1. Introduction
Locoregional anaesthesia is employed in a multimodal analgesic protocol to manage pain, decrease the consumption of systemic analgesic drugs (i.e., opioids) and their potential adverse effects, and potentially decrease the hospital length of stay [
1]. The ultrasound (US)-guided
transversus abdominis plane (TAP) block is a locoregional technique used in humans and animals to provide perioperative analgesia by blocking the sensory component of the nerves supplying the abdominal wall and the underlying parietal peritoneum [
2,
3,
4,
5,
6,
7]. The TAP is the intermuscular fascial plane between the muscle (m.)
transversus abdominis, the innermost muscle of the abdominal wall, and m.
obliquus internus or m.
rectus abdominis and contains the ventral branches of the thoracolumbar (T9–L3) spinal nerves supplying the skin, the abdominal wall and the underlying peritoneum [
8]. An adequate volume of local anaesthetic injected into the TAP is expected to provide sensory blockade of the cranial and middle regions of the abdomen. Therefore, the TAP block provides somatic analgesia for surgical procedures involving the abdominal wall (e.g., hernia or laceration repairs, mastectomy, celiotomy [
3,
4,
9]). The veterinary literature is still limited in recommending the TAP block versus other locoregional anaesthetic techniques such as epidural,
quadratus lumborum block or intraperitoneal administration of local anaesthetics. However, the TAP block does not cause pelvic limb motor impairment or urinary retention that can be observed after epidural administration of local anaesthetics and opioids.
In veterinary medicine, an US-guided TAP block was firstly described in a Canadian lynx as a bilateral 1-point injection of local anaesthetic within the mm.
transversus abdominis and
obliquus internus abdominis performed at the midpoint between the last rib and the iliac crest using a lateral approach with a transversal needle orientation [
9]. The US-guided TAP block has now been studied in small animals. In dogs, a TAP injection using a single lateral injection failed to stain all target nerves because the injectate solution tended to stay near the injection point, even when large volumes of injectate were used [
10,
11]. To obtain a wide local anaesthetic spread within the TAP, and consequently an enhanced nerve blockade, multiple-point TAP injections have been used [
2,
8,
12]. Nevertheless, a cadaveric study in dogs revealed that thoracic nerve staining (T9–T12) was rare following a 2-point lateral TAP approach [
12]. To target these nerves, a subcostal approach to the TAP has been suggested, which consists of the injection of local anaesthetic in the plane between the mm.
transversus abdominis and
rectus abdominis [
13]. Recently, an investigation into canine cadavers showed that a combination of subcostal and lateral TAP approaches provides a more consistent staining of the thoracolumbar nerves (T9–L2). However, a gap staining was observed at the level of T13 and it is not known whether needle orientation (longitudinal versus transversal) affects the injectate spread following lateral TAP injection [
14]. These studies show that there is no current consensus on the volume of injection (as volumes from 0.3 to 1 mL have been used) or the most appropriate technique (lateral-transversal versus lateral-longitudinal versus subcostal combined with lateral) to perform the TAP block in small animals.
In cats, a recent cadaveric study concluded that a 3-point TAP injection with a combination of subcostal and lateral-transversal TAP approaches is necessary to stain the thoracolumbar spinal nerve branches that provide sensory abdominal innervation [
8]. A clinical study showed that a bilateral 1-point lateral-longitudinal TAP block with 1.5 mL of a lidocaine–bupivacaine solution per hemiabdomen (0.3–0.46 mL/kg/point) provided analgesia in cats undergoing ovariectomy [
4]. The literature is scarce in cats, and different techniques and volumes of administration are employed in this species. Therefore, anatomical and computed tomography studies may provide further evidence of nerve staining and possible complication of different TAP block techniques. These cadaveric studies may constitute the basis for in vivo future studies involving the pharmacokinetics of local anaesthetics and clinical trials investigating postoperative analgesia following the TAP block in cats.
The objective of this study was to compare the distribution pattern of 0.5 mL/kg of a solution administered by either a 1-point lateral-longitudinal TAP injection (TAP-L approach) or a 2-point subcostal and lateral–longitudinal TAP injection (TAP-SL approach) in cat cadavers using anatomical dissection and computed tomography.
The hypothesis was that TAP-SL approach would result in a wider spread of injectate and staining of thoracolumbar spinal nerve branches compared with the TAP-L approach.
2. Materials and Methods
This prospective, randomized, blinded, cadaveric study was approved by the Institutional animal care and use committee (Comité d’éthique de l’utilisation des animaux) of the Université de Montréal (no. 21-Rech-2111).
A total of 10 frozen cadavers of adult cats (eight domestic shorthair, three males and five females; two female domestic longhair) euthanized for reasons unrelated to this study were used. Exclusion criteria included body weight < 2 kg or >6 kg, body condition score > 7 or <3 on a scale from 1 to 9, and any evidence of skeletal and/or muscle injuries. Each cadaver was thawed at room temperature for 48 h prior to the experiment, and the thoracic and abdominal areas were clipped.
The study was divided in two phases: gross anatomy and sonoanatomy study (phase I), and injectate spread evaluation and nerve staining after TAP injections by computed tomography (CT) and dissection (phase II).
2.1. Phase I: Anatomical and Ultrasound Study
Two cadavers were used to describe the relevant gross anatomy of the abdominal wall and spinal nerves (T9–L3). Dissections were performed on one hemiabdomen. The skin, the m.
obliquus externus abdominis, the m.
rectus abdominis, and the m.
obliquus internus abdominis were dissected and reflected laterally to expose the nerves innervating the abdominal wall. Subsequently, dissections were performed to identify the thoracolumbar nerve branches up to their origin at the level of the intervertebral foramina. Afterwards, the contralateral hemiabdomen was sonographically evaluated and used to identify ultrasonographic landmarks for the TAP injection. Once the investigators (M.G. and S.M.) became familiar with the sonoanatomy, a 50 mm and 22-gauge spinal needle (BD Spinal Needle; BD Medical, Franklin Lakes, NJ, USA) was used to test an US-guided in-plane TAP subcostal and lateral approach (US settings described below). Two dye solutions, yellow and lime, were prepared using 0.5 mL of permanent tissue marking dye (Davison Marking System; Bradley Products Inc., Minneapolis, MN, USA) diluted in 50 mL of 2% lidocaine (Lurocaine, Vetoquinol, Lavaltrie, QC, Canada) as previously described [
15]. A volume of 0.1 mL of the yellow-coloured solution was injected under sonographic visualization (Edge II; Sonosite Inc., Bothell, WA, USA) between the mm.
rectus abdominis and
transversus abdominis, and between the mm.
transversus abdominis and
obliquus internus abdominis, whereas the same volume of the lime-coloured solution was injected between the mm.
obliquus internus abdominis and
obliquus externus abdominis to identify the anatomical landmarks. Finally, the hemiabdomen was dissected to confirm injectate spread.
2.2. Phase II: Injectate Spread Evaluation after Ultrasound-Guided TAP Injections
Eight cat cadavers were used to describe the US-guided injection of a bupivacaine–iopamidol–dye solution into the TAP by two approaches and evaluate the injectate spread and nerve staining by CT and dissection.
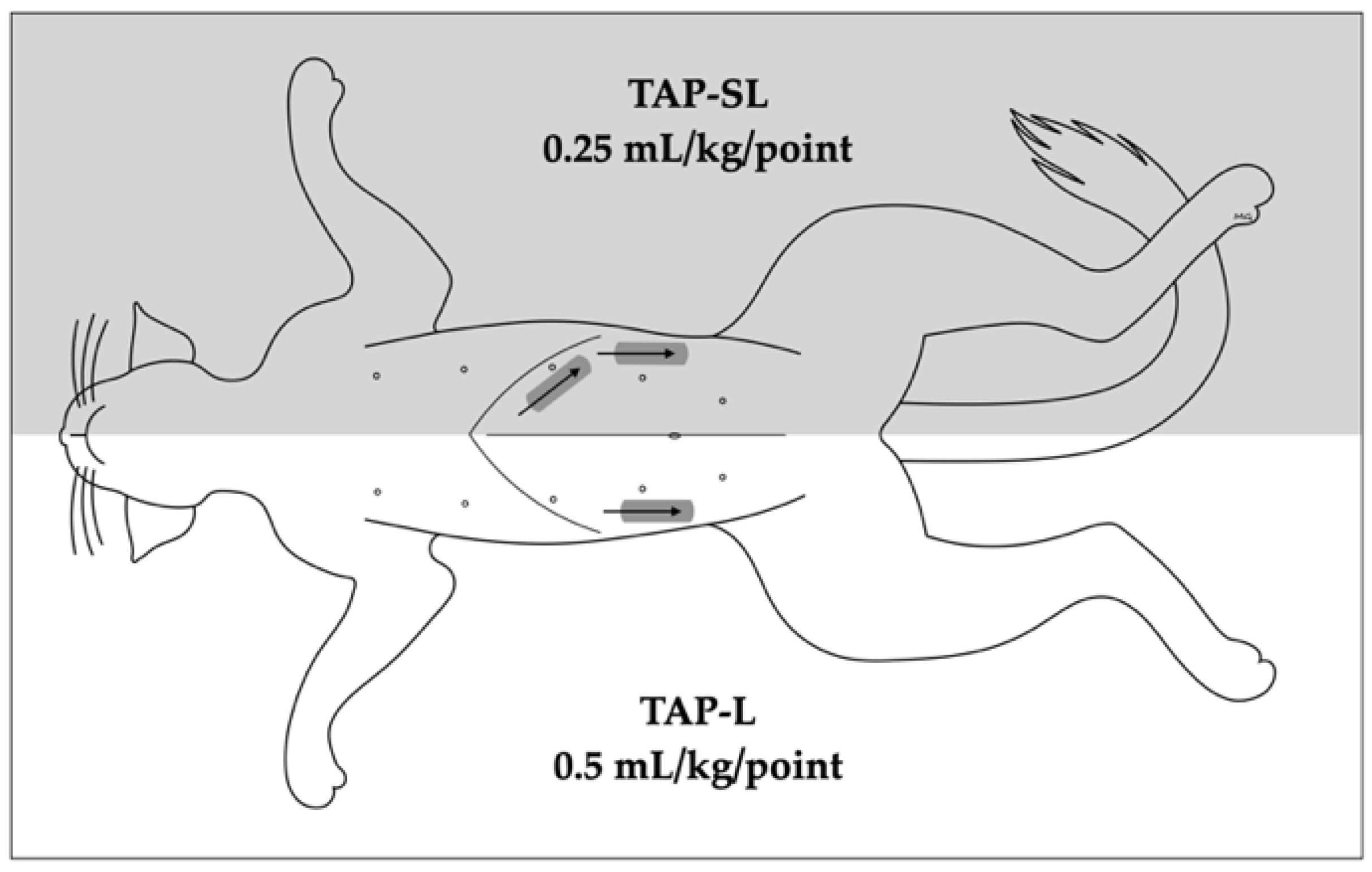
In each cat, a 1-point TAP injection was performed on one hemiabdomen (lateral approach, TAP-L), whereas a 2-point TAP injection was performed on the contralateral hemiabdomen (subcostal and lateral approach, TAP-SL;
Figure 1). The side allocation and the TAP block approach were randomly selected by one of the investigators (M.G.) using a randomization order by Microsoft Excel for Mac Version 16.13 (Microsoft Corporation, Redmond, WA, USA).
The TAP-L was performed injecting 0.5 mL/kg of solution, whereas the TAP-SL was performed using the same volume divided in two aliquots of 0.25 mL/kg per injection point (
Figure 1). The injectate solution was prepared diluting 0.2 mL of permanent tissue dye (Davison Marking System; Bradley Products Inc., Minneapolis, MN, USA) and 2 mL of iopamidol (Isovue 300, Bracco Imaging Canada, Montréal, QC, Canada) with 20 mL of bupivacaine hydrochloride 0.25% (Bupivacaine Injection BP 0.25%; SteriMax Inc., Oakville, ON, Canada).
2.2.1. Ultrasound-Guided TAP Injection
Sonographic imaging was performed using a 38 mm, 13–6 MHz, linear transducer connected to a portable US machine (Edge II; Sonosite Inc., Bothell, WA, USA). The US setting was adjusted at the minimum depth (1.9 mm) while the gain was adjusted to optimize the image. A 22-gauge, 50 mm Quincke spinal needle (BD spinal needle; Becton Dickinson & Co., Franklin Lakes, NJ, USA) connected by a T-port (Med-RX extension set with t-connector; CHS Ltd., Oakville, ON, Canada) to a prefilled 5 mL syringe, was used to perform the TAP injections. All the injections were performed with the cat in dorsal recumbency by an anaesthesiologist familiar with US-guided TAP blocks (M.G.).
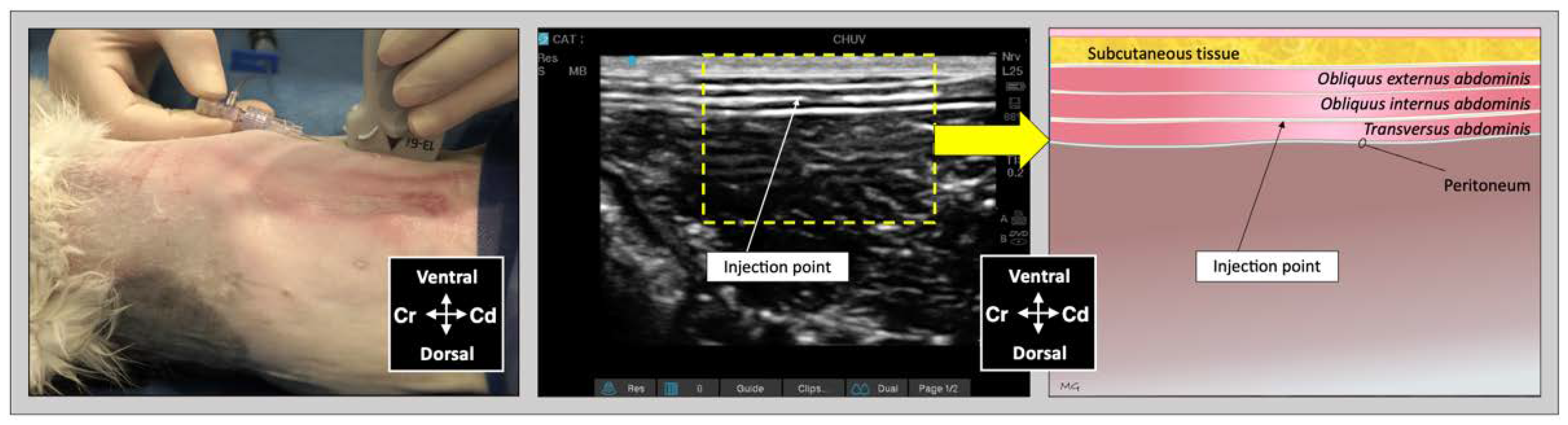
For the lateral TAP injection, the transducer was positioned parallel to the long axis of the cat (longitudinal plane), at 3–4 cm lateral to the abdominal midline and about 1 cm lateral to the mammary line, immediately caudal to the last rib (
Figure 2). The transducer was slide laterally until the belly of the m.
obliquus internus abdominis could be identified between the mm.
obliquus externus and
transversus abdominis. The needle was introduced in-plane in a ventrocranial-to-dorsocaudal orientation and continually visualized until its tip was advanced into the TAP between the m.
obliquus internus abdominis and the m.
transversus abdominis (
Figure 2). Then, the assigned injectate volume was administered (0.5 mL/kg in TAP-L versus 0.25 mL/kg in TAP-SL).
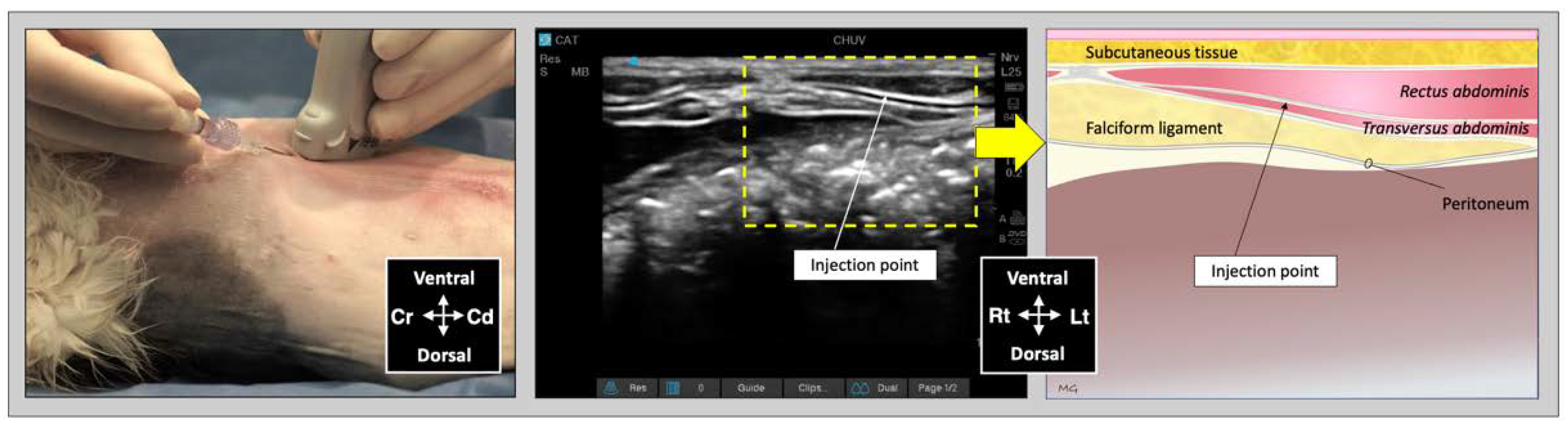
For the subcostal TAP injection, the transducer was initially positioned perpendicular to the long axis of the cat, immediately caudal to the xiphoid process, over the ventral midline. After identification of the
linea alba, the transducer was slid laterally and parallel to the costal arch and oblique to the midline (
Figure 3). The needle was introduced in-plane in a ventromedial-to-dorsolateral orientation and continuously visualized while advanced towards the TAP until its tip was inserted into the fascial plane between the mm.
rectus abdominis and
transversus abdominis. Then, an injectate volume of 0.25 mL/kg was administered by TAP-SL.
To improve the sonographic visualization of the needle tip within the fascial planes, the US image was zoomed in proximity of the injection site. Correct positioning of the needle tip was confirmed by visualizing the hydrodissection of the TAP following injection of 0.1 mL injectate solution, prior to the assigned volume. If hydrodissection of the target plane was not observed, the needle was redirected, and the test volume repeated until correct positioning was achieved. The quality of needle visualization was rated as good when both shaft and tip of the needle could be visualized; poor when only the needle tip was clearly visualized; or absent, when the needle tip could not be visualized. US images of each injection were recorded. Complications of the TAP-L and TAP-SL techniques, including needle advancement within the peritoneum or abdominal cavity, or spread of injectate to non-target fascial planes, were assessed and noted.
2.2.2. Computed Tomography Study
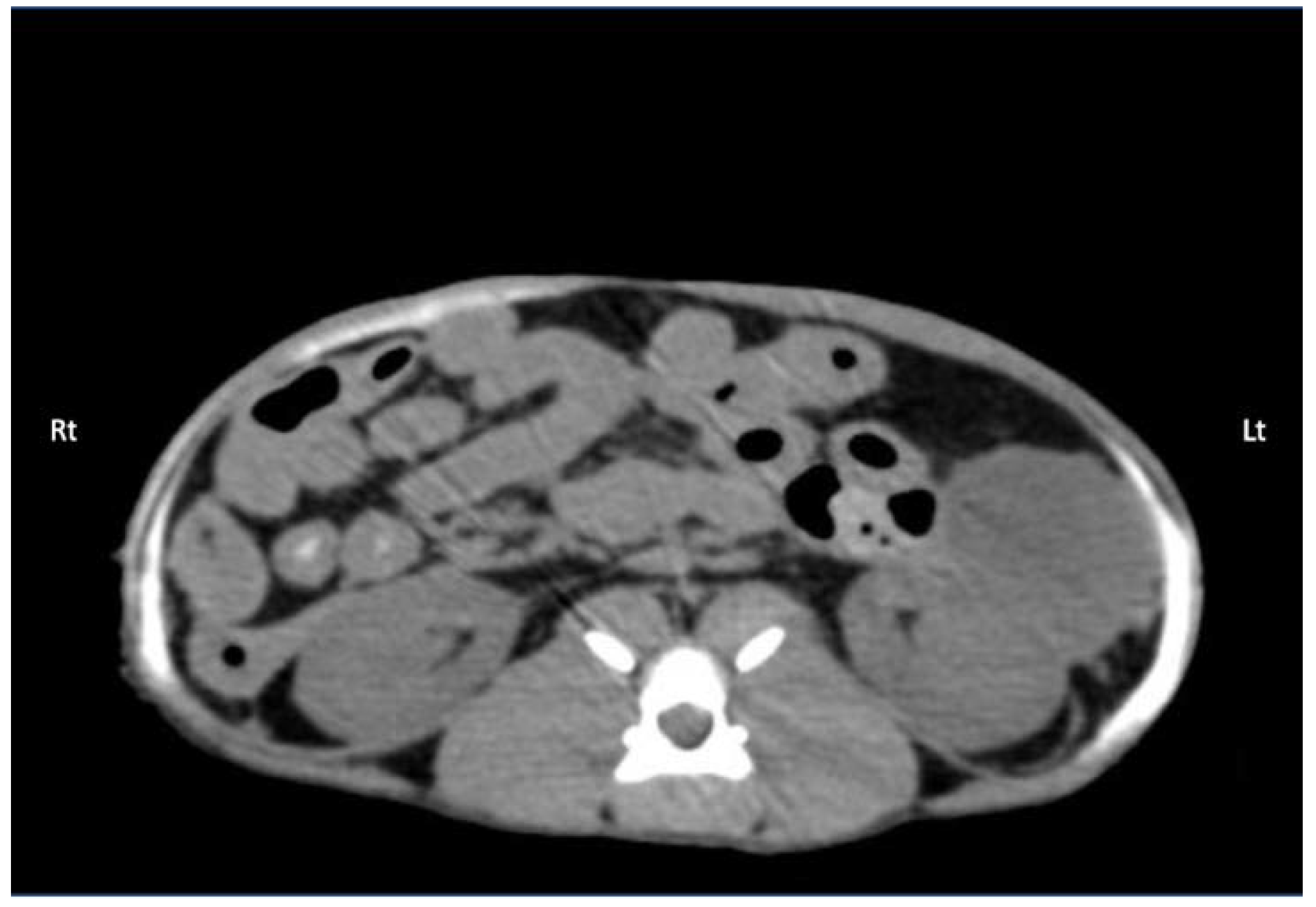
Thirty minutes after the last TAP injection, each cadaver underwent a full body CT examination in dorsal recumbency to observe contrast distribution. Scans were obtained using a 16-slice multidetector CT scanner (Hi-speed ZXi; General Electric, Milwaukee, WI, USA). The CT images were obtained in a helical mode at 100 kVp, 300 mA, slice thickness 0.625 mm, and reconstructed using low-pass and high-pass filter algorithms. A board-certified radiologist (C.F.) who was blinded to the treatments provided image interpretation using the Horos™ software (Horosproject.org, Nimble Co LLC d/b/a Purview in Annapolis, MD, USA). The following CT features were recorded: total length (mm) of the contrast spread within the TAP in all directions (cranial-to-caudal, dorsal-to-ventral, and medial-to-lateral), and any intra-abdominal contrast migration (yes/no). The predominant distribution patterns throughout the abdominal musculature for TAP-L and TAP-SL approaches were also described.
2.2.3. Anatomical Dissection
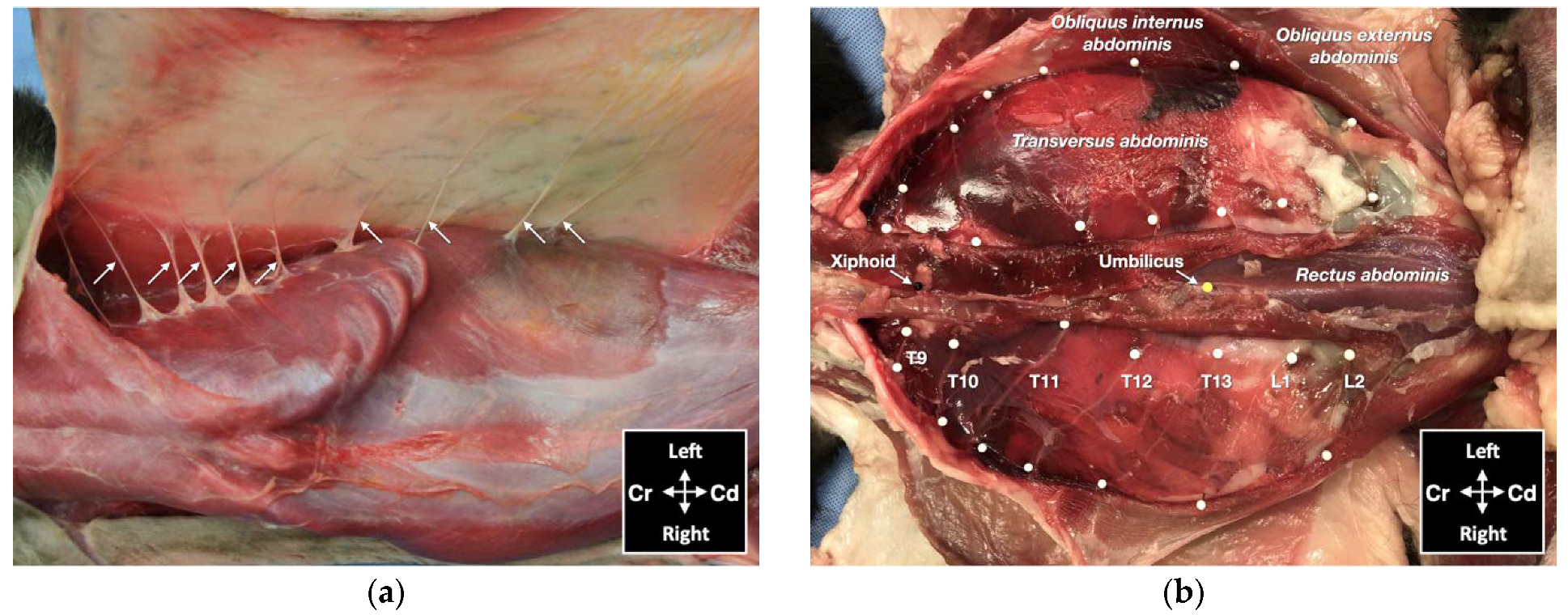
About 30 min after the end of CT examination, the cadavers were dissected to observe dye distribution and thoracolumbar spinal nerve ventral branch staining within the TAP. Two researchers (S.M. and M.G.) performed the dissections and the evaluation of the dye spread. Each cadaver was initially positioned in dorsal recumbency, and the skin was incised along the ventral midline from the mid-thorax to the pubis. The rectus sheath was dissected from the linea alba. The aponeurosis of the m. obliquus externus abdominis was identified, dissected, and the belly of the muscle was reflected laterally to expose the aponeurosis of the m. obliquus internus abdominis. The m. rectus abdominis was detached from its lateral margin and dissected medially to expose the m. transversus abdominis and its aponeurosis. At this point, the cadaver was placed in lateral recumbency with the hemiabdomen to be dissected uppermost. The aponeurosis of the m. obliquus internus was dissected and reflected dorsally to expose the ventromedial branches of the thoracolumbar spinal nerves and the m. transversus abdominis.
The ventromedial branches of the spinal nerves T10–L2 were considered the target nerves. Successful staining was defined as circumferential nerve staining with a length ≥ 1 cm [
16]. Location and staining of the ventromedial branches of T9 and L3 nerve branches were also noted. The cat was then positioned in the opposite lateral recumbency, and the dissection was repeated in the contralateral hemiabdomen.
After nerve staining evaluation, the cat was repositioned in dorsal recumbency, the abdominal cavity was exposed by incision of the linea alba, and the abdominal organs were carefully inspected to assess the presence of the bupivacaine–iopamidol–dye solution. Other complications such as presence of dye intramuscularly or in non-target fascial plane or in the abdominal cavity were recorded.
2.3. Statistical Analysis
The number of cats enrolled in the study as well as the total volume of 1 mL/kg of injectate solution per cat, corresponding to 0.5 mL/kg of solution per hemiabdomen, were based on similar cadaveric studies in animals [
8,
14,
15]. Statistical analyses were performed using SPSS Version 28.0 (IBM SPSS Statistic; IBM Corp., Armonk, NY, USA). The Shapiro–Wilk test was used to assess data normality. Data are presented as mean ± standard deviation when normally distributed and as median (range) when not normally distributed. The spread of contrast following TAP-L versus TAP-SL was compared using the Student’s
t test. The number of ventral nerve branches successfully stained were compared with the Mann–Whitney U test, whereas any difference in the single nerve staining was compared with the Fisher’s exact test. Differences were considered statistically significant when
p < 0.05.
4. Discussion
This study described the anatomical and sonographic findings of the TAP in cats. Injectate distribution and consistency of nerve staining was also assessed by ultrasonography, CT and dissection after a TAP injection using two approaches. This information allows better understanding of potential local anaesthetic distribution, complications and limitations of this locoregional technique in the clinical setting.
In this study, a significant difference in cranial-to-caudal injectate spread was found between the 1-point TAP-L approach and the 2-point TAP-SL approach. Nevertheless, the most cranial and caudal margins of the solution spread corresponded to the nerve T10 and L2, with either approach. On the other hand, the TAP-SL approach resulted in a more consistent staining of the target nerves than TAP-L, which could represent a more predictable distribution of an anaesthetic solution in cats.
The ventromedial branch of L2 was stained only in 12.5% of cases after TAP-SL and TAP-L. Therefore, the caudal portion of the abdominal wall could possibly not be desensitized, depending on the incision size, by a local anaesthetic. The lateral TAP injection was performed inserting the needle immediately caudal to the last ribs at 3–4 cm from the ventral midline; it is possible that a more caudal injection point (about 1–1.5 cm from the caudal margin of the last rib—in proximity of the umbilicus) would lead to a better caudal injectate distribution and consistent staining of L2 than the present technique. Further anatomical studies are warranted to confirm or reject this hypothesis.
The ventromedial branch of T10, T11, T12, T13, L1 and L2 was stained in 87.5%, 87.5%, 100%, 100%, 100% and 12.5% of cases with our lateral-longitudinal TAP-SL approach when compared with 57.1%, 100%, 87.5%, 28.5%, 42.8% and 87.5%, respectively, by a previously reported lateral-transversal TAP-SL approach [
8]. This shows that the needle orientation influences nerve staining after a lateral TAP injection, as the lateral-longitudinal TAP injection resulted in a more consistent nerve staining from T10 to L1 than the lateral-transversal TAP injection. However, the lateral-transversal TAP-SL resulted in a consistent staining of L2. This previous study obtained more consistent staining of L2 branches than with the 2-point lateral-transversal TAP-SL technique when the same injectate volume was divided in three aliquots and injected using a 3-point lateral-transversal TAP-SL technique [
8]. At this point, a clinical trial is necessary to assess which TAP injection technique would be more effective (i.e., a 2-point TAP-SL with a lateral-longitudinal approach or a 3-point TAP-SL with a lateral-transversal approach).
Individual variability in the pathway of the thoracolumbar nerve branches in the TAP was observed as reported in similar cadaveric studies in cats [
8] and in dogs [
17]. T9 branches were identified within the TAP in 75% of our cats versus 56.2% in a previous study [
8]. L3 branches were observed within the TAP in all hemiabdomens. However, in 87.5% of cadavers, it ended within the belly of the m.
transversus abdominis, whereas in one out of eight cadavers, it reached the midline at the level of the pubis. Neither of the TAP approaches employed in this study stained the ventral branches of T9 and L3. These findings demonstrate that the efficacy of a TAP block may vary due to anatomical individual differences independently of similar volumes of administration and techniques. It is possible that the efficacy of a TAP block may also vary according to the extent (size) and location (cranial versus caudal) of a laparotomy.
In this study, the ventrolateral branches of the thoracolumbar nerves responsible for the abdominal cutaneous dermatome innervation were also stained. This phenomenon was also observed in a previous study [
8]; however, the clinical relevance of this finding is unknown. Although the TAP injections were all performed in the target fascial plane, partial intramuscular contrast spread in the m.
transversus abdominis and suspected presence of contrast between the mm.
obliquus internus and
externus abdominis were observed in the CT. During gross anatomical dissection, the m.
obliquus internus abdominis and sometimes the m.
obliquus externus abdominis were partially stained by the dye solution, as previously reported [
8]. Diffusion of injectate through the thin abdominal musculature may commonly occur after a TAP block in cats without any clinical significance.
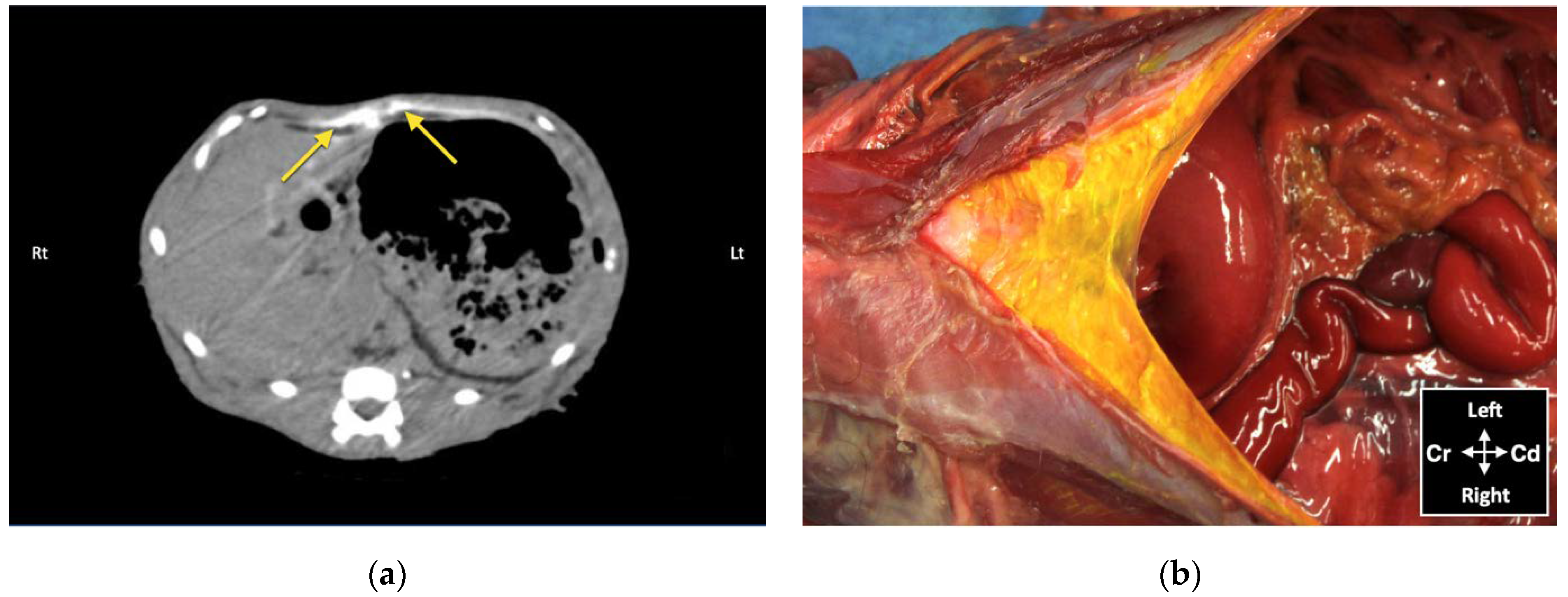
Although no bupivacaine–iopamidol–dye solution was observed free in the abdominal cavity, a total of 4 out of 24 injections (16.7%) resulted in intraperitoneal spread of injectate solution. Both CT and dissection revealed the presence of injectate solution into the falciform ligament following two subcostal injections. In these two cases, distribution of injectate solution both in the TAP and ventral to the m. transversus abdominis was observed with the US. Nevertheless, gentle repositioning of the needle tip under US visualization resulted in partial intramuscular injection in the m. rectus abdominis, suggesting that the bevel of the needle was larger than the TAP and the m. transversus abdominis. Analysis of CT images also revealed focal intraperitoneal injections that were not observed with real-time US visualization and could not be identified by gross dissection following two lateral TAP injections. These two focal infiltrations of contrast solution could be explained by puncture of the peritoneum with the needle tip, without evident needle perforation of the abdominal cavity. The Quincke needle used in this study has a 20-degree-long cutting bevel, and the m. transversus abdominis of the three cats, in which these complications were observed, was about 0.2 mm thick. It is possible that a different type of needle with a shorter bevel (e.g., an insulated needle for peripheral nerve block or a Touhy epidural needle with a rounder tip) would be more appropriate to perform TAP injections in cats.
The prevalence of intraperitoneal injection reported herein (16.7%) was lower than that observed in similar investigations into dog (23%) and human cadavers (25%) using CT studies [
11,
18]. Therefore, CT might be a more sensitive method to assess contrast misplacement and to identify intra-abdominal injections than gross anatomical dissection. However, the use of CT to assess the injectate spread before a clinical procedure is not feasible. Hence, particular attention should be taken when performing an US-guided TAP block in small patients, because intraperitoneal injection is a possible risk. In this case, there could be a risk of organ perforation or partial anaesthetic block with this technique.
The injectate volume used was based on a previous cadaveric study in cats [
8]. Moreover, a volume of 1 mL/kg of anaesthetic solution is considered a reasonable clinical applicable volume and easily divided in four aliquots. The anaesthetic solution should be diluted before administration without exceeding recommended doses of local anaesthetic. Our group published a recent pharmacokinetic study on bupivacaine administered by TAP block in healthy cats showing that a dose of 2.5 mg/kg is safe [
19]; this dose is comparably higher than previously recommended in cats [
20], allowing high volumes of administration. A cadaveric study in dogs assessing different volumes from 0.5 to 1 mL/kg, administered by 1-point lateral-transversal TAP block, showed a correlation between volume and number of stained nerves [
21]. However, two other cadaveric studies in dogs showed that volumes higher than 0.5 mL/kg/point tend to be pooled in the injection site rather than distributed cranio-caudally along the TAP [
10,
11].
The findings obtained by cadaveric studies should be carefully applied to a clinical setting. The physical properties of the bupivacaine–iopamidol–dye solution employed in this investigation differ from local anaesthetics, and it is possible that this might have influenced the results. The contrast was diluted to a proportion of 1:10 as previously described in cadaveric studies in humans and dogs [
11,
18]. A further dilution of the contrast might have reduced the quality of the CT images and the spread analysis. On the other hand, this cadaver study was able to identify potential complications and limitations of this technique in the clinical setting. Indeed, this study allowed us to later perform a pharmacokinetic study [
19] of bupivacaine in cats undergoing ovariohysterectomy in which we demonstrated that the TAP-SL is safe and bupivacaine concentrations (2 or 2.5 mg/kg) were below toxic levels. Our group is now conducting a prospective, randomised, masked clinical trial investigating the analgesic efficacy of bupivacaine following a TAP block in cats.
This investigation has limitations, and some have already been discussed. The injectate distribution in a cadaver model may differ from the distribution encountered in live animals because of tissue modifications related to freezing and thawing, systemic drug absorption according to the tissue temperature, and changes in abdominal pressure with breathing. A further limitation is that one researcher performing the dissections was not blinded, which may have biased the interpretation of results. Finally, it is unknown how the results and potential complications would have been changed if the TAP block had been performed by an inexperienced veterinarian.