1. Introduction
World aquaculture currently provides most of the world’s food fish consumption, demonstrating its crucial role in global food security [
1]. However, fish production, especially intensive fish farming, depends heavily on the supply of artificial feed [
2]. Furthermore, marine fish requires more and a higher quality of feed protein than terrestrial animals do. Processed products of marine catches such as fish meal (FM) are the preferred animal protein source in the feed formulations of marine fish due to their high nutritional value [
2]. Given the global constrained supply of FM, reducing the dependence of aquafeeds on FM is widely accepted as an important strategy for ensuring the developments of aquaculture in the future [
3].
One of the major sources of alternatives to FM is soybean by-products, and they have been widely used in aquafeeds as the main protein for most farmed fish because soybean has a higher protein quality, steady supply, and low price compared with other vegetable proteins [
4]. It has been reported that the use of a high-soybean diet can cause poor growth performance [
5,
6,
7], induce oxidative stress [
8,
9], increase histological damage in the intestine [
10,
11], lead to microbiota dysbiosis [
7,
8,
9] and induce intestinal inflammation [
12,
13,
14]. However, the occurrence of the negative effects of SBM is caused by the existence of anti-nutritional factors (ANFs) [
15,
16]. Soy antigen proteins (SAPs), a form of globulin, are one of the main ANFs in soybean by-products [
17,
18]. This protein has high thermal stability [
19] and strong immunogenicity [
20], and it is difficult to eliminate by conventional treatment methods [
21]. This kind of protein is believed to be the cause of enteritis of both terrestrial animals and fish [
21]. SAPs can be subdivided into four subtypes, including glycinin, α-conglycinin, β-conglycinin and γ-conglycinin, by the sedimentation rates of the proteins, of which glycinin and β-conglycinin account for about 40% and 31% of total soy globulin, respectively. Therefore, the majority of the research focuses on the sensitization of the two types of SAPs in animals and their underlying mechanisms. Recent results showed that SAPs exhibit high antigenic activity [
22,
23,
24,
25,
26], induce specific antigen–antibody reactions and delay hypersensitivity mediated by T lymphoid cells—by stimulating the immune system [
15,
22,
27,
28]; trigger reactive oxygen, leading to oxidative stress in intestinal epithelial cells [
5,
27,
29,
30] and intestinal epithelial cell apoptosis and damage [
15,
28,
31,
32]; and develop into symptoms such as enteritis and allergic reactions [
22,
24,
26,
33], eventually leading to poor growth [
34]. In terms of oxidative stress, many factors in diets, such as polypropylene microplastics [
35] and nitrite [
36], can cause oxidative stress in fish and injure tissues and organs. Fish, especially farmed marine fish, have lower tolerance to SAPs than do terrestrial animals, which is evidenced by a number of experimental results in FM replacement by an excessive dietary addition of soybean by-products [
4,
34]. Although SAPs have a negative impact on fish growth and health, the underlying exact mechanism involving enteritis and allergy induced by SAPs is still unclear. In the future, more relevant research work needs to be carried out in order to find better feeding strategies and feed formulations to ultimately improve the health and growth of fish.
Grouper aquaculture has developed into a large-scale aquaculture industry along the coast of Southeast Asia [
37], with the largest aquaculture production scale in China. China’s output of farming grouper reached 204,119 tons in 2021 [
38], ranking it as the third-largest mariculture fish in China. The grouper is highly favored by Chinese people, not only because of their traditional consumption habits, but also because of its rapid growth and high-quality flesh. Similar to other carnivorous fish, grouper is also highly sensitive to soybean by-products [
4,
10,
39]. In these studies, feeding high soybean by-products caused the decline of both the feed intake and the growth of the grouper, accompanied by obvious symptoms of intestinal mucosal structure damage. Recently, the effects of dietary glycinin and β-conglycinin on the immunity and microbiota in the intestine of hybrid grouper (
Epinephelus fuscoguttatus ♀ ×
Epinephelus lanceolatus ♂) were investigated [
40]. However, the effects of SAPs on apoptosis and permeability in the intestine of grouper have not been investigated yet. In this study, we sought to investigate the effects of dietary glycinin levels on the growth, intestinal barrier function, intestinal cell apoptosis and intestinal microbiotia in the intestine of orange-spotted grouper (
Epinephelus coioides). With these results, the relationship between intestinal health and SAPs in the fish species is better understood and its mechanism of action is clarified, which provides a clue to reasonably develop and utilize soybean by-products for grouper feed. This study provides a new basis for the prevention of SAP-induced physiological responses in this fish species.
4. Discussion
Dietary high-SBM inclusion resulted in poor growth performance of fish in the present study and in previous studies [
4,
12,
39,
49,
50]. In this study, two inclusion levels (4.5% and 10%) of glycinin identical to the SBM substitution for 30% and 60% FM protein, respectively, were used to investigate whether dietary glycinin addition affects the growth performance of orange-spotted grouper. Unexpectedly, 10% glycinin addition achieved better growth rate and feed utilization than did the SBM diet and had a growth rate and feed utilization similar to that of the control, although its growth rate and feed utilization were inferior to that of 4.5% glycinin addition. Similarly, the reduced growth rate and feed utilization were observed in grass carp (
Ctenopharyngodon idella) [
51], hybrid yellow catfish (
Pelteobagrus fulvidraco♀ ×
Pelteobaggrus vachelli♂) [
34] and hybrid grouper [
25] when they were fed the diets containing 6–8% glycinin, and the opposite results occurred for those fed the diets containing 2–4% glycinin, as compared with FM diets. These findings indicate that dietary high-glycinin content addition does not constrain growth, and the intermediate-glycinin content addition exhibits superior growth performance to that of the control diet. Our current study also showed that both the 4.5% and the 10% glycinin diets achieved feed intake comparable to the control. However, the hepatosomatic index of fish fed the 4.5% and 10% glycinin diets was lower than that of the control. Furthermore, the lower nutritional status was found to be generally parallel to the higher content of whole-body moisture and lower whole-body contents of crude lipid and ash of fish receiving the SBM diet and that of the 10% glycinin diet when comparing the control. Reduced whole-body ash and lipid contents in fish fed high-SBM diets were also observed as compared with those of FM diets [
4,
6,
12]. Similarly, increased whole-body moisture content [
52,
53] and lowered whole-body lipid and/or ash contents [
52] of fish fed diets containing diets with high glycinin were observed as compared to those of the control. These results indicate that fish fed high-glycinin diets may be suffering from a certain degree of malnutrition, similar to those fish receiving high-SBM diets.
SBM diets showed lower liver values for T-AOC, SOD and GSH-Px, and higher MDA contents in comparison with the FM diet in this study. These reflected high-level hepatic lipid peroxidation caused by high SBM, and indirectly reflected a certain degree of oxidative damage in the liver. The enhanced effect of liver lipid peroxidation in terms of T-AOC value and GSH-Px activity also occurred in fish fed 10% glycinin diets, but not in fish fed 4.5% glycinin diets, which indicates the reduced stress effects of 4.5% glycinin diets compared to 10% glycinin diets. Similarly, 8–12% glycinin diets reduced the T-AOC value and the activities of SOD and/or CAT, and increased MDA contents in the intestine of grass carp [
30], Jian Carp (
Cyprinus carpiovar Jian) [
21] and golden crucian carp (
Cyprinus carpio ×
Carassius auratus) [
52]. Therefore, dietary high-glycinin contents could enhance the hepatic and intestinal lipid peroxidation of fish.
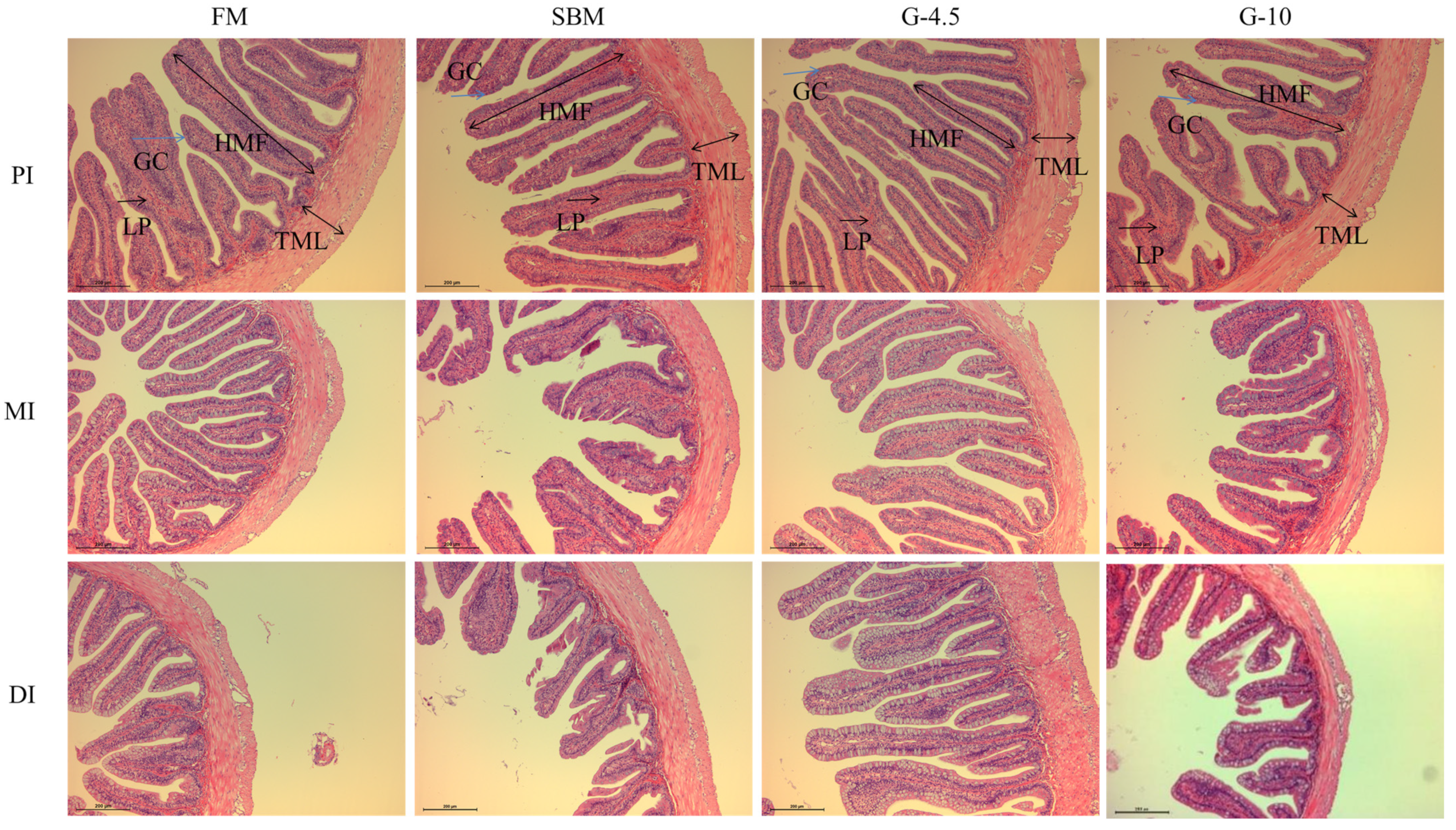
It is well known that the intestine is the place for fish to digest and absorb nutrients [
7]. The intestinal histomorphological integrity is the prerequisite for keeping a good digestion and absorption state, and intestinal disease resistance is a crucial barrier against exogenous pathogens in the immune defense of fish [
46]. The height and length of intestinal plica and the TML are important indicators for measuring the intestinal digestion and absorption function. In previous studies, 8% glycinin diets reduced the plica heights of PI and MI of Jian carp [
21], and the TML and HMF of DI of
Rhynchocypris lagowskii [
53] vs. FM diets, which was supported by our current results and previous results that 10% glycinin diets reduced the TML and HMF of DI of hybrid grouper [
40] and hybrid yellow catfish [
34]. The findings showed the MI and DI were more susceptible to glycinin than was the PI. However, the HMF and TML were greater in fish fed 2% glycinin diets than in fish fed 8% glycinin diets [
25]. The above observations showed that dietary high-glycinin content inclusion could potentially cause adverse alterations in the intestinal morphology of fish, thereby affecting the absorption of nutrients by the intestine.
Studies have shown that dietary high-SBM can also damage the intestinal structural integrity of fish [
12,
54], leading to the occurrence of SBM-induced enteritis, and that the severity of enteritis correlates in a dose-dependent manner with increasing SBM levels. In this study, the effects of dietary glycinin on the intestinal structural integrity were determined through the tight junction function in the intestine of grouper at the molecular level. The down-regulated expression levels of
claudin-3,
occludin and/or
ZO-1 genes were observed in the SBM and 10% glycinin diets in our current study, and in the 8% glycinin diets [
21,
26] and high-SBM diets [
13,
14] in previous studies in comparison with the FM diets, indicating that dietary high-glycinin contents could damage the intestinal intercellular structure by disrupting the intestinal tight junction structure of fish, including grouper [
26].
As an important barrier against external bacterial invasion, the intestine plays an extremely significant role in maintaining animal homeostasis [
55]. In this study, both SBM diets and 10% glycinin diets increased plasma diamine oxidase activity, D-lactic acid and endotoxin contents. The enhancement effect of high-glycinin (8% glycinin) content addition on the indices was also observed in previous studies with grass carp [
26] and
Rhynchocypris lagowskii [
56,
57]. This indicates that the damage of the intestinal mucosal barrier is associated with the increased permeability of intestinal mucosa of fish caused by high-glycinin contents. This may be a secondary effect of dietary high glycinin, causing foodborne allergic reactions in fish. However, the intermediate glycinin level (4.5% glycinin) did not trigger the adverse effect in orange-spotted grouper.
The Caspase family mediates intestinal cell apoptosis induced by dietary high-glycinin content inclusion [
57]. The dietary high-glycinin (6–8%) contents were found to trigger mRNA expression of
caspase-3,
caspase-8 and/or
caspase-9 genes in MI for grass carp [
26], and in DI for Jian carp [
21] and hybrid yellow catfish [
34]. In the present study, we determined the effects of dietary glycinin on intestinal apoptosis of orange-spotted groupers by using TUNEL fluorescence staining and gene expression levels of intestinal
caspase-3,
caspase-8 and
caspase-9. We observed that fish fed SBM and 10% glycinin diets exhibited an increase in the number of intestinal apoptotic cells and an up-regulation of intestinal expression levels of
caspase-3,
caspase-8 and
caspase-9 genes vs. those fish fed the FM diets, but did not observe this effect on fish fed 4.5% glycinin diets. Therefore, apoptosis of intestinal cells of fish could be induced by dietary high-glycinin contents by promoting the intestinal caspases activation but not by intermediate-glycinin contents.
In addition to the caspase-mediated intestinal apoptosis, we also conducted further research on the NF-κB signaling pathway regulating intestinal apoptosis. The activated NF-κB can up-regulate the expression levels of
bcl-2 and
bcl-xL genes, thereby leading to the reduction of mitochondrial membrane permeability and obstruction of cytochrome C release, and thus inhibits apoptosis [
58]. The cytokine TNF-α acts as an activator of the NF-κB signaling pathway. The up-regulation of TNF-α will stimulate
NF-κB1/
RelA to enter the nucleus and activate those genes containing DNA binding sites for NF-κB. This will lead to apoptosis occurrence, which in turn induces down-regulation of expressions of downstream
bcl-2 and
bcl-xL genes [
59]. In this study, the mRNA expressions of
NF-κB1 and
TNF-α genes were up-regulated, but the mRNA expressions of
bcl-2 and
bcl-xL genes were down-regulated by SBM and 10% glycinin diets vs. the FM diets. Similarly, down-regulated intestinal
bcl-2 mRNA expressions were observed in grass carp [
26] and hybrid yellow catfish [
34] that received 8% glycinin diets compared with those fish fed the control diets. The results indicate that the occurrence of intestinal apoptosis triggered by dietary high-glycinin content addition is associated with the down-regulated expression of anti-apoptosis genes
bcl-2 and
bcl-xL and up-regulated expression of pro-apoptosis genes
TNF-α and/or
caspase-3, both in high-SBM and high-glycinin diets [
8,
57], by activation of the NF-κB signaling pathway.
There is an interdependent relationship between intestinal microbiota and the host, and intestinal microbiota is inseparable from nutrition, immunity and metabolism, forming a relatively independent intestinal microbiota homeostasis [
60]. The abundance levels of the intestinal microbial communities of fish are influenced by various environmental factors such as the source, composition, and type of feed, etc. [
61]. For example, at the phylum level, the decreased Firmicutes and Actinobacteria abundance and the increased Cyanobacteria and Tenericutes abundance, as well as the lowered Fictibacillus Lactobacillus abundance and increased Mycoplasma abundance at the genus level were observed in the intestine of largemouth bass (
Micropterus salmoides) fed high-SBM diets vs. the control diets [
9]. In this study, the intestinal dominant phyla of fish were Proteobacteria (73.2%), Tenericutes (7.0%), Actinobacteria (10.9%), Firmicutes (3.7%), Bacteroidetes (2.2%) and Cyanobacteria (0.9%). Interestingly, like SBM diets, 10% glycinin diets generally promoted the abundance of phylum Proteobacteria in the intestine of orange-spotted grouper vs. FM diets. The increased intestinal Proteobacteria abundance was also observed in largemouth bass fed SBM diets [
62] and in hybrid grouper fed 10% glycinin diets [
40]. Proteobacteria has many pathogenic bacteria that can secrete a large amount of lipopolysaccharides [
63], and higher lipopolysaccharides levels exacerbate stroke by activating NF-κB and thus produce large amounts of pro-inflammatory cytokines [
64]. On the contrary, the intestinal Tenericutes abundance was superior in FM and 4.5% glycinin diets to those of the SBM and 10% glycinin diets. It is still unknown whether Tenericutes belongs to a pathogenic bacterial at the phylum level in fish, a question which requires further research [
65]. The intestinal phylum Proteobacteria enrichment and phylum Tenericutes reduction may indicate the imbalance of intestinal microbiota [
66]. However, differing from our current observations, 10% glycinin diets tended to reduce the abundance of Bacteroides, Actinobacteria and Firmicutes, though not differently from the FM diet [
40]. The variation in intestinal bacterial abundance between spotted-orange and hybrid grouper may be due to differences in fish species and feed composition.
In the present study, SBM and 10% glycinin diets generally promoted the abundance of genus
Achromobacter and/or
Vibrio in the intestine of orange-spotted grouper, in comparison with FM diets. The increased abundance of genus
Vibrio in the intestine of hybrid grouper was observed [
40]. The genus
Achromobacter is considered opportunistic pathogens of low virulence in humans [
67] and the genus
Vibrio, such as
Vibrio anguallanim, is another opportunistic pathogen that can cause hemorrhagic septicemia and vibriosis in many fish species [
68]. Furthermore, the intestinal abundance was highest, respectively, in SBM diets for the former and in 10% glycinin diets for the latter among the treatments (
Figure 8A) in this study. This might partly explain the reason for the induction of enteritis of fish caused by dietary high-SBM contents and glycinin addition through providing favorable conditions for opportunistic pathogens to colonize the intestine. In addition, the unidentified bacterial genera in SBM diets were higher than those in other groups, which may be another trigger for enteritis in the case of feeding high-SBM contents.
It is clear that that the intestine of fish is also an important organ that performs immune defense functions. Under the stimulation of pathogens, a series of complex intestinal inflammatory reactions are triggered and activated through cytokines and complementary factors secreted by macrophages and leukocytes [
69]. NF-kB as a central mediator of pro-inflammatory gene induction also participates in the inflammation regulation through inducing the expression of various pro-inflammatory genes, including those encoding
TNF-α,
IL-1β,
IL-6,
IL-12 and cyclooxygenase-2 [
70]. TAK1 activates the downstream IKK complex and phosphorylates the NF-κB inhibitor IκBα, thereby mediating NF-κB activation [
71]. In addition, NF-κB activation mediates immune and inflammatory responses triggered by MyD88 [
72]. The NF-κB dimer RelA undergoes rapid and transient nuclear translocation under the action of phosphorylated IκBα via IKK activation [
71]. Inflammatory responses are associated with aberrant T cell activation due to aberrant activation of NF-κB [
73]. After activation, CD4+T cells develop into effector T cells (inflammatory T cells), which secrete cytokines and mediate aberrant inflammatory responses [
74].
Previous studies showed that dietary high-SBM content triggers significant changes in the expression of inflammatory-factor genes in fish [
7,
12,
46]. Massive production of pro-inflammatory cytokines was observed in fish fed high-SBM diets [
8,
11,
13]. In contrast, the down-regulated expressions of anti-inflammatory genes
IL-10 and
TGF-β were observed in the liver and spleen of fish fed high-SBM diets [
14]. In the present study, the SBM and 10% glycinin diets up-regulated the expression levels of
TNF-α,
IL-1β,
IL-8,
MyD88,
IKK-α,
TAK1,
RelA and
NF-κB1 genes, but down-regulated the expression levels of
IκBα,
TGF-β1 and
IL-10 genes, compared to FM diets. Similarly, an up-regulation of pro-inflammatory genes, including
TNF-α,
IL-1β and/or
IL-8, and a down-regulation of anti-inflammatory genes
IL-10 and/or
TGF-β in the intestine were observed in recent studies with fish fed 8% glycinin diets [
21,
26,
53,
56]. The above results show that NF-κB induces the expression of pro-inflammatory genes and also regulates inflammatory responses caused by high glycinin diets.