Biogenic Amine Content Analysis of Three Chicken-Based Dry Pet Food Formulations
Abstract
:Simple Summary
Abstract
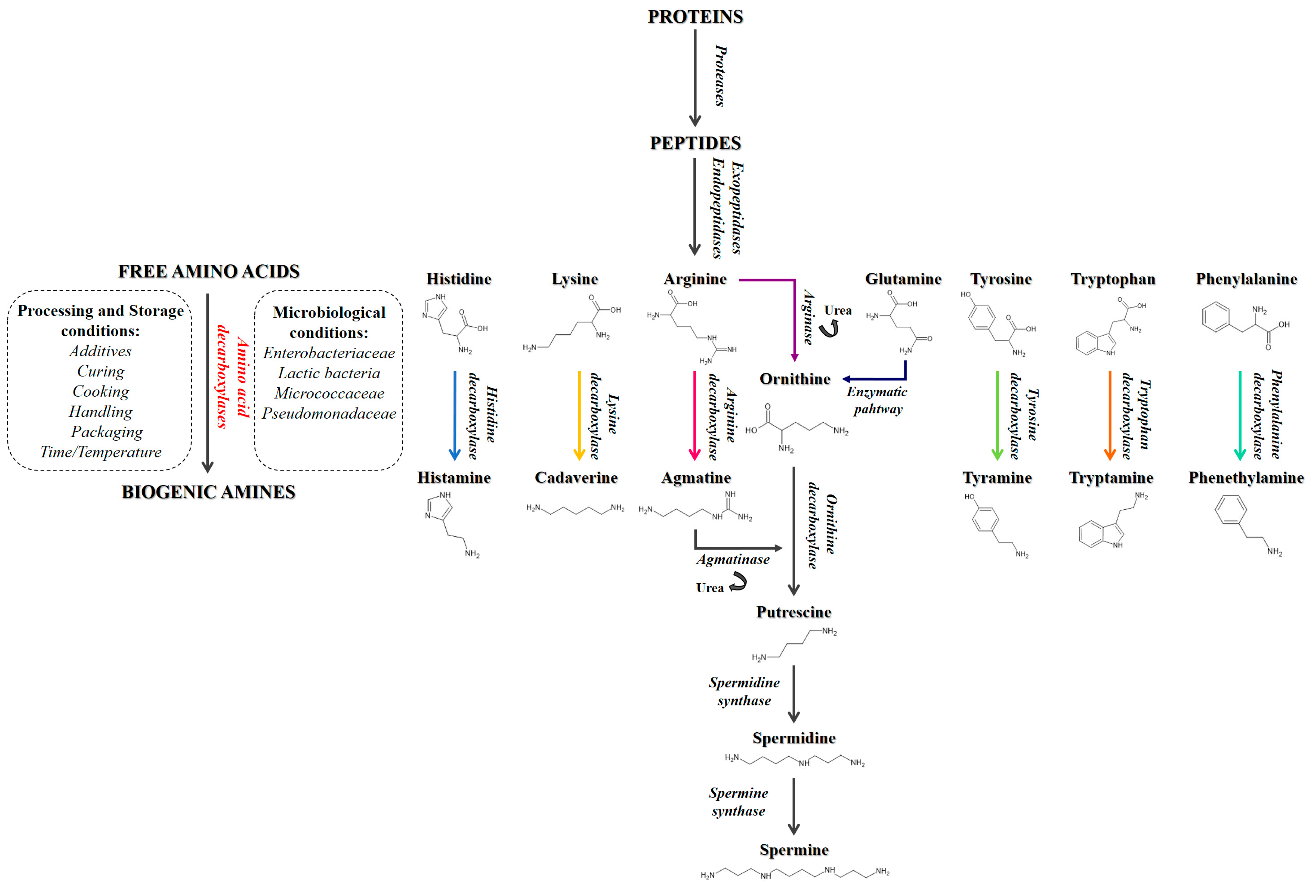
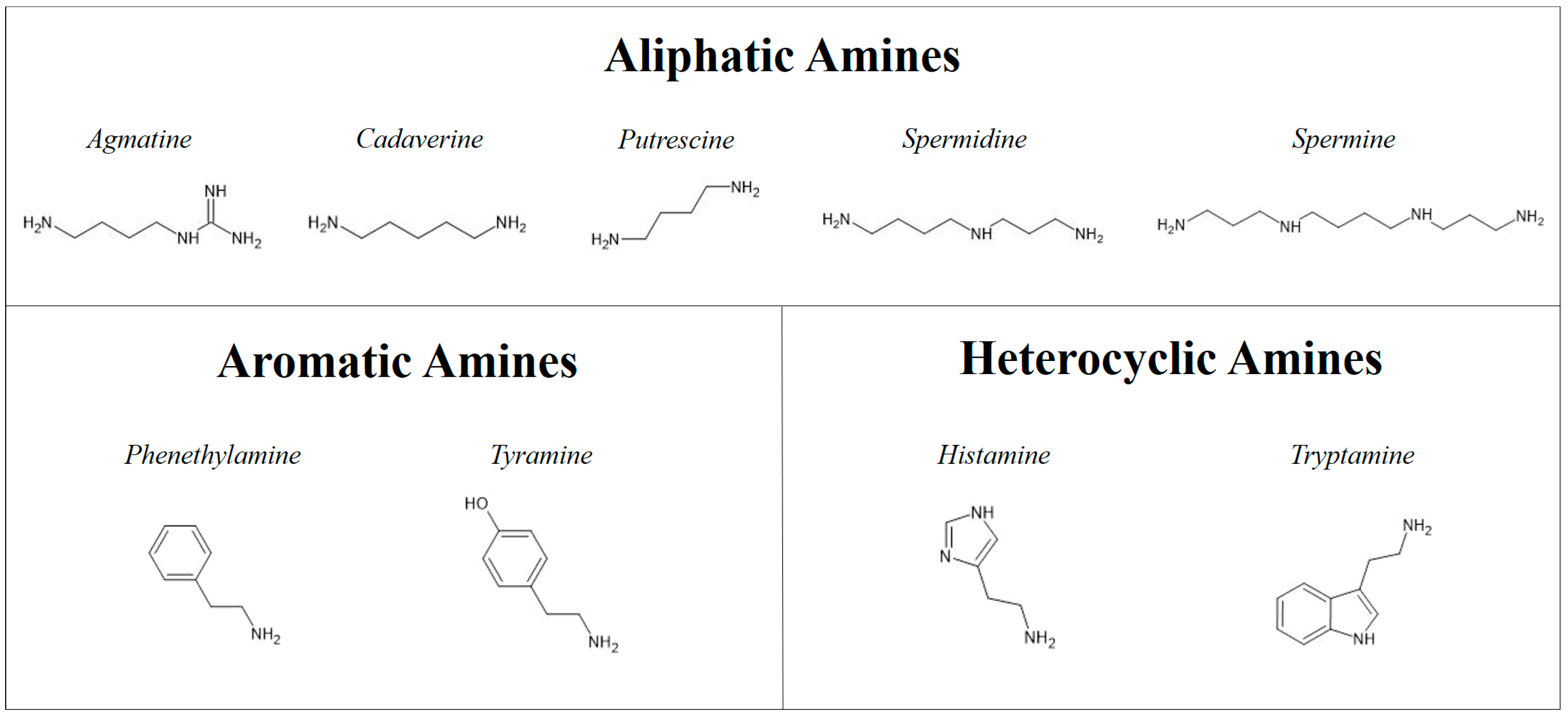
1. Introduction
2. Materials and Methods
2.1. Dry Pet Foods
2.2. Sample Preparation
2.3. Determination of Biogenic Amines and Free Amino Acids
2.4. Microbiological Analysis
2.5. Statistical Analysis
3. Results
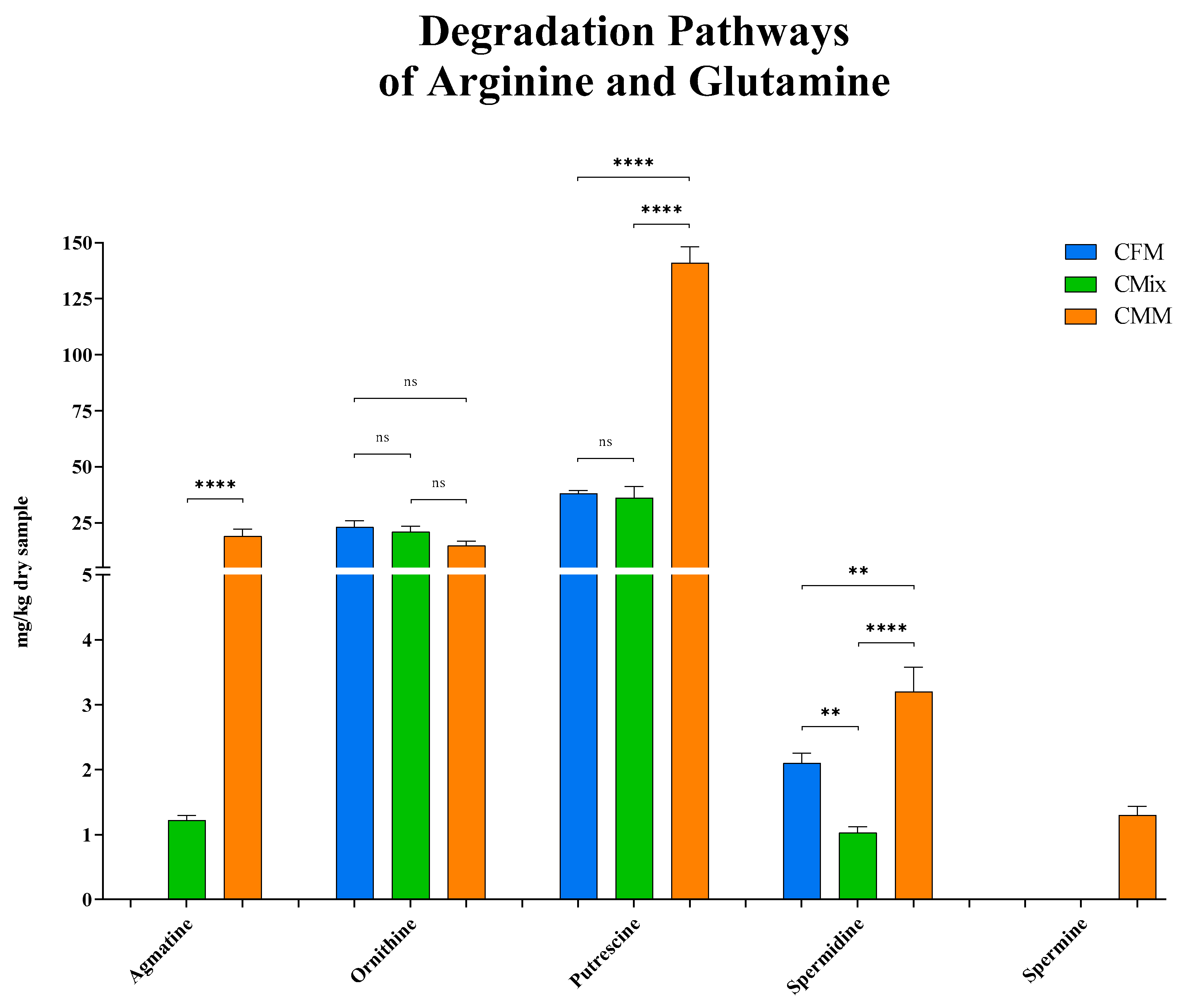
3.1. Biogenic Amines
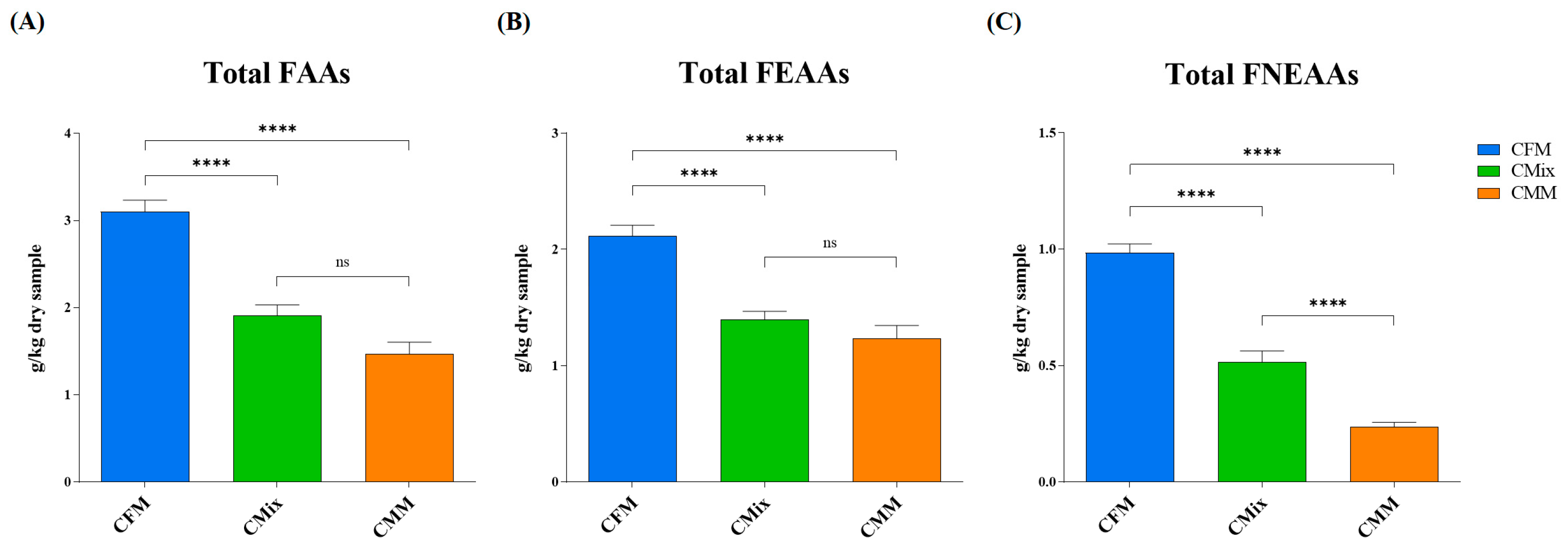
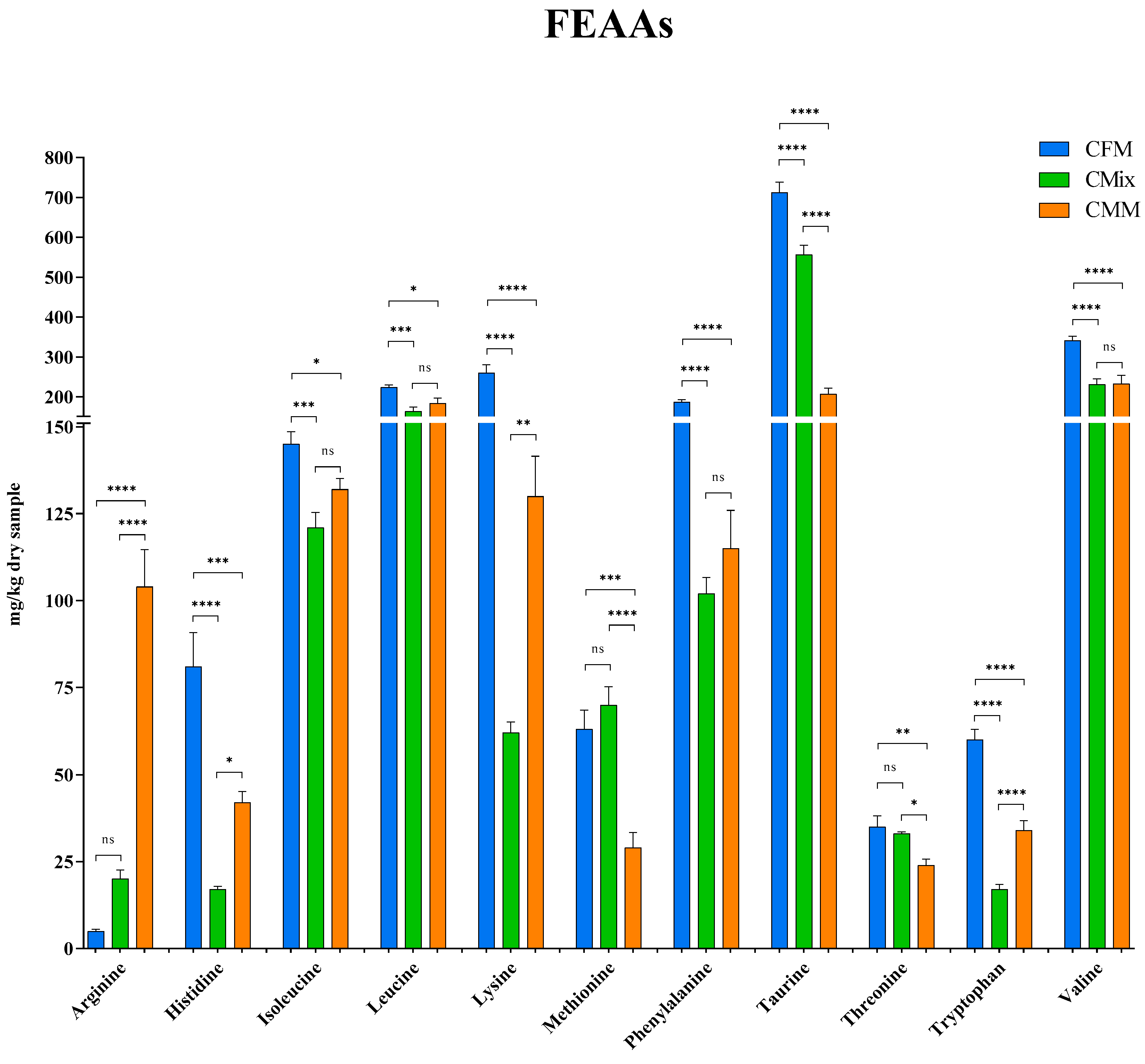
3.2. Free Amino Acids
3.3. Microbiological Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Gibson, M.W.; Sajid, A. Pet Food Processing: Understanding Transformations in Starch during Extrusion and Baking. Cereal Foods World 2013, 58, 232–236. [Google Scholar] [CrossRef] [Green Version]
- Aldrich, G. Rendered Products in Pet Food. Essent. Render. 2006, 159–178. [Google Scholar]
- Zicker, S.C. Evaluating Pet Foods: How Confident Are You When You Recommend a Commercial Pet Food? Top. Companion Anim. Med. 2008, 23, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Montegiove, N.; Calzoni, E.; Cesaretti, A.; Pellegrino, R.M.; Emiliani, C.; Pellegrino, A.; Leonardi, L. The Hard Choice about Dry Pet Food: Comparison of Protein and Lipid Nutritional Qualities and Digestibility of Three Different Chicken-Based Formulations. Animals 2022, 12, 1538. [Google Scholar] [CrossRef]
- Di Donfrancesco, B.; Koppel, K.; Swaney-Stueve, M.; Chambers, E. Consumer Acceptance of Dry Dog Food Variations. Animals 2014, 4, 313–330. [Google Scholar] [CrossRef] [PubMed]
- Morelli, G.; Stefanutti, D.; Ricci, R. A Survey among Dog and Cat Owners on Pet Food Storage and Preservation in the Households. Animals 2021, 11, 273. [Google Scholar] [CrossRef]
- Rombach, M.; Dean, D.L. It Keeps the Good Boy Healthy from Nose to Tail: Understanding Pet Food Attribute Preferences of US Consumers. Animals 2021, 11, 3301. [Google Scholar] [CrossRef]
- Learey, J.J.; Crawford-Clark, S.; Bowen, B.J.; Barrow, C.J.; Adcock, J.L. Detection of Biogenic Amines in Pet Food Ingredients by RP-HPLC with Automated Dansyl Chloride Derivatization. J. Sep. Sci. 2018, 41, 4430–4436. [Google Scholar] [CrossRef]
- Kovacova-Hanuskova, E.; Buday, T.; Gavliakova, S.; Plevkova, J. Histamine, Histamine Intoxication and Intolerance. Allergol. Immunopathol. 2015, 43, 498–506. [Google Scholar] [CrossRef]
- del Rio, B.; Redruello, B.; Linares, D.M.; Ladero, V.; Fernandez, M.; Martin, M.C.; Ruas-Madiedo, P.; Alvarez, M.A. The Dietary Biogenic Amines Tyramine and Histamine Show Synergistic Toxicity towards Intestinal Cells in Culture. Food Chem. 2017, 218, 249–255. [Google Scholar] [CrossRef]
- Montegiove, N.; Calzoni, E.; Cesaretti, A.; Alabed, H.; Pellegrino, R.M.; Emiliani, C.; Pellegrino, A.; Leonardi, L. Biogenic Amine Analysis in Fresh Meats and Meat Meals Used as Raw Materials for Dry Pet Food Production. Sci. Bull. Ser. F Biotechnol. 2020, 24, 33–42. [Google Scholar]
- Linares, D.M.; del Rio, B.; Redruello, B.; Ladero, V.; Martin, M.C.; Fernandez, M.; Ruas-Madiedo, P.; Alvarez, M.A. Comparative Analysis of the in Vitro Cytotoxicity of the Dietary Biogenic Amines Tyramine and Histamine. Food Chem. 2016, 197, 658–663. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Del Rio, B.; Redruello, B.; Linares, D.M.; Ladero, V.; Ruas-Madiedo, P.; Fernandez, M.; Martin, M.C.; Alvarez, M.A. The Biogenic Amines Putrescine and Cadaverine Show in Vitro Cytotoxicity at Concentrations That Can Be Found in Foods. Sci. Rep. 2019, 9, 120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzzi, G.; Torriani, S. Editorial: Biogenic Amines in Foods. Front. Microbiol. 2015, 6, 472. [Google Scholar] [CrossRef] [Green Version]
- Tabanelli, G. Biogenic Amines and Food Quality: Emerging Challenges and Public Health Concerns. Foods 2020, 9, 859. [Google Scholar] [CrossRef]
- Schirone, M.; Esposito, L.; D’Onofrio, F.; Visciano, P.; Martuscelli, M.; Mastrocola, D.; Paparella, A. Biogenic Amines in Meat and Meat Products: A Review of the Science and Future Perspectives. Foods 2022, 11, 788. [Google Scholar] [CrossRef]
- Martuscelli, M.; Esposito, L.; Mastrocola, D. Biogenic Amines’ Content in Safe and Quality Food. Foods 2021, 10, 100. [Google Scholar] [CrossRef]
- Vasconcelos, H.; Coelho, L.C.C.; Matias, A.; Saraiva, C.; Jorge, P.A.S.; de Almeida, J.M.M.M. Biosensors for Biogenic Amines: A Review. Biosensors 2021, 11, 82. [Google Scholar] [CrossRef]
- Vasconcelos, H.C.A.S.G.; Marques Martins de Almeida, J.M.; Mendes, J.P.; Dias, B.; Jorge, P.A.d.S.; Saraiva, C.M.T.; Coelho, L.C.C. Optical Biosensor for the Detection of Biogenic Amines. IEEE Sens. J. 2022, 22, 20484–20491. [Google Scholar] [CrossRef]
- Vasconcelos, H.; de Almeida, J.M.M.M.; Matias, A.; Saraiva, C.; Jorge, P.A.S.; Coelho, L.C.C. Detection of Biogenic Amines in Several Foods with Different Sample Treatments: An Overview. Trends Food Sci. Technol. 2021, 113, 86–96. [Google Scholar] [CrossRef]
- Altafini, A.; Roncada, P.; Sonfack, G.M.; Guerrini, A.; Romeo, G.A.; Fedrizzi, G.; Caprai, E. Occurrence of Histamine in Commercial Cat Foods under Different Storage Conditions. Vet. Sci. 2022, 9, 270. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Biological Hazards (BIOHAZ). Scientific Opinion on Risk Based Control of Biogenic Amine Formation in Fermented Foods. EFSA J. 2011, 9, 2393. [Google Scholar] [CrossRef] [Green Version]
- Li, B.; Lu, S. The Importance of Amine-Degrading Enzymes on the Biogenic Amine Degradation in Fermented Foods: A Review. Process. Biochem. 2020, 99, 331–339. [Google Scholar] [CrossRef]
- Erdag, D.; Merhan, O.; Yildiz, B. Biochemical and Pharmacological Properties of Biogenic Amines. In Biogenic Amines; IntechOpen: London, UK, 2018; ISBN 978-1-78984-134-3. [Google Scholar]
- del Rio, B.; Redruello, B.; Fernandez, M.; Martin, M.C.; Ladero, V.; Alvarez, M.A. The Biogenic Amine Tryptamine, Unlike β-Phenylethylamine, Shows in Vitro Cytotoxicity at Concentrations That Have Been Found in Foods. Food Chem. 2020, 331, 127303. [Google Scholar] [CrossRef]
- Berry, M.D. Mammalian Central Nervous System Trace Amines. Pharmacologic Amphetamines, Physiologic Neuromodulators. J. Neurochem. 2004, 90, 257–271. [Google Scholar] [CrossRef]
- Raasch, W.; Schäfer, U.; Chun, J.; Dominiak, P. Biological Significance of Agmatine, an Endogenous Ligand at Imidazoline Binding Sites. Br. J. Pharm. 2001, 133, 755–780. [Google Scholar] [CrossRef] [Green Version]
- Sezer, A.; Güçlü, B.; Kazancı, B.; Çakır, M.; Çoban, M.K. Neuroprotective Effects of Agmatine In Experimental Peripheral Nerve Injury In Rats: A Prospective Randomized and Placebo-Controlled Trial. In Sıçanlarda Deneysel Periferik Sinir Yaralanmasında Agmatinin Nöroprotektif Etkileri: Prospektif, Randomize, Plasebo Kontrollü Çalışma; Turkish Neurological Society: Ankara, Turkey, 2014; Volume 24, pp. 196–201. [Google Scholar]
- Liu, G.; Mei, H.; Chen, M.; Qin, S.; Li, K.; Zhang, W.; Chen, T. Protective Effect of Agmatine against Hyperoxia-Induced Acute Lung Injury via Regulating LncRNA Gadd7. Biochem. Biophys. Res. Commun. 2019, 516, 68–74. [Google Scholar] [CrossRef]
- Li, X.; Liu, Z.; Jin, H.; Fan, X.; Yang, X.; Tang, W.; Yan, J.; Liang, H. Agmatine Protects against Zymosan-Induced Acute Lung Injury in Mice by Inhibiting NF-κB-Mediated Inflammatory Response. BioMed Res. Int. 2014, 2014, e583736. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.F.; Gong, Z.H.; Cao, J.B.; Wang, H.L.; Luo, Z.P.; Li, J. Antidepressant-like Effect of Agmatine and Its Possible Mechanism. Eur. J. Pharmacol. 2003, 469, 81–88. [Google Scholar] [CrossRef]
- Galgano, F.; Caruso, M.; Condelli, N.; Favati, F. Focused Review: Agmatine in Fermented Foods. Front. Microbiol. 2012, 3, 199. [Google Scholar] [CrossRef] [Green Version]
- Fan, P.; Song, P.; Li, L.; Huang, C.; Chen, J.; Yang, W.; Qiao, S.; Wu, G.; Zhang, G.; Ma, X. Roles of Biogenic Amines in Intestinal Signaling. Curr. Protein Pept. Sci. 2017, 18, 532–540. [Google Scholar] [CrossRef]
- Ladero, V.; Calles-Enriquez, M.; Fernandez, M.; Alvarez, M.A. Toxicological Effects of Dietary Biogenic Amines. Curr. Nutr. Food Sci. 2010, 6, 145–156. [Google Scholar] [CrossRef]
- Montegiove, N.; Pellegrino, R.M.; Emiliani, C.; Pellegrino, A.; Leonardi, L. An Alternative Approach to Evaluate the Quality of Protein-Based Raw Materials for Dry Pet Food. Animals 2021, 11, 458. [Google Scholar] [CrossRef] [PubMed]
- Montegiove, N.; Calzoni, E.; Cesaretti, A.; Alabed, H.; Pellegrino, R.M.; Emiliani, C.; Pellegrino, A.; Leonardi, L. Comprehensive Evaluation of Lipidic Content in Dry Pet Food Raw Materials: Comparison between Fresh Meats and Meat Meals. Sci. Bull. Ser. F Biotechnol. 2020, 24, 122–129. [Google Scholar]
- Thompson, A. Ingredients: Where Pet Food Starts. Top. Companion Anim. Med. 2008, 23, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Montegiove, N.; Calzoni, E.; Cesaretti, A.; Pellegrino, R.M.; Emiliani, C.; Pellegrino, A.; Leonardi, L. Soluble Protein Content Assessment in Dry Pet Food Raw Materials: Comparison between Fresh Meat and Meat Meal Formulations. Sci. Bull. Ser. F Biotechnol. 2021, 25, 54–60. [Google Scholar]
- FEDIAF. Guide to Good Practice for the Manufacture of Safe Pet Foods; FEDIAF: Brussels, Belgium, 2018. [Google Scholar]
- Leiva, A.; Molina, A.; Redondo-Solano, M.; Artavia, G.; Rojas-Bogantes, L.; Granados-Chinchilla, F. Pet Food Quality Assurance and Safety and Quality Assurance Survey within the Costa Rican Pet Food Industry. Animals 2019, 9, 980. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kępińska-Pacelik, J.; Biel, W. Microbiological Hazards in Dry Dog Chews and Feeds. Animals 2021, 11, 631. [Google Scholar] [CrossRef]
- Girio, T.M.S.; Filho, A.N.; Junior, O.D.R.; Amaral, L.A.; Girio, R.J.S. Microbiological Quality of Dog Feed Sold in Sealed Packages and in Bulk. Ars Vet. 2012, 28, 036–040. [Google Scholar]
- Kazimierska, K.; Biel, W.; Witkowicz, R.; Karakulska, J.; Stachurska, X. Evaluation of Nutritional Value and Microbiological Safety in Commercial Dog Food. Vet. Res. Commun. 2021, 45, 111–128. [Google Scholar] [CrossRef]
- ISO 7218:2007; Microbiology of Food and Animal Feeding Stuffs—General Requirements and Guidance for Microbiological Examinations. ISO: Geneva, Switzerland, 2007. Available online: https://www.iso.org/standard/36534.html (accessed on 8 March 2023).
- Ruiz-Capillas, C.; Jiménez-Colmenero, F. Biogenic Amines in Meat and Meat Products. Crit. Rev. Food Sci. Nutr. 2005, 44, 489–599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- White, M.V. The Role of Histamine in Allergic Diseases. J. Allergy Clin. Immunol. 1990, 86, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, A. Role of Histamine in Allergic Conjunctivitis. Acta Ophthalmol. Scand. 2000, 78, 18–21. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.L.; Eitenmiller, R.R. Histamine Food Poisoning: Toxicology and Clinical Aspects. CRC Crit. Rev. Toxicol. 1986, 17, 91–128. [Google Scholar] [CrossRef] [PubMed]
- Maintz, L.; Novak, N. Histamine and Histamine Intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altafini, A.; Roncada, P.; Guerrini, A.; Sonfack, G.M.; Accurso, D.; Caprai, E. Development of Histamine in Fresh and Canned Tuna Steaks Stored under Different Experimental Temperature Conditions. Foods 2022, 11, 4034. [Google Scholar] [CrossRef]
- Visciano, P.; Schirone, M.; Paparella, A. An Overview of Histamine and Other Biogenic Amines in Fish and Fish Products. Foods 2020, 9, 1795. [Google Scholar] [CrossRef]
- ten Brink, B.; Damink, C.; Joosten, H.M.; Huis in ’t Veld, J.H. Occurrence and Formation of Biologically Active Amines in Foods. Int. J. Food Microbiol. 1990, 11, 73–84. [Google Scholar] [CrossRef]
- Papavergou, E.J.; Savvaidis, I.N.; Ambrosiadis, I.A. Levels of Biogenic Amines in Retail Market Fermented Meat Products. Food Chem. 2012, 135, 2750–2755. [Google Scholar] [CrossRef]
- Feddern, V.; Mazzuco, H.; Fonseca, F.N.; Lima, G.J.M.M. de A Review on Biogenic Amines in Food and Feed: Toxicological Aspects, Impact on Health and Control Measures. Anim. Prod. Sci. 2019, 59, 608–618. [Google Scholar] [CrossRef]
- Wójcik, W.; Łukasiewicz-Mierzejewska, M.; Damaziak, K.; Bień, D. Biogenic Amines in Poultry Meat and Poultry Products: Formation, Appearance, and Methods of Reduction. Animals 2022, 12, 1577. [Google Scholar] [CrossRef] [PubMed]
- Danchuk, A.I.; Komova, N.S.; Mobarez, S.N.; Doronin, S.Y.; Burmistrova, N.A.; Markin, A.V.; Duerkop, A. Optical Sensors for Determination of Biogenic Amines in Food. Anal. Bioanal. Chem. 2020, 412, 4023–4036. [Google Scholar] [CrossRef] [PubMed]
- Geornaras, I.; Dykes, G.A.; von Holy, A. Biogenic Amine Formation by Poultry-Associated Spoilage and Pathogenic Bacteria. Lett. Appl. Microbiol. 1995, 21, 164–166. [Google Scholar] [CrossRef]
- Til, H.P.; Falke, H.E.; Prinsen, M.K.; Willems, M.I. Acute and Subacute Toxicity of Tyramine, Spermidine, Spermine, Putrescine and Cadaverine in Rats. Food Chem. Toxicol. 1997, 35, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.A. Lewis’ Dictionary of Toxicology; CRC Press: Boca Raton, FL, USA, 1998; ISBN 978-1-56670-223-2. [Google Scholar]
- Kukier, E.; Goldsztejn, M.; Grenda, T.; Kwiatek, K.; Wasyl, D.; Hoszowski, A. Microbiological Quality of Compound Feed Used in Poland. J. Vet. Res. 2012, 56, 349–354. [Google Scholar] [CrossRef] [Green Version]
- Tittarelli, R.; Mannocchi, G.; Pantano, F.; Saverio Romolo, F. Recreational Use, Analysis and Toxicity of Tryptamines. Curr. Neuropharmacol. 2015, 13, 26–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mousseau, D.D. Tryptamine: A Metabolite of Tryptophan Implicated in Various Neuropsychiatric Disorders. Metab. Brain Dis. 1993, 8, 1–44. [Google Scholar] [CrossRef]
- Mossoba, M.E.; Vohra, S.N.; Wiesenfeld, P.L.; Sprando, R.L. Nephrotoxicity of Combining 2-Phenethylamine and N, N-Dimethyl-β-Phenethylamine. Appl. Vitr. Toxicol. 2016, 2, 49–55. [Google Scholar] [CrossRef]
- Wu, G.; Haynes, T.E.; Li, H.; Meininger, C.J. Glutamine Metabolism in Endothelial Cells: Ornithine Synthesis from Glutamine via Pyrroline-5-Carboxylate Synthase. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2000, 126, 115–123. [Google Scholar] [CrossRef]
- de Vera, N.; Serratosa, J.; Artigas, F.; Martínez, E. Toxic Effects of Putrescine in Rat Brain: Polyamines Can Be Involved in the Action of Excitotoxins. Amino Acids 1992, 3, 261–269. [Google Scholar] [CrossRef]
- Wunderlichová, L.; Buňková, L.; Koutný, M.; Jančová, P.; Buňka, F. Formation, Degradation, and Detoxification of Putrescine by Foodborne Bacteria: A Review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 1012–1030. [Google Scholar] [CrossRef]
- Halaris, A.; Plietz, J. Agmatine: Metabolic pathway and spectrum of activity in brain. CNS Drugs 2007, 21, 885–900. [Google Scholar] [CrossRef] [PubMed]
- Bardócz, S. Polyamines in Food and Their Consequences for Food Quality and Human Health. Trends Food Sci. Technol. 1995, 6, 341–346. [Google Scholar] [CrossRef]
- Slemr, J.; Beyermann, K. Concentration Profiles of Diamines in Fresh and Aerobically Stored Pork and Beef. J. Agric. Food Chem. 1985, 33, 336–339. [Google Scholar] [CrossRef]
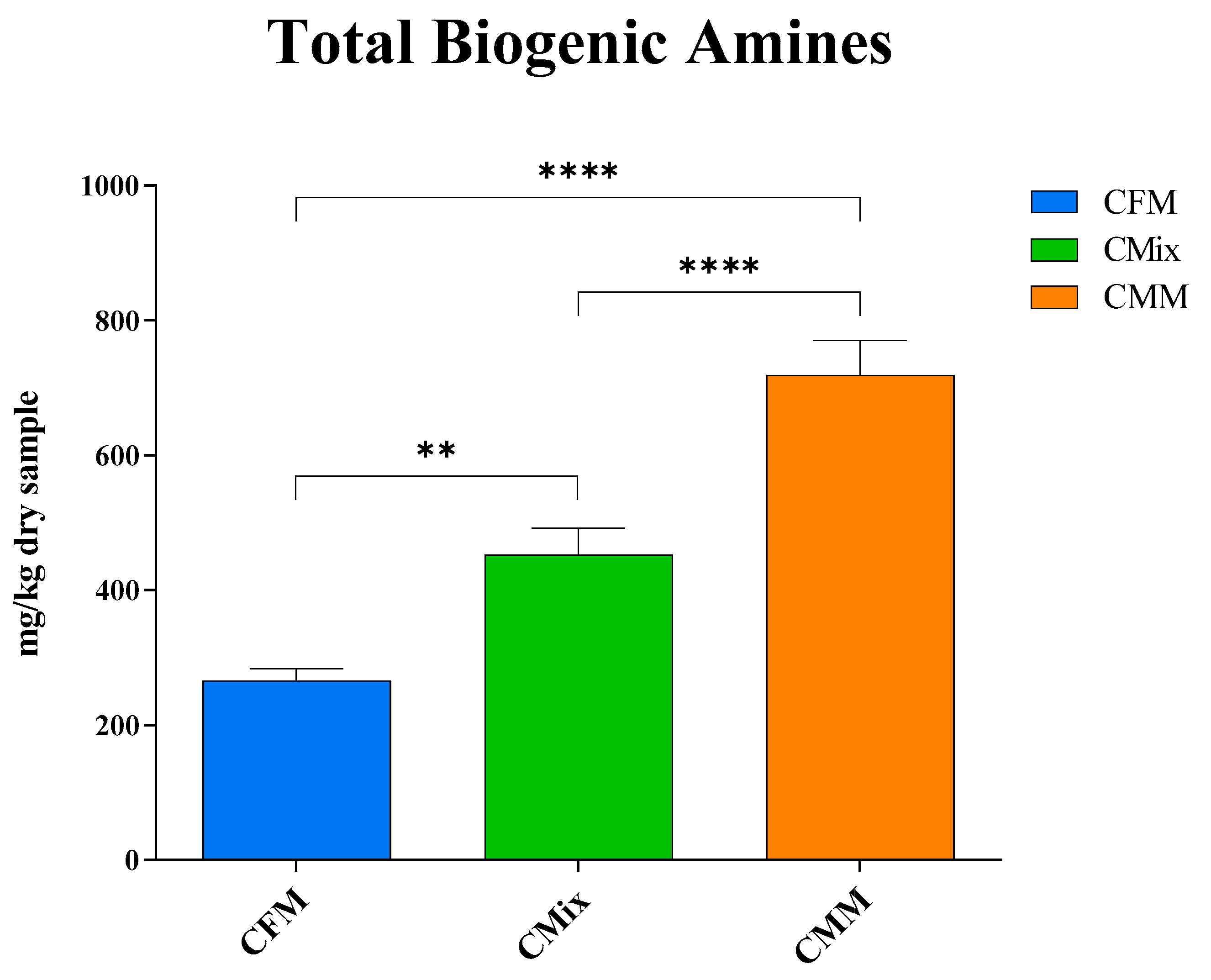


| Dry Pet Food | TAMBC (CFU/g of Dry Sample) |
|---|---|
| CFM | 900 ± 40 |
| CMix | 1500 ± 70 |
| CMM | 2900 ± 160 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Montegiove, N.; Leonardi, L.; Cesaretti, A.; Pellegrino, R.M.; Pellegrino, A.; Emiliani, C.; Calzoni, E. Biogenic Amine Content Analysis of Three Chicken-Based Dry Pet Food Formulations. Animals 2023, 13, 1945. https://doi.org/10.3390/ani13121945
Montegiove N, Leonardi L, Cesaretti A, Pellegrino RM, Pellegrino A, Emiliani C, Calzoni E. Biogenic Amine Content Analysis of Three Chicken-Based Dry Pet Food Formulations. Animals. 2023; 13(12):1945. https://doi.org/10.3390/ani13121945
Chicago/Turabian StyleMontegiove, Nicolò, Leonardo Leonardi, Alessio Cesaretti, Roberto Maria Pellegrino, Alessia Pellegrino, Carla Emiliani, and Eleonora Calzoni. 2023. "Biogenic Amine Content Analysis of Three Chicken-Based Dry Pet Food Formulations" Animals 13, no. 12: 1945. https://doi.org/10.3390/ani13121945
APA StyleMontegiove, N., Leonardi, L., Cesaretti, A., Pellegrino, R. M., Pellegrino, A., Emiliani, C., & Calzoni, E. (2023). Biogenic Amine Content Analysis of Three Chicken-Based Dry Pet Food Formulations. Animals, 13(12), 1945. https://doi.org/10.3390/ani13121945









