Follicular Dynamics during Estrous Cycle of Pubertal, Mature and Postpartum Crossbred (Nili Ravi × Jianghan) Buffaloes
Abstract
:Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Ethics Statement
2.2. Experimental Animals
2.3. Estrus Detection and Ovarian Ultrasonography
2.4. Follicular Dynamics
2.5. Statistical Analyses
3. Results
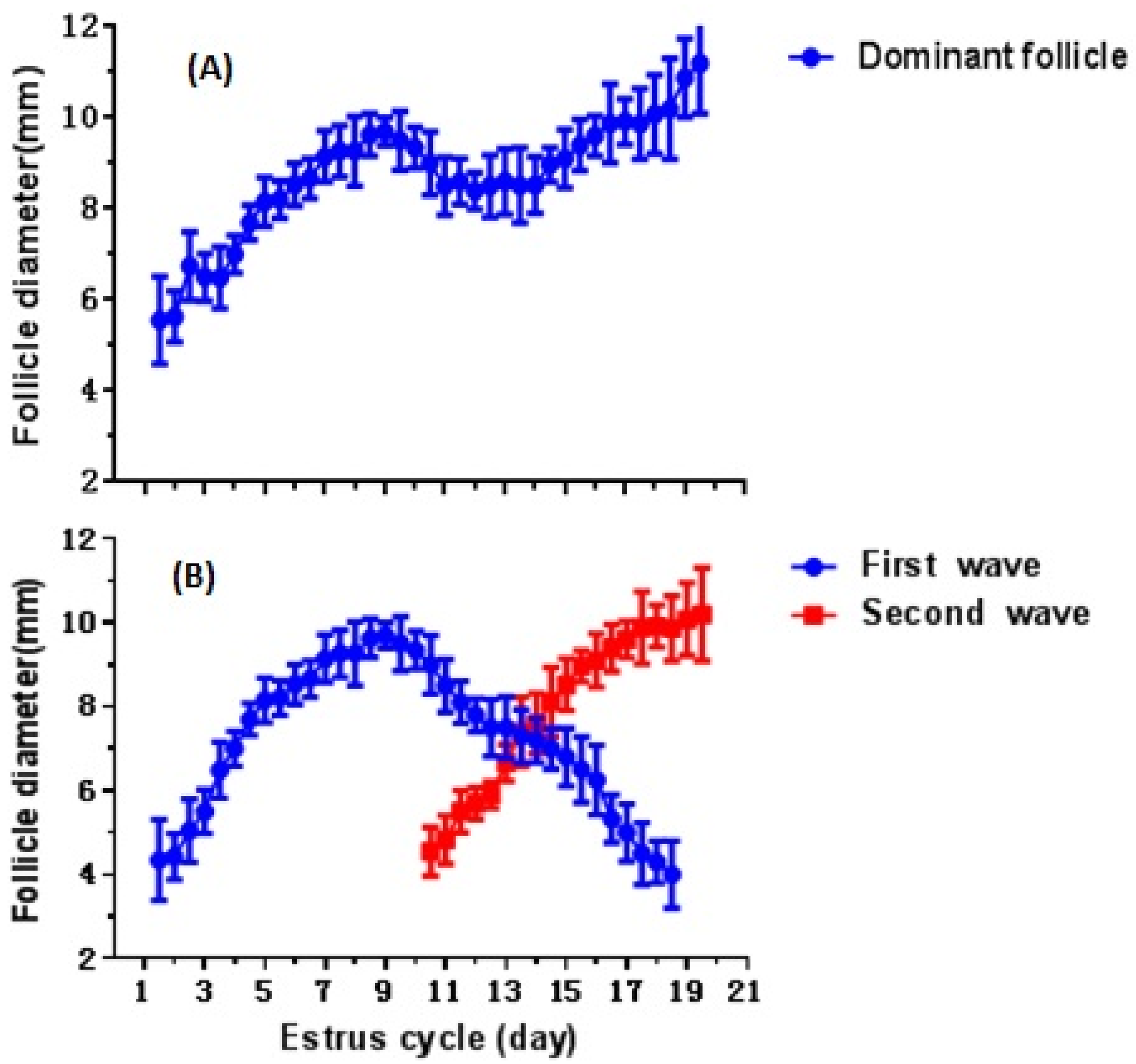
3.1. Distribution of Follicular Waves in Estrous Cycle of Crossbred Buffaloes
3.2. Estrous Cycle Length and Emergence of Follicular Waves in Different Follicle Wave
3.3. Follicular Dynamics in Pubertal Crossbred Buffalo Heifers
3.4. Follicular Dynamics in Sexually Mature Crossbred Buffaloes
3.5. Follicular Dynamics in Postpartum Crossbred Buffaloes
3.6. Relationship between Different Factors (Ovary Location, Physiological Stage and Follicle Wave) Effects on DF Diameter at Either the First Detection or Maximal Size and Diameter Increase in an Estrous Cycle
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cruz, L.C. Transforming swamp buffaloes to producers of milk and meat through crossbreeding and backcrossing. Japs. J. Anim. Plant. Sci. 2014, 22, 157–168. [Google Scholar]
- Jinlong, H. Exploitation and utilization of unique river-type milk buffalo-Binglangjiang buffalo in China. Chin. Cattle Sci. 2010, 36, 59. [Google Scholar] [CrossRef]
- Yuan, L. The only river-type water buffalo species in my country—Binglangjiang River water buffalo. Rural. Best. 2012, 18, 47. [Google Scholar] [CrossRef]
- Changqing, W. Development prospects and countermeasures of my country’s buffalo milk industry. China Anim. Husb. Mag. 2004, 40, 37–39. [Google Scholar]
- Hooda, O.; Singh, S.; Kundu, S.; Vaidya, M. Follicular dynamics its regulation and heat stress effects on folliculogenesis: A review. Wayamba. J. Anim. Sci. 2011, 18, P103–P106. [Google Scholar]
- Ali, A.; Abdel-Razek, A.K.; Abdel-Ghaffar, S.; Glatzel, P. Ovarian follicular dynamics in buffalo cows (Bubalus bubalis). Reprod. Domest. Anim. 2010, 38, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Glatzel, P.S.; Ali, A.; Gilles, M.; Fiedlak, C. Zur feststellung der frühträchtigkeit bei 30 wasserbüffelfärsen (Bubalus bublis) durch die transrektale palpation mit und ohne ultrasonographie. Tieraerzt. Umschau. 2000, 55, 329–332. [Google Scholar]
- Mishra, S.R.; Thakur, N.; Somal, A.; Parmar, M.S.; Yadav, V.P.; Bharati, J.; Bharti, M.K.; Paul, A.; Verma, M.R.; Chouhan, V.S.; et al. Expression and localization of angiopoietin family in buffalo ovarian follicles during different stages of development and modulatory role of angiopoietins on steroidogenesis and survival of cultured buffalo granulosa cells. Theriogenology 2016, 86, 1818–1833. [Google Scholar] [CrossRef]
- Baruselli, P.S.; Soares, J.G.; Gimenes, L.U.; Monteiro, B.M.; Olazarri, M.J.; Carvalho, N.A.T. Control of buffalo follicular dynamics for artificial insemination, superovulation and in vitro embryo production. Buff. Bull. 2013, 32, 160–176. [Google Scholar]
- Noseir, W.M.B.; El-Bawab, I.E.; Hassan, W.R.; Fadel, M.S. Ovarian follicular dynamics in buffaloes during different estrus synchronization protocols. J. Vet. Sci. Dev. 2014, 4, 25–29. [Google Scholar] [CrossRef] [Green Version]
- Ginther, O.J.; Kastelic, J.P.; Knopf, L. Intraovarian relationships among dominant and subordinate follicles and the corpus luteum in heifers. Theriogenology 1989, 32, 787–795. [Google Scholar] [CrossRef]
- Evans, A.C.O. Characteristics of ovarian follicle development in domestic animals. Reprod. Domest. Anim. 2003, 38, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Stagg, K.; Diskin, M.G.; Sreenan, J.M.; Roche, J.F. Follicular development in long-term anoestrous suckler beef cows fed two levels of energy postpartum. Anim. Reprod. Sci. 1995, 38, 49–61. [Google Scholar] [CrossRef]
- Warriach, H.; McGill, D.; Bush, R.; Wynn, P.; Chohan, K.R. A review of recent developments in buffalo reproduction—A review. Asian-Australas. J. Anim. Sci. 2015, 28, 451. [Google Scholar] [CrossRef] [PubMed]
- Riaz, U.; Hassan, M.; Husnain, A.; Naveed, M.I.; Singh, J.; Ahmad, N. Effect of timing of artificial insemination in relation to onset of standing estrus on pregnancy per AI in Nili-Ravi buffalo. Anim. Reprod. 2018, 15, 1231–1235. [Google Scholar] [CrossRef] [Green Version]
- Bodla, M.T.; Anwar, M.; Ahmad, E.; Naseer, Z.; Ahsan, U. Effect of two management systems and mineral feeding on age at puberty in Nili-Ravi buffalo heifers. Buffalo Bull. 2017, 36, 27–33. [Google Scholar]
- Abulaiti, G.H.A.; Ahmad, H.I.; Yang, L. Puberty, ovarian cycle, ovulation and post-partum uterus recovery in river and swamp type crossbred buffaloes. Int. J. Agric. Biol. 2018, 20, 1839–1845. [Google Scholar]
- Abulaiti, A.; Naseer, Z.; Ahmed, Z.; Wang, D.; Hua, G.; Yang, L. Effect of different synchronization regimens on reproductive variables of crossbred (swamp × riverine) nulliparous and multiparous buffaloes during peak and low breeding seasons. Animals 2022, 12, 415–426. [Google Scholar] [CrossRef]
- Warriach, H.M.; Ahmad, N. Follicular waves during the oestrous cycle in Nili-Ravi buffaloes undergoing spontaneous and PGF2α induced luteolysis. Anim. Reprod. Sci. 2007, 101, 332–337. [Google Scholar] [CrossRef]
- Shah, S.N.H. Prolonged calving intervals in the Nili Ravi buffalo. Ital. J. Anim. Sci. 2010, 6, 694–696. [Google Scholar] [CrossRef]
- Mirmahmoudi, R.; Prakash, B.S. The endocrine changes, the timing of ovulation and the efficacy of the Doublesynch protocol in the Murrah buffalo (Bubalus bubalis). Gen. Comp. Endocrinol. 2012, 177, 153. [Google Scholar] [CrossRef] [PubMed]
- Mirmahmoudi, R.; Souri, M.; Prakash, B.S. Comparison of endocrine changes, timing of ovulations, ovarian follicular growth, and efficacy associated with Estradoublesynch and Heatsynch protocols in Murrah buffalo cows (Bubalus bubalis). Theriogenology 2014, 82, 1012–1020. [Google Scholar] [CrossRef] [PubMed]
- Lohan, I.S.; Kaker, M.L.; Malik, R.K.; Arora, K.L. Ovarian follicular dynamics in Murrah buffalo (Bubalus bubalis) heifers during puberty and sexual maturity. Ital. J. Anim. Sci. 2003, 73, 517–518. [Google Scholar]
- Petrovas, G.; Kosior, M.; Presicce, G.; Russo, M.; Zullo, G.; Albero, G.; Alkan, S.; Gasparrini, B. FSH stimulation with short withdrawal improves oocyte competence in Italian Mediterranean buffalo (Bubalus bubalis). Animals 2020, 10, 1997. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Li, H.; Rehman, Z.U.; Dan, X.; Liu, X.; Bhattarai, D.; Yang, L. The efficacy of an inhibin DNA vaccine delivered by attenuated Salmonella choleraesuis on follicular development and ovulation responses in crossbred buffaloes. Anim. Reprod. Sci. 2016, 172, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Sirois, J.; Fortune, J.E. Ovarian follicular dynamics during the estrous cycle in heifers monitored by real-time ultrasonography. Bio. Reprod. 1988, 39, 308–317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moley, K.H.; Schreiber, J.R. Ovarian follicular growth, ovulation and atresia. Endocrine, paracrine and autocrine regulation. Adv. Exp. Med. Biol. 1995, 377, 103–119. [Google Scholar]
- Manik, R.S.; Palta, P.; Singla, S.K.; Sharma, V. Folliculogenesis in buffalo (Bubalus bubalis): A review. Reprod. Fertil. Dev. 2002, 14, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Barkawi, A.H.; Hafez, Y.M.; Ibrahim, S.A.; Ashour, G.; El-Asheeri, A.K.; Ghanem, N. Characteristics of ovarian follicular dynamics throughout the estrous cycle of Egyptian buffaloes. Anim. Reprod. Sci. 2009, 110, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, M.; Kavani, F.; Siddiquee, G.; Dhami, A.; Sarvaiya, N. Characterization of postpartum follicular development through ultrasound scanning and luteal function in suckled Mehsana buffaloes. Indian J. Anim. Sci. 2007, 77, 10–33. [Google Scholar]
- Honparkhe, M.; Gandotra, V.K.; Matharoo, J.S.; Ghuman, S.P.; Dadarwal, D.; Singh, J. Synchronization of follicular wave emergence following ultrasound-guided transvaginal follicle ablation or estradiol-17beta administration in water buffalo (Bubalus bubalis). Anim. Reprod. Sci. 2014, 146, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Valipour, M. Analysis of potential evapotranspiration using limited weather data. Appl. Water Sci. 2014, 7, 187–197. [Google Scholar] [CrossRef] [Green Version]
- Valipour, M. Variations of land use and irrigation for next decades under different scenarios. Irriga 2016, 1, 262. [Google Scholar] [CrossRef] [Green Version]
- Dimauro, C.; Catillo, G.; Bacciu, N.; Macciotta, N.P.P. Fit of different linear models to the lactation curve of Italian water buffalo. Indian J. Anim. Sci. 2010, 4, 22–24. [Google Scholar] [CrossRef]
- Das, S.; Das, M.C.; Paul, R. Swarna Bhasma in cancer: A prospective clinical study. Ayu 2012, 33, 365–367. [Google Scholar] [CrossRef]
- Hossein-Zadeh, N.G. Modelling lactation curve for milk fat to protein ratio in Iranian buffaloes (Bubalus bubalis) using non-linear mixed models. J. Dairy Res. 2016, 83, 334–340. [Google Scholar] [CrossRef]
- Siqueira, J.B.; Leal, L.S.; Oba, E. Ovarian follicular dynamics in buffaloes. Rev. Bra. Reprod. Anim. 2009, 19, 139–148. [Google Scholar]
- Promdireg, A.; Presicce, G.A.; Rensis, F.D.; Singlor, J.; Techakumphu, M. Follicular dynamics following estrus synchronization in swamp buffalo cows (Bubalus bubalis). Thai. Vet. Med. 2008, 38, 25–34. [Google Scholar]
- Baruselli, P.S.; Mucciolo, R.G.; Visintin, J.A.; Viana, W.G.; Arruda, R.P.; Madureira, E.H.; Oliveira, C.A.; Molero-Filho, J.R. Ovarian follicular dynamics during the estrous cycle in buffalo (Bubalus bubalis). Theriogenology 1997, 47, 1531–1547. [Google Scholar] [CrossRef]
- Taneja, M.; Ali, A.; Singh, G. Ovarian follicular dynamics in water buffalo. Theriogenology 1996, 46, 121–130. [Google Scholar] [CrossRef]
- Manik, R.S.; Madan, M.L.; Singla, S.K. Ovarian follicular dynamics in water buffaloes (Bubalus bubalis): Ultrasonically monitoring individual follicles for wave hypothesis. Theriogenology 1994, 41, 246. [Google Scholar] [CrossRef]
- Presicce, G.A.; Parmeggiani, A.; Senatore, E.M.; Stecco, R.; Barile, V.L.; Mauro, G.J.D.; Santis, G.D.; Terzano, G.M. Hormonal dynamics and follicular turnover in prepuberal Mediterranean Italian buffaloes (Bubalus bubalis). Theriogenology 2003, 60, 485–493. [Google Scholar] [CrossRef]
- Campanile, G.; Baruselli, P.S.; Neglia, G.; Vecchio, D.; Gasparrini, B.; Gimenes, L.U.; Zicarelli, L.; D’Occhio, M.J. Ovarian function in the buffalo and implications for embryo development and assisted reproduction. Anim. Reprod. Sci. 2010, 121, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Neglia, G.; Gasparrini, B.; Salzano, A.; Vecchio, D.; Carlo, E.D.; Cimmino, R.; Balestrieri, A.; Michael, J.D.; Campanile, G. Relationship between the ovarian follicular response at the start of an Ovsynch-TAI program and pregnancy outcome in the Mediterranean river buffalo. Theriogenology 2016, 86, 2328–2333. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Fahmy, S. Ovarian dynamics and milk progesterone concentrations in cycling and non-cycling buffalo-cows (Bubalus bubalis) during Ovsynch program. Theriogenology 2007, 68, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Presicce, G.A.; Bella, A.; Terzano, G.M.; Santis, G.D.; Senatore, E.M. Postpartum ovarian follicular dynamics in primiparous and pluriparous Mediterranean Italian buffaloes (Bubalus bubalis). Theriogenology 2005, 63, 1430–1439. [Google Scholar] [CrossRef]
- Ginther, O.J.; Siddiqui, M.A.; Baldrighi, J.M. Effects of conversion of follicular activity from wave 1 to wave 2 and proximity of wave 2 follicles to CL in heifers. Theriogenology 2015, 83, 1241–1248. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Dhaliwal, G.S.; Malik, V.S.; Dadarwal, D.; Honparkhe, M.; Singhal, S.; Brar, P.S. Comparison of follicular dynamics, superovulatory response, and embryo recovery between estradiol based and conventional superstimulation protocol in buffaloes (Bubalus bubalis). Vet. World 2015, 8, 983–988. [Google Scholar] [CrossRef] [Green Version]
- Mokhtari, A.; Kafi, M.; Zamiri, M.J.; Akbari, R. Factors affecting the size of ovulatory follicles and conception rate in high-yielding dairy cows. Theriogenology 2016, 85, 747–753. [Google Scholar] [CrossRef]
- Karen, A.M.; Darwish, S.A. Efficacy of Ovsynch protocol in cyclic and acyclic Egyptian buffaloes in summer. Anim. Reprod. Sci. 2010, 119, 17–23. [Google Scholar] [CrossRef]
- Khan, F.A.; Das, G.K. Follicular characteristics and intrafollicular concentrations of nitric oxide and ascorbic acid during ovarian acyclicity in water buffalo (Bubalus bubalis). Trop. Anim. Health Prod. 2012, 44, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Yindee, M.; Techakumphu, M.; Lohachit, C.; Sirivaidyapong, S.; Na-Chiangmai, A.; Rodriguez-Martinez, H.; Weyden, G.V.D.; Colenbrander, B. Follicular Dynamics and Oestrous Detection in Thai Postpartum Swamp Buffaloes (Bubalus bubalis). Reprod. Domest. Anim. 2011, 46, e91–e96. [Google Scholar] [CrossRef] [PubMed]
- Kanai, Y.; Shimizu, H. Changes in plasma concentrations of luteinizing hormone, progesterone and oestradiol-17β during the periovulatory period in cyclic swamp buffaloes (Bubalus bubalis). Anim. Reprod. Sci. 1986, 11, 17–24. [Google Scholar] [CrossRef]
- Thatcher, W.W.; Macmillan, K.L.; Hansen, P.J.; Drost, M. Concepts for regulation of corpus luteum function by the conceptus and ovarian follicles to improve fertility. Theriogenology 1989, 31, 149–164. [Google Scholar] [CrossRef]
- Satheshkumar, S.; Subramanian, A.; Devanathan, T.G.; Kathiresan, D.; Veerapandian, C.; Palanisamy, A. Follicular and endocrinological turnover associated with GnRH induced follicular wave synchronization in Indian crossbred cows. Theriogenology 2012, 77, 1144–1150. [Google Scholar] [CrossRef]
- Diaz, D.; Galina, C.S.; Fiordelisio, T.; Rubio, I.; Alarcon, M.A.; Rodriguez, A.D.; Orihuela, A. Relationship between growth of the preovulatory follicle and its steroidogenic activity on the onset and expression of estrus behavior in CIDR-treated Bos indicus cows: An observational study. Physiol. Behav. 2012, 107, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, N.A.; Soares, J.G.; Baruselli, P.S. Strategies to overcome seasonal anestrus in water buffalo. Theriogenology 2016, 86, 200–206. [Google Scholar] [CrossRef]
- Miura, R.; Izumi, T. Relationship of the conception rate and the side (left or right) of preovulatory follicle location at artificial insemination in dairy heifers. Anim. Sci. J. 2018, 89, 328–331. [Google Scholar] [CrossRef]


| Physiological Status | Follicular Waves Pattern | ||
|---|---|---|---|
| One-Wave (%) | Two-Wave (%) | Three-Wave (%) | |
| Pubertal (n = 28) | 3/28 (10.71) | 25/28 (89.29) | - |
| Sexually mature (n = 22) | - | 20/22 (90.90) | 2/22 (9.09) |
| Postpartum (n = 18) | 2/18 (11.11) | 14/18 (77.77) | 2/18 (11.11) |
| Overall (n = 68) | 5/68 (7.35) | 59/68 (86.76) | 4/68 (5.88) |
| Characteristics | Puberty | Sexual Maturity | Postpartum |
|---|---|---|---|
| Estrous cycle duration in one-wave cycle | |||
| DF present on right-side ovary (days) | 17.3 ± 1.1 | - | 18.8 ± 1.9 |
| DF present on left-side ovary (days) | 16.8 ± 1.3 | - | 17.6 ± 1.1 |
| Average estrous cycle duration (days) | 17.1 ± 1.2 | - | 18.2 ± 1.6 (n = 2) |
| Estrous cycle duration in two-wave cycle | |||
| DF present on right-side ovary (days) | 19.5 ± 0.6 | 22.1 ± 0.5 | 21.8 ± 0.6 |
| DF present on left-side ovary (days) | 19.3 ± 0.5 | 21.1 ± 0.4 | 22.1 ± 0.6 |
| Average estrous cycle duration (days) | 19.4 ± 0.4 b (n = 25) | 21.5 ± 0.3 a (n = 20) | 21.9± 0.4 a (n = 14) |
| Estrous cycle duration in three-wave cycle | |||
| DF present on right-side ovary (days) | - | 22.3 ± 0.2 | 23.4 ± 0.1 |
| DF present on left-side ovary (days) | - | 21.2 ± 0.8 | 22.8 ± 0.5 |
| Average estrous cycle duration (days) | - | 21.8 ± 0.5 (n = 2) | 23.1 ± 0.3 (n = 2) |
| Emergence of follicular waves during one-wave cycle | |||
| First follicular wave (day) | 1.1 ± 0.4 (n = 3) | - | 1.5 ± 0.5 (n = 2) |
| Emergence of follicular waves during two-wave cycle | |||
| First follicular wave(day) | 1.8 ± 0.6 (n = 25) | 1.7 ± 0.5 (n = 20) | 1.5 ± 0.6 (n = 14) |
| Second follicular wave(day) | 10.3 ± 1.4 (n = 25) | 11.4 ± 1.3 (n = 20) | 12.4 ± 2.3 (n = 14) |
| Emergence of follicular waves during three-wave cycle | |||
| First follicular wave (day) | - | 1.5 ± 0.5 (n = 2) | 1.6 ± 0.4 (n = 2) |
| Second follicular wave (day) | - | 9.1 ± 1.4 (n = 2) | 9.5 ± 1.2 (n = 2) |
| Third follicular wave (day) | - | 15.8 ± 1.1 (n = 2) | 16.3 ± 1.3 (n = 2) |
| Variables | Puberty | Sexual Maturity | Postpartum |
|---|---|---|---|
| Initial diameter of DF | |||
| On right ovary during one-wave (mm) | 5.4 ± 0.2 (n = 15) | - | 5.4 ± 0.1 (n = 8) |
| On left ovary during one-wave (mm) | 5.5 ± 0.2 (n = 13) | - | 5.2 ± 0.2 (n = 6) |
| Average initial diameter DF during one-wave (mm) | 5.5 ± 0.1 (n = 28) | - | 5.3 ± 0.1 b (n = 14) |
| On right ovary during two-wave (mm) | 5.7 ± 0.2 (n = 15) | 6.7 ± 0.2 (n = 7) | 6.2 ± 0.3 (n = 8) |
| On left ovary during two-wave (mm) | 5.8 ± 0.2 (n = 10) | 5.8 ± 0.3 (n = 13) | 6.1 ± 0.3 (n = 6) |
| Average initial diameter DF during two-wave (mm) | 5.7 ± 0.2 (n = 25) | 6.2 ± 0.2 (n = 20) | 6.1 ± 0.2 a (n = 14) |
| On right ovary during three-wave (mm) | - | 5.8 ± 1.1 (n = 2) | 6.1 ± 0.9 (n = 2) |
| On left ovary during three-wave (mm) | - | 6.7 ± 0.7 (n = 2) | 6.2 ± 1.0 (n = 2) |
| Average DF during three-wave (mm) | - | 6.2 ± 1.0 (n = 4) | 6.1 ± 1.0 (n = 4) |
| Maximum diameter of DF | |||
| On right ovary during one-wave (mm) | 8.6 ± 0.3 (n = 12) | - | 9.9 ± 0.8 (n = 6) |
| On left ovary during one-wave (mm) | 9.0 ± 0.4 (n = 13) | - | 10.7 ± 0.5 (n = 8) |
| Average maximum diameter during one-wave (mm) | 8.8 ± 0.2 (n = 25) | - | 10.3 ± 0.5 b (n = 14) |
| On right ovary during two-wave (mm) | 9.6 ± 2.0 (n = 13) | 9.5 ± 0.5 (n = 8) | 10.9 ± 0.5 (n = 6) |
| On left ovary during two-wave (mm) | 9.5 ± 0.5 (n = 12) | 10.6 ± 0.5 (n = 12) | 12.6 ± 0.7 (n = 8) |
| Average maximum diameter of DF during two-wave (mm) | 9.5 ± 0.3 (n = 25) | 10.2 ± 0.4 (n = 20) | 11.7 ± 0.4 a (n = 14) |
| On right ovary during three-wave (mm) | - | 11.2 ± 0.8 (n = 2) | 11.9 ± 0.7 (n = 2) |
| On right ovary during three-wave (mm) | - | 12.4 ± 0.5 (n = 2) | 12.2 ± 0.9 (n = 2) |
| Average maximum diameter of DF during three-wave (mm) | - | 11.8 ± 0.6 (n = 4) | 12.05 ± 0.8 (n = 4) |
| Variables | Puberty | Sexual Maturity | Postpartum |
|---|---|---|---|
| Growth rate of DF | |||
| On right ovary during one-wave (mm/day) | 0.73 ± 0.13 | - | 0.85 ± 0.06 |
| On left ovary during one-wave (mm/day) | 0.66 ± 0.07 | - | 0.92 ± 0.11 |
| Average growth of DF during one-wave (mm/day) | 0.69 ± 0.069 b | - | 0.68 ± 0.09 |
| On right ovary during two-wave (mm/day) | 1.06 ± 0.06 | 0.84 ± 0.06 | 0.89 ± 0.11 |
| On left ovary during two-wave (mm/day) | 1.12 ± 0.04 | 0.98 ± 0.05 | 0.98 ± 0.09 |
| Average growth of DF during two-wave (mm/day) | 1.08 ± 0.04 a | 0.92 ± 0.04 | 0.9 ± 0.07 |
| On right ovary during three-wave (mm/day) | - | 0.83 ± 0.07 | 0.87 ± 0.05 |
| On left ovary during three-wave (mm/day) | - | 0.89 ± 0.03 | 0.95 ± 0.95 |
| Average DF growth during three-wave (mm/day) | - | 0.87 ± 0.05 | 0.79 ± 0.08 |
| Growth duration of DF | |||
| On right ovary during one-wave (day) | 9.05 ± 0.49 | - | 11.67 ± 0.31 |
| On left ovary during one-wave (day) | 9.18 ± 0.87 | - | 11.42 ± 0.45 |
| Average growth duration during one-wave (day) | 9.12 ± 0.47 a | - | 11.54 ± 0.36 |
| On right ovary during two-wave (day) | 10.45 ± 0.38 | 11.67 ± 0.36 | 11.58 ± 0.23 |
| On left ovary during two-wave (day) | 10.71 ± 0.29 | 10.84 ± 0.46 | 11.42 ± 0.36 |
| Average growth duration during two-wave (day) | 10.56 ± 0.25 b | 11.16 ± 0.32 | 11.0 ± 0.23 |
| On right ovary during three-wave (day) | - | 11.63 ± 0.29 | 10.93 ± 0.30 |
| On left ovary during three-wave (day) | - | 11.13 ± 0.41 | 11.15 ± 0.43 |
| Average growth duration in three-wave (day) | - | 11.08 ± 0.28 | 10.89 ± 0.34 |
| Atresia rate of DF | |||
| On right ovary during one-wave (mm/day) | −0.93 ± 0.06 | - | −0.71 ± 0.06 b |
| On left ovary during one-wave (mm/day) | −1.39 ± 0.32 | - | −1.12 ± 0.12 a |
| Average atresia rate in one-wave (mm/day) | −1.12 ± 0.14 | - | −0.96 ± 0.08 |
| On right ovary during second-wave (mm/day) | - | 0.83 ± 0.09 | 0.77 ± 0.07 |
| On left ovary during second-wave (mm/day) | - | 0.96 ± 0.07 | 1.04 ± 0.06 |
| Average atresia rate in second-wave (mm/day) | 0.91 ± 0.08 | 0.93 ± 0.1 | |
| Atresia duration of DF | |||
| On right ovary during one-wave (day) | 9.52 ± 0.24 | - | 12.50 ± 0.15 a |
| On left ovary during one-wave (day) | 9.00 ± 0.35 | - | 10.50 ± 0.26 b |
| Average atresia rate of DF during one-wave (day) | 9.71 ± 0.20 | - | 11.50 ± 0.26 |
| On right ovary during second-wave (day) | - | 10.63 ± 0.26 | 11.56 ± 0.19 |
| On left ovary during second-wave (day) | - | 10.13 ± 0.22 | 10.31 ± 0.24 |
| Average DF atresia duration in second-wave (day) | - | 10.44 ± 0.23 | 10.97 ± 0.20 |
| Factors (Y)Parameters | Follicle Location over Ovary | Physiological State | Follicle Wave | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Left | Right | p-Value | Puberty | Sexually Mature | Postpartum | p-Value | First | Second | Third | p-Value | |
| DF diameter at first detection (mm) * | 5.75 ± 0.19 | 6.54 ± 0.16 | 0.653 | 6.70 ± 0.16 | 6.03 ± 0.24 | 5.70 ± 0.24 | 0.135 | 5.75 ± 0.16 | 6.55 ± 0.17 | 6.21 ± 0.11 | 0.0003 |
| Maximal diameter of DF (mm) | 10.34 ± 0.19 | 9.70 ± 0.20 | 0.023 | 9.16 ± 0.20 | 9.89 ± 0.25 | 11.01 ± 0.27 | 0.0001 | 9.63 ± 00.19 | 10.41 ± 0.19 | 12.17 ± 0.12 | 0.003 |
| Increase in DF diameter (mm) | 4.92 ± 0.27 | 5.71 ± 0.25 | 0.076 | 5.79 ± 0.24 | 4.72 ± 0.35 | 5.43 ± 0.36 | 0.001 | 4.94 ± 0.24 | 5.68 ± 0.25 | 6.65 ± 0.27 | 0.044 |
| Growth rate of DF (mm/day) | 0.729 ± 0.12 | 0.766 ± 0.13 | 0.051 | 0.716 ± 0.15 | 0.732 ± 0.15 | 0.793 ± 0.16 | 0.0009 | 0.666 ± 0.13 | 0.828 ± 0.12 | 0.829 ± 0.11 | 0.0001 |
| Atresia rate of DF (mm/day) | 0.798 ± 0.15 | 0.804 ± 0.13 | 0.225 | 0.714 ± 0.13 | 0.797 ± 0.13 | 0.892 ± 0.28 | 0.0005 | 0.769 ± 0.12 | 0.833 ± 0.16 | 0.885 ± 0.17 | 0.648 |
| Duration of growth (day) | 9.20 ± 0.28 | 9.46 ± 0.29 | 0.519 | 9.30 ± 0.32 | 10.36 ± 0.36 | 11.34 ± 0.38 | 0.884 | 1.70 ± 0.27 | 9.97 ± 0.31 | 15.96 ± 0.28 | 0.001 |
| Duration of atresia (day) | 8.26 ± 0.24 | 9.95 ± 0.26 | 0.280 | 9.67 ± 0.26 | 10.40 ± 0.32 | 11.24 ± 0.36 | 0.168 | 1.61 ± 0.18 | 4.11 ± 0.32 | - | 0.0002 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abulaiti, A.; Riaz, U.; Naseer, Z.; Ahmed, Z.; Hua, G.; Yang, L. Follicular Dynamics during Estrous Cycle of Pubertal, Mature and Postpartum Crossbred (Nili Ravi × Jianghan) Buffaloes. Animals 2022, 12, 1208. https://doi.org/10.3390/ani12091208
Abulaiti A, Riaz U, Naseer Z, Ahmed Z, Hua G, Yang L. Follicular Dynamics during Estrous Cycle of Pubertal, Mature and Postpartum Crossbred (Nili Ravi × Jianghan) Buffaloes. Animals. 2022; 12(9):1208. https://doi.org/10.3390/ani12091208
Chicago/Turabian StyleAbulaiti, Adili, Umair Riaz, Zahid Naseer, Zulfiqar Ahmed, Guohua Hua, and Liguo Yang. 2022. "Follicular Dynamics during Estrous Cycle of Pubertal, Mature and Postpartum Crossbred (Nili Ravi × Jianghan) Buffaloes" Animals 12, no. 9: 1208. https://doi.org/10.3390/ani12091208







