Ketamine–Propofol Coadministration for Induction and Infusion Maintenance in Anesthetized Dogs: Effects on Electroencephalography and Antinociception
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
- Phase 0: The study began with six healthy awake dogs.
- Phase 1: Anesthesia induction commenced using ketofol, a combination of ketamine and propofol, administered for 10 min, followed by a bolus of ketamine.
- Phases 2–4: Anesthesia maintenance was carried out using ketofol for 45 min, with this period divided into three 15 min segments. Each segment involved the administration of ketofol at different doses, categorized as high, moderate and low.
- Phase 5: Following the anesthesia maintenance phases, CRI was terminated. Subsequently, the dogs underwent extubation.
- Phase 6: The dogs were monitored until they reached sternal recumbency after extubation.
2.2. Animals
2.3. Drug Administration
2.4. EEG and EMG Instrumentation, Measurement and Analysis
2.5. Antinociception Tests
2.6. Behavioral Assessment
2.7. Statistics
3. Results
3.1. EEG and Behavior Changes during Induction
3.2. EEG Indices, Hemodynamic and Antinociceptive Data during CRI Maintenance
3.3. EEG and Hemodynamic Values and Behavior Signs during Recovery
3.4. Individual Dog Sensitivity and Responses to Ketamine–Propofol CRI Anesthesia Based on EEG Indices
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Anesthetic Plane | Depth Score | Pupil Size | Globe Position | Eye Reflex (P: Palpebral; C: Corneal) | Heart Rate, Blood Pressure, Breathing | Third Eyelid Position | Jaw Tone, Body/Limb Movement or Swallowing |
|---|---|---|---|---|---|---|---|
| Very light | 5 | Large or small, moving | Central | P: active C: Active | Increased, spontaneous, breathing attempts | Retracted | Strong |
| Medium-light | 4 | Large or small | Central or ventromedial | P: active or mildly depressed C: depressed | Increased, spontaneous breathing attempts | Retracted or partially prolapsed | Medium |
| Medium | 3 | Small or medium | Ventromedial | P: depressed C: partially depressed | Normal | Completely prolapsed | None or barely |
| Medium-deep | 2 | Medium | Central | P: depressed or markedly depressed C: depressed | Normal or depressed | Completely or partially prolapsed | None |
| Very deep | 1 | Large | Central | P: absent C: markedly depressed or absent | Depressed | Partially depressed | None |
References
- Glen, J.B. Animal studies of the anaesthetic activity of ICI 35 868. Br. J. Anaesth. 1980, 52, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Sebel, P.S.; Lowdon, J.D. Propofol: A new intravenous anesthetic. Anesthesiology 1989, 71, 260–277. [Google Scholar] [CrossRef] [PubMed]
- Marik, P.E. Propofol: Therapeutic Indications and Side-Effects. Curr. Pharm. Des. 2004, 10, 3639–3649. [Google Scholar] [CrossRef] [PubMed]
- Sahinovic, M.M.; Struys, M.M.R.F.; Absalom, A.R. Clinical Pharmacokinetics and Pharmacodynamics of Propofol. Clin. Pharmacokinet. 2018, 57, 1539–1558. [Google Scholar] [PubMed]
- Vasileiou, I.; Xanthos, T.; Koudouna, E.; Perrea, D.; Klonaris, C.; Katsargyris, A.; Papadimitriou, L. Propofol: A review of its non-anaesthetic effects. Eur. J. Pharmacol. 2009, 605, 1–8. [Google Scholar]
- Li, L.; Vlisides, P.E. Ketamine: 50 Years of Modulating the Mind. Front. Hum. Neurosci. 2016, 10, 612. [Google Scholar]
- White, P.F.; Way, W.L.; Trevor, A.J. Ketamine—Its Pharmacology and Therapeutic Uses. Anesthesiology 1982, 56, 119–136. [Google Scholar]
- Andolfatto, G.; Abu-Laban, R.B.; Zed, P.J.; Staniforth, S.M.; Stackhouse, S.; Moadebi, S.; Willman, E. Ketamine-Propofol Combination (Ketofol) Versus Propofol Alone for Emergency Department Procedural Sedation and Analgesia: A Randomized Double-Blind Trial. Ann. Emerg. Med. 2012, 59, 504–512.e2. [Google Scholar]
- Smischney, N.J.; Beach, M.L.; Loftus, R.W.; Dodds, T.M.; Koff, M.D. Ketamine/propofol admixture (ketofol) is associated with improved hemodynamics as an induction agent: A randomized, controlled trial. J. Trauma Acute Care Surg. 2012, 73, 94–101. [Google Scholar] [CrossRef]
- Jalili, M.; Bahreini, M.; Doosti-Irani, A.; Masoomi, R.; Arbab, M.; Mirfazaelian, H. Ketamine-propofol combination (ketofol) vs propofol for procedural sedation and analgesia: Systematic review and meta-analysis. Am. J. Emerg. Med. 2016, 34, 558–569. [Google Scholar] [CrossRef]
- Gonz, A. Corticoadrenal and Cardiorespiratory Responses to Administration of Propofol Combined with Dexmedetomidine or Ketamine in Rabbits. J. Am. Assoc. Lab. Anim. Sci. 2018, 57, 278–281. [Google Scholar]
- Hashemi, S.R.; Vesal, N. Ketamine–propofol for total intravenous anaesthesia in rabbits: A comparison of premedication with acepromazine–medetomidine, acepromazine–midazolam or acepromazine–morphine. Vet. Anaesth. Analg. 2023, 50, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Boudreau, A.E.; Bersenas, A.M.E.; Kerr, C.L.; Holowaychuk, M.K.; Johnson, R.J. A comparison of 3 anesthetic protocols for 24 h of mechanical ventilation in cats. J. Vet. Emerg. Crit. Care 2012, 22, 239–252. [Google Scholar] [CrossRef]
- Ravasio, G.; Gallo, M.; Beccaglia, M.; Comazzi, S.; Gelain, M.E.; Fonda, D.; Bronzo, V.; Zonca, A. Evaluation of a ketamine-propofol drug combination with or without dexmedetomidine for intravenous anesthesia in cats undergoing ovariectomy. J. Am. Vet. Med. Assoc. 2012, 241, 1307–1313. [Google Scholar] [CrossRef] [PubMed]
- Jarrett, M.A.; Bailey, K.M.; Messenger, K.M.; Prange, T.; Gaines, B.; Posner, L.P. Recovery of horses from general anesthesia after induction with propofol and ketamine versus midazolam and ketamine. J. Am. Vet. Med. Assoc. 2018, 253, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, M.J.; Smith, L.J. A comparison of cardiopulmonary function, recovery quality, and total dosages required for induction and total intravenous anesthesia with propofol versus a propofol-ketamine combination in healthy Beagle dogs. Vet. Anaesth. Analg. 2015, 42, 350–359. [Google Scholar] [CrossRef]
- Mair, A.R.; Pawson, P.; Courcier, E.; Flaherty, D. A comparison of the effects of two different doses of ketamine used for co-induction of anaesthesia with a target-controlled infusion of propofol in dogs. Vet. Anaesth. Analg. 2009, 36, 532–538. [Google Scholar] [CrossRef]
- Lee, M.; Kim, S.; Moon, C.; Park, J.; Lee, H.; Jeong, S.M. Anesthetic Effect of Different Ratio of Ketamine and Propofol in Dogs. J. Vet. Clin. 2017, 34, 234–240. [Google Scholar] [CrossRef]
- Lerche, P.; Reid, J.; Nolan, A.M. Comparative study of propofol or propofol and ketamine for the induction of anaesthesia in dogs. Vet. Rec. 2000, 146, 571–574. [Google Scholar] [CrossRef]
- Martinez-Taboada, F.; Leece, E.A. Comparison of propofol with ketofol, a propofol-ketamine admixture, for induction of anaesthesia in healthy dogs. Vet. Anaesth. Analg. 2014, 41, 575–582. [Google Scholar] [CrossRef]
- Henao-Guerrero, N.; Riccó, C.H. Comparison of the cardiorespiratory effects of a combination of ketamine and propofol, propofol alone, or a combination of ketamine and diazepam before and after induction of anesthesia in dogs sedated with acepromazine and oxymorphone. Am. J. Vet. Res. 2014, 75, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Wamaitha, M.; Mogoa, E.; Mande, J. Evaluation of anesthesia produced by ketofol in acepromazine- or medetomidine-sedated dogs. J. Adv. Vet. Anim. Res. 2019, 6, 215. [Google Scholar] [CrossRef] [PubMed]
- Seliškar, A.; Nemec, A.; Roškar, T.; Butinar, J. Total intravenous anaesthesia with propofol or propofol/ketamine in spontaneously breathing dogs premedicated with medetomidine. Vet. Rec. 2007, 160, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.; Antunes, L. Electroencephalogram-based anaesthetic depth monitoring in laboratory animals. Lab. Anim. 2012, 46, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Drinkenburg, W.H.; Ahnaou, A.; Ruigt, G.S. Pharmaco-EEG Studies in Animals: A History-Based Introduction to Contemporary Translational Applications. Neuropsychobiology 2015, 72, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Murillo, C.; Weil, A.B.; Moore, G.E.; Kreuzer, M.; Ko, J.C. Electroencephalographic and Cardiovascular Changes Associated with Propofol Constant Rate of Infusion Anesthesia in Young Healthy Dogs. Animals 2023, 13, 664. [Google Scholar] [CrossRef]
- Hayashi, K.; Tsuda, N.; Sawa, T.; Hagihira, S. Ketamine increases the frequency of electroencephalographic bicoherence peak on the α spindle area induced with propofol. Br. J. Anaesth. 2007, 99, 389–395. [Google Scholar] [CrossRef]
- Sakai, T.; Singh, H.; Mi, W.D.; Kudo, T.; Matsuki, A. The effect of ketamine on clinical endpoints of hypnosis and EEG variables during propofol infusion. Acta Anaesthesiol. Scand. 1999, 43, 212–216. [Google Scholar] [CrossRef]
- Murillo, C.; Weng, H.-Y.; Weil, A.B.; Kreuzer, M.; Ko, J.C. Perioperative Brain Function Monitoring with Electroencephalography in Horses Anesthetized with Multimodal Balanced Anesthetic Protocol Subjected to Surgeries. Animals 2022, 12, 2851. [Google Scholar] [CrossRef]
- Krimins, R.A.; Ko, J.C.; Weil, A.B.; Payton, M.E. Evaluation of anesthetic, analgesic, and cardiorespiratory effects in dogs after intramuscular administration of dexmedetomidine–butorphanol–tiletamine-zolazepam or dexmedetomidine-tramadol-ketamine drug combinations. Am. J. Vet. Res. 2012, 73, 1707–1714. [Google Scholar] [CrossRef]
- Von Dincklage, F.; Jurth, C.; Schneider, G.; Garcia, P.S.; Kreuzer, M. Technical considerations when using the EEG export of the SEDLine Root device. J. Clin. Monit. Comput. 2021, 35, 1047–1054. [Google Scholar] [CrossRef] [PubMed]
- Purdon, P.L.; Sampson, A.; Pavone, K.J.; Brown, E.N. Clinical Electroencephalography for Anesthesiologists. Anesthesiology 2015, 123, 937–960. [Google Scholar] [CrossRef] [PubMed]
- Purdon, P.L.; Pierce, E.T.; Mukamel, E.A.; Prerau, M.J.; Walsh, J.L.; Wong KF, K.; Salazar-Gomez, A.F.; Harrell, P.G.; Sampson, A.L.; Cimenser, A.; et al. Electroencephalogram signatures of loss and recovery of consciousness from propofol. Proc. Natl. Acad. Sci. USA 2023, 110, E1142–E1151. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.N.; Purdon, P.L.; Van Dort, C.J. General Anesthesia and Altered States of Arousal: A Systems Neuroscience Analysis. Annu. Rev. Neurosci. 2011, 34, 601–628. [Google Scholar] [CrossRef] [PubMed]
- Seamans, J. Losing inhibition with ketamine. Nat. Chem. Biol. 2008, 4, 91–93. [Google Scholar]
- Homayoun, H.; Moghaddam, B. NMDA Receptor Hypofunction Produces Opposite Effects on Prefrontal Cortex Interneurons and Pyramidal Neurons. J. Neurosci. 2007, 27, 11496–11500. [Google Scholar] [CrossRef]
- Domino, E.F.; Warner, D.S. Taming the Ketamine Tiger. Anesthesiology 2010, 113, 678–684. [Google Scholar] [CrossRef]
- Akeju, O.; Song, A.H.; Hamilos, A.E.; Pavone, K.J.; Flores, F.J.; Brown, E.N.; Purdon, P.L. Electroencephalogram signatures of ketamine anesthesia-induced unconsciousness. Clin. Neurophysiol. 2016, 127, 2414–2422. [Google Scholar] [CrossRef]
- Bojak, I.; Day, H.C.; Liley, D.T.J. Ketamine, Propofol, and the EEG: A Neural Field Analysis of HCN1-Mediated Interactions. Front. Comput. Neurosci. 2013, 7, 22. [Google Scholar] [CrossRef]
- Ko, J.C.; Raffe, M.R.; Knels, O.; Inoue, T.; Weil, A.B. Analgesia, Sedation and Anesthesia: Making the Switch from Medetomidine to Dexmedetomidine. Compend. Contin. Educ. Vet. 2009, 31, 1–24. Available online: https://s3.amazonaws.com/assets.prod.vetlearn.com/00/092570e29811dfb5427c6d628b17ab/file/fileDexmedetomidineFAQ.pdf (accessed on 17 September 2023).
- Ko, J.C.H.; Fox, S.M.; Mandsager, R.E. Sedative and cardiorespiratory effects of medetomidine, medetomidine-butorphanol, and medetomidine-ketamine in dogs. J. Am. Vet. Med. Assoc. 2000, 216, 1578–1583. [Google Scholar] [CrossRef] [PubMed]
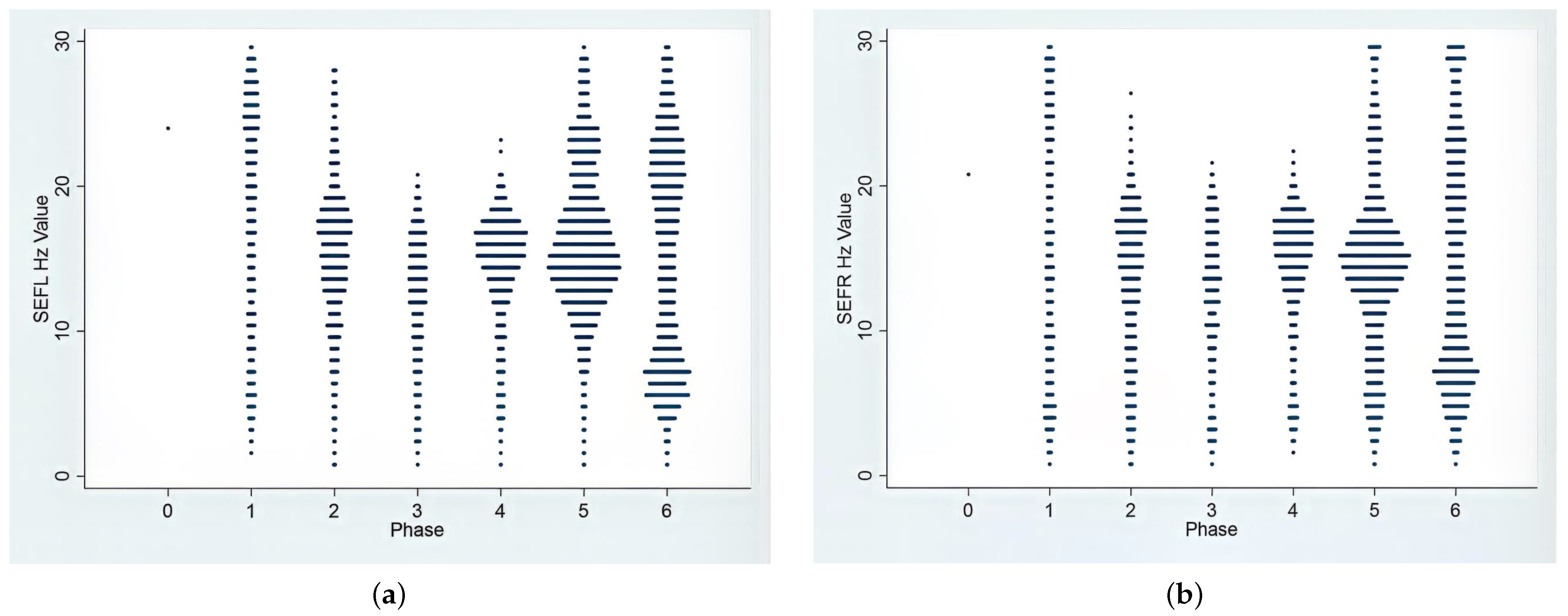
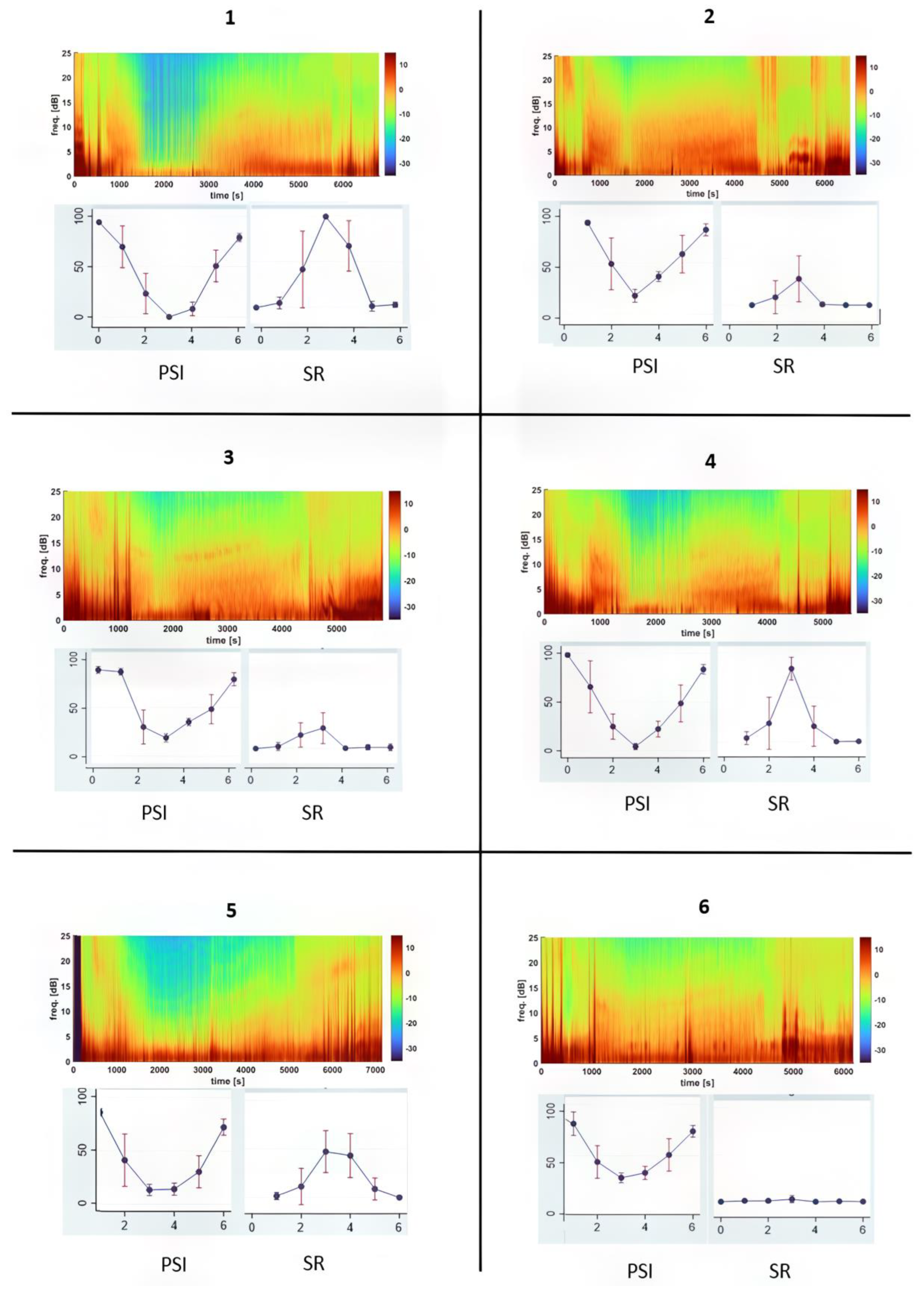
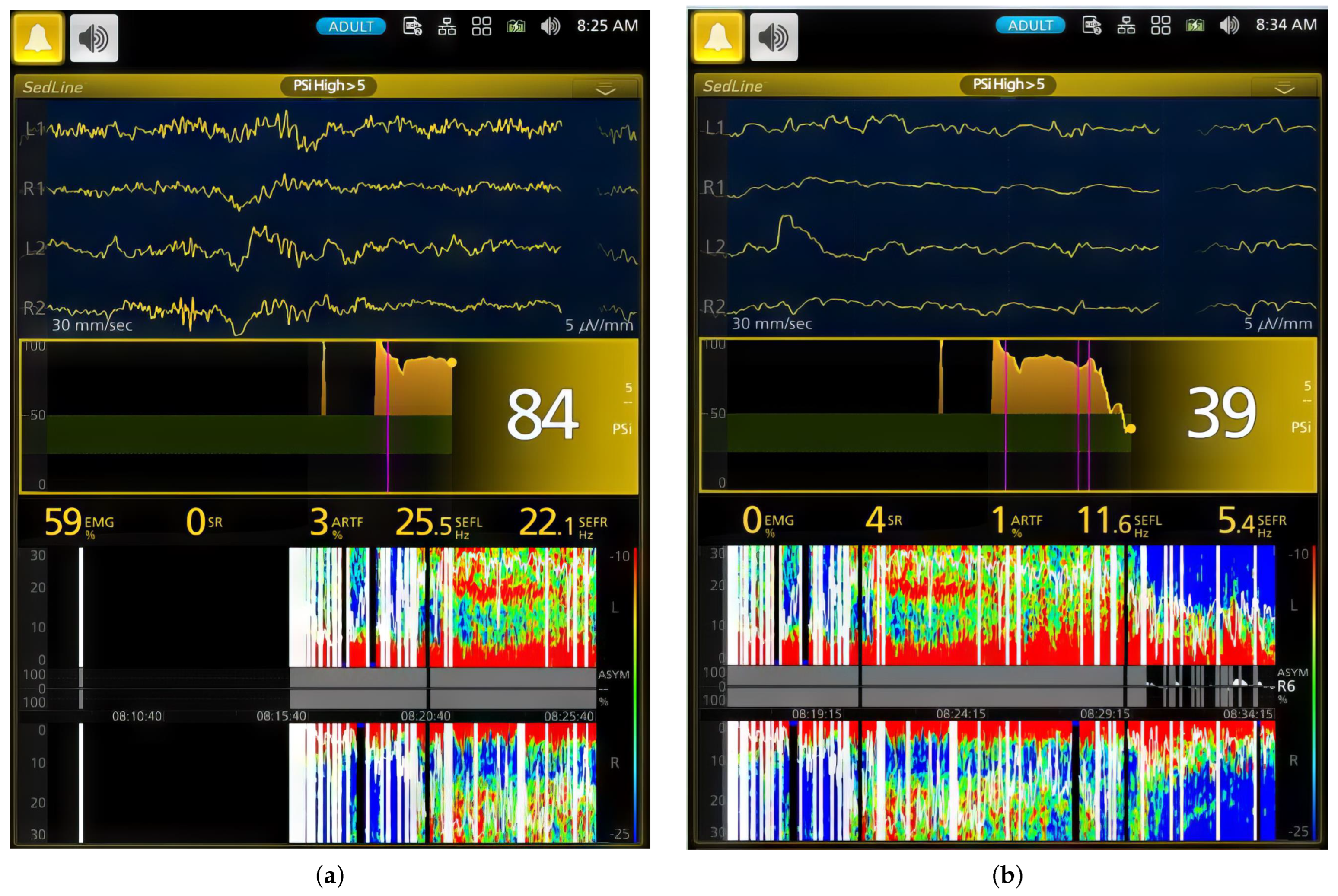
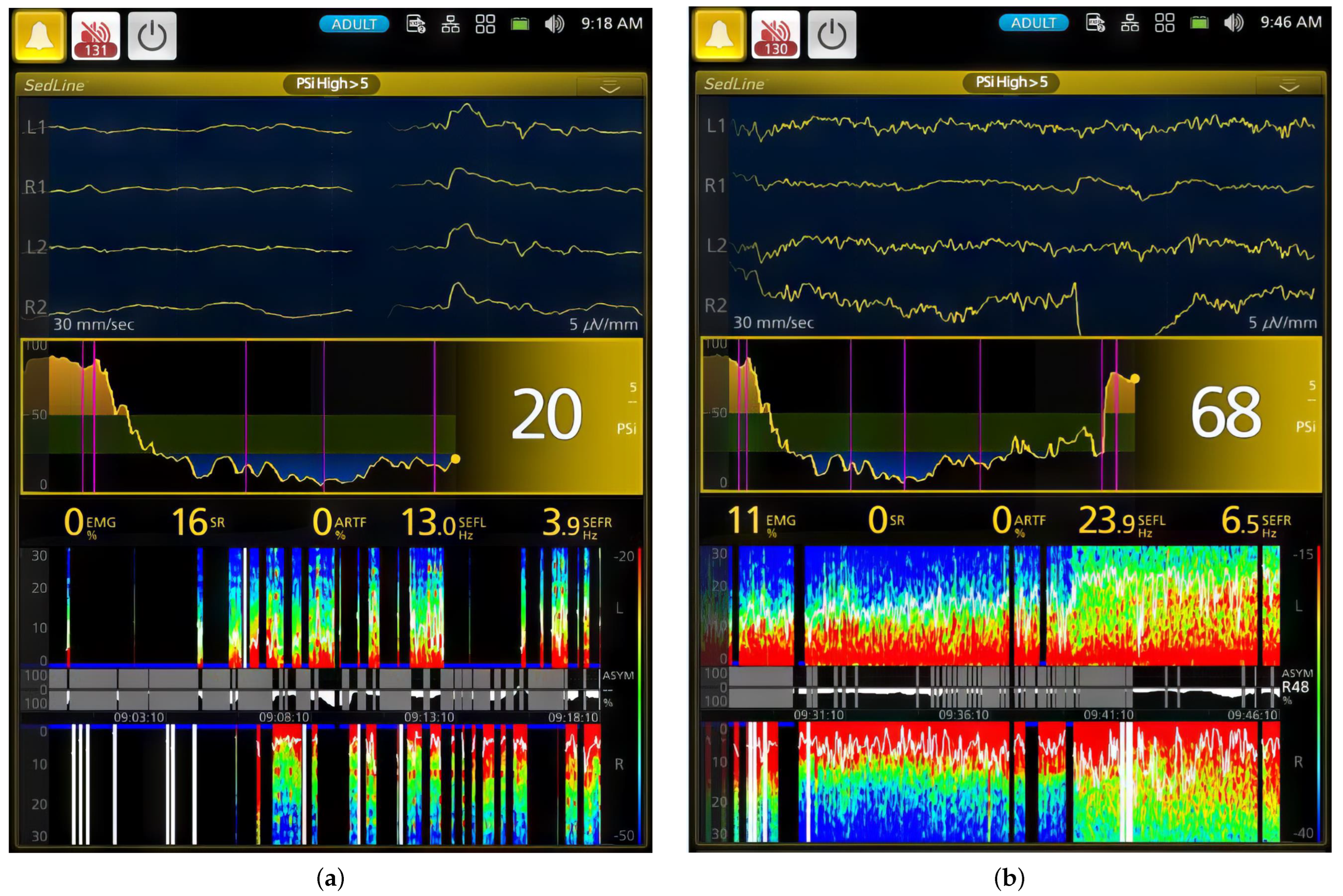
| Phase | PSI | SR (%) | EMG (%) | ARTF (%) | SEFR (Hz) | SEFL (Hz) |
|---|---|---|---|---|---|---|
| 0 | 92.7 ± 5.1 a | 0.0 ± 0.0 a | 52.4 ±30.0 | 9.8 ± 9.6 | 20.8 ± 0.0 | 23.6 ± 0.0 |
| 1 | 79.9 ± 17.1 a | 2.4 ± 5.0 b | 57.9 ± 28.6 | 5.4 ± 10.2 | 15.3 ± 8.4 | 18.0 ± 7.6 |
| 2 | 35.7 ± 22.5 b | 16.6 ± 27.4 b | 7.8 ± 14.0 | 4.7 ± 14.3 | 13.3 ± 5.0 | 15.3 ± 4.8 |
| 3 | 14.0 ± 10.4 c | 46.7 ± 37.2 c | 1.0 ± 1.8 | 0.1 ± 0.8 | 12.3 ± 4.8 | 12.4 ± 4.2 |
| 4 | 25.0 ± 12.7 b | 21.7 ± 31.0 b | 1.9 ± 3.1 | 0.4 ± 2.2 | 14.2 ± 4.1 | 14.4 ± 3.7 |
| 5 | 47.4 ± 1 8.7 b | 2.4 ± 6.6 b | 12.4 ± 17.3 | 0.6 ± 3.0 | 14.4 ± 5.7 | 16.4 ± 4.9 |
| 6 | 78.1 ± 7.4 a | 0.6 ± 2.1 a | 32.5 ± 22.8 | 10.4 ± 17.1 | 14.0 ± 7.8 | 15.1 ± 7.4 |
| Phase | HR (bpm) | SBP (mmHg) | MBP (mmHg) | DBP (mmHg) c | Depth Score d | Electrical Stimulation (Hz) e |
|---|---|---|---|---|---|---|
| 0 | 108.9 ± 21.0 | 165.5 ± 22.4 | 114.5 ± 15.9 | 96.7 ± 15.1 | 5.0 ± 0.0 a | NA |
| 1 | 135.7 ± 35.1 | 164.1 ± 17.9 | 114.4 ± 22.6 | 97.3 ± 25.4 | 4.4 ± 0.6 a | NA |
| 2 | 142.9 ± 21.3 | 149.3 ± 28.6 | 105.1 ± 25.4 a | 89 ± 26.8 a | 2.7 ± 0.7 b | 845.4 ± 126.2 a |
| 3 | 131.2 ± 12.6 | 123.6 ± 21.5 a | 81.5 ± 14.7 a | 67.4 ± 14.6 a | 2.2 ± 0.4 b | 875 ± 56.8 a |
| 4 | 146.1 ± 15.2 | 144.4 ± 25.0 | 110.3 ± 23.3 | 93.9 ± 21.0 a | 2.4 ± 0.5 b | 803.0 ± 170.4 a |
| 5 | 140.9 ± 22.3 | 171.7 ± 17.5 b | 137.7 ± 13.6 b | 118.8 ± 12.8 b | 3.2 ± 0.6 c | 629.1 ± 231.8 a |
| 6 | 162.0 ± 25.1 | 187.2 ± 32.9 b | 147.2 ± 29.2 b | 133.3 ± 26.9 c | 4.9 ± 0.2 a | 150 ± 0.0 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ko, J.C.; Murillo, C.; Weil, A.B.; Kreuzer, M.; Moore, G.E. Ketamine–Propofol Coadministration for Induction and Infusion Maintenance in Anesthetized Dogs: Effects on Electroencephalography and Antinociception. Animals 2023, 13, 3391. https://doi.org/10.3390/ani13213391
Ko JC, Murillo C, Weil AB, Kreuzer M, Moore GE. Ketamine–Propofol Coadministration for Induction and Infusion Maintenance in Anesthetized Dogs: Effects on Electroencephalography and Antinociception. Animals. 2023; 13(21):3391. https://doi.org/10.3390/ani13213391
Chicago/Turabian StyleKo, Jeff C., Carla Murillo, Ann B. Weil, Matthias Kreuzer, and George E. Moore. 2023. "Ketamine–Propofol Coadministration for Induction and Infusion Maintenance in Anesthetized Dogs: Effects on Electroencephalography and Antinociception" Animals 13, no. 21: 3391. https://doi.org/10.3390/ani13213391





