Nanoliposome-Loaded Phenolics from Nasturtium officinale Improves Health Parameters in a Colorectal Cancer Mouse Model
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethic Statement
2.2. Plant Material and Reagents
2.3. Fractionation and Total Phenolic Determination
2.4. Nanoliposomes Preparation
2.5. Characterization of Nanoliposomes
2.6. Phenolic Profiling of Nanoliposomes
2.7. Animal Trial
2.8. Blood Parameters, Liver Enzymes and Lipid Peroxidation Assay
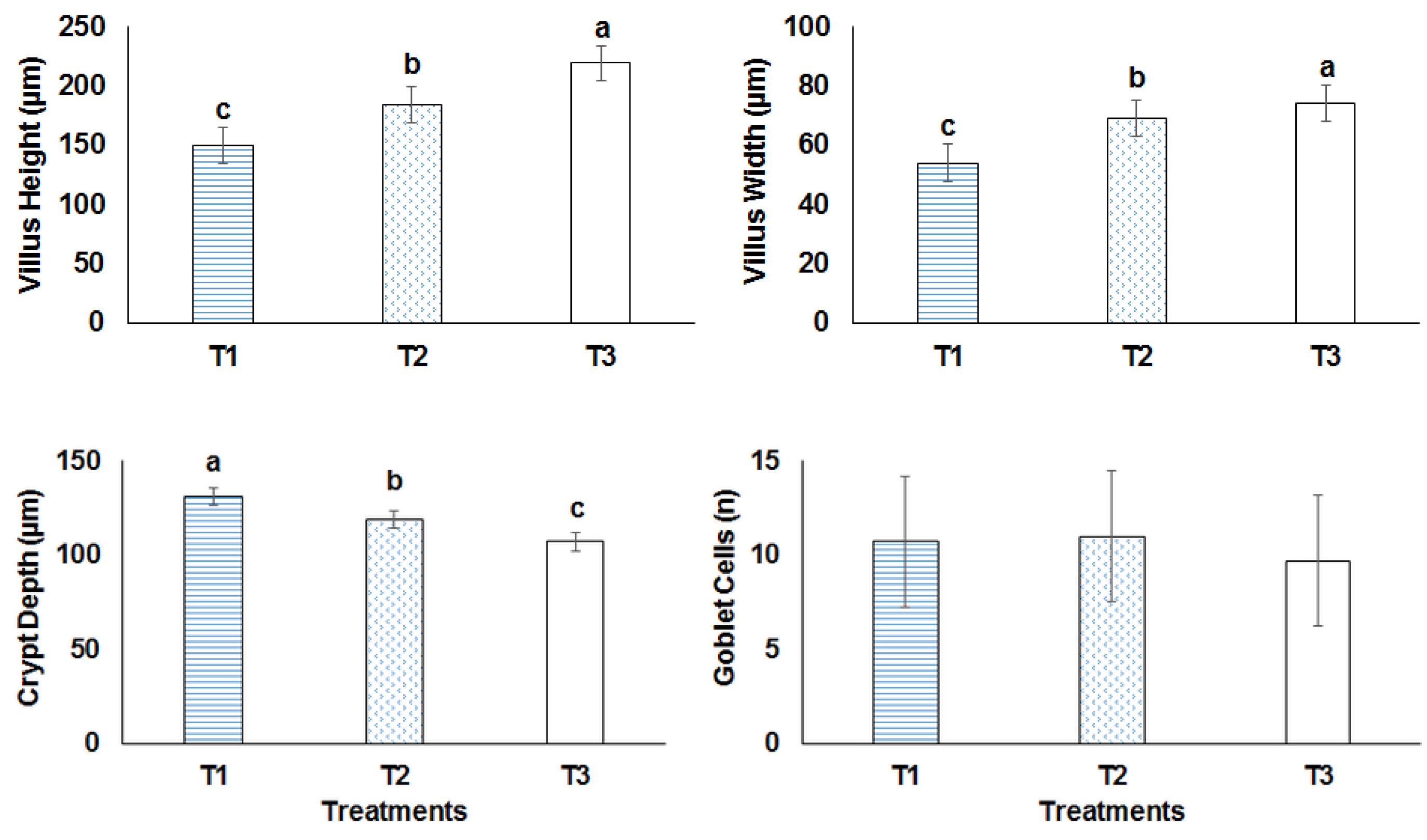
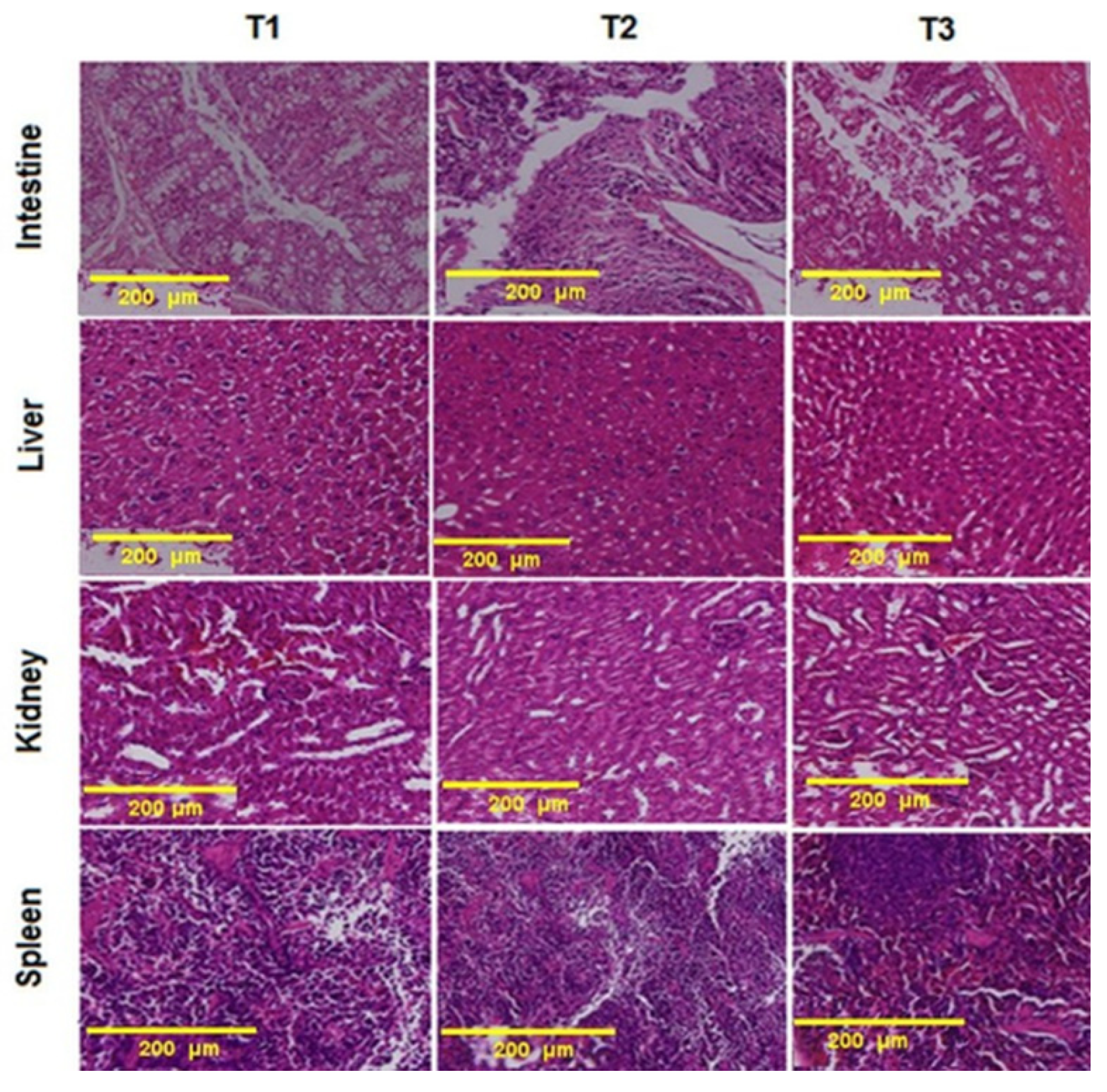
2.9. Histopathology and Morphometric Analyses
2.10. Gene Expression Analysis
2.11. Statistical Analysis
3. Results
3.1. Fractionation and Total Phenolic Determination
3.2. Physicochemical Characteristics of PRF Loaded Nanoliposome
3.3. Analysis of Phenolic Compounds
3.4. Weight and Feed Intake Alteration
3.5. Analysis of Serum and Blood Biomarkers
3.6. Histopathological Examination
3.7. Gene Expression Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fadaka, A.; Ajiboye, B.; Ojo, O.; Adewale, O.; Olayide, I.; Emuowhochere, R. Biology of glucose metabolization in cancer cells. J. Oncol. Sci. 2017, 3, 45–51. [Google Scholar] [CrossRef]
- Giovannucci, E. Diet, body weight, and colorectal cancer: A summary of the epidemiologic evidence. J. Women. Health 2003, 12, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Testa, U.; Pelosi, E.; Castelli, G. Colorectal cancer: Genetic abnormalities, tumor progression, tumor heterogeneity, clonal evolution and tumor-initiating cells. Med. Sci. 2018, 6, 31. [Google Scholar] [CrossRef]
- Hossain, M.S.; Karuniawati, H.; Jairoun, A.A.; Urbi, Z.; Ooi, D.J.; John, A.; Lim, Y.C.; Kibria, K.K.; Mohiuddin, A.; Ming, L. Colorectal Cancer: A Review of Carcinogenesis, Global Epidemiology, Current Challenges, Risk Factors, Preventive and Treatment Strategies. Cancers 2022, 14, 1732. [Google Scholar] [CrossRef]
- Shakeri, M.; Le, H.H. Deleterious Effects of Heat Stress on Poultry Production: Unveiling the Benefits of Betaine and Polyphenols. Poultry 2022, 1, 147–156. [Google Scholar] [CrossRef]
- Zeb, A.J.S. Phenolic profile and antioxidant potential of wild watercress (Nasturtium officinale L.). SpringerPlus 2015, 4, 714. [Google Scholar] [CrossRef] [PubMed]
- Sefidkon, F.; Torabi Sagvand, B.; Naderi, M.; Ghooshegir, S. Comparison of anticancer effects of nanocapsules of Nasturtium officinalis (L.) R. Br. extract with methanolic extract and its fractions. Iranian J. Med. Aromat. Plants Res. 2013, 29, 35–50. [Google Scholar]
- Fallah, N.; Ebrahimi, S. The anti-cancer effect of watercress (rorripa nasturtium aquaticum) extract on breast cancer cells. Zahedan J. Res. Med. Sci. 2016, 18. [Google Scholar] [CrossRef]
- Adlravan, E.; Nejati, K.; Karimi, M.A.; Mousazadeh, H.; Abbasi, A.; Dadashpour, M. Potential activity of free and PLGA/PEG nanoencapsulated nasturtium officinale extract in inducing cytotoxicity and apoptosis in human lung carcinoma A549 cells. J. Drug Delivery Sci. Tech. 2021, 61, 102256. [Google Scholar] [CrossRef]
- Mazandarani, M.; Momeji, A.; Zarghami, M.P. Evaluation of phytochemical and antioxidant activities from different parts of Nasturtium officinale R. Br. Maz. 2013, 659–664. [Google Scholar]
- Lagoa, R.; Silva, J.; Rodrigues, J.R.; Bishayee, A. Advances in phytochemical delivery systems for improved anticancer activity. Biotechnol. Adv. 2020, 38, 107382. [Google Scholar] [CrossRef] [PubMed]
- Slika, L.; Moubarak, A.; Borjac, J.; Baydoun, E.; Patra, D.J. Preparation of curcumin-poly (allyl amine) hydrochloride based nanocapsules: Piperine in nanocapsules accelerates encapsulation and release of curcumin and effectiveness against colon cancer cells. Mater. Sci. Eng. 2020, 109, 110550. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Li, J.-S.; Tang, D.-N.J.J.B. Potential of utilization of albumin as a delivery module in cancer model. J. Buon 2019, 24, 347–353. [Google Scholar] [PubMed]
- Karimi, E.; Mehrabanjoubani, P.; Es-Haghi, A.; Chamani, J. Phenolic compounds of endemic buxus plants in caspian hyrcanian forest (Buxus Hyrcana Pojark) and their biological activities. Pharm. Chem. J. 2019, 53, 741–747. [Google Scholar] [CrossRef]
- Oskoueian, E.; Karimi, E.; Noura, R.; Ebrahimi, M.; Shafaei, N.; Karimi, E. Nanoliposomes encapsulation of enriched phenolic fraction from pistachio hulls and its antioxidant, anti-inflammatory, and anti-melanogenic activities. J. Microencapsul. 2020, 37, 1–13. [Google Scholar] [CrossRef]
- Mingxin, Z.; Yan, L.; Hongbo, W.; Jianhua, Z.; Hongyan, L.; He, L.; Hongqi, X.; Sen, Z.; Xiaoguang, C. The antitumor activity of meisoindigo against human colorectal cancer HT-29 cells in vitro and in vivo. J. Chemother. 2008, 20, 728–733. [Google Scholar] [CrossRef]
- Shafaei, N.; Barkhordar, S.M.A.; Rahmani, F.; Nabi, S.; Idliki, R.B.; Alimirzaei, M.; Karimi, E.; Oskoueian, E. Protective effects of Anethum graveolens seed’s oil nanoemulsion against cadmium-induced oxidative stress in mice. Biol. Trace Elem. Res. 2020, 198, 583–591. [Google Scholar] [CrossRef]
- Navarrete, J.; Vásquez, B.; Del Sol, M. Morphoquantitative analysis of the Ileum of C57BL/6 mice (Mus musculus) fed with a high-fat diet. Int. J. Exp. Pathol. 2015, 8, 14649. [Google Scholar]
- Kathirvel, E.; Chen, P.; Morgan, K.; French, S.W.; Morgan, T. Oxidative stress and regulation of anti-oxidant enzymes in cytochrome P4502E1 transgenic mouse model of non-alcoholic fatty liver. J. Gastroenterol. Hepatol. 2010, 25, 1136–1143. [Google Scholar] [CrossRef]
- Anitha, A.; Maya, S.; Sivaram, A.J.; Mony, U.; Jayakumar, R. Combinatorial nanomedicines for colon cancer therapy. Nanomed. Nanobiotechnology 2016, 8, 151–159. [Google Scholar] [CrossRef]
- Wang, T.; Liu, Z.; Zhang, Z.; Tang, S.; Yue, M.; Feng, S.; Hu, M.; Xuan, L.; Chen, Y. Evaluation of antitumor activity of survivin short interfering RNA delivered by lipid nanoparticles in colon cancer in vitro and in vivo. Oncol. Lett. 2017, 14, 2001–2008. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Hasanzadeh Davarani, F.; Javanmard, R.; Dokhani, A.; Khorasani, S.; Mozafari, M.J.P. Impact of particle size and polydispersity index on the clinical applications of lipidic nanocarrier systems. Pharmaceutics 2018, 10, 57. [Google Scholar] [CrossRef] [PubMed]
- Bulbake, U.; Doppalapudi, S.; Kommineni, N.; Khan, W.J.P. Liposomal formulations in clinical use: An updated review. Pharmaceutics 2017, 9, 12. [Google Scholar] [CrossRef] [PubMed]
- Refai, H.; Hassan, D.; Abdelmonem, R. Development and characterization of polymer-coated liposomes for vaginal delivery of sildenafil citrate. Drug Deliv. 2017, 24, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Yazdanparast, R.; Bahramikia, S.; Ardestani, A.J.C.-B. Nasturtium officinale reduces oxidative stress and enhances antioxidant capacity in hypercholesterolaemic rats. Chem. Biol. Interact. 2008, 172, 176–184. [Google Scholar] [CrossRef]
- Bahramikia, S.; Yazdanparast, R.J.J.o.A.; Studies, M. Antioxidant efficacy of Nasturtium officinale extracts using various in vitro assay systems. J. Acupunct. Meridian Stud. 2010, 3, 283–290. [Google Scholar] [CrossRef]
- Bahramikia, S.; Yazdanparast, R. Effect of hydroalcoholic extracts of Nasturtium officinale leaves on lipid profile in high-fat diet rats. J. Ethnopharmacol. 2008, 115, 116–121. [Google Scholar] [CrossRef]
- Rašić, I.; Rašić, A.; Akšamija, G.; Radović, S. The relationship between serum level of malondialdehyde and progression of colorectal cancer. Acta Clin. Croat. 2018, 57, 411–416. [Google Scholar] [CrossRef]
- Shakeri, M.; Cottrell, J.J.; Wilkinson, S.; Le, H.H.; Suleria, H.A.; Warner, R.D.; Dunshea, F. Dietary betaine reduces the negative effects of cyclic heat exposure on growth performance, blood gas status and meat quality in broiler chickens. Agriculture 2020, 10, 176. [Google Scholar] [CrossRef]
- Cong, Z.; Zhao, Q.; Yang, B.; Cong, D.; Zhou, Y.; Lei, X.; Zhang, X.J.P. Ginsenoside Rh3 inhibits proliferation and induces apoptosis of colorectal cancer cells. Pharmacology 2020, 105, 329–338. [Google Scholar] [CrossRef]
- Nazeri, M.; Mirzaie-Asl, A.; Saidijam, M.; Moradi, M.J.M.B.R. Methanolic extract of Artemisia absinthium prompts apoptosis, enhancing expression of Bax/Bcl-2 ratio, cell cycle arrest, caspase-3 activation and mitochondrial membrane potential destruction in human colorectal cancer HCT-116 cells. Mol. Biol. Rep. 2020, 47, 8831–8840. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Ma, L.; Li, W.; Ding, L.; Gao, H. MicroRNA-498 reduces the proliferation and invasion of colorectal cancer cells via targeting Bcl-2. FEBS Open Bio. 2020, 10, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Benkhelifa, S.; Rafa, H.; Belhadef, S.; Ait-Kaci, H.; Medjeber, O.; Belkhelfa, M.; Hetit, S.; Ait-Younes, S.; Moralès, O.; Mahfouf, H.J. Aberrant up-regulation of iNOS/NO system is correlated with an increased abundance of Foxp3+ cells and reduced effector/memory cell markers expression during colorectal cancer: Immunomodulatory effects of cetuximab combined with chemotherapy. Inflammopharmacology 2019, 27, 685–700. [Google Scholar] [CrossRef] [PubMed]
- Egler, R.A.; Ahuja, S.P.; Matloub, Y. L-asparaginase in the treatment of patients with acute lymphoblastic leukemia. Inflammopharmacology 2016, 7, 62–71. [Google Scholar]
- Doustimotlagh, A.H.; Kokhdan, E.P.; Vakilpour, H.; Khalvati, B.; Barmak, M.J.; Sadeghi, H.; Asfaram, A.J. Protective effect of Nasturtium officinale R. Br and quercetin against cyclophosphamide-induced hepatotoxicity in rats. Mol. Biol. Rep. 2020, 47, 5001–5012. [Google Scholar] [CrossRef]
- Azarmehr, N.; Afshar, P.; Moradi, M.; Sadeghi, H.; Sadeghi, H.; Alipoor, B.; Khalvati, B.; Barmoudeh, Z.; Abbaszadeh-Goudarzi, K.; Doustimotlagh, A. Hepatoprotective and antioxidant activity of watercress extract on acetaminophen-induced hepatotoxicity in rats. Mol. Biol. Rep. 2019, 5, e02072. [Google Scholar] [CrossRef]
- Martínez-Sánchez, A.; Gil-Izquierdo, A.; Gil, M.I.; Ferreres, F. A comparative study of flavonoid compounds, vitamin C, and antioxidant properties of baby leaf Brassicaceae species. J. Agric. Food Chem. 2008, 56, 2330–2340. [Google Scholar] [CrossRef]
- Asadi, M.; Mirvaghefei, A.; Nematollahi, M.; Banaee, M.; Ahmadi, K. Effects of Watercress (Nasturtium nasturtium) extract on selected immunological parameters of rainbow trout (Oncorhynchus mykiss). Open Vet. J. 2012, 2, 32–39. [Google Scholar]
- Shakeri, M.; Shakeri, M.; Omidi, A. Effect of garlic supplementation to diet on performance and intestinal morphology of broiler chickens under high stocking density. Istanb. Univ. Vet. Fak. Derg 2014, 41, 212–217. [Google Scholar]
- Giannenas, I.; Bonos, E.; Skoufos, I.; Tzora, A.; Stylianaki, I.; Lazari, D.; Tsinas, A.; Christaki, E.; Florou-Paneri, P. Effect of herbal feed additives on performance parameters, intestinal microbiota, intestinal morphology and meat lipid oxidation of broiler chickens. Br. Poult. Sci. 2018, 59, 545–553. [Google Scholar] [CrossRef]


| Gene | Forward (5′ to 3′) | Reverse (5′ to 3′) |
|---|---|---|
| Caspase 3 | CACAATAGCACCCATCCG | GGGACATCAGTCGCTTCA-3 |
| Bax | AATGCCCGTTCATCTCAG | GGGACATCAGTCGCTTCA |
| Bcl2 | GGCACTGTCTTGACCCAC | TCATAAACCCTGCTTGCTG |
| iNOS | CACCTTGGAGTTCACCCAGT | ACCACTCGTACTTGGGATGC |
| SOD | GAGACCTGGGCAATGTGACT | GTTTACTGCGCAATCCCAAT |
| β-actin 1 | CCTGAACCCTAAGGCCAACC | CAGCTGTGGTGGTGAAGCTG |
| Particle Size (nm) | Polydispersity Index (PDI) | Zeta Potential (mV) |
|---|---|---|
| 189.2 ± 11.68 | 0.29 ± 0.04 | −58.2 ± 6.82 |
| Phenolic Compounds Contents (µg/g DW) | ||||
|---|---|---|---|---|
| GA 1 | SA | SY | CA | EA |
| 438.3 ± 2.7 | 206.2 ± 5.1 | 287.3 ± 1.0 | 681.8 ± 4.5 | 702.5 ± 6.2 |
| Average 1 | T1 | T2 | T3 | SEM |
|---|---|---|---|---|
| Average daily weight gain (mg) | 39.1 c | 46.5 b | 51.9 a | 2.13 |
| Average daily feed intake (g) | 1.46 c | 1.54 b | 1.63 a | 0.03 |
| RBC 1 (106/µL) | WBC (103/µL) | NEU (%) | Lymph (%) | Mno (%) | Eo (%) | |
|---|---|---|---|---|---|---|
| T1 2 | 7.18 | 4.9 | 35 | 58 | 5 | 2 |
| T2 | 7.44 | 5.9 | 36 | 57 | 4 | 3 |
| T3 | 7.56 | 6.1 | 35 | 61 | 3 | 1 |
| SEM | 0.92 | 7.63 | 2.48 | 2.75 | 0.48 | 0.61 |
| Gene Expression (Fold Changes) | SEM | |||
|---|---|---|---|---|
| Genes | T1 1 | T2 | T3 | |
| Up-regulated genes | ||||
| Caspase-3 | 1.0 c | 1.7 b | 2.4 a | 0.08 |
| Bax | 1.0 c | 2.9 b | 3.2 a | 0.11 |
| SOD | 1.0 c | 1.8 b | 2.5 a | 0.09 |
| Down-regulated genes | ||||
| Bcl2 | 1.0 c | 1.6 b | 2.2 a | 0.12 |
| iNOS | 1.0 c | 1.9 b | 2.7 a | 0.15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taghavinia, F.; Teymouri, F.; Farokhrouz, F.; Bagherabad, E.H.; Farjami, S.; Karimi, E.; Oskoueian, E.; Le, H.H.; Shakeri, M. Nanoliposome-Loaded Phenolics from Nasturtium officinale Improves Health Parameters in a Colorectal Cancer Mouse Model. Animals 2022, 12, 3492. https://doi.org/10.3390/ani12243492
Taghavinia F, Teymouri F, Farokhrouz F, Bagherabad EH, Farjami S, Karimi E, Oskoueian E, Le HH, Shakeri M. Nanoliposome-Loaded Phenolics from Nasturtium officinale Improves Health Parameters in a Colorectal Cancer Mouse Model. Animals. 2022; 12(24):3492. https://doi.org/10.3390/ani12243492
Chicago/Turabian StyleTaghavinia, Fatemeh, Fatemeh Teymouri, Fatemeh Farokhrouz, Elahe Hashemi Bagherabad, Sarasadat Farjami, Ehsan Karimi, Ehsan Oskoueian, Hieu Huu Le, and Majid Shakeri. 2022. "Nanoliposome-Loaded Phenolics from Nasturtium officinale Improves Health Parameters in a Colorectal Cancer Mouse Model" Animals 12, no. 24: 3492. https://doi.org/10.3390/ani12243492
APA StyleTaghavinia, F., Teymouri, F., Farokhrouz, F., Bagherabad, E. H., Farjami, S., Karimi, E., Oskoueian, E., Le, H. H., & Shakeri, M. (2022). Nanoliposome-Loaded Phenolics from Nasturtium officinale Improves Health Parameters in a Colorectal Cancer Mouse Model. Animals, 12(24), 3492. https://doi.org/10.3390/ani12243492






