Does Direct MRI Tenography Improve the Diagnostic Performance of Low-Field MRI to Identify Artificially Created Soft-Tissue Lesions within the Equine Cadaveric Digital Flexor Tendon Sheath?
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Specimens
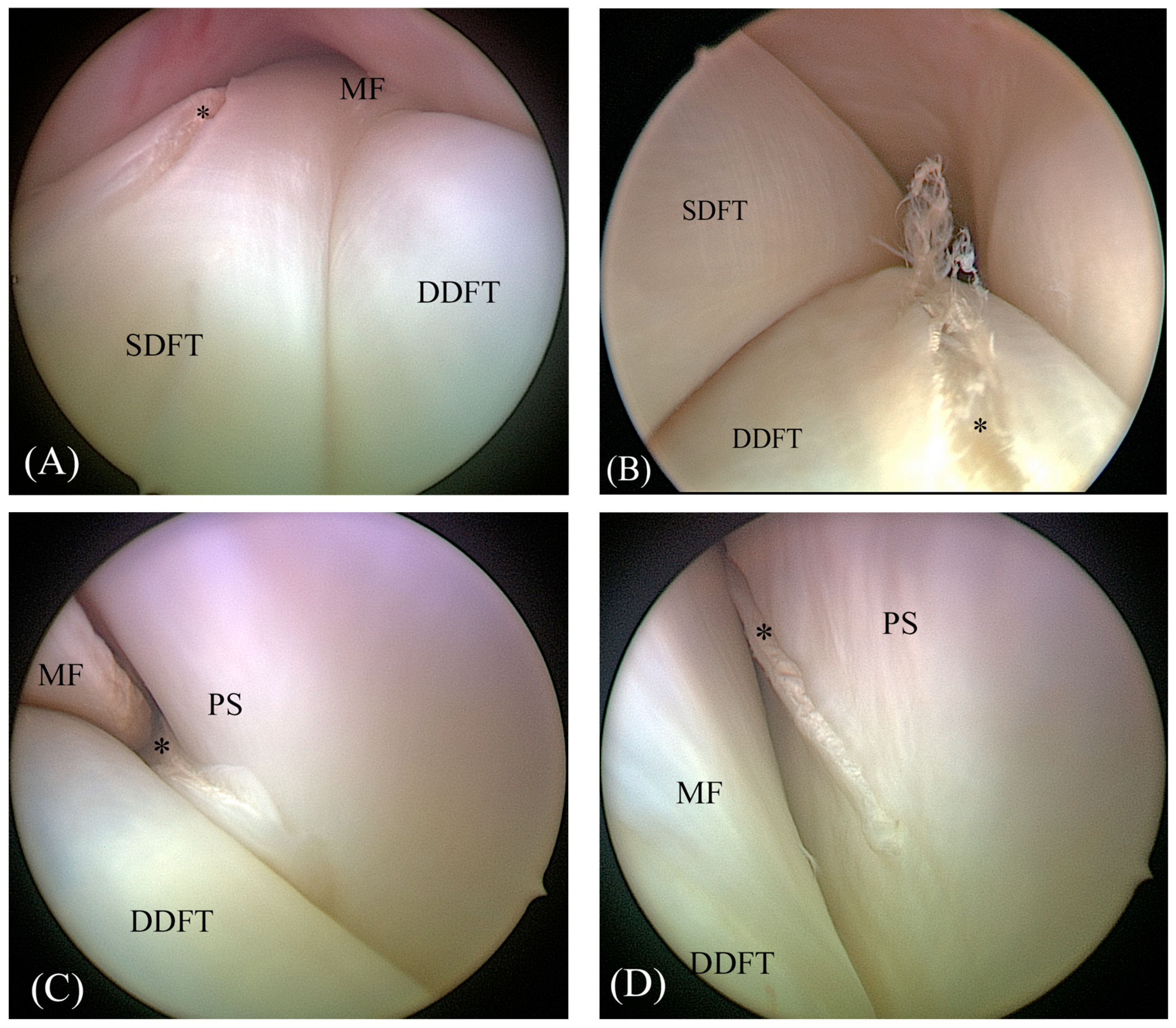
2.2. Tenoscopic Procedure
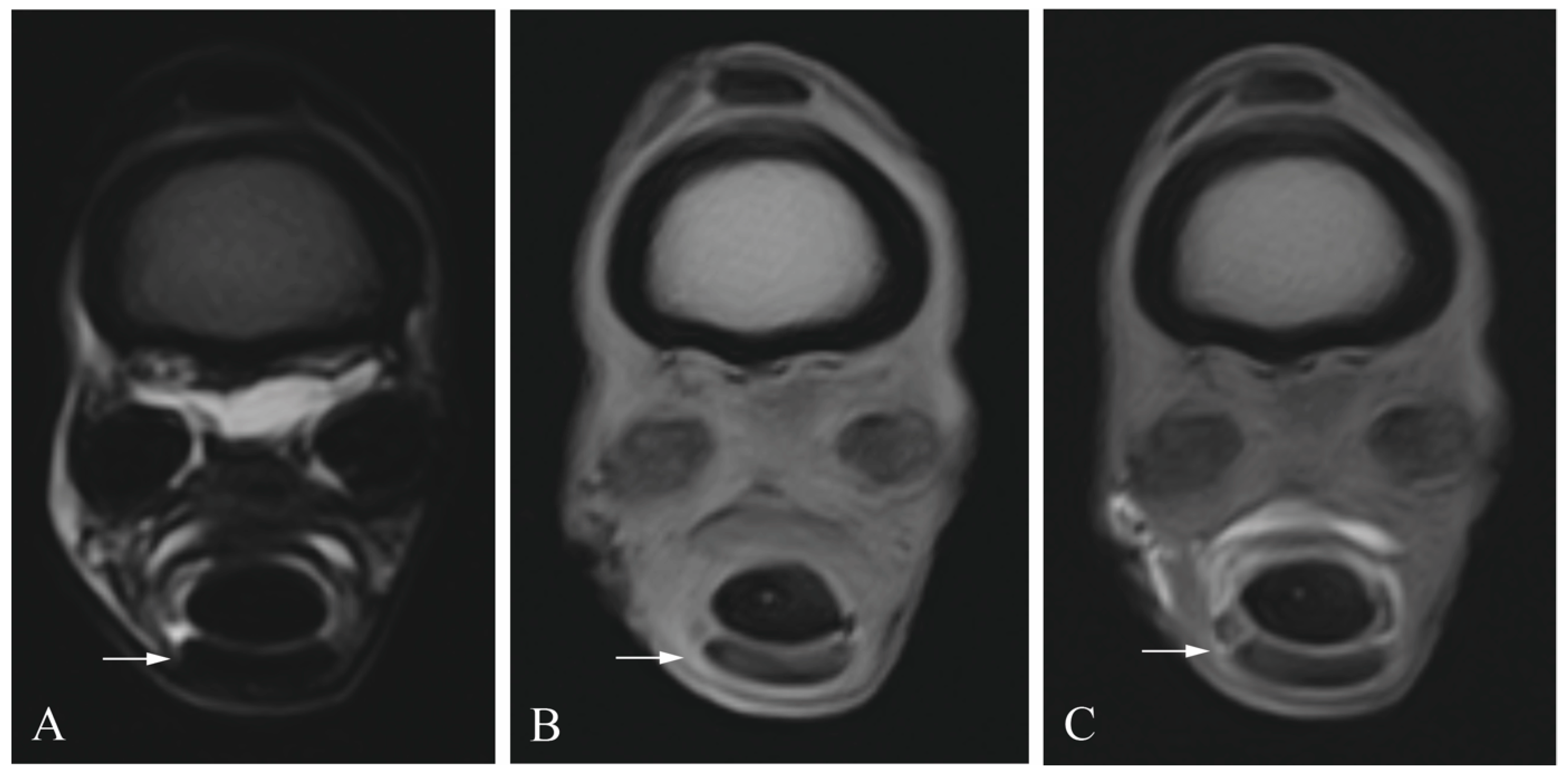
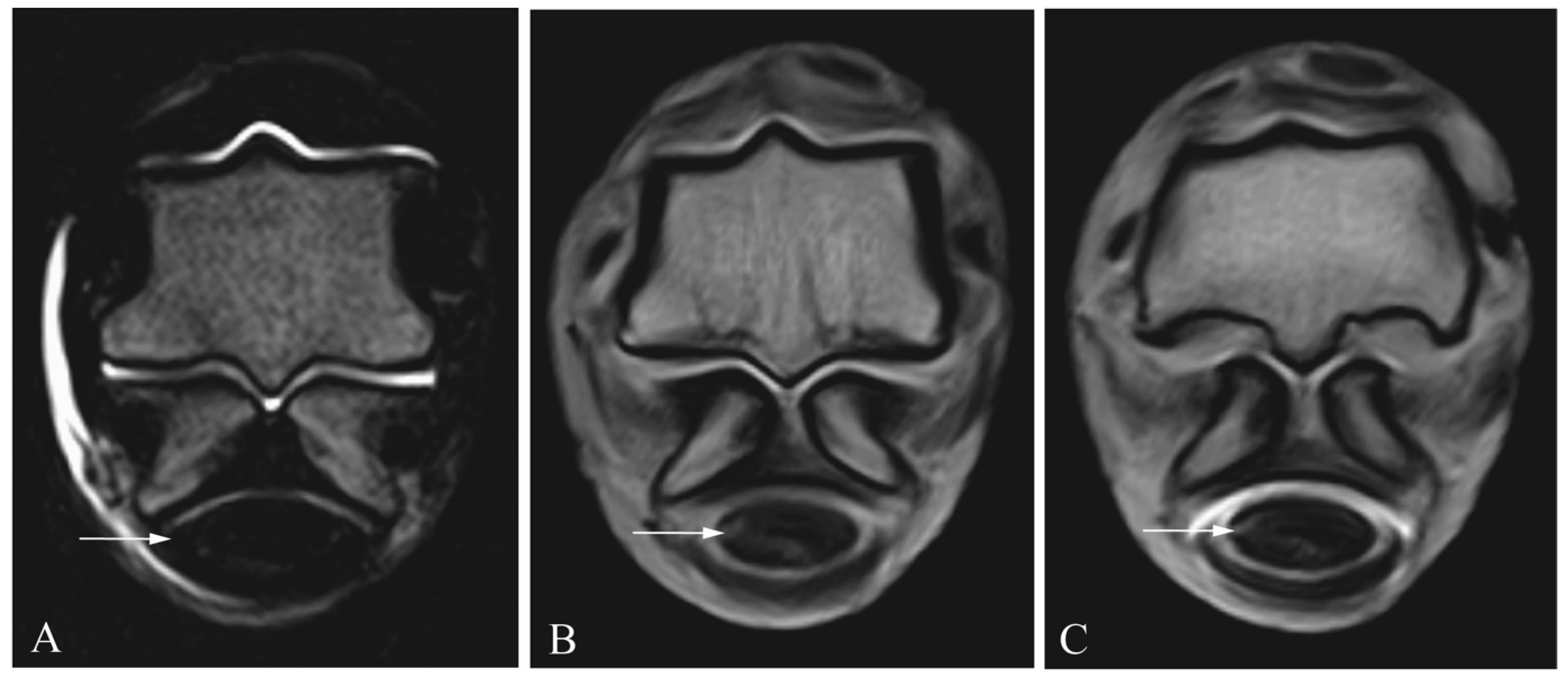
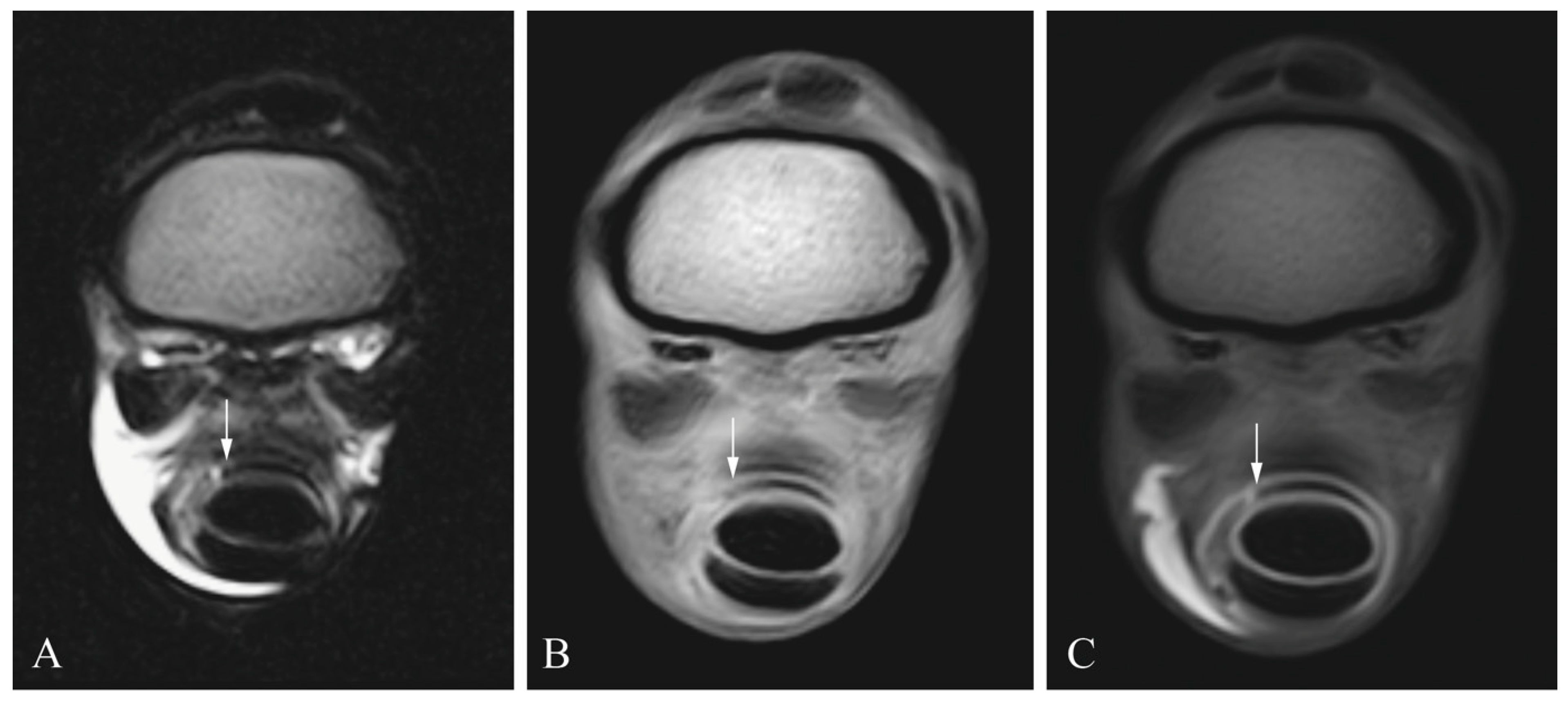
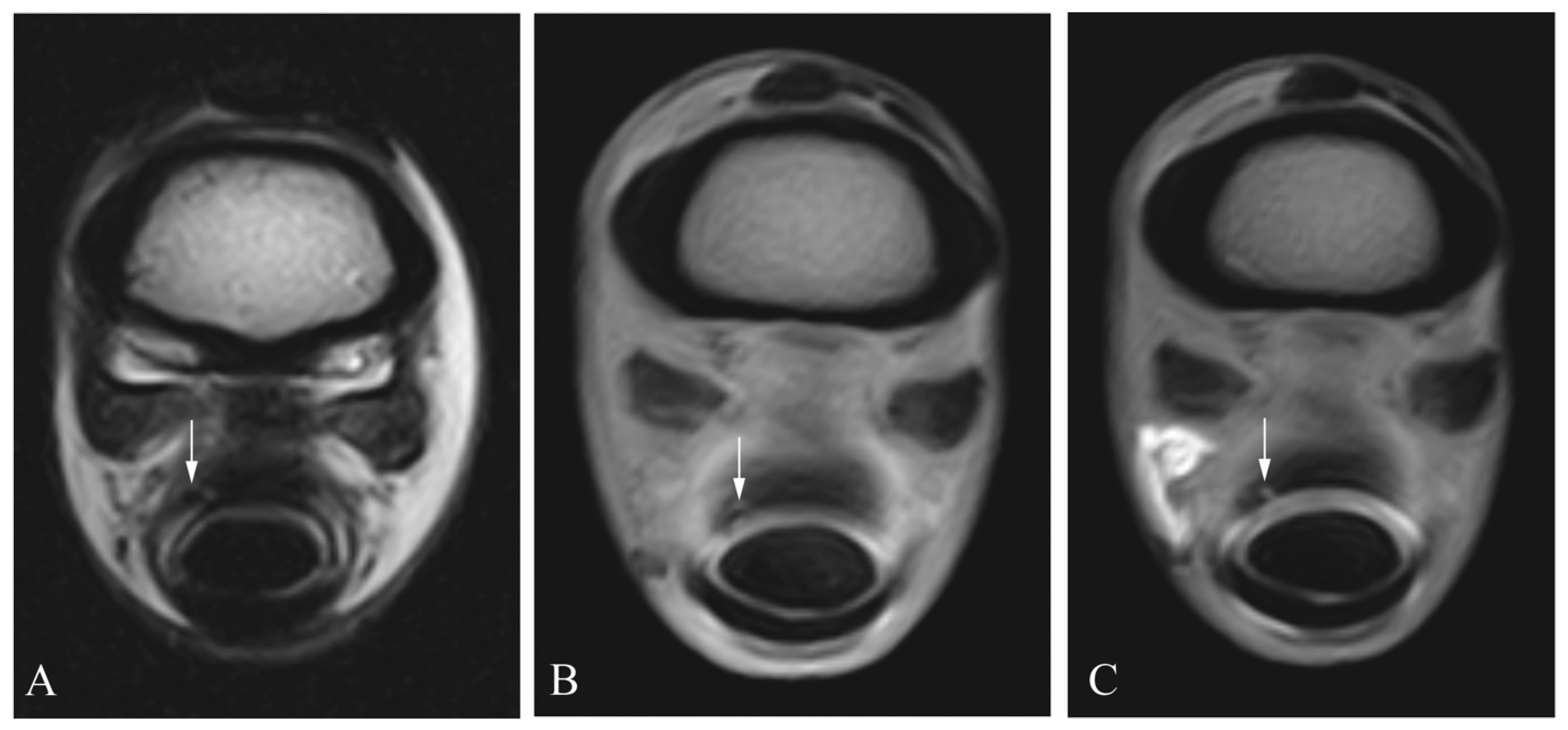
2.3. Diagnostic Imaging
2.4. Image Analysis
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Arensburg, L.; Wilderjans, H.; Simon, O.; Dewulf, J.; Boussauw, B. Nonseptic tenosynovitis of the digital flexor tendon sheath caused by longitudinal tears in the digital flexor tendons: A retrospective study of 135 tenoscopic procedures. Equine Vet. J. 2011, 43, 660–668. [Google Scholar] [CrossRef] [PubMed]
- Wilderjans, H.; Boussauw, B.; Madder, K.; Simon, O. Tenosynovitis of the digital flexor tendon sheath and annular ligament constriction syndrome caused by longitudinal tears in the deep digital flexor tendon: A clinical and surgical report of 17 cases in Warmblood horses. Equine Vet. J. 2003, 35, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.R.; Wright, I.M. Noninfected tenosynovitis of the digital flexor tendon sheath: A retrospective analysis of 76 cases. Equine Vet. J. 2006, 38, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Findley, J.A.; De Oliveira, F.; Bladon, B. Tenoscopic surgical treatment of tears of the manica flexoria in 53 horses. Vet. Surg. 2012, 41, 924–930. [Google Scholar] [CrossRef] [PubMed]
- Edinger, J.; Möbius, G.; Ferguson, J. Comparison of tenoscopic and ultrasonographic methods of examination of the digital flexor tendon sheath in horses. Vet. Comp. Orthop. Traumatol. 2005, 18, 209–214. [Google Scholar] [PubMed]
- Garcia da Fonseca, R.M.; Evrard, L.; Rabba, S.; Salciccia, A.; Busoni, V. Dynamic flexion/extension and non-weight bearing ultrasonography is helpful for identifying manica flexoria tears in horses. Vet. Radiol. Ultrasound 2019, 60, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Hibner-Szaltys, M.; Cavallier, F.; Cantatore, F.; Withers, J.M.; Marcatili, M. Ultrasonography can be used to predict the location of manica flexoria tears in horses. Equine Vet. Educ. 2022, 35, e200–e207. [Google Scholar] [CrossRef]
- Kent, A.V.; Chesworth, M.J.; Wells, G.; Gerdes, C.; Bladon, B.M.; Smith, R.K.W.; Fiske-Jackson, A.R. Improved diagnostic criteria for digital flexor tendon sheath pathology using contrast tenography. Equine Vet. J. 2019, 52, 205–221. [Google Scholar] [CrossRef]
- Fiske-Jackson, A.R.; Barker, W.H.; Eliashar, E.; Foy, K.; Smith, R.K. The use of intrathecal analgesia and contrast radiography as preoperative diagnostic methods for digital flexor tendon sheath pathology. Equine Vet. J. 2013, 45, 36–40. [Google Scholar] [CrossRef]
- Nixon, A.J.; McIlwraith, C.W.; Wright, I.M. Arthroscopic Surgery of the Carpal and Digital Tendon Sheaths, 4th ed.; Mosby: Maryland Heights, MI, USA, 2015. [Google Scholar]
- Gonzalez, L.M.; Schramme, M.C.; Robertson, I.D.; Thrall, D.E.; Redding, R.W. MRI features of metacarpo (tarso) phalangeal region lameness in 40 horses. Vet. Radiol. Ultrasound 2010, 51, 404–414. [Google Scholar] [CrossRef]
- King, J.N.; Zubrod, C.J.; Schneider, R.K.; Sampson, S.N.; Roberts, G. MRI findings in 232 horses with lameness localized to the metacarpo (tarso) phalangeal region and without a radiographic diagnosis. Vet. Radiol. Ultrasound 2013, 54, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Dyson, S.; Murray, R. Magnetic resonance imaging of the equine fetlock. Clin. Tech. Equine Pract. 2007, 6, 62–77. [Google Scholar] [CrossRef]
- Daniel, A.; Leise, B.; Selberg, K.; Barrett, M. Enhanced ultrasonographic imaging of the equine distal limb using saline injection of the digital flexor tendon sheath: A cadaver study. Vet. J. 2019, 247, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Steinbach, L.S.; Palmer, W.E.; Schweitzer, M.E. Special focus session: MR arthrography. Radiographics 2002, 22, 1223–1246. [Google Scholar] [CrossRef] [PubMed]
- Bergin, D.; Schweitzer, M. Indirect magnetic resonance arthrography. Skelet. Radiol. 2003, 32, 551–558. [Google Scholar] [CrossRef]
- Bittersohl, B.; Hosalkar, H.S.; Werlen, S.; Trattnig, S.; Siebenrock, K.A.; Mamisch, T.C. Intravenous versus intra-articular delayed gadolinium-enhanced magnetic resonance imaging in the hip joint: A comparative analysis. Investig. Radiol. 2010, 45, 538–542. [Google Scholar] [CrossRef] [PubMed]
- Şahin, G.; Demirtaş, M. An overview of MR arthrography with emphasis on the current technique and applicational hints and tips. Rev. Clin. Imaging 2007, 31, 73. [Google Scholar] [CrossRef]
- Kopka, L.; Funke, M.; Fischer, U.; Keating, D.; Oestmann, J.; Grabbe, E. MR arthrography of the shoulder with gadopentetate dimeglumine: Influence of concentration, iodinated contrast material, and time on signal intensity. AJR. Am. J. Roentgenol. 1994, 163, 621–623. [Google Scholar] [CrossRef]
- McCauley, T.R.; Elfar, A.; Moore, A.; Haims, A.H.; Jokl, P.; Lynch, J.K.; Ruwe, P.A.; Katz, L.D. MR arthrography of anterior cruciate ligament reconstruction grafts. Am. J. Roentgenol. 2003, 181, 1217–1223. [Google Scholar] [CrossRef]
- Robinson, P.; White, L.; Salonen, D.; Ogilvie-Harris, D. Anteromedial impingement of the ankle: Using MR arthrography to assess the anteromedial recess. Am. J. Roentgenol. 2002, 178, 601–604. [Google Scholar] [CrossRef]
- Sciulli, R.L.; Boutin, R.D.; Brown, R.; Nguyen, K.D.; Muhle, C.; Lektrakul, N.; Pathria, M.N.; Pedowitz, R.; Resnick, D. Evaluation of the postoperative meniscus of the knee: A study comparing conventional arthrography, conventional MR imaging, MR arthrography with iodinated contrast material, and MR arthrography with gadolinium-based contrast material. Skelet. Radiol. 1999, 28, 508–514. [Google Scholar] [CrossRef]
- Saveraid, T.C.; Judy, C.E. Use of intravenous gadolinium contrast in equine magnetic resonance imaging. The Veterinary clinics of North America. Equine Pract. 2012, 28, 617–636. [Google Scholar] [CrossRef] [PubMed]
- De Zani, D.; Rabbogliatti, V.; Ravasio, G.; Pettinato, C.; Di Giancamillo, M.; Zani, D.D. Contrast enhanced magnetic resonance imaging of the foot in horses using intravenous versus regional intraarterial injection of gadolinium. Open Vet. J. 2018, 8, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Aarsvold, S.; Solano, M.; Garcia-Lopez, J. Magnetic resonance imaging following regional limb perfusion of gadolinium contrast medium in 26 horses. Equine Vet. J. 2018, 50, 649–657. [Google Scholar] [CrossRef] [PubMed]
- Nelson, B.; Goodrich, L.; Barrett, M.; Grinstaff, M.; Kawcak, C. Use of contrast media in computed tomography and magnetic resonance imaging in horses: Techniques, adverse events and opportunities. Equine Vet. J. 2017, 49, 410–424. [Google Scholar] [CrossRef] [PubMed]
- Zhalniarovich, Y.; Przyborowska-Zhalniarovich, P.; Mieszkowska, M.; Adamiak, Z. Direct magnetic resonance arthrography of the canine elbow. Acta Vet. Brno 2017, 86, 85–89. [Google Scholar] [CrossRef]
- Banfield, C.M.; Morrison, W.B. Magnetic resonance arthrography of the canine stifle joint technique and applications in eleven military dogs. Vet. Radiol. Ultrasound 2000, 41, 200–213. [Google Scholar] [CrossRef]
- Van Zadelhoff, C.; Schwarz, T.; Smith, S.; Engerand, A.; Taylor, S. Identification of naturally occurring cartilage damage in the equine distal interphalangeal joint using low-field magnetic resonance imaging and magnetic resonance arthrography. Front. Vet. Sci. 2020, 6, 508. [Google Scholar] [CrossRef]
- Bischofberger, A.S.; Fürst, A.E.; Torgerson, P.R.; Carstens, A.; Hilbe, M.; Kircher, P. Use of a 3-telsa magnet to perform delayed gadolinium-enhanced magnetic resonance imaging of the distal interphalangeal joint of horses with and without naturally occurring osteoarthritis. Am. J. Vet. Res. 2018, 79, 287–298. [Google Scholar] [CrossRef]
- Carstens, A.; Kirberger, R.M.; Velleman, M.; Dahlberg, L.E.; Fletcher, L.; Lammentausta, E. Feasibility for mapping cartilage T1 relaxation times in the distal metacarpus3/metatarsus3 of thoroughbred racehorses using delayed gadolinium-enhanced magnetic resonance imaging of cartilage (dGEMRIC): Normal cadaver study. Vet. Radiol. Ultrasound 2013, 54, 365–372. [Google Scholar] [CrossRef]
- McIlwraith, C.W.; Nixon, A.J.; Wright, I.M. Diagnostic and Surgical Arthroscopy in the Horse, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Aßmann, A.D.; Ohlerth, S.; Torgerson, P.R.; Bischofberger, A.S. Sensitivity and specificity of 3 Tesla magnetic resonance imaging and multidetector computed tomographic tenography to identify artificially induced soft tissue lesions in the equine cadaveric digital flexor tendon sheath. Equine Vet. Educ. 2023, 35, e507–e516. [Google Scholar] [CrossRef]
- Findley, J.A.; Ricci, E.E.; Singer, E.E. An anatomical and histological study of the equine proximal manica flexoria. Vet. Comp. Orthop. Traumatol. 2017, 30, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.P.; Brown, J.J. MR contrast agents: Physical and pharmacologic basics. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2007, 25, 884–899. [Google Scholar] [CrossRef] [PubMed]
- Murray, R.C.; Mair, T.S.; Sherlock, C.E.; Blunden, A.S. Comparison of high-field and low-field magnetic resonance images of cadaver limbs of horses. Vet. Rec. 2009, 165, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Vallance, S.; Bell, R.; Spriet, M.; Kass, P.H.; Puchalski, S. Comparisons of computed tomography, contrast enhanced computed tomography and standing low-field magnetic resonance imaging in horses with lameness localised to the foot. Part 1: Anatomic visualisation scores. Equine Vet. J. 2012, 44, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Porter, E.G.; Werpy, N.M. New concepts in standing advanced diagnostic equine imaging. Vet. Clin. Equine Pract. 2014, 30, 239–268. [Google Scholar] [CrossRef]
- Werpy, N.M. Magnetic resonance imaging of the equine patient: A comparison of high-and low-field systems. Clin. Tech. Equine Pract. 2007, 6, 37–45. [Google Scholar] [CrossRef]
- Carrino, J.A.; Morrison, W.B.; Zou, K.H.; Steffen, R.T.; Snearly, W.N.; Murray, P.M. Noncontrast MR imaging and MR arthrography of the ulnar collateral ligament of the elbow: Prospective evaluation of two-dimensional pulse sequences for detection of complete tears. Skelet. Radiol. 2001, 30, 625–632. [Google Scholar] [CrossRef]
- Zanetti, M.; Bräm, J.; Hodler, J. Triangular fibrocartilage and intercarpal ligaments of the wrist: Does MR arthrography improve standard MRI? J. Magn. Reson. Imaging 1997, 7, 590–594. [Google Scholar] [CrossRef]
- Stecco, A.; Brambilla, M.; Puppi, A.M.; Lovisolo, M.; Boldorini, R.; Carriero, A. Shoulder MR arthrography: In vitro determination of optimal gadolinium dilution as a function of field strength. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2007, 25, 200–207. [Google Scholar] [CrossRef]
- Andreisek, G.; Froehlich, J.M.; Hodler, J.; Weishaupt, D.; Beutler, V.; Pfirrmann, C.W.; Boesch, C.; Nanz, D. Direct MR arthrography at 1.5 and 3.0 T: Signal dependence on gadolinium and iodine concentrations—Phantom study. Radiology 2008, 247, 706–716. [Google Scholar] [CrossRef] [PubMed]
- Murphy, S.E.; Ballegeer, E.A.; Forrest, L.J.; Schaefer, S.L. Magnetic resonance imaging findings in dogs with confirmed shoulder pathology. Vet. Surg. 2008, 37, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, S.L.; Baumel, C.A.; Gerbig, J.R.; Forrest, L.J. Direct magnetic resonance arthrography of the canine shoulder. Vet. Radiol. Ultrasound 2010, 51, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Ugas, M.A.; Huynh, B.H.; Fox, M.G.; Patrie, J.T.; Gaskin, C.M. MR arthrography: Impact of steroids, local anesthetics, and iodinated contrast material on gadolinium signal intensity in phantoms at 1.5 and 3.0 T. Radiology 2014, 272, 475–483. [Google Scholar] [CrossRef]
- Montgomery, D.D.; Morrison, W.B.; Schweitzer, M.E.; Weishaupt, D.; Dougherty, L. Effects of iodinated contrast and field strength on gadolinium enhancement: Implications for direct MR arthrography. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2002, 15, 334–343. [Google Scholar] [CrossRef]
| T2W_FSE_TRA_MI Pre-Contrast | T1W_GRE_TRA_MI Pre-Contrast | T1W_GRE_TRA_MI Post-Contrast | |
|---|---|---|---|
| TR | 1544 | 52 | 52 |
| TE | 88 | 8 | 8 |
| Flip angle | 90 | 50 | 50 |
| Echo | 8 | 1 | 1 |
| NEX | 1 | 1 | 1 |
| FOV | 170 | 171 | 171 |
| Slice thickness | 5 mm | 5 mm | 5 mm |
| Gap width | 1 mm | 1 mm | 1 mm |
| Number of slices | 8 | 8 | 8 |
| Time to run (min) | 2:10 | 2:10 | 2:10 |
| SNR | 94 | 99 | 99 |
| Spacing between slices | 6.0 | 6.0 | 6.0 |
| Lesion | Sensitivity (%) (95% ^), (n) | Specificity (%) (95% ^), (n) |
|---|---|---|
| MRI SDFT | 39% (0.22–0.59) (11) | 93% (0.83–0.98) (52) |
| MRIt SDFT | 54% (0.34–0.72) (15) | 96% (0.88–0.99) (54) |
| MRI DDFT | 34% (0.19–0.51) (13) | 100% (0.92–1) (46) |
| MRIt DDFT | 32% (0.18–0.49) (12) | 98% (0.88–0.99) (45) |
| MRI MF | 61% (0.36–0.83) (11) | 96% (0.79–0.99) (23) |
| MRIt MF | 50% (0.26–0.74) (9) | 96% (0.79–0.99) (23) |
| MRI scutum | 38% (0.22–0.56) (13) | 96% (0.86–0.99) (48) |
| MRIt scutum | 50% (0.32–0.68) (17) | 96% (0.86–0.99) (48) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aßmann, A.; Ohlerth, S.; Hartmann, S.; Torgerson, P.; Bischofberger, A. Does Direct MRI Tenography Improve the Diagnostic Performance of Low-Field MRI to Identify Artificially Created Soft-Tissue Lesions within the Equine Cadaveric Digital Flexor Tendon Sheath? Animals 2023, 13, 3772. https://doi.org/10.3390/ani13243772
Aßmann A, Ohlerth S, Hartmann S, Torgerson P, Bischofberger A. Does Direct MRI Tenography Improve the Diagnostic Performance of Low-Field MRI to Identify Artificially Created Soft-Tissue Lesions within the Equine Cadaveric Digital Flexor Tendon Sheath? Animals. 2023; 13(24):3772. https://doi.org/10.3390/ani13243772
Chicago/Turabian StyleAßmann, Anton, Stefanie Ohlerth, Silvana Hartmann, Paul Torgerson, and Andrea Bischofberger. 2023. "Does Direct MRI Tenography Improve the Diagnostic Performance of Low-Field MRI to Identify Artificially Created Soft-Tissue Lesions within the Equine Cadaveric Digital Flexor Tendon Sheath?" Animals 13, no. 24: 3772. https://doi.org/10.3390/ani13243772
APA StyleAßmann, A., Ohlerth, S., Hartmann, S., Torgerson, P., & Bischofberger, A. (2023). Does Direct MRI Tenography Improve the Diagnostic Performance of Low-Field MRI to Identify Artificially Created Soft-Tissue Lesions within the Equine Cadaveric Digital Flexor Tendon Sheath? Animals, 13(24), 3772. https://doi.org/10.3390/ani13243772




