2.2. Slaughter Process
The entire slaughter process and data collection followed a strict pre-established protocol. However, the height of the shooter position and type of ammunition were modified over the study period to achieve the best possible stun quality.
Approximately 1 h before shooting, four to seven animals were gathered in the corral (
Figure 1). The shooter was partly concealed in the adjacent building and waited for the right moment for a clean and safe shot at one of the animals. The shooter was instructed to shoot at any one of the animals in the corral and aim at a point on the forehead 2 cm caudally to (above) the cross-section of two straight lines from the horn base to the contralateral outer eye angle. The shot should be in the midline and as perpendicular to the forehead as possible [
35]. Depending on the shooter’s position, animal’s location and animal head’s position, the resulting shooting distance and angle to the animal’s forehead varied (
Figure 2). For the first eight animals, the rifle was fired from 2.9 m above the ground. Attempting to increase the shooting angle, the height was increased to 4.1 m for the remaining animals. The shooter was standing on a firm platform with elbows resting on an aiming support.
Shooting was done using a hunting rifle (Marlin Firearms, Madison, North Carolina, USA) equipped with a red-dot sight (Acro C-1TM 3.5 MOA, Aimpoint AB, Malmö, Sweden). Two types of small-calibre ammunition (.22 Magnum rimfire) were tested in a systematic order. Twelve animals (#3–#14) were shot with semi-jacketed 3.24 g hollow-point bullets with a muzzle velocity of 511 m/s and muzzle energy of 425 J (.22 Win. Mag. Game-Shok JHP, Federal Premium Ammunition, Anoka, MN, USA) and eight animals (#1–#2 and #15–#20) were shot with 2.59 g jacketed soft-point bullets with a muzzle velocity of 572 m/s and muzzle energy of 423 J (Maxi-Mag Game Point JSP, CCI, Lewiston, Idaho, USA). A Carl Gustaf 2000 hunting rifle (Bofors Carl Gustaf AB, Eskilstuna, Sweden) and large-bore ammunition with a muzzle velocity of 850 m/s and muzzle energy of 3975 J (Norma Tipstrike 30-06 Springfield, 11 g, Norma Precision AB, Åmotfors, Sweden) was used for reshooting. No silencer was used. A cartridge-driven captive bolt gun was kept as a reserve weapon, but never used. After a successful shot and clearance from the shooter, the remaining animals were immediately released from the corral and the shot animal was checked for stun quality.
As soon as possible after stunning, a loader entered the corral and hoisted the shot animal by the hind legs. It was then bled through thoracic sticking, by first opening the skin and then cutting the thoracic blood vessels using two different sterilized knives. The blood was collected in a tub and later discarded. During bleeding, samples were taken for chemical analysis of blood cortisol, glucose and lactate as stress indicators [
36,
37,
38].
The carcass was placed on an open standard car trailer, wrapped in a clean tarpaulin, the feet covered with protective plastic bags to prevent contamination of the carcass, and transported 2 km to on-farm slaughter premises. Approximately half an hour after stunning, the carcass was lifted off the trailer and onto a wheeled cradle at the entrance to the slaughter premises (
Figure 3 and
Figure 4), where the first part of the dressing was performed, i.e., removal of hide, feet and head. The dehided head was recovered for measurements and dissection later the same day. Evisceration and further dressing were done with the carcass hanging by the hind legs.
Unclean and clean processes were separated, and only dressed and trimmed carcasses were allowed into the clean part of the slaughter premises. The facility was washed and disinfected before and after each carcass was dressed out. The microbiological quality of the tap water in the facility was checked shortly before the study was started and found acceptable for human consumption.
2.3. Data Collection and Analyses
Pre-slaughter conditions and events in the stun corral, as well as the total time the animals were kept in the corral, the time from start of observations to shooting, the stun-to-stick-interval, as well as the time for bleeding, dressing and microbiological sampling, were all recorded by direct observations. Accidentally, the time of entry to the corral was not recorded for animals #1 and #2. The entire stunning and bleeding process was videotaped from the shooter’s location using a portable action camera (GoPro Hero 5 Black, GoPro, Inc., San Mateo, CA, USA) mounted to the wall opening. From the time that the shooter was in position (‘start of recording’) until the remaining animals left the corral, the behaviour of the animals was observed continuously by one person from a concealed location at the corner of the building, approximately 2.5 m above ground level. Behaviours were characterized as walking, listening, exploring or other behaviour (running, turning around, butting, vocalizing, performing flehmen, defecating or urinating). At the start of each recording, the outdoor temperature and relative humidity were measured and the predominant weather conditions were recorded on three binary scales as sunny, windy and rainy. Shooting distance was measured with a portable laser range finder (Strike Inno 600, Optisan Optics Europe GmbH, Berlin, Germany).
Stun quality was assessed in accordance with previous research on captive-bolt stunning of cattle [
20,
22,
39] and a protocol adapted for the gunshot method by Schiffer et al. [
26]. Vital behavioural, eye-related and respiratory signs of inadequate stun (
Table 1) were noted directly after shooting and the behaviours were later confirmed from the video recordings. The stun was classified as ‘deep’ if: (i) posture, (ii) righting reflex at hoisting, (iii) eye direction and movements, (iv) blinking and (v) corneal reflex were of type a and no other signs were of type c; as ‘poor’ if (i) posture, (ii) righting reflex at hoisting, (iii) eye direction and movements, (iv) blinking, (v) corneal reflex, (vi) breathing or (vii) vocalization was of type c; and as ‘ambiguous’ in all other cases.
Lactate was analysed with portable measuring equipment (Lactate Plus, Nova Biomedical Corp., Waltham, MA, USA), and the median of three separate samples was used. Blood serum was separated, frozen and stored for later chemical analyses of cortisol and glucose at a laboratory. Cortisol was analysed twice with Immulite 2000 (Siemens Healthcare Diagnostics, Erlangen, Germany) and the median value was used. The concentration of glucose was determined with Architect c4000 (Abbott Laboratories, Chicago, IL, USA).
On arrival at the slaughter premises, the trailer with the shot animal and the collected blood was weighed on a weighbridge, to estimate the live weight. In the cases of three animals, a significant amount of blood had been spilled in the stun corral; the weight of the lost blood was therefore estimated subjectively, and the recorded live weight was corrected accordingly. After slaughter, the warm carcass was weighed on a digital slaughter scale (Brecom AS, Årnes, Norway), and the air temperature of the slaughter premises was recorded. Bleeding success was calculated as 100 times the estimated weight of drawn blood divided by the estimated live weight [
40].
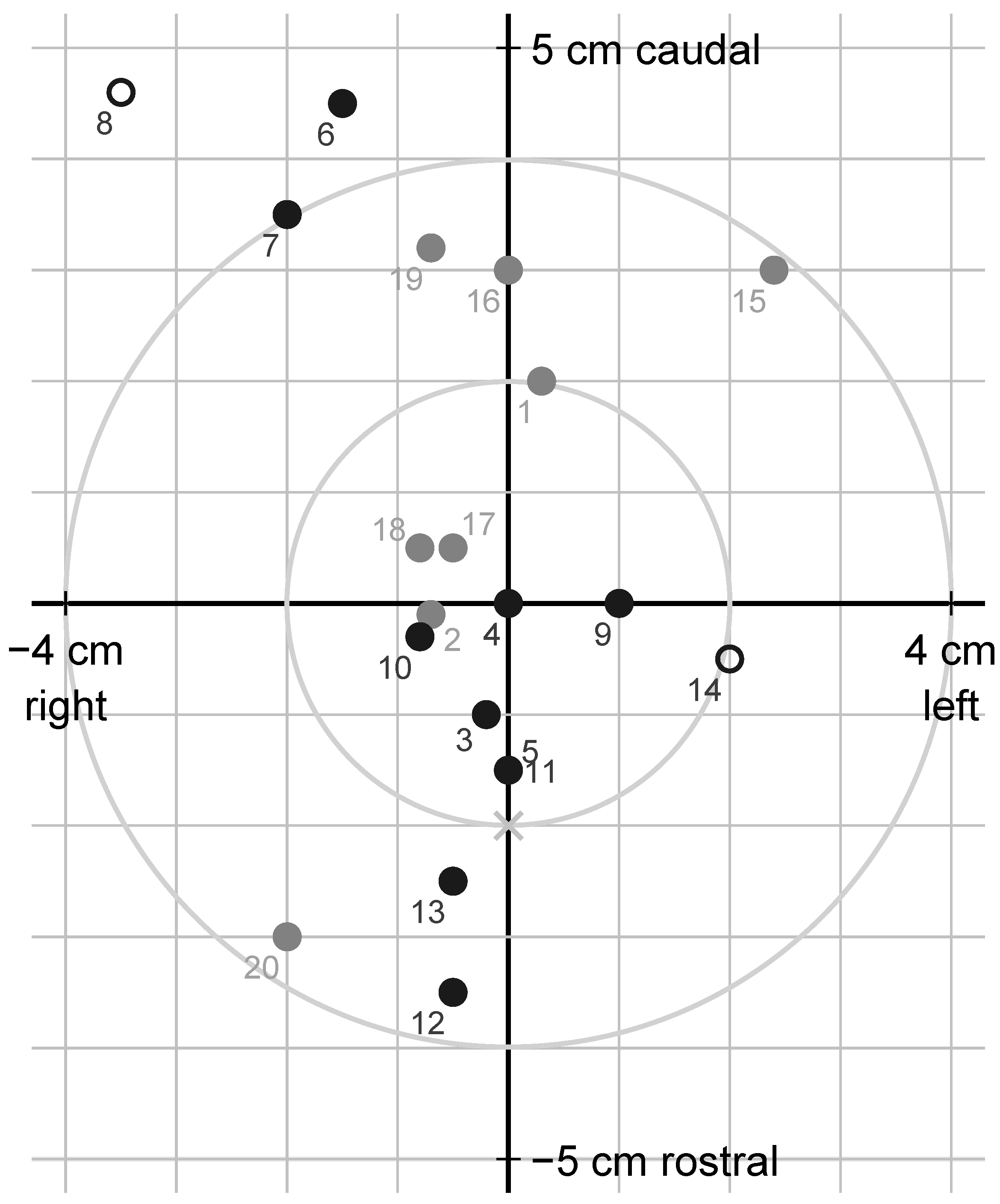
At dissection, the dimensions of the skull and location of the shot (
Figure 5), the diameter of the bullet entry hole through the skull, the depth of the bullet canal and the shot angle relative to the forehead rostrally to the hole were measured. The roof of the skull was removed using a saw, and the brain was extracted from the skull and split into hemispheres. The bullet or bullet fragments were recovered, if possible, and weighed, and their positions in the brain were noted. A dorsal photograph was taken of the dehided head. Photographs were also taken of the brain dorsally in situ after removal of the skull roof, ventrally after extraction, and from the medial aspect of both hemispheres after midsagittal section. The measurements of the shot location were later repeated and corrected using the photographs.
In line with previous research [
26,
35], the extent and degree of lesions and haemorrhages in the brain were assessed from the photographs. Brain tissue damage was characterized as ‘severe’ when there was a complete destruction of the brainstem (composed of the midbrain, the pons and the medulla oblongata), and ‘marginal’ otherwise. Extra-axial brain haemorrhages were assessed on the dorsal and ventral aspects of the cerebrum and the ventral aspect of the brainstem. They were each denoted as ‘moderate’ if covering more than 25% of the visible brain surface, as ‘severe’ if more than 70%, or otherwise as ‘slight’. Intra-ventricular haemorrhages were assessed after midsagittal section and characterized as ‘considerable’ if more than a few small blood clots were found in the ventricles, or otherwise as ‘slight’. The cerebellum, and in some cases a part of the medulla, were not clearly visible in the photographs and therefore not assessed.
The microbiological contamination of carcasses was evaluated after trimming, prior to chilling. The carcasses were sampled by the destructive method, as described by EU Commission Regulation (EC) 2073/2005 and standard ISO 17604:2015 [
41]. Four meat samples, approximately 2 mm thick and with a diameter of 2.5 cm, were excised aseptically using a sterile cork drill from the neck, brisket, flank or rump of both carcass halves at approximately 2 cm from the cutting edge. The pooled samples, representing a total area of 20 cm
2, were placed in a sterile plastic bag and transported to a laboratory for analysis. Total bacterial counts and counts of
Enterobacteriaceae,
Escherichia coli and coagulase-positive
Staphylococcus spp. were calculated as CFU/cm
2 of sampled area and expressed on the log 10 scale. The presence of two important foodborne pathogens,
Listeria monocytogenes and verotoxin-producing
E. coli (VTEC), was analysed but not quantified.
The microbiological samples were put into a blender bag (Grade Packaging, VWR International AB, Spånga, Sweden), mixed with 100 mL of buffered peptone water and stomached in a blender (easyMIX®, AES Laboratoire, Combourg, France) for 2 min. Of the resulting primary homogenate, 25 mL was incubated at 41.5 °C for 20 ± 2 h and analysed for VTEC the following day. To analyse Listeria, 25 mL of the primary homogenate was mixed with 225 mL of Half Fraser broth (Oxoid, Basingstoke, UK) and incubated at 30 °C for 24 ± 2 h. After incubation, 20 µL of the mixture was plated on Chromogenic Listeria Agar ISO plate (Oxoid, Basingstoke, UK) and 20 µL on blood agar (National Veterinary Institute, Uppsala, Sweden), and both were incubated at 37 °C for 48 ± 4 h. Another 100 µL of the same mixture was diluted with 10 mL of Fraser broth (Oxoid, Basingstoke, UK) and incubated at 37 °C for 48 ± 4 h. After incubation, 20 µL was plated on Chromogenic Listeria Agar ISO plate and 20 µL on blood agar (National Veterinary Institute, Uppsala, Sweden), and both were incubated at 37 °C for 48 ± 4 h. All incubations were performed under aerobic conditions.
Appropriate serial decimal dilutions of the remaining primary homogenate were made in peptone saline (0.1% peptone in 0.85% NaCl; Dilucups, LabRobot Products AB, Stenungsund, Sweden) and 0.1 mL of each dilution was plated on 3M™ Petrifilm™ (3M Health Care, Eden Prairie, MN, USA) and incubated at 30 °C for 72 ± 6 h for total aerobic bacteria, at 37 °C for 24 h for Enterobacteriaceae and at 44 °C for 48 h for E. coli. Of each dilution, 0.1 mL was plated on Baird Parker agar (Oxoid, Basingstoke, UK) and incubated at 37 °C for the quantification of coagulase-positive Staphylococcus spp. Again, all incubations were performed under aerobic conditions.
In order to detect VTEC, DNA from 200 µL overnight incubated primary homogenate was extracted using the EZ1 DNA Tissue kit (Qiagen, Hilden, Germany) and analysed for verotoxin genes
stx1 and
stx2 and virulence genes
eae and
saa. Genes
stx1,
stx2 and
eae were detected using the polymerase chain reaction (PCR) with primers and probes according to ISO/TS 13136:2012. The PCR reactions consisted of PerfeCTa qPCR ToughMix with Low ROX (Quantabio, Beverly, MA, USA), 13.3 nM of each
eae primer, 26.6 nM of each
stx primer and 100 nM of
eae,
stx1 and
stx2 probe and 2 µL sample extract, amounting to a total reaction volume of 15 µL. The probes were labeled with HEX, CY5 and FAM fluorophores, respectively. For detection of
saa, primers and probe were used according to the method of Nielsen & Andersen [
42]. The PCR reactions consisted of PerfeCTa qPCR ToughMix with Low ROX (Quantabio), 500 nM of each primer, 100 nM of FAM-labelled probe and 2 µL of sample extract, amounting to a total reaction volume of 15 µL. All PCR analyses were performed using an ABI 7500 Fast Thermocycler (Life Technologies, Carlsbad, CA, USA) and the following thermal profile: 50 °C for 10 min, 95 °C for 3 min and 45 cycles of 95 °C for 3 s and 60 °C for 30 s.
Standard descriptive statistics were produced using Excel 2016 (Microsoft Corp., Redmond, Washington, DC, USA) and Stata IC 15 (StataCorp, College Station, TX, USA). Shot precision, stun quality, levels of stress-related blood constituents, tissue damage, brain haemorrhages and bacterial findings on the carcass were described in detail, looking for possible associations with the recorded animal and environmental factors. The association of stun quality with shooting order on the same day, age, live weight, time from entry to shot, shooting distance, ammunition, shot deviation, skull thickness, brainstem haemorrhage and blood lactate was checked with Spearman rank correlation.