Metabolic Responses and Resilience to Environmental Challenges in the Sedentary Batrachoid Halobatrachus didactylus (Bloch & Schneider, 1801)
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sourcing and Housing
2.2. Metabolic Rates Measurement Procedure
2.3. Standard, Routine and Maximum Metabolic Rate
2.4. Mass-Scaling Exponent
2.5. Critical Thermal Maximum (CTmax) Trial
2.6. Temperature Ramp
2.7. Critical Oxygen Tension Trial
2.8. Data Analysis
2.8.1. Metabolic Rates
2.8.2. CTmax and Acute Thermal Tolerance
2.8.3. Pcrit
3. Results
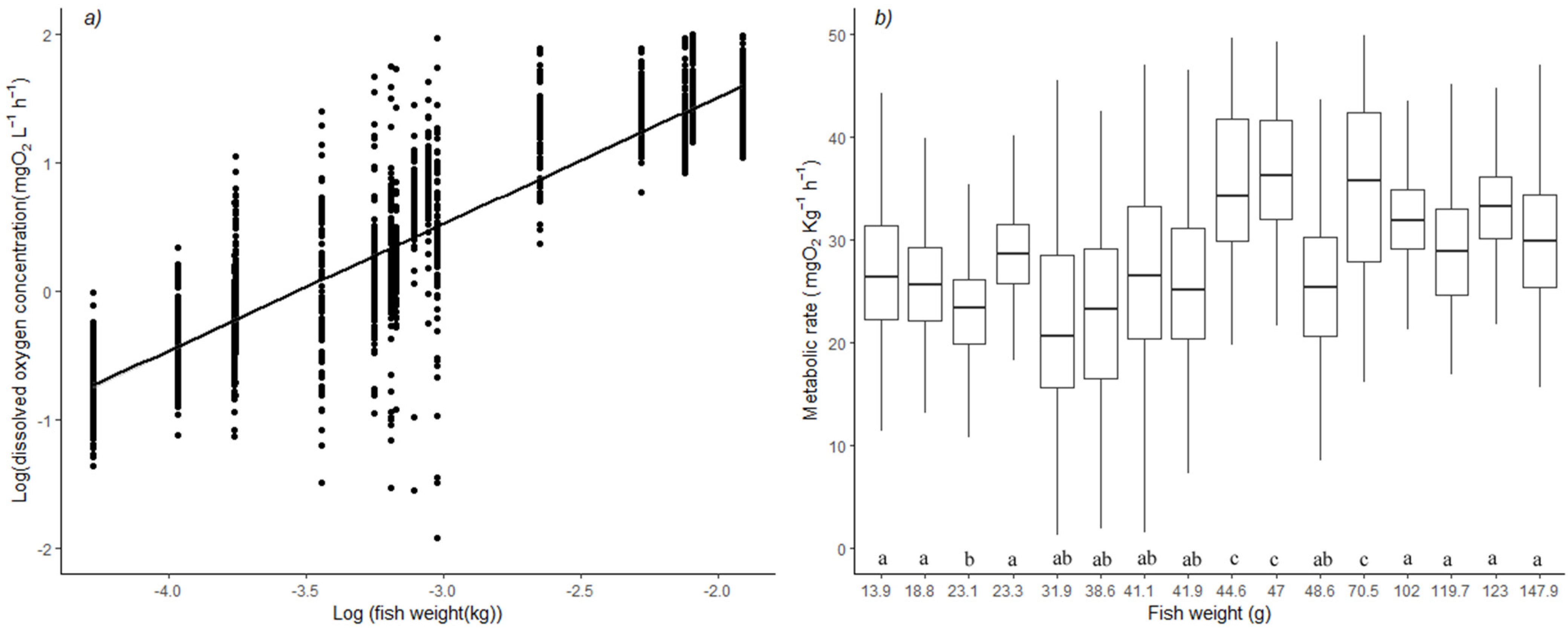
3.1. Mass-Scaling Exponent of Oxygen Consumption
3.2. Metabolic Rates and Aerobic Scope
3.3. CTmax
3.4. Temperature Ramp
3.5. Pcrit
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sunday, J.M.; Bates, A.E.; Dulvy, N.K. Thermal Tolerance and the Global Redistribution of Animals. Nat. Clim. Chang. 2012, 2, 686–690. [Google Scholar] [CrossRef]
- Pörtner, H.-O.; Karl, D.M.; Boyd, P.W.; Cheung, W.; Lluch−Cota, S.E.; Nojiri, Y.; Schmidt, D.N.; Zavialov, P.O.; Alheit, J.; Aristegui, J. Ocean Systems. In Climate Change 2014: Impacts, Adaptation, and Vulnerability. Part A: Global and Sectoral Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2014; pp. 411–484. [Google Scholar]
- Ern, R.; Norin, T.; Gamperl, A.K.; Esbaugh, A.J. Oxygen Dependence of Upper Thermal Limits in Fishes. J. Exp. Biol. 2016, 219, 3376–3383. [Google Scholar] [CrossRef] [Green Version]
- Parmesan, C.; Pörtner, H.-O.; Roberts, D.C. IPCC 2022: Summary for Policymakers. In Climate Change 2022: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2022. [Google Scholar]
- Koenigstein, S.; Mark, F.C.; Gößling−Reisemann, S.; Reuter, H.; Poertner, H.−O. Modelling Climate Change Impacts on Marine Fish Populations: Process−Based Integration of Ocean Warming, Acidification and Other Environmental Drivers. Fish Fish. 2016, 17, 972–1004. [Google Scholar] [CrossRef] [Green Version]
- Ribeiro, D.; Gkenas, C.; Gago, J.; Ribeiro, F. Variation in Diet Patterns of the Invasive Top Predator Sander lucioperca (Linnaeus, 1758) across Portuguese Basins. Water 2021, 13, 2053. [Google Scholar] [CrossRef]
- Pérez−Ruzafa, A.; Pérez−Ruzafa, I.M.; Newton, A.; Marcos, C. Coastal Lagoons: Environmental Variability, Ecosystem Complexity, and Goods and Services Uniformity. In Coasts and Estuaries; Elsevier: Amsterdam, Netherlands, 2019; pp. 253–276. [Google Scholar]
- Moyano, M.; Candebat, C.; Ruhbaum, Y.; Alvarez−Fernandez, S.; Claireaux, G.; Zambonino−Infante, J.-L.; Peck, M.A. Effects of Warming Rate, Acclimation Temperature and Ontogeny on the Critical Thermal Maximum of Temperate Marine Fish Larvae. PLoS ONE 2017, 12, e0179928. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chabot, D.; Steffensen, J.F.; Farrell, A.P. The Determination of Standard Metabolic Rate in Fishes. J. Fish Biol. 2016, 88, 81–121. [Google Scholar] [CrossRef] [PubMed]
- Lutterschmidt, W.I.; Hutchison, V.H. The Critical Thermal Maximum: History and Critique. Can. J. Zool. 1997, 75, 1561–1574. [Google Scholar] [CrossRef]
- Morgan, R.; Finnøen, M.H.; Jutfelt, F. CTmax Is Repeatable and Doesn’t Reduce Growth in Zebrafish. Sci. Rep. 2018, 8, 7099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campos, D.F.D.; Val, A.L.; Almeida−Val, V.M.F. The Influence of Lifestyle and Swimming Behavior on Metabolic Rate and Thermal Tolerance of Twelve Amazon Forest Stream Fish Species. J. Therm. Biol. 2018, 72, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Madeira, D.; Costa, P.M.; Vinagre, C.; Diniz, M.S. When Warming Hits Harder: Survival, Cellular Stress and Thermal Limits of Sparus aurata Larvae under Global Change. Marine Biology 2016, 163, 91. [Google Scholar] [CrossRef]
- Madeira, D.; Vinagre, C.; Diniz, M.S. Are Fish in Hot Water? Effects of Warming on Oxidative Stress Metabolism in the Commercial Species. Sparus Aurata. Ecol. Indic. 2016, 63, 324–331. [Google Scholar]
- Vinagre, C.; Leal, I.; Mendonça, V.; Flores, A.A. Effect of Warming Rate on the Critical Thermal Maxima of Crabs, Shrimp and Fish. J. Therm. Biol. 2015, 47, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Bennett, W.A.; Beitinger, T.L. Temperature Tolerance of the Sheepshead Minnow, Cyprinodon variegatus. Copeia 1997, 77–87. [Google Scholar] [CrossRef]
- Bennett, W.A.; Judd, F.W. Comparison of Methods for Determining Low Temperature Tolerance: Experiments with Pinfish, Lagodon rhomboides. Copeia 1992, 1059–1065. [Google Scholar] [CrossRef]
- Ultsch, G.R.; Jackson, D.C.; Moalli, R. Metabolic Oxygen Conformity among Lower Vertebrates: The Toadfish Revisited. J. Comp. Physiol. 1981, 142, 439–443. [Google Scholar] [CrossRef]
- Rogers, N.J.; Urbina, M.A.; Reardon, E.E.; McKenzie, D.J.; Wilson, R.W. A New Analysis of Hypoxia Tolerance in Fishes Using a Database of Critical Oxygen Level (Pcrit). Conserv. Physiol. 2016, 4. [Google Scholar] [CrossRef]
- Reemeyer, J.E.; Rees, B.B. Standardizing the Determination and Interpretation of Pcrit in Fishes. J. Exp. Biol. 2019, 222, jeb210633. [Google Scholar] [CrossRef] [Green Version]
- Claireaux, G.; Chabot, D. Responses by Fishes to Environmental Hypoxia: Integration through Fry’s Concept of Aerobic Metabolic Scope. J. Fish Biol. 2016, 88, 232–251. [Google Scholar] [CrossRef]
- Snyder, S.; Nadler, L.E.; Bayley, J.S.; Svendsen, M.B.S.; Johansen, J.L.; Domenici, P.; Steffensen, J.F. Effect of Closed v. Intermittent−Flow Respirometry on Hypoxia Tolerance in the Shiner Perch Cymatogaster aggregata. J. Fish Biol. 2016, 88, 252–264. [Google Scholar] [CrossRef] [Green Version]
- Wood, C.M. The Fallacy of the P Crit—Are There More Useful Alternatives? J. Exp. Biol. 2018, 221, jeb163717. [Google Scholar] [CrossRef] [Green Version]
- Seibel, B.A.; Andres, A.; Birk, M.A.; Burns, A.L.; Shaw, C.T.; Timpe, A.W.; Welsh, C.J. Oxygen Supply Capacity Breathes New Life into Critical Oxygen Partial Pressure (Pcrit). J. Exp. Biol. 2021, 224. [Google Scholar] [CrossRef]
- Gamito, S.; Pires, A.; Pita, C.; Erzini, K. Food Availability and the Feeding Ecology of Ichthyofauna of a Ria Formosa (South Portugal) Water Reservoir. Estuaries 2003, 26, 938–948. [Google Scholar] [CrossRef]
- Costa, J.L.; Costa, M.J. Distribution and Abundance of the Lusitanian Toadfish Halobatrachus didactylus (Bloch & Schneider, 1801) in Portugal with Some Remarks on Its Population Fragmentation. Rev. Biol. 2002, 20, 156–167. [Google Scholar]
- Bauchot, M.L. Poissons Osseux. In Fiches FAO d’identification Pour Les Besoins de La Pêche. (Rev. 1). Méditerranée et Mer Noire. Zone de Pêche 37; Fischer, M.S., Bauchot, M.L., Schneider, M., Eds.; Commission des Communautés Européennes and FAO: Rome, Italy, 1987; Volume 2. [Google Scholar]
- Veiga, P.; Ribeiro, J.; Gonçalves, J.M.S.; Erzini, K. Quantifying Recreational Shore Angling Catch and Harvest in Southern Portugal (North−East Atlantic Ocean): Implications for Conservation and Integrated Fisheries Management. J. Fish Biol. 2010, 76, 2216–2237. [Google Scholar] [CrossRef]
- Greenfield, D.W.; Winterbottom, R.; Collette, B.B. Review of the Toadfish Genera (Teleostei: Batrachoididae); California Academy of Sciences: San Francisco, CA, USA, 2008. [Google Scholar]
- Roux, C. Fiches FAO d’identifcation Des Espèces Pour Les Besoins de La Pêche. In Fiches Fao D’Identifcation Des Espèces Pour Les Besoins de la Pêche.; Fischer, W., Bianchi, G., Scott, W.B., Eds.; Atlantique Centreest: Zones de Pêche; Canada Fonds de Dépôt: Ottawa, ON, Canada, 1987; Volume 1. [Google Scholar]
- Muzavor, S.; Arruda, L.M.; Andrade, J.P. Roteiro Ecológico Da Ria Formosa; Peixes; Foco Editora: Faro, Portugal, 1993; Volume 2. [Google Scholar]
- Palazón−Fernández, J.L.; Arias, A.M.; Sarasquete, C. Aspects of the Reproductive Biology of the Toadfish, Halobatrachus didactylus (Schneider, 1801) (Pisces: Batrachoididae). Sci. Mar. 2001, 65, 131–138. [Google Scholar] [CrossRef] [Green Version]
- Modesto, T.; Canário, A.V. Hormonal Control of Swimbladder Sonic Muscle Dimorphism in the Lusitanian Toadfish Halobatrachus didactylus. J. Exp. Biol. 2003, 206, 3467–3477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vasconcelos, R.O.; Ladich, F. Development of Vocalization, Auditory Sensitivity and Acoustic Communication in the Lusitanian Toadfish Halobatrachus didactylus. J. Exp. Biol. 2008, 211, 502–509. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pereira, T.J.; Almeida, P.R.; Quintella, B.R.; Gronningsaeter, A.; Costa, M.J.; Marques, J.P.; Costa, J.L. Fine−Scale Behaviour of the Lusitanian Toadfish Assessed in Situ with the AccelTag. Anim. Biotelemetry 2021, 9, 1–9. [Google Scholar] [CrossRef]
- Clarke, A. Is There a Universal Temperature Dependence of Metabolism? Funct. Ecol. 2004, 18, 252–256. [Google Scholar] [CrossRef] [Green Version]
- Burton, T.; Killen, S.S.; Armstrong, J.D.; Metcalfe, N.B. What Causes Intraspecific Variation in Resting Metabolic Rate and What Are Its Ecological Consequences? Proc. R. Soc. B Biol. Sci. 2011, 278, 3465–3473. [Google Scholar] [CrossRef] [Green Version]
- Stoffels, R.J. Physiological Trade−Offs along a Fast−Slow Lifestyle Continuum in Fishes: What Do They Tell Us about Resistance and Resilience to Hypoxia? PLoS ONE 2015, 10, e0130303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Killen, S.S.; Glazier, D.S.; Rezende, E.L.; Clark, T.D.; Atkinson, D.; Willener, A.S.; Halsey, L.G. Ecological Influences and Morphological Correlates of Resting and Maximal Metabolic Rates across Teleost Fish Species. Am. Nat. 2016, 187, 592–606. [Google Scholar] [CrossRef] [Green Version]
- Baptista, V.; Silva, P.L.; Relvas, P.; Teodósio, M.A.; Leitão, F. Sea Surface Temperature Variability along the Portuguese Coast since 1950. Int. J. Climatol. 2018, 38, 1145–1160. [Google Scholar] [CrossRef]
- Ribeiro, J.; Monteiro, C.C.; Monteiro, P.; Bentes, L.; Coelho, R.; Gonçalves, J.M.; Lino, P.G.; Erzini, K. Long−Term Changes in Fish Communities of the Ria Formosa Coastal Lagoon (Southern Portugal) Based on Two Studies Made 20 Years Apart. Estuar. Coast. Shelf Sci. 2008, 76, 57–68. [Google Scholar] [CrossRef]
- Ribeiro, J.; Bentes, L.; Coelho, R.; Gonçalves, J.M.; Lino, P.G.; Monteiro, P.; Erzini, K. Seasonal, Tidal and Diurnal Changes in Fish Assemblages in the Ria Formosa Lagoon (Portugal). Estuar. Coast. Shelf Sci. 2006, 67, 461–474. [Google Scholar] [CrossRef]
- Svendsen, M.B.S.; Bushnell, P.G.; Steffensen, J.F. Design and Setup of Intermittent−Flow Respirometry System for Aquatic Organisms. J. Fish Biol. 2016, 88, 26–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zimmermann, C.; Kunzmann, A. Baseline Respiration and Spontaneous Activity of Sluggish Marine Tropical Fish of the Family Scorpaenidae. Mar. Ecol. Prog. Ser. 2001, 219, 229–239. [Google Scholar] [CrossRef]
- Kühnhold, H.; Steinmanna, N.; Huanga, Y.-H.; Indrianab, L.; Meyera, A.; Kunzmanna, A. Temperature−Induced Aerobic Capacity and Hsp70 Expression in the Sea Cucumber Holothuria svcabra. PLoS ONE 2019, 74. [Google Scholar]
- Norin, T.; Clark, T.D. Measurement and Relevance of Maximum Metabolic Rate in Fishes. J. Fish Biol. 2016, 88, 122–151. [Google Scholar] [CrossRef] [Green Version]
- Hatry, C.; Thiem, J.D.; Binder, T.R.; Hatin, D.; Dumont, P.; Stamplecoskie, K.M.; Molina, J.M.; Smokorowski, K.E.; Cooke, S.J. Comparative Physiology and Relative Swimming Performance of Three Redhorse (Moxostoma spp.) Species: Associations with Fishway Passage Success. Physiol. Biochem. Zool. 2014, 87, 148–159. [Google Scholar] [CrossRef] [Green Version]
- Andersson, M.L.; Sundberg, F.; Eklöv, P. Chasing Away Accurate Results: Exhaustive Chase Protocols Underestimate Maximum Metabolic Rate Estimates in European Perch Perca fluviatilis. J. Fish Biol. 2020, 97, 1644–1650. [Google Scholar] [CrossRef] [PubMed]
- Prinzing, T.S.; Zhang, Y.; Wegner, N.C.; Dulvy, N.K. Analytical Methods Matter Too: Establishing a Framework for Estimating Maximum Metabolic Rate for Fishes. Ecol. Evol. 2021, 11, 9987–10003. [Google Scholar] [CrossRef]
- Rangel, R.E.; Johnson, D.W. Metabolic Responses to Temperature in a Sedentary Reef Fish, the Bluebanded Goby (Lythrypnus dalli, Gilbert). J. Exp. Mar. Biol. Ecol. 2018, 501, 83–89. [Google Scholar] [CrossRef]
- Crawley, M.J. The R Book; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Clark, T.D.; Sandblom, E.; Jutfelt, F. Aerobic Scope Measurements of Fishes in an Era of Climate Change: Respirometry, Relevance and Recommendations. J. Exp. Biol. 2013, 216, 2771–2782. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fox, J. Applied Regression Analysis and Generalized Linear Models; Sage Publications: Sauzend Oaks, CA, USA, 2015. [Google Scholar]
- Callaghan, N.I.; Tunnah, L.; Currie, S.; MacCormack, T.J. Metabolic Adjustments to Short−Term Diurnal Temperature Fluctuation in the Rainbow Trout (Oncorhynchus mykiss). Physiol. Biochem. Zool. 2016, 89, 498–510. [Google Scholar] [CrossRef]
- Molina, J.M.; Finotto, L.; Walker, T.I.; Reina, R.D. The Effect of Gillnet Capture on the Metabolic Rate of Two Shark Species with Contrasting Lifestyles. J. Exp. Mar. Biol. Ecol. 2020, 526, 151354. [Google Scholar] [CrossRef]
- Ogle, D.H. Introductory Fisheries Analyses with R; Chapman and Hall/CRC: London, UK, 2018. [Google Scholar]
- Strobl, C.; Malley, J.; Tutz, G. An Introduction to Recursive Partitioning: Rationale, Application, and Characteristics of Classification and Regression Trees, Bagging, and Random Forests. Psychol. Methods 2009, 14, 323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- R Core Team, R. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Barber, M.L.; Walsh, P.J. Interactions of Acid−Base Status and Nitrogen Excretion and Metabolism in the Ureogenic Teleost Opsanus beta. J. Exp. Biol. 1993, 185, 87–105. [Google Scholar] [CrossRef]
- Gilmour, K.M.; Perry, S.F.; Wood, C.M.; Henry, R.P.; Laurent, P.; Pärt, P.; Walsh, P.J. Nitrogen Excretion and the Cardiorespiratory Physiology of the Gulf Toadfish, Opsanus beta. Physiol. Zool. 1998, 71, 492–505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amorim, M.C.P.; McCracken, M.L.; Fine, M.L. Metabolic Costs of Sound Production in the Oyster Toadfish, Opsanus tau. Can. J. Zool. 2002, 80, 830–838. [Google Scholar] [CrossRef]
- McDonald, M.D.; Gilmour, K.M.; Walsh, P.J.; Perry, S.F. Cardiovascular and Respiratory Reflexes of the Gulf Toadfish (Opsanus beta) during Acute Hypoxia. Respir. Physiol. Neurobiol. 2010, 170, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Bucking, C.; Edwards, S.L.; Tickle, P.; Smith, C.P.; McDonald, M.D.; Walsh, P.J. Immunohistochemical Localization of Urea and Ammonia Transporters in Two Confamilial Fish Species, the Ureotelic Gulf Toadfish (Opsanus beta) and the Ammoniotelic Plainfin Midshipman (Porichthys Notatus). Cell Tissue Res. 2013, 352, 623–637. [Google Scholar] [CrossRef] [PubMed]
- Hall, F.G. The Influence of Varying Oxygen Tensions upon the Rate of Oxygen Consumption in Marine Fishes. Am. J. Physiol.−Leg. Content 1929, 88, 212–218. [Google Scholar] [CrossRef] [Green Version]
- Haschemeyer, A.E. Oxygen Consumption of Temperature−Acclimated Toadfish, Opsanus tau. Biol. Bull. 1969, 136, 28–32. [Google Scholar] [CrossRef]
- Kunzmann, A. First Record of Stonefish Metabolism: Baseline Respiration and Spontaneous Activity of Tropical Marine Synanceia verrucosa. Mar. Biol. Res. 2021, 17, 475–485. [Google Scholar] [CrossRef]
- Silva, G.; Almeida, P.R.; Costa, M.J.; Costa, J.L. Activity End Diet of “Halobatrachus didactylus” (Bloch & Schneider, 1801) Adults in the Tagus Estuary. Thalass. Int. J. Mar. Sci. 2000, 16, 21–25. [Google Scholar]
- Costa, J.L. A Biologia Do Xarroco, Halobatrachus didactylus (Bloch & Schneider, 1801), Eo Seu Papel Na Estruturação e Funcionamento Das Comunidades Em Que Se Insere; Universidade de Lisboa: Lisbon, Portugal, 2004. [Google Scholar]
- Killen, S.S.; Atkinson, D.; Glazier, D.S. The Intraspecific Scaling of Metabolic Rate with Body Mass in Fishes Depends on Lifestyle and Temperature. Ecol. Lett. 2010, 13, 184–193. [Google Scholar] [CrossRef]
- Marras, S.; Claireaux, G.; McKenzie, D.J.; Nelson, J.A. Individual Variation and Repeatability in Aerobic and Anaerobic Swimming Performance of European Sea Bass, Dicentrarchus labrax. J. Exp. Biol. 2010, 213, 26–32. [Google Scholar] [CrossRef] [Green Version]
- Norin, T.; Malte, H. Repeatability of Standard Metabolic Rate, Active Metabolic Rate and Aerobic Scope in Young Brown Trout during a Period of Moderate Food Availability. J. Exp. Biol. 2011, 214, 1668–1675. [Google Scholar] [CrossRef] [Green Version]
- Svendsen, J.C.; Genz, J.; Anderson, W.G.; Stol, J.A.; Watkinson, D.A.; Enders, E.C. Evidence of Circadian Rhythm, Oxygen Regulation Capacity, Metabolic Repeatability and Positive Correlations between Forced and Spontaneous Maximal Metabolic Rates in Lake Sturgeon Acipenser fulvescens. PLoS ONE 2014, 9, e94693. [Google Scholar] [CrossRef] [Green Version]
- Pörtner, H.O.; Knust, R. Climate Change Affects Marine Fishes through the Oxygen Limitation of Thermal Tolerance. Science 2007, 315, 95–97. [Google Scholar] [CrossRef] [Green Version]
- Farrell, A.P. Cardiorespiratory Performance during Prolonged Swimming Tests with Salmonids: A Perspective on Temperature Effects and Potential Analytical Pitfalls. Philos. Trans. R. Soc. B Biol. Sci. 2007, 362, 2017–2030. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eliason, E.J.; Clark, T.D.; Hague, M.J.; Hanson, L.M.; Gallagher, Z.S.; Jeffries, K.M.; Gale, M.K.; Patterson, D.A.; Hinch, S.G.; Farrell, A.P. Differences in Thermal Tolerance among Sockeye Salmon Populations. Science 2011, 332, 109–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Volkoff, H.; Rønnestad, I. Effects of Temperature on Feeding and Digestive Processes in Fish. Temperature 2020, 7, 307–320. [Google Scholar] [CrossRef] [PubMed]
- Di Santo, V.; Lobel, P.S. Size Affects Digestive Responses to Increasing Temperature in Fishes: Physiological Implications of Being Small under Climate Change. Mar. Ecol. 2016, 37, 813–820. [Google Scholar] [CrossRef]
- Hölker, F. The Metabolic Rate of Roach in Relation to Body Size and Temperature. J. Fish Biol. 2003, 62, 565–579. [Google Scholar] [CrossRef]
- Cotter, J.C.; Pereira, T.J.; Costa, M.J.; Costa, J.L. Distribution, Abundance, Population Structure and Activity of Halobatrachus didactylus in the Tagus Estuary (Portugal) and Adjacent Coastal Waters. J. Mar. Biol. Assoc. U. K. 2013, 93, 405–412. [Google Scholar] [CrossRef]
- Little, A.G.; Loughland, I.; Seebacher, F. What Do Warming Waters Mean for Fish Physiology and Fisheries? J. Fish Biol. 2020, 97, 328–340. [Google Scholar] [CrossRef]
- Diaz, R.J.; Rosenberg, R. Spreading Dead Zones and Consequences for Marine Ecosystems. Science 2008, 321, 926–929. [Google Scholar] [CrossRef]
- Rabalais, N.N.; Diaz, R.J.; Levin, L.A.; Turner, R.E.; Gilbert, D.; Zhang, J. Dynamics and Distribution of Natural and Human−Caused Hypoxia. Biogeosciences 2010, 7, 585–619. [Google Scholar] [CrossRef] [Green Version]
- Clark, T.D. Respirometry. In Methods for Fish Biology; Midway, S., Hasler, C., Chakrabarty, P., Eds.; American Fisheries Society: Bethesda, Maryland, 2022; pp. 247–268. [Google Scholar]
- Craig, P.M.; Fitzpatrick, J.L.; Walsh, P.J.; Wood, C.M.; McClelland, G.B. Coping with Aquatic Hypoxia: How the Plainfin Midshipman (Porichthys notatus) Tolerates the Intertidal Zone. Environ. Biol. Fishes 2014, 97, 163–172. [Google Scholar] [CrossRef]
- LeMoine, C.M.; Bucking, C.; Craig, P.M.; Walsh, P.J. Divergent Hypoxia Tolerance in Adult Males and Females of the Plainfin Midshipman (Porichthys notatus). Physiol. Biochem. Zool. 2014, 87, 325–333. [Google Scholar] [CrossRef] [Green Version]
- Johnson, D.L.; Metcalf, M.T. Causes and Controls of Freshwater Drum Mortality during Transportation. Trans. Am. Fish. Soc. 1982, 111, 58–62. [Google Scholar] [CrossRef]
- Rees, B.B.; Sudradjat, F.A.; Love, J.W. Acclimation to Hypoxia Increases Survival Time of Zebrafish, Danio rerio, during Lethal Hypoxia. J. Exp. Zool. 2001, 289, 266–272. [Google Scholar] [CrossRef]
- Wu, R.S. Effects of Hypoxia on Fish Reproduction and Development. In Fish Physiology; Elsevier: Amsterdam, The Netherlands, 2009; Volume 27, pp. 79–141. [Google Scholar]
- Cook, K.V.; Lennox, R.J.; Hinch, S.G.; Cooke, S.J. Fish out of Water: How Much Air Is Too Much? Fisheries 2015, 40, 452–461. [Google Scholar] [CrossRef]
- Martin, K.L. Aerial Release of CO2 and Respiratory Exchange Ratio in Intertidal Fishes out of Water. Environ. Biol. Fishes 1993, 37, 189–196. [Google Scholar] [CrossRef]
- Martin, K.L. Time and Tide Wait for No Fish: Intertidal Fishes out of Water. Environ. Biol. Fishes 1995, 44, 165–181. [Google Scholar] [CrossRef]
- Houpt, N.; Borowiec, B.G.; Bose, A.P.; Brown, N.A.; Scott, G.R.; Balshine, S. Parental Males of the Plainfin Midshipman Are Physiologically Resilient to the Challenges of the Intertidal Zone. Physiol. Biochem. Zool. 2020, 93, 111–128. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, G.E. Gill Remodeling in Fish–a New Fashion or an Ancient Secret? J. Exp. Biol. 2007, 210, 2403–2409. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brix, O.; Clements, K.D.; Wells, R.M.G. Haemoglobin Components and Oxygen Transport in Relation to Habitat Distribution in Triplefin Fishes (Tripterygiidae). J. Comp. Physiol. B 1999, 169, 329–334. [Google Scholar] [CrossRef]
- Farrell, A.P.; Richards, J.G. Defining Hypoxia: An Integrative Synthesis of the Responses of Fish to Hypoxia. In Fish Physiology; Elsevier: Amsterdam, The Netherlands, 2009; Volume 27, pp. 487–503. [Google Scholar]
- Dwyer, G.K.; Stoffels, R.J.; Pridmore, P.A. Morphology, Metabolism and Behaviour: Responses of Three Fishes with Different Lifestyles to Acute Hypoxia. Freshw. Biol. 2014, 59, 819–831. [Google Scholar] [CrossRef]
- Sokolova, I. Bioenergetics in Environmental Adaptation and Stress Tolerance of Aquatic Ectotherms: Linking Physiology and Ecology in a Multi−Stressor Landscape. J. Exp. Biol. 2021, 224, jeb236802. [Google Scholar] [CrossRef]
- Delaney, R.G.; Lahiri, S.; Fishman, A.P. Aestivation of the African Lungfish Protopterus aethiopicus: Cardiovascular and Respiratory Functions. J. Exp. Biol. 1974, 61, 111–128. [Google Scholar] [CrossRef] [PubMed]
- Stecyk, J.A.; Galli, G.L.; Shiels, H.A.; Farrell, A.P. Cardiac Survival in Anoxia−Tolerant Vertebrates: An Electrophysiological Perspective. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2008, 148, 339–354. [Google Scholar] [CrossRef] [PubMed]
- Richards, J.G. Metabolic Rate Suppression as a Mechanism for Surviving Environmental Challenge in Fish. Aestivation 2010, 49, 113–139. [Google Scholar]
- Seibel, B.A. Critical Oxygen Levels and Metabolic Suppression in Oceanic Oxygen Minimum Zones. J. Exp. Biol. 2011, 214, 326–336. [Google Scholar] [CrossRef] [Green Version]
- Hochachka, P.W. Oxygen—A Key Regulatory Metabolite in Metabolic Defense against Hypoxia. Am. Zool. 1997, 37, 595–603. [Google Scholar] [CrossRef] [Green Version]
- Guppy, M.; Withers, P. Metabolic Depression in Animals: Physiological Perspectives and Biochemical Generalizations. Biol. Rev. 1999, 74, 1–40. [Google Scholar] [CrossRef]
- Bickler, P.E.; Buck, L.T. Hypoxia Tolerance in Reptiles, Amphibians, and Fishes: Life with Variable Oxygen Availability. Annu. Rev. Physiol. 2007, 69, 145–170. [Google Scholar] [CrossRef]
- Nilsson, G.E.; Renshaw, G.M. Hypoxic Survival Strategies in Two Fishes: Extreme Anoxia Tolerance in the North European Crucian Carp and Natural Hypoxic Preconditioning in a Coral−Reef Shark. J. Exp. Biol. 2004, 207, 3131–3139. [Google Scholar] [CrossRef] [Green Version]
- Frick, L.H.; Walker, T.I.; Reina, R.D. Immediate and Delayed Effects of Gill−Net Capture on Acid–Base Balance and Intramuscular Lactate Concentration of Gummy Sharks, Mustelus antarcticus. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2012, 162, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Regan, M.D.; Mandic, M.; Dhillon, R.S.; Lau, G.Y.; Farrell, A.P.; Schulte, P.M.; Seibel, B.A.; Speers−Roesch, B.; Ultsch, G.R.; Richards, J.G. Don’t Throw the Fish out with the Respirometry Water. J. Exp. Biol. 2019, 222, jeb200253. [Google Scholar] [CrossRef] [Green Version]
- Rijnsdorp, A.D.; Peck, M.A.; Engelhard, G.H.; Möllmann, C.; Pinnegar, J.K. Resolving the Effect of Climate Change on Fish Populations. ICES J. Mar. Sci. 2009, 66, 1570–1583. [Google Scholar] [CrossRef]
- Froese, R.; Papaioannou, E.; Scotti, M. Climate Change or Mismanagement? Environ. Biol. Fishes 2022, 105, 1363–1380. [Google Scholar] [CrossRef]
- Félix, P.M.; Amorim, M.C.P.; Pereira, T.J.; Fonseca, P.J.; Sousa−Santos, C.; Costa, J.L. Feeding Ecology and Life−History Strategy of Nesting Males in a Fish with Long Parental Care, Lusitanian Toadfish (Halobatrachus didactylus, Batrachoididae). J. Mar. Biol. Assoc. U. K. 2016, 96, 657–665. [Google Scholar] [CrossRef]
- Campos, M.C.; Costa, J.L.; Quintella, B.R.; Costa, M.J.; Almeida, P.R. Activity and Movement Patterns of the Lusitanian Toadfish Inferred from Pressure−Sensitive Data−Loggers in the Mira Estuary (Portugal). Fish. Manag. Ecol. 2008, 15, 449–458. [Google Scholar] [CrossRef]
- Robalo, J.I.; Crespo, A.M.; Castilho, R.; Francisco, S.M.; Amorim, M.C.; Almada, V.C. Are Local Extinctions and Recolonizations Continuing at the Colder Limits of Marine Fish Distributions? Halobatrachus didactylus (Bloch & Schneider, 1801), a Possible Candidate. Mar. Biol. 2013, 160, 2461–2467. [Google Scholar]
- Bañón, R.; Tejerina, R.; Morales, X.; Alonso−Fernandez, A.; Barros−García, D.; Carlos, A.D. Unusual Occurrences of Fishes along the Northeast Atlantic: New Biological and Distributional Data. Mediterr. Mar. Sci. 2019, 20, 189–196. [Google Scholar] [CrossRef] [Green Version]
- Argyriou, A.; Kaspiris, P.; Vertsiotis, A. New Record of Halobatrachus didactylus (Bloch and Schneider, 1801) in the Coastal Waters of the Ionian Sea, Western Greece. J. Appl. Ichthyol. 2006, 22, 238–239. [Google Scholar] [CrossRef]
- Di Santo, V. Intraspecific Variation in Physiological Performance of a Benthic Elasmobranch Challenged by Ocean Acidification and Warming. J. Exp. Biol. 2016, 219, 1725–1733. [Google Scholar] [CrossRef] [Green Version]
- McArley, T.J.; Hickey, A.J.; Herbert, N.A. Chronic Warm Exposure Impairs Growth Performance and Reduces Thermal Safety Margins in the Common Triplefin Fish (Forsterygion lapillum). J. Exp. Biol. 2017, 220, 3527–3535. [Google Scholar] [CrossRef] [Green Version]
- Pandey, P.; Hooda, O.K.; Kumar, S. Impact of Heat Stress and Hypercapnia on Physiological, Hematological, and Behavioral Profile of Tharparkar and Karan Fries Heifers. Vet. World 2017, 10, 1146. [Google Scholar] [CrossRef] [Green Version]
- Hilton, Z.; Wellenreuther, M.; Clements, K.D. Physiology Underpins Habitat Partitioning in a Sympatric Sister−Species Pair of Intertidal Fishes. Funct. Ecol. 2008, 22, 1108–1117. [Google Scholar] [CrossRef]
- Johnston, I.A.; Clarke, A.; Ward, P. Temperature and Metabolic Rate in Sedentary Fish from the Antarctic, North Sea and Indo−West Pacific Ocean. Mar. Biol. 1991, 109, 191–195. [Google Scholar] [CrossRef]
- Sloman, K.A.; Mandic, M.; Todgham, A.E.; Fangue, N.A.; Subrt, P.; Richards, J.G. The Response of the Tidepool Sculpin, Oligocottus maculosus, to Hypoxia in Laboratory, Mesocosm and Field Environments. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2008, 149, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Speers−Roesch, B.; Norin, T.; Driedzic, W.R. The Benefit of Being Still: Energy Savings during Winter Dormancy in Fish Come from Inactivity and the Cold, Not from Metabolic Rate Depression. Proc. R. Soc. B Biol. Sci. 2018, 285, 20181593. [Google Scholar] [CrossRef] [PubMed] [Green Version]

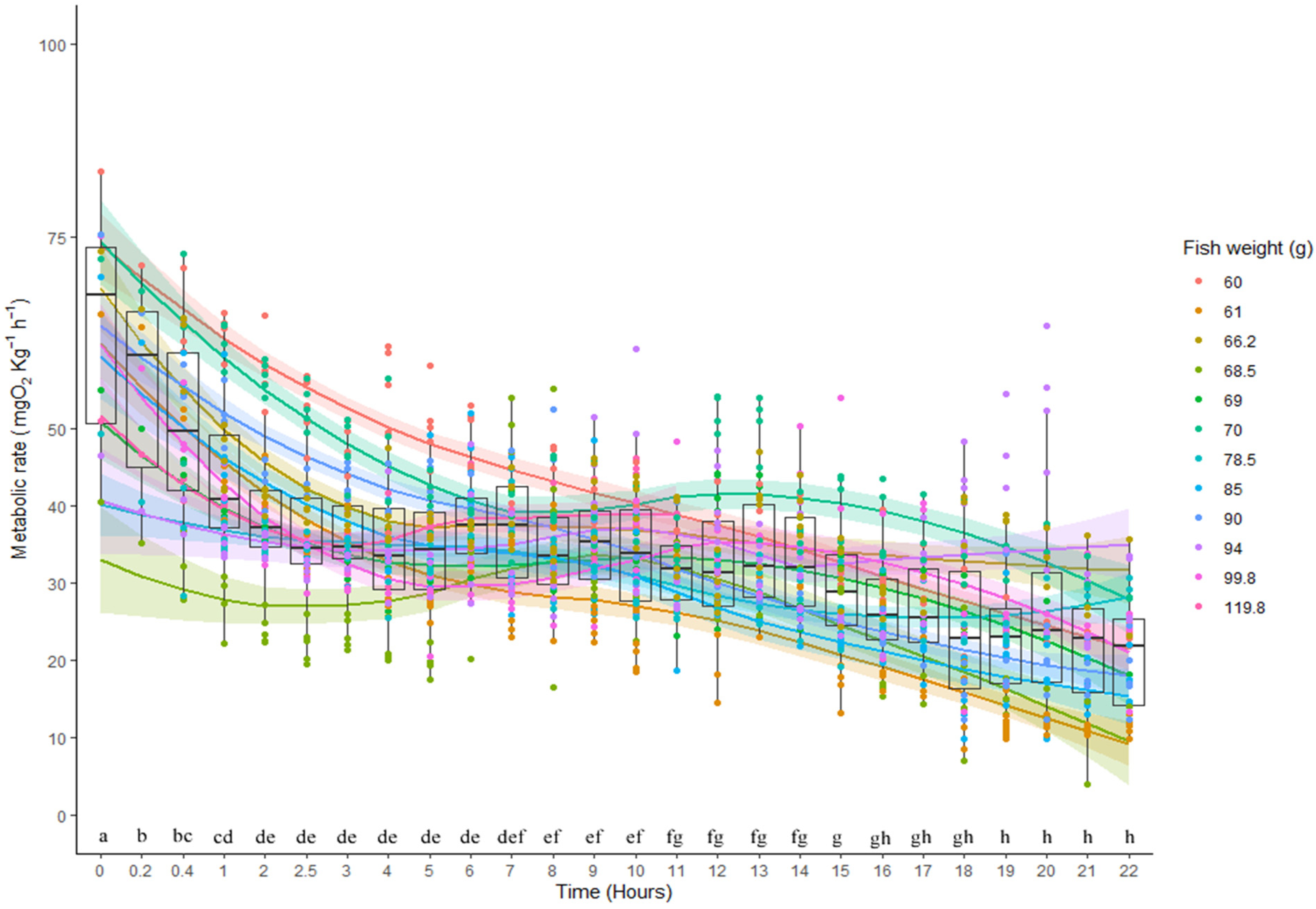



| Fish No. | Weight | MMR | RMR | SMR | %M/S | %R/S | Time |
|---|---|---|---|---|---|---|---|
| 1 | 60 | 83.4 | 29.7 | 22.6 | 73.0 | 24.0 | 18.0 |
| 2 | 61 | 64.9 | 17.8 | 10.6 | 83.7 | 40.6 | 23.0 |
| 3 | 66.2 | 73.1 | 35.8 | 28.5 | 61.0 | 20.4 | 15.0 |
| 4 | 68.5 | 40.6 | 24.1 | 12.4 | 69.4 | 48.6 | 30.0 |
| 5 | 69 | 55.1 | 28.1 | 19.1 | 65.3 | 32.0 | 15.0 |
| 6 | 70 | 72.1 | 37.0 | 29.1 | 59.6 | 21.3 | 25.0 |
| 7 | 78.5 | 49.4 | 27.1 | 22.9 | 53.7 | 15.4 | 26.0 |
| 8 | 85 | 69.8 | 22.0 | 13.5 | 80.7 | 38.5 | 19.0 |
| 9 | 90 | 75.3 | 24.5 | 16.0 | 78.8 | 34.6 | 28.0 |
| 10 | 94 | 46.6 | 33.5 | 27.2 | 41.7 | 18.8 | 16.0 |
| 11 | 99.8 | 75.1 | 29.6 | 23.4 | 68.9 | 21.2 | 24.0 |
| 12 | 119.8 | 51.1 | 38.9 | 18.3 | 64.2 | 53.0 | 21.0 |
| Median | 74.3 | 67.3 | 28.9 | 20.8 | 69.1 | 27.8 | 22.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molina, J.M.; Kunzmann, A.; Reis, J.P.; Guerreiro, P.M. Metabolic Responses and Resilience to Environmental Challenges in the Sedentary Batrachoid Halobatrachus didactylus (Bloch & Schneider, 1801). Animals 2023, 13, 632. https://doi.org/10.3390/ani13040632
Molina JM, Kunzmann A, Reis JP, Guerreiro PM. Metabolic Responses and Resilience to Environmental Challenges in the Sedentary Batrachoid Halobatrachus didactylus (Bloch & Schneider, 1801). Animals. 2023; 13(4):632. https://doi.org/10.3390/ani13040632
Chicago/Turabian StyleMolina, Juan Manuel, Andreas Kunzmann, João Pena Reis, and Pedro Miguel Guerreiro. 2023. "Metabolic Responses and Resilience to Environmental Challenges in the Sedentary Batrachoid Halobatrachus didactylus (Bloch & Schneider, 1801)" Animals 13, no. 4: 632. https://doi.org/10.3390/ani13040632






