cold Water | Lukewarm Water | WF | WT | WF × WT | ||||
| 21 d | ||||||||||
| Head °C | 7:00 | 8.96 b | 9.73 ab | 10.30 ab | 10.78 a | 0.54 | 0.091 | 0.039 | 0.261 | 0.796 |
| 14:00 | 9.07 b | 8.93 b | 10.30 ab | 11.72 a | 0.48 | 0.020 | 0.001 | 0.199 | 0.125 | |
| Ear °C | 7:00 | 7.60 b | 8.92 a | 8.80 ab | 8.65 ab | 0.40 | 0.078 | 0.163 | 0.260 | 0.084 |
| 14:00 | 9.58 | 10.12 | 10.92 | 11.07 | 0.47 | 0.125 | 0.026 | 0.691 | 0.480 | |
| Abdomen °C | 7:00 | 12.87 | 12.70 | 11.83 | 12.70 | 0.34 | 0.171 | 0.149 | 0.322 | 0.149 |
| 14:00 | 10.97 b | 11.60 b | 13.03 a | 13.52 a | 0.47 | 0.003 | 0.004 | 0.252 | 0.876 | |
| Leg °C | 7:00 | 11.92 b | 12.68 b | 14.52 a | 15.58 a | 0.46 | <0.001 | <0.001 | 0.542 | 0.032 |
| 14:00 | 13.75 | 13.10 | 13.55 | 14.28 | 0.61 | 0.580 | 0.426 | 0.946 | 0.267 | |
| Rectal temperature °C | 7:00 | 36.43 b | 36.45 b | 36.85 a | 36.92 a | 0.13 | 0.021 | 0.003 | 0.847 | 0.747 |
| 14:00 | 37.35 b | 37.38 b | 37.45 ab | 37.75 a | 0.12 | 0.093 | 0.062 | 0.174 | 0.272 | |
| Respiratory rate | 7:00 | 10.85 | 11.50 | 11.45 | 11.38 | 0.49 | 0.772 | 0.560 | 0.628 | 0.475 |
| 14:00 | 12.46 | 12.00 | 11.60 | 13.28 | 0.54 | 0.189 | 0.286 | 0.681 | 0.070 | |
| 42 d | ||||||||||
| Head °C | 7:00 | 10.37 b | 10.83 b | 11.23 b | 12.47 a | 0.38 | 0.005 | 0.003 | 0.035 | 0.321 |
| 14:00 | 9.63 b | 9.68 b | 10.10 ab | 10.87 a | 0.31 | 0.038 | 0.201 | 0.260 | 0.015 | |
| Ear °C | 7:00 | 9.23 | 9.50 | 9.65 | 10.37 | 0.52 | 0.444 | 0.216 | 0.339 | 0.659 |
| 14:00 | 9.88 | 10.50 | 10.68 | 10.85 | 0.41 | 0.394 | 0.179 | 0.354 | 0.592 | |
| Abdomen °C | 7:00 | 10.17 | 11.03 | 10.85 | 11.20 | 0.40 | 0.301 | 0.141 | 0.298 | 0.523 |
| 14:00 | 11.70 b | 11.67 b | 12.00 b | 13.35 a | 0.35 | 0.008 | 0.010 | 0.059 | 0.071 | |
| Leg °C | 7:00 | 10.43 b | 11.15 ab | 11.92 a | 12.15 a | 0.46 | 0.059 | 0.013 | 0.310 | 0.602 |
| 14:00 | 12.20 b | 13.13 ab | 13.33 ab | 14.20 a | 0.51 | 0.077 | 0.042 | 0.090 | 0.948 | |
| Rectal temperature °C | 7:00 | 36.58 | 36.77 | 36.85 | 36.87 | 0.12 | 0.361 | 0.149 | 0.423 | 0.503 |
| 14:00 | 37.60 | 37.78 | 37.58 | 37.72 | 0.07 | 0.189 | 0.043 | 0.576 | 0.737 | |
| Respiratory rate | 7:00 | 10.77 | 11.33 | 11.17 | 11.22 | 0.28 | 0.533 | 0.290 | 0.623 | 0.373 |
| 14:00 | 10.98 b | 11.17 b | 11.43 ab | 12.48 a | 0.39 | 0.055 | 0.127 | 0.277 | 0.034 | |
| Items | Time | No Windproof f. | Windproof f. | SEM | p | p | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cold Water | Lukewarm Water | Cold Water | Lukewarm Water | WF | WT | WF × WT | ||||
| 21 d | ||||||||||
| TP g/L | 7:00 | 55.33 b | 58.17 b | 68.33 a | 71.75 a | 1.65 | 0.041 | 0.037 | 0.444 | 0.568 |
| 14:00 | 55.33 | 59.83 | 61.33 | 62.67 | 2.22 | 0.155 | 0.053 | 0.432 | 0.177 | |
| ALB g/L | 7:00 | 19.67 | 20.83 | 22.50 | 24.00 | 0.72 | 0.193 | 0.366 | 0.051 | 0.909 |
| 14:00 | 19.83 | 20.33 | 21.17 | 23.00 | 1.13 | 0.830 | 0.414 | 0.632 | 0.784 | |
| TG mmol/L | 7:00 | 0.15 b | 0.17 b | 0.21 a | 0.22 a | 0.01 | 0.046 | 0.033 | 0.647 | 0.844 |
| 14:00 | 0.27 | 0.28 | 0.32 | 0.35 | 0.02 | 0.336 | 0.214 | 0.143 | 0.697 | |
| CHO mmol/L | 7:00 | 1.52 | 1.57 | 1.58 | 1.58 | 0.05 | 0.973 | 0.714 | 0.789 | 0.814 |
| 14:00 | 1.51 | 1.61 | 1.77 | 2.06 | 0.10 | 0.036 | 0.007 | 0.787 | 0.159 | |
| BUN mmol/L | 7:00 | 5.44 b | 5.59 b | 6.21 a | 6.65 a | 0.22 | 0.043 | 0.012 | 0.855 | 0.944 |
| 14:00 | 5.47 | 5.52 | 6.00 | 6.11 | 0.23 | 0.332 | 0.078 | 0.771 | 0.557 | |
| GLU mmol/L | 7:00 | 7.05 a | 6.44 ab | 5.53 b | 5.23 b | 0.14 | 0.042 | 0.027 | 0.599 | 0.795 |
| 14:00 | 4.49 | 4.72 | 4.90 | 4.98 | 0.32 | 0.278 | 0.062 | 0.824 | 0.518 | |
| LDL-C mmol/L | 7:00 | 0.16 | 0.11 | 0.14 | 0.11 | 0.01 | 0.282 | 0.260 | 0.443 | 0.159 |
| 14:00 | 0.12 | 0.11 | 0.15 | 0.17 | 0.01 | 0.143 | 0.032 | 0.795 | 0.424 | |
| HDL-C mmol/L | 7:00 | 1.18 b | 1.19 b | 1.43 a | 1.49 a | 0.05 | 0.043 | 0.025 | 0.477 | 0.115 |
| 14:00 | 1.53 | 1.52 | 1.62 | 1.66 | 0.04 | 0.479 | 0.126 | 0.957 | 0.889 | |
| 42 d | ||||||||||
| TP g/L | 7:00 | 69.00 a | 67.50 a | 57.00 b | 56.67 b | 1.34 | 0.049 | 0.038 | 0.878 | 0.775 |
| 14:00 | 59.33 | 58.50 | 56.50 | 56.00 | 1.76 | 0.830 | 0.852 | 0.458 | 0.609 | |
| ALB g/L | 7:00 | 20.33 | 20.50 | 20.83 | 21.17 | 0.46 | 0.920 | 0.531 | 0.928 | 0.788 |
| 14:00 | 20.50 | 20.67 | 20.17 | 21.17 | 0.66 | 0.959 | 0.950 | 0.661 | 0.754 | |
| TG mmol/L | 7:00 | 0.19b | 0.20 b | 0.29 a | 0.34 a | 0.01 | 0.038 | 0.034 | 0.813 | 0.348 |
| 14:00 | 0.34 | 0.35 | 0.37 | 0.45 | 0.03 | 0.074 | 0.060 | 0.975 | 0.057 | |
| CHO mmol/L | 7:00 | 1.40 b | 1.43 b | 1.60 a | 1.65 a | 0.06 | 0.043 | 0.037 | 0.272 | 0.882 |
| 14:00 | 1.63 | 1.65 | 1.76 | 1.80 | 0.08 | 0.297 | 0.081 | 0.566 | 0.362 | |
| BUN mmol/L | 7:00 | 5.52 a | 5.29 a | 4.71 b | 4.15 b | 0.14 | 0.045 | 0.028 | 0.319 | 0.355 |
| 14:00 | 4.95 | 4.93 | 4.83 | 4.58 | 0.24 | 0.785 | 0.427 | 0.538 | 0.899 | |
| GLU mmol/L | 7:00 | 6.02 a | 5.88 a | 5.11 b | 5.02b | 0.10 | 0.047 | 0.022 | 0.777 | 0.671 |
| 14:00 | 4.61 | 4.56 | 4.46 | 4.39 | 0.23 | 0.689 | 0.942 | 0.327 | 0.500 | |
| LDL-C mmol/L | 7:00 | 0.07 | 0.06 | 0.11 | 0.11 | 0.01 | 0.174 | 0.031 | 0.764 | 0.819 |
| 14:00 | 0.21 | 0.25 | 0.19 | 0.23 | 0.02 | 0.786 | 0.686 | 0.355 | 0.958 | |
| HDL-C mmol/L | 7:00 | 1.23 | 1.27 | 1.38 | 1.35 | 0.05 | 0.099 | 0.059 | 0.836 | 0.776 |
| 14:00 | 1.53 | 1.53 | 1.67 | 1.71 | 0.06 | 0.092 | 0.068 | 0.194 | 0.179 | |
| Items | Time | No Windproof f. | Windproof f. | SEM | p | p | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cold Water | Lukewarm Water | Cold Water | Lukewarm Water | WF | WT | WF × WT | ||||
| 21 d | ||||||||||
| ACTH pg/mL | 7:00 | 64.22 a | 59.28 a | 53.97 b | 51.10 b | 1.81 | 0.045 | 0.034 | 0.693 | 0.319 |
| 14:00 | 47.99 | 48.31 | 44.76 | 43.17 | 1.75 | 0.178 | 0.243 | 0.082 | 0.332 | |
| COR ng/mL | 7:00 | 26.17 a | 25.23 a | 22.15 b | 21.99 b | 0.60 | 0.039 | 0.014 | 0.302 | 0.496 |
| 14:00 | 20.43 | 19.77 | 19.99 | 19.15 | 0.45 | 0.727 | 0.331 | 0.798 | 0.678 | |
| ADPN μg/mL | 7:00 | 9.26 | 9.66 | 9.10 | 10.08 | 0.14 | 0.141 | 0.660 | 0.034 | 0.345 |
| 14:00 | 8.37 | 8.65 | 8.05 | 8.18 | 0.19 | 0.077 | 0.013 | 0.114 | 0.337 | |
| Insulin mIU/L | 7:00 | 8.50 | 8.61 | 8.70 | 8.86 | 0.18 | 0.935 | 0.581 | 0.744 | 0.943 |
| 14:00 | 8.17 | 8.35 | 8.31 | 8.76 | 0.18 | 0.709 | 0.505 | 0.748 | 0.452 | |
| Leptin ng/mL | 7:00 | 2.67 | 2.65 | 2.86 | 2.82 | 0.07 | 0.651 | 0.222 | 0.851 | 0.931 |
| 14:00 | 2.88 | 2.91 | 3.06 | 3.35 | 0.09 | 0.314 | 0.122 | 0.416 | 0.514 | |
| T3 nmol/L | 7:00 | 2.25 a | 2.10 a | 1.83b | 1.75 b | 0.04 | 0.036 | 0.022 | 0.863 | 0.492 |
| 14:00 | 1.82 | 1.81 | 1.75 | 1.72 | 0.03 | 0.113 | 0.018 | 0.792 | 0.884 | |
| T4 nmol/L | 7:00 | 43.51 a | 42.87 a | 38.95 b | 38.53 b | 0.93 | 0.043 | 0.028 | 0.832 | 0.779 |
| 14:00 | 37.86 | 37.56 | 36.43 | 36.55 | 0.53 | 0.310 | 0.066 | 0.814 | 0.897 | |
| 42 d | ||||||||||
| ACTH pg/mL | 7:00 | 50.89 | 50.16 | 48.80 | 47.59 | 0.95 | 0.480 | 0.703 | 0.247 | 0.325 |
| 14:00 | 45.01 | 44.53 | 42.16 | 41.47 | 1.27 | 0.507 | 0.640 | 0.307 | 0.311 | |
| COR ng/mL | 7:00 | 20.77 | 20.58 | 19.51 | 19.59 | 0.28 | 0.328 | 0.935 | 0.077 | 0.825 |
| 14:00 | 17.74 | 17.67 | 17.35 | 17.06 | 0.41 | 0.739 | 0.460 | 0.994 | 0.413 | |
| ADPN μg/mL | 7:00 | 8.53 | 7.91 | 8.85 | 8.01 | 0.18 | 0.254 | 0.584 | 0.067 | 0.764 |
| 14:00 | 7.23 | 6.69 | 7.40 | 7.13 | 0.21 | 0.144 | 0.069 | 0.817 | 0.146 | |
| Insulin mIU/L | 7:00 | 8.12 | 8.08 | 8.28 | 8.40 | 0.15 | 0.161 | 0.101 | 0.834 | 0.664 |
| 14:00 | 8.08 | 8.02 | 8.12 | 8.15 | 0.19 | 0.617 | 0.829 | 0.329 | 0.389 | |
| Leptin ng/mL | 7:00 | 2.10 | 1.90 | 2.08 | 1.93 | 0.06 | 0.524 | 0.996 | 0.150 | 0.820 |
| 14:00 | 2.24 | 2.04 | 2.19 | 2.05 | 0.05 | 0.521 | 0.838 | 0.154 | 0.798 | |
| T3 nmol/L | 7:00 | 1.59 | 1.49 | 1.55 | 1.51 | 0.03 | 0.766 | 0.918 | 0.363 | 0.637 |
| 14:00 | 1.39 | 1.39 | 1.35 | 1.42 | 0.04 | 0.906 | 0.937 | 0.615 | 0.599 | |
| T4 nmol/L | 7:00 | 40.77 a | 40.57 a | 36.58 b | 35.89 b | 0.88 | 0.046 | 0.026 | 0.678 | 0.599 |
| 14:00 | 35.12 | 35.48 | 33.09 | 32.84 | 0.69 | 0.148 | 0.910 | 0.085 | 0.136 | |
| Items | No Windproof f. | Windproof f. | SEM | p | p | ||||
|---|---|---|---|---|---|---|---|---|---|
| Cold Water | Lukewarm Water | Cold Water | Lukewarm Water | WF | WT | WF × WT | |||
| 21 d | |||||||||
| IL-1β pg/mL | 128.6 a | 94.54 b | 80.46 c | 73.61 c | 2.38 | <0.001 | <0.001 | 0.001 | 0.012 |
| IL-4 pg/mL | 6.83 b | 6.96 b | 9.28 a | 10.69 a | 0.29 | <0.001 | <0.001 | 0.281 | 0.196 |
| TNF-α pg/mL | 26.32 a | 26.37 a | 18.61 b | 19.98 b | 0.74 | 0.001 | <0.001 | 0.637 | 0.657 |
| IgA μg/mL | 35.54 b | 35.48 b | 51.30 a | 57.12 a | 1.83 | 0.001 | <0.001 | 0.442 | 0.433 |
| IgG μg/mL | 43.64 b | 49.31 b | 69.76 a | 71.83 a | 1.73 | <0.001 | <0.001 | 0.278 | 0.610 |
| IgM μg/mL | 17.88 b | 18.94 b | 23.39 a | 25.37 a | 0.45 | <0.001 | <0.001 | 0.108 | 0.617 |
| 42 d | |||||||||
| IL-1β pg/mL | 124.73 a | 115.69 a | 78.44 b | 76.89 b | 4.93 | 0.004 | <0.001 | 0.004 | 0.006 |
| IL-4 pg/mL | 6.58 b | 7.65 b | 9.06 a | 10.47 a | 0.24 | <0.001 | <0.001 | 0.002 | 0.180 |
| TNF-α pg/mL | 29.09 a | 28.14 a | 19.97 b | 19.71 b | 0.88 | 0.010 | <0.001 | 0.152 | 0.008 |
| IgA μg/mL | 37.80 b | 44.27 b | 54.38 a | 61.52 a | 2.62 | 0.009 | 0.005 | 0.220 | 0.951 |
| IgG μg/mL | 50.06 b | 55.18 b | 73.85 a | 79.91 a | 2.08 | 0.002 | <0.001 | 0.206 | 0.914 |
| IgM μg/mL | 18.79 b | 20.94 b | 32.15 a | 32.37 a | 0.61 | <0.001 | <0.001 | 0.390 | 0.481 |
| Items | No Windproof f. | Windproof f. | SEM | p | p | ||||
|---|---|---|---|---|---|---|---|---|---|
| Cold Water | Lukewarm Water | Cold Water | Lukewarm Water | WF | WT | WF × WT | |||
| 21 d | |||||||||
| T-AOC mM | 0.38 | 0.38 | 0.38 | 0.42 | 0.01 | 0.599 | 0.386 | 0.591 | 0.372 |
| MDA nmol/mL | 1.52 a | 1.44 a | 0.94 b | 0.84 b | 0.03 | <0.001 | <0.001 | 0.161 | 0.333 |
| T-SOD U/mL | 89.19 b | 91.16 b | 106.4 a | 108.0 a | 2.76 | 0.045 | 0.006 | 0.977 | 0.746 |
| CAT U/mL | 1.38 | 1.43 | 1.58 | 1.62 | 0.11 | 0.842 | 0.987 | 0.388 | 0.831 |
| GPx U/mL | 410.9 b | 487.3 b | 572.7 a | 613.2 a | 25.8 | 0.031 | 0.019 | 0.102 | 0.363 |
| 42 d | |||||||||
| T-AOC mM | 0.39 | 0.37 | 0.40 | 0.38 | 0.01 | 0.754 | 0.749 | 0.312 | 0.979 |
| MDA nmol/mL | 1.52 a | 1.43 a | 1.00 b | 0.89 b | 0.09 | 0.009 | 0.005 | 0.288 | 0.243 |
| T-SOD U/mL | 88.26 b | 92.47 b | 118.0 a | 124.0 a | 3.08 | 0.010 | <0.001 | 0.416 | 0.386 |
| CAT U/mL | 1.41 b | 1.55 b | 1.92 a | 2.06 a | 0.01 | 0.023 | 0.009 | 0.310 | 0.295 |
| GPx U/mL | 501.6 | 521.8 | 557.3 | 611.4 | 20.9 | 0.296 | 0.098 | 0.386 | 0.491 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, R.; Shi, L.; Guo, W.; Xu, Y.; Jin, X.; Yan, S.; Shi, B. Effects of Housing and Management Systems on the Growth, Immunity, Antioxidation, and Related Physiological and Biochemical Indicators of Donkeys in Cold Weather. Animals 2022, 12, 2405. https://doi.org/10.3390/ani12182405
Gao R, Shi L, Guo W, Xu Y, Jin X, Yan S, Shi B. Effects of Housing and Management Systems on the Growth, Immunity, Antioxidation, and Related Physiological and Biochemical Indicators of Donkeys in Cold Weather. Animals. 2022; 12(18):2405. https://doi.org/10.3390/ani12182405
Chicago/Turabian StyleGao, Ruiheng, Lulu Shi, Wenliang Guo, Yuanqing Xu, Xiao Jin, Sumei Yan, and Binlin Shi. 2022. "Effects of Housing and Management Systems on the Growth, Immunity, Antioxidation, and Related Physiological and Biochemical Indicators of Donkeys in Cold Weather" Animals 12, no. 18: 2405. https://doi.org/10.3390/ani12182405
APA StyleGao, R., Shi, L., Guo, W., Xu, Y., Jin, X., Yan, S., & Shi, B. (2022). Effects of Housing and Management Systems on the Growth, Immunity, Antioxidation, and Related Physiological and Biochemical Indicators of Donkeys in Cold Weather. Animals, 12(18), 2405. https://doi.org/10.3390/ani12182405





