4.1. DM, DOM, and iNDF Intake
Dry matter intake (
Table 3) increased quadratically from level 0 to 355.43 g SC+UAS/kg DM, probably due to the inclusion of SC, which has a highly effective ruminal DM degradability (711 g/kg DM) [
8] as a result of its low NDF and lignin concentrations (148 and 10 g/kg DM, respectively), and its high NFC concentration (712 g/kg DM;
Table 1). In addition, the SC+UAS inclusion resulted in decreased iNDFI to levels below 150 g/kg DM, a value above which DM intake can be limited [
31].
Reduction in DM and DOM intakes (
Table 3) were possibly due to metabolic regulation as a consequence of the increased propionate concentrations observed in this experiment (
Table 4 and
Figure 2). Farningham and Whyte [
32] reported that propionate flow on the portal system has an important role in regulating intake, which is independent of changes regarding plasma insulin level. Oba and Allen [
33] observed that increasing propionate concentrations through ruminal infusions decreased DM intake, metabolizable energy intake, meal size, and meal frequency. Propionate uptake by the liver can be used for gluconeogenesis, utilization of ATP, or oxidation in the tricarboxylic acid cycle through acetyl CoA. Propionate uptake during meals stimulates oxidation of acetyl CoA to CO
2, rapidly generating ATP and stimulating satiety [
34].
4.2. Ruminal Fermentation
Quadratic decrease in pH with collection time (
Table 4) was caused by dietary carbohydrate fermentation [
35], which generated the same quadratic response in total VFA concentration (
Table 4). Subsequently, pH increased gradually as a consequence of rumen acids being extracted either via liquid phase passage or by absorption through the rumen wall. The linear reduction in rumen pH with increasing SC+UAS inclusion can be an effect of the fermentation of the high NFC levels in the SC diets (
Table 2), resulting in greater total VFA production. Moreover, SC inclusion diminished rumination [
7] due to low NDF concentration, which reduces saliva secretion and, consequently, rumen buffering capacity [
36]. Ruminal pH did not reach values lower than 6.0 with SC+UAS inclusion. Therefore, cellulolysis [
37,
38] and NDF digestibility could not be affected, as reported by Siqueira et al. [
9].
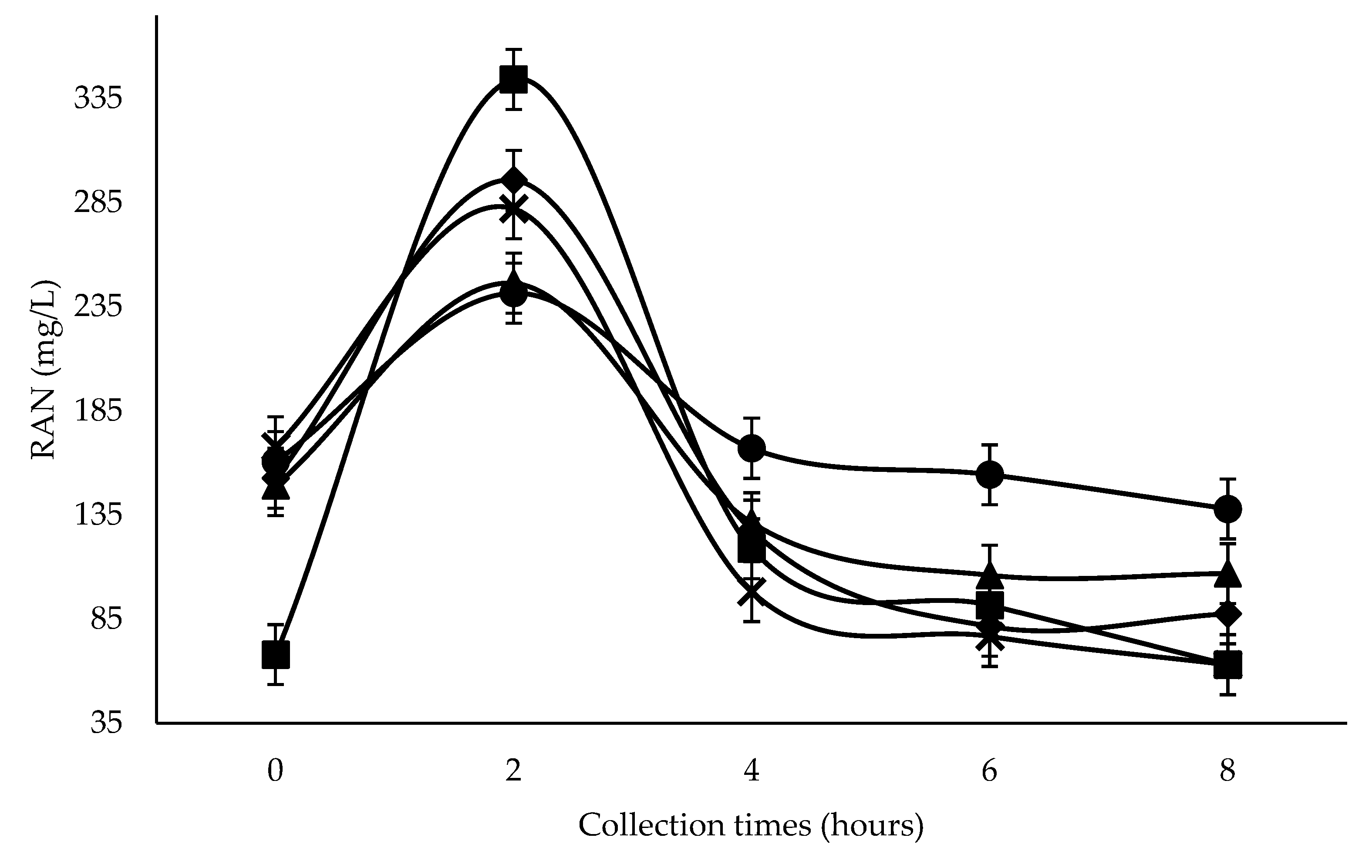
The greatest RAN concentration was seen at 600 g SC+UAS/kg DM two hours after feeding, despite showing the lowest RAN concentration at zero hours (
Figure 1), possibly due to the greater UAS proportion and its high solubility in the rumen. The greater reduction at four hours with 300–600 g SC+UAS/kg DM was possibly due to greater ruminal synchrony of N to readily fermentable carbohydrate [
39], which could be a consequence of a greater NFC content of SC, plus the use of two rapidly rumen-degradable non-protein N-sources, such as UAS. The use of that source in diets with rapidly rumen-fermentable energy promotes greater RAN efficiency by rumen microbes in microbial protein synthesis [
40,
41].
Ruminal pH reduction resulting from SC+UAS inclusion could influence the lower RAN concentration from 300 g SC+UAS/kg DM level after the peak at two hours following the first feeding (
Figure 1). The ruminal wall permeability toward ammonia absorption as NH
3 is 175 times greater than as NH
4+ when the ruminal pH is below 6.4. However, NH
3 concentration is lower than NH
4+; therefore, 70% of the ammonia will be absorbed through the rumen wall as NH
4+ by potassium-facilitated transport [
42]. Thus, NH
4+ ions cannot rapidly be removed through the rumen wall into the blood [
43], meaning that they are stored for longer in the rumen. Therefore, NH
4+ ions can be captured by microbes for protein synthesis [
44], consequently decreasing RAN.
Inclusion levels of SC at all collection times promoted RAN concentrations greater than 50 mg/L (
Figure 1). According to Satter and Slyter [
45], this value is enough to support an adequate rumen bacterial growth rate. Two hours after feeding, all inclusion levels reached RAN concentrations greater than 235 mg/L, a value suggested by Mehrez et al. [
46] to be the point of maximal dietary OM degradation. Detmann et al. [
47] suggested that for diets based on medium- to-high-quality forages, supplementation should increase RAN concentrations above 160 mg/L, a value reached from 0–4 h at 0 g SC+UAS/kg DM.
Acetate concentration usually increases with structural carbohydrate fermentation in the diet [
48]. However, in the present experiment there was a linear increase in acetate concentration with increasing SC+UAS inclusion (
Table 4) as NDF decreased (
Table 2), differing by 18% between the 0 and 600 g SC+UAS/kg DM treatments. The increase in the acetate concentration with SC inclusion could be due to its high content of fructans and
β-glucans [
49], both of which are constituents of the neutral detergent-soluble fiber [
50]. Fructans are fermented by some rumen bacteria that produce acetate [
51].
The increase of propionate concentration with increasing SC+UAS inclusion, with the highest values observed after feeding with 300 g SC+UAS/kg DM (
Figure 2), could be a consequence of NFC fermentation [
48]. Pectin is a constituent of NFC [
50] and is found in SC [
49]. It is rich in rhamnose [
52], the anaerobic fermentation of which generates ethanol, acetone, and 1,2-propanediol [
53]. The latter is fermented by rumen bacteria that produce propionate [
54]. Greater propionic acid production with SC+UAS inclusion promotes a linear decrease in the ratio of acetate to propionate (
Table 4), which is also related to rumen pH decrease [
35].
4.3. Nitrogen Balance, Nitrogen Compounds, and Blood Glucose
The quadratic increase in N-intake (g/d and g/kg BW
0.75) and its excretion through feces (
Table 5) was possibly a consequence of the same response observed for DM intake (
Table 3). Greater DM intake implies higher intake of concentrate consisting mainly of corn (
Table 2). This ingredient increases the amount of microbial-originated N via feces, due to its greater fermentative activity in the large intestine [
55].
Using diets higher in rumen-degradable protein increases the N-excretion via urine [
56]. In this experiment, the inclusion of UAS to adjust the crude protein concentration of diets increased the amount of degradable N in the rumen, although urine N-excretion decreased (
Table 5). The greatest N-intake was observed at 363 g SC+UAS/kg DM, although its excretion in feces decreased from 244 g SC+UAS/kg DM, in addition to the decreased urinary N-excretion. This response allows the inference of a greater N-use by animals at that level of SC+UAS inclusion in the diet. A greater use of energy and a protein diet for microbial protein synthesis generates lower N-loss [
57], and the greater utilization was up to 438 g SC+UAS/kg DM when the greatest N-retention was reached.
Inclusion levels of 0 and 150 g SC+UAS/kg DM showed consumptions in excess of 33.6 g N/kg DOM; therefore, losses of protein or incomplete net transfer could occur [
58], which explains the lower N-retention at these levels of inclusion. At 300 g SC+UAS/kg DM, consumption was higher than 16 g N/kg DOM; hence, there was no N-limitation for microbial protein synthesis [
59]. However, retained N decreased from inclusion level of 438 g SC+UAS/kg DM; therefore, other factors generated the decrease in the use of N from this level of inclusion.
The dietary N-usage was also reflected on both serum urea and plasma ammonia (
Table 5), with a linear decrease in their concentrations with increasing SC+UAS inclusion. Blood urea values were within the normal range for sheep (24–60 mg/dL) [
60], and plasma ammonia concentrations were lower than the limit for intoxication (10–40 mg/L) [
61].
Urine urea concentration and its daily excretion (
Table 5) were not influenced by SC+UAS inclusion, although blood concentration levels differed. The amount of urea excreted in the urine is determined by the amount of urea filtered at the kidney glomeruli [
62]. However, N-intake between 26.0 and 31.6 g/d (
Table 5) could generate the same rate of clearance of urea from the plasma by the kidneys, as observed by Cocimano and Leng [
63]; despite the greater concentration of urea in blood and similar amount in the urine (
Table 6), a similar urea excretion was generated.
Szanyiová et al. [
64] reported when sheep were fed high- or low-N diets (28.71 vs. 9.32 g/d), the N excreted through urine as urea as a proportion of total urinary N was not affected (74 vs. 69%). In the present experiment, N-excretion in the urine as urea as a proportion of the total urinary N showed a quadratic response, despite daily urea excretion in grams was similar between treatments; the difference is possibly due to the total N-excretion through urine being reduced.
Ammonia concentration and daily N-excretion as ammonia through urine (
Table 5) showed the same response as serum urea and plasma ammonia. Free ammonia in extrahepatic tissues binds with glutamate to produce glutamine, which transports ammonia via blood to the liver to produce urea, or even to the kidneys, where glutamine degradation generates ammonia release in the urine [
65]. Therefore, there was possibly a decrease in ammonia production by extrahepatic tissues with increasing SC+UAS inclusion.
Glucose concentrations were within the reference levels of 43–76 mg/dL, as suggested by Contreras et al. [
60]. The linear increase in glucose concentration with increasing SC+UAS inclusion (
Table 5) may be a consequence of the same response observed in rumen propionic acid concentration, since this acid is a substrate for gluconeogenesis and the main glucose source for ruminants [
48,
66]. Moreover, the dietary glucose absorption is greatly reduced in ruminants [
67].
4.4. Urinary Volume, PD, and MPS
Vieira et al. [
68] reported that the diuretic effect of SC is promoted by its high K and low Na contents, despite this inclusion having no influence on urinary volume (
Table 6). This response was different from those reported by other authors when SC was included in the diet [
69,
70].
The quadratic increase in PD excretion with increasing SC+UAS inclusion (
Table 6) could be a consequence of the same effect, as observed for DM intake, given the correlation between them when DM intake is adjusted for BW [
16,
71]. For example, greater PD excretion was observed at 345 g SC+UAS/kg DM, which matched the greater DM intake (g/kg BW) at 343 g SC+UAS/kg DM.
According to Van Soest [
36], greater DM intake reduces the energetic cost of maintaining rumen microbes because the residence time in the rumen is also decreased. In addition, due to the greater DM intake, rumen particle flow is increased and the number of bacteria adhering to feed that escapes from rumen to abomasum and duodenum is increased, so the flow of microbial N is greater, promoting higher microbial purine absorption [
15]. At 335 g SC+UAS/kg DM, the maximum MNS was produced, at which the RAN concentration was estimated at 147 mg/L (RAN = 164.96 − 0.0537 × SC + UAS), close to that observed by Detmann et al. [
72], who reported maximum MNS at a RAN concentration of 145 mg/L.
In the present experiment, a quadratic increase in MPS was observed. In contrast, Cardoso et al. [
5] and Barros et al. [
73] found a linear increase in MPS when SC was included in lamb (450 g/kg DM) and heifer (500 g/kg DM) diets, respectively. This different response may be due to the roughage/concentrate ratio being 50:50 in the experiments mentioned above, and 70:30 in our research.
The gradual increase of MPS until the inclusion reached 418 g SC+UAS/kg DM could be due to the use of the energy contained in the NFC of the SC, mostly as a result of rapid ruminal degradation [
49], and the energy provided by slow degradation of the structural carbohydrates of Tifton hay (cellulose and hemicellulose). These two different rates of carbohydrate degradation are able to maintain a constant supply of ATP for microbial growth [
74]. Additionally, the feed contained non-protein N-sources for rapid ruminal degradation (a UAS mix) that maximize microbial growth, and bacteria that ferment NFC grow faster than those that ferment structural carbohydrates [
75,
76].
In addition to the DM intake reduction from the level 356 g DM/kg, which could negatively influence microbial protein synthesis, the decrease in rumen pH (close to 6.0) would contribute to lower MPS, even when a reduction in RAN with SC+UAS inclusion is associated with greater microbial protein utilization. Ruminal pH values of 6.0 generate lower ATP synthesis because bacteria use it for non-growth functions, such as intracellular pH maintenance [
77].
With decreases in RAN, blood urea and ammonia concentrations (as indicators of dietary N-utilization), and consumption over 16 g N/kg DOM, the lower MPS as from 418 g SC+UAS/kg DM would be directly associated with the rumen environment. The great amounts of fermentable carbohydrates in diets with high SC+UAS levels increase VFAs and consequently decrease rumen pH, which increases bacterial energy demand. This lower production of MPS could explain the decrease in retained N (g/d).
Despite SC+UAS inclusion having an influence on MNS and MPS, the efficiency of MPS was not influenced by inclusion (
Table 6). The values of the efficiency of MPS per kg of DOM intake and kg of DOMR were lower than those recommended by Balch [
78] (130 g MPS/kg DOM, or 200 g MPS/kg DOMR). Pereira et al. [
79] also observed low MPS efficiency (47.4 g/kg total digestible nutrient intake) in sheep fed ad libitum under tropical conditions, a value lower than that recommended by NRC [
14] (130 g/kg).