Growth and Welfare Status of Giant Freshwater Prawn (Macrobrachium rosenbergii) Post-Larvae Reared in Aquaponic Systems and Fed Diets including Enriched Black Soldier Fly (Hermetia illucens) Prepupae Meal
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Feeding Substrate Preparation and Insect Rearing
2.2. Prawns’ Diets Production
2.3. Aquaponic Systems and Experimental Design
2.4. Survival Rate, Growth Performances, and Morphological Features
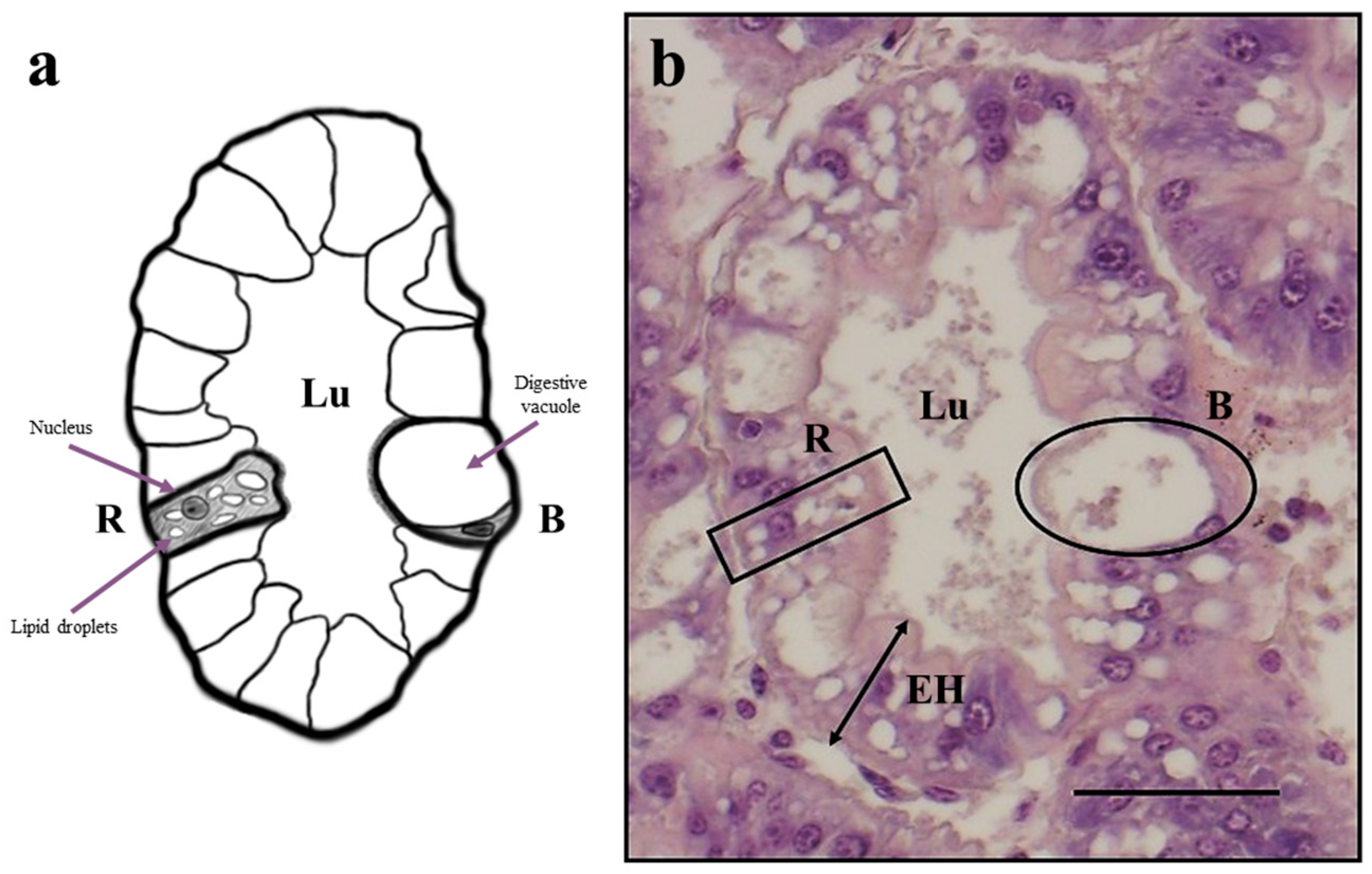
2.5. Histological Analysis
2.6. RNA Extraction and cDNA Synthesis
2.7. Real-Time PCR
2.8. Physical and Chemical Analyses
2.9. Statistical Analysis
3. Results
3.1. Water Quality Parameters
3.2. Survival Rate, Growth Performance and Morphological Features
3.3. Histological Analysis
3.4. Gene Expression
3.5. Physical and Chemical Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gupta, A.; Sehgal, H.S.; Sehgal, G.K. Growth and carcass composition of giant freshwater prawn, Macrobrachium rosenbergii (De Man), fed different isonitrogenous and isocaloric diets. Aquac. Res. 2007, 38, 1355–1363. [Google Scholar] [CrossRef]
- Schwantes, V.S.; Diana, J.S.; Yi, Y. Social, economic, and production characteristics of giant river prawn Macrobrachium rosenbergii culture in Thailand. Aquaculture 2009, 287, 120–127. [Google Scholar] [CrossRef]
- Bakhsh, H.K.; Chopin, T. Water quality and nutrient aspects in recirculating aquaponic production of the freshwater prawn, Macrobrachium rosenbergii and the lettuce, Lactuca sativa. Int. J. Recirc. Aquac. 2011, 12, 13–33. [Google Scholar] [CrossRef]
- Paulraj, A.; Devi, K.; Kathiravan; Mohamed, M.J.; Musthafa, M.S.; Peixoto, S.; Altaff, K. Role of mesophilic fungal biomass and diversity in the pond soil for the productivity of freshwater prawn Macrobrachium rosenbergii. Aquaculture 2021, 545, 737250. [Google Scholar] [CrossRef]
- Kamarudin, M.S.; Roustaian, P. Growth and fatty acid composition of freshwater prawn, Macrobrachium rosenbergii, larvae fed diets containing various ratios of cod liver oil–corn oil mixture. J. Appl. Ichthyol. 2002, 18, 148–153. [Google Scholar] [CrossRef]
- Nguyen, N.H.Y.; Trinh, L.T.; Chau, D.T.; Baruah, K.; Lundh, T.; Kiessling, A. Spent brewer’s yeast as a replacement for fishmeal in diets for giant freshwater prawn (Macrobrachium rosenbergii), reared in either clear water or a biofloc environment. Aquac. Nutr. 2019, 25, 970–979. [Google Scholar] [CrossRef]
- Anh, N.T.N.; Hien, T.T.T.; Mathieu, W.; Van Hoa, N.; Sorgeloos, P. Effect of fishmeal replacement with Artemia biomass as a protein source in practical diets for the giant freshwater prawn Macrobrachium rosenbergii. Aquac. Res. 2009, 40, 669–680. [Google Scholar] [CrossRef]
- Bijoy, V.M.; Sabu, S.; Harikrishnan, M. Fish meal replacement with squilla (Oratosquilla nepa, Latreille) silage in a practical diet for the juvenile giant freshwater prawn, Macrobrachium rosenbergii de man, 1879. Aquac. Int. 2018, 26, 1229–1245. [Google Scholar] [CrossRef]
- Du, L.; Niu, C.J. Effects of dietary substitution of soya bean meal for fish meal on consumption, growth, and metabolism of juvenile giant freshwater prawn, Macrobrachium rosenbergii. Aquac. Nutr. 2003, 9, 139–143. [Google Scholar] [CrossRef]
- Hu, X.; Yang, H.L.; Yan, Y.Y.; Zhang, C.X.; Ye, J.D.; Lu, K.L.; Hu, L.H.; Zhang, J.J.; Ruan, L.; Sun, Y.Z. Effects of fructooligosaccharide on growth, immunity and intestinal microbiota of shrimp (Litopenaeus vannamei) fed diets with fish meal partially replaced by soybean meal. Aquac. Nutr. 2019, 25, 194–204. [Google Scholar] [CrossRef]
- Tidwell, J.H.; Webster, C.D.; Yancey, D.H.; D’Abramo, L.R. Partial and total replacement of fish meal with soybean meal and distillers’ by-products in diets for pond culture of the freshwater prawn (Macrobrachium rosenbergii). Aquaculture 1993, 118, 119–130. [Google Scholar] [CrossRef]
- Zhu, Y.Z.; Yang, G.H. Preliminary studies on suitable dietary protein level and soybean meal as dietary protein source of the freshwater prawn Macrobrachium rosenbergii -. Fish. Sci. Technol. 1995, 22, 125–1278. [Google Scholar]
- Ding, Z.; Zhou, D.; Zheng, J.; Chen, X.; Kong, Y.; Qi, C.; Liu, Y.; Tang, Q.; Yang, G.; Ye, J. Replacing fishmeal with soybean meal affects survival, antioxidant capacity, intestinal microbiota, and mRNA expression of TOR and S6K1 in Macrobrachium rosenbergii. J. Oceanol. Limnol. 2022, 40, 805–817. [Google Scholar] [CrossRef]
- Gao, X.; Jiang, Z.; Zhang, S.; Chen, Q.; Tong, S.; Liu, X.; Jiang, Q.; Yang, H.; Wei, W.; Zhang, X. Transcriptome analysis and immune-related genes expression reveals the immune responses of Macrobrachium rosenbergii infected by Enterobacter cloacae. Fish Shellfish Immunol. 2020, 101, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Pasookhush, P.; Hindmarch, C.; Sithigorngul, P.; Longyant, S.; Bendena, W.G.; Chaivisuthangkura, P. Transcriptomic analysis of Macrobrachium rosenbergii (giant fresh water prawn) post-larvae in response to M. rosenbergii nodavirus (MrNV) infection: De novo assembly and functional annotation. BMC Genom. 2019, 20, 762. [Google Scholar] [CrossRef] [PubMed]
- Kaleo, I.V.; Gao, Q.; Liu, B.; Sun, C.; Zhou, Q.; Zhang, H.; Shan, F.; Xiong, Z.; Bo, L.; Song, C. Effects of Moringa oleifera leaf extract on growth performance, physiological and immune response, and related immune gene expression of Macrobrachium rosenbergii with Vibrio anguillarum and ammonia stress. Fish Shellfish Immunol. 2019, 89, 603–613. [Google Scholar] [CrossRef]
- Miao, S.; Han, B.; Zhao, C.; Hu, J.; Zhu, J.; Zhang, X.; Sun, L. Effects of dietary Pediococcus acidilactici GY2 single or combined with Saccharomyces cerevisiae or/and β-glucan on the growth, innate immunity response and disease resistance of Macrobrachium rosenbergii. Fish Shellfish Immunol. 2020, 98, 68–76. [Google Scholar] [CrossRef]
- Mohan, K.; Muralisankar, T.; Uthayakumar, V.; Chandirasekar, R.; Karthick Rajan, D. Dietary Ganoderma lucidum polysaccharides to enhance the growth, immune response and disease resistance of freshwater prawn Macrobrachium rosenbergii. Aquac. Rep. 2019, 14, 100203. [Google Scholar] [CrossRef]
- Gamboa-Delgado, J.; Márquez-Reyes, J.M. Potential of microbial-derived nutrients for aquaculture development. Rev. Aquac. 2018, 10, 224–246. [Google Scholar] [CrossRef]
- Somayeh, S.M.; Jiun, Z.; Loh, Y.; Mousavi, S.; Loh, J.-Y.; Zahedinezhad, S. A review on insect meals in aquaculture: The immunomodulatory and physiological effects. Int. Aquat. Res. 2020, 12, 100–115. [Google Scholar] [CrossRef]
- Maliwat, G.C.F.; Velasquez, S.F.; Buluran, S.M.D.; Tayamen, M.M.; Ragaza, J.A. Growth and immune response of pond-reared giant freshwater prawn Macrobrachium rosenbergii post larvae fed diets containing Chlorella vulgaris. Aquac. Fish. 2021, 6, 465–470. [Google Scholar] [CrossRef]
- Maliwat, G.C.; Velasquez, S.; Robil, J.L.; Chan, M.; Traifalgar, R.F.; Tayamen, M.; Ragaza, J.A. Growth and immune response of giant freshwater prawn Macrobrachium rosenbergii (De Man) postlarvae fed diets containing Chlorella vulgaris (Beijerinck). Aquac. Res. 2017, 48, 1666–1676. [Google Scholar] [CrossRef]
- Radhakrishnan, S.; Seenivasan, C.; Muralisankar, T. Effect of dietary replacement of fishmeal with Chlorella vulgaris on growth performance, energy utilization and digestive enzymes in Macrobrachium rosenbergii postlarvae. Int. J. Fish. Aquac. 2015, 7, 62–70. [Google Scholar] [CrossRef]
- Feng, P.; He, J.; Lv, M.; Huang, G.; Chen, X.; Yang, Q.; Wang, J.; Wang, D.; Ma, H. Effect of dietary Tenebrio molitor protein on growth performance and immunological parameters in Macrobrachium rosenbergii. Aquaculture 2019, 511, 734247. [Google Scholar] [CrossRef]
- Ahmad Ansari, F.; Nasr, M.; Guldhe, A.; Kumar Gupta, S.; Rawat, I.; Bux, F. Techno-economic feasibility of algal aquaculture via fish and biodiesel production pathways: A commercial-scale application. Sci. Total Environ. 2020, 704, 135259. [Google Scholar] [CrossRef]
- Alfiko, Y.; Xie, D.; Astuti, R.T.; Wong, J.; Wang, L. Insects as a feed ingredient for fish culture: Status and trends. Aquac. Fish. 2022, 7, 166–178. [Google Scholar] [CrossRef]
- Borrelli, L.; Varriale, L.; Dipineto, L.; Pace, A.; Menna, L.F.; Fioretti, A. Insect derived lauric acid as promising alternative strategy to antibiotics in the antimicrobial resistance scenario. Front. Microbiol. 2021, 12, 330. [Google Scholar] [CrossRef]
- Xia, J.; Ge, C.; Yao, H. Antimicrobial peptides from black soldier fly (Hermetia illucens) as potential antimicrobial factors representing an alternative to antibiotics in livestock farming. Animals 2021, 11, 1937. [Google Scholar] [CrossRef]
- Zarantoniello, M.; Randazzo, B.; Cardinaletti, G.; Truzzi, C.; Chemello, G.; Riolo, P.; Olivotto, I. Possible dietary effects of insect-based diets across zebrafish (Danio rerio) generations: A multidisciplinary study on the larval phase. Animals 2021, 11, 751. [Google Scholar] [CrossRef]
- Liland, N.S.; Biancarosa, I.; Araujo, P.; Biemans, D.; Bruckner, C.G.; Waagbø, R.; Torstensen, B.E.; Lock, E.J. Modulation of nutrient composition of black soldier fly (Hermetia illucens) larvae by feeding seaweed-enriched media. PLoS One 2017, 12, e0183188. [Google Scholar] [CrossRef]
- Truzzi, C.; Giorgini, E.; Annibaldi, A.; Antonucci, M.; Illuminati, S.; Scarponi, G.; Riolo, P.; Isidoro, N.; Conti, C.; Zarantoniello, M.; et al. Fatty acids profile of black soldier fly (Hermetia illucens): Influence of feeding substrate based on coffee-waste silverskin enriched with microalgae. Anim. Feed Sci. Technol. 2020, 259, 114309. [Google Scholar] [CrossRef]
- Chemello, G.; Zarantoniello, M.; Randazzo, B.; Gioacchini, G.; Truzzi, C.; Cardinaletti, G.; Riolo, P.; Olivotto, I. Effects of black soldier fly (Hermetia illucens) enriched with Schizochytrium sp. on zebrafish (Danio rerio) reproductive performances. Aquaculture 2022, 550, 737853. [Google Scholar] [CrossRef]
- Ratti, S.; Zarantoniello, M.; Chemello, G.; Giammarino, M.; Palermo, F.A.; Cocci, P.; Mosconi, G.; Tignani, M.V.; Pascon, G.; Cardinaletti, G.; et al. Spirulina-enriched substrate to rear black soldier fly (Hermetia illucens) prepupae as alternative aquafeed ingredient for rainbow trout (Oncorhynchus mykiss) diets: Possible effects on zootechnical performances, gut and liver health status, and fillet quality. Animals 2023, 13, 173. [Google Scholar] [PubMed]
- Rosas, V.T.; Poersch, L.H.; Romano, L.A.; Tesser, M.B. Feasibility of the use of Spirulina in aquaculture diets. Rev. Aquac. 2019, 11, 1367–1378. [Google Scholar] [CrossRef]
- Aracati, M.F.; Rodrigues, L.F.; de Oliveira, S.L.; Rodrigues, R.A.; Conde, G.; Cavalcanti, E.N.F.; Borba, H.; Charlie-Silva, I.; Fernandes, D.C.; Eto, S.F.; et al. Astaxanthin improves the shelf-life of tilapia fillets stored under refrigeration. J. Sci. Food Agric. 2022, 102, 4287–4295. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, F.O.; Tramonte, V.L.C.G.; Parisenti, J.; Lima-Garcia, J.F.; Maraschin, M.; Da Silva, E.L. Litopenaeus vannamei muscle carotenoids versus astaxanthin: A comparison of antioxidant activity and in vitro protective effects against lipid peroxidation. Food Biosci. 2015, 9, 12–19. [Google Scholar] [CrossRef]
- Pulido-Rodriguez, L.F.; Cardinaletti, G.; Secci, G.; Randazzo, B.; Bruni, L.; Cerri, R.; Olivotto, I.; Tibaldi, E.; Parisi, G. Appetite regulation, growth performances and fish quality are modulated by alternative dietary protein ingredients in gilthead sea bream (Sparus aurata) culture. Animals 2021, 11, 1919. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, S.; Belal, I.E.H.; Seenivasan, C.; Muralisankar, T.; Bhavan, P.S. Impact of fishmeal replacement with Arthrospira platensis on growth performance, body composition and digestive enzyme activities of the freshwater prawn, Macrobrachium rosenbergii. Aquac. Rep. 2016, 3, 35–44. [Google Scholar] [CrossRef]
- Radhakrishnan, S.; Saravana Bhavan, P.; Seenivasan, C.; Shanthi, R.; Muralisankar, T. Replacement of fishmeal with Spirulina platensis, Chlorella vulgaris and Azolla pinnata on non-enzymatic and enzymatic antioxidant activities of Macrobrachium rosenbergii. J. Basic Appl. Zool. 2014, 67, 25–33. [Google Scholar] [CrossRef]
- Mccallum, M.L.; Weston, S.D.; Tilahun, Y. Performance of early juvenile giant river prawns (Macrobrachium rosenbergii) fed fish, soybean, shrimp and four insect based diets while under low temperature stress. Proc. Okla. Acad. Sci 2020, 100, 5–15. [Google Scholar]
- Vogt, G. Functional cytology of the hepatopancreas of decapod crustaceans. J. Morphol. 2019, 280, 1405–1444. [Google Scholar] [CrossRef]
- Fadhilah, N.; Estim, A.; Fatihah, S.N. Water quality and growth performance of giant freshwater prawn, Macrobrachium rosenbergii and green bean, Phaseolus vulgaris in aquaponics system at different flow rates. Trans. Sci. Technol. 2019, 6, 1–8. [Google Scholar]
- Rebecca, A.A.; Bhavan, P.S. Growth performance of Macrobrachium rosenbergii post larvae fed with vegetable wastes and palmolein supplemented formulated feeds. Recent Res. Sci. Technol. 2011, 3, 69–76. [Google Scholar]
- Makkar, H.P.S.; Tran, G.; Heuzé, V.; Ankers, P. State-of-the-art on use of insects as animal feed. Anim. Feed Sci. Technol. 2014, 197, 1–33. [Google Scholar] [CrossRef]
- Barragan-Fonseca, K.B.; Dicke, M.; van Loon, J.J.A. Influence of larval density and dietary nutrient concentration on performance, body protein, and fat contents of black soldier fly larvae (Hermetia illucens). Entomol. Exp. Appl. 2018, 166, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Spranghers, T.; Ottoboni, M.; Klootwijk, C.; Ovyn, A.; Deboosere, S.; De Meulenaer, B.; Michiels, J.; Eeckhout, M.; De Clercq, P.; De Smet, S. Nutritional composition of black soldier fly (Hermetia illucens) prepupae reared on different organic waste substrates. J. Sci. Food Agric. 2017, 97, 2594–2600. [Google Scholar] [CrossRef] [PubMed]
- Diener, S.; Zurbrügg, C.; Tockner, K. Conversion of organic material by black soldier fly larvae: Establishing optimal feeding rates. Waste Manag. Res. 2009, 27, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Osimani, A.; Ferrocino, I.; Corvaglia, M.R.; Roncolini, A.; Milanović, V.; Garofalo, C.; Aquilanti, L.; Riolo, P.; Ruschioni, S.; Jamshidi, E.; et al. Microbial dynamics in rearing trials of Hermetia illucens larvae fed coffee silverskin and microalgae. Food Res. Int. 2021, 140, 110028. [Google Scholar] [CrossRef] [PubMed]
- Goda, A.M.A.S. Effect of dietary protein and lipid levels and protein-energy ratio on growth indices, feed utilization and body composition of freshwater prawn, Macrobrachium rosenbergii (de Man 1879) post larvae. Aquac. Res. 2008, 39, 891–901. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of Official Analytical Chemists, 20th ed.; George, W., Latimer, A.A.V.V., Jr., Eds.; AOAC: Washington, DC, USA, 2006; ISBN 0-935584-87-0. [Google Scholar]
- Nozzi, V.; Graber, A.; Schmautz, Z.; Mathis, A.; Junge, R. Nutrient management in aquaponics: Comparison of three approaches for cultivating lettuce, mint and mushroom herb. Agronomy 2018, 8, 27. [Google Scholar] [CrossRef]
- Posadas, B.C. Effects of two pelletized feed formulations on experimental freshwater prawn, Macrobrachium rosenbergii, pond production, processing, and costs. J. Appl. Aquac. 2004, 16, 155–165. [Google Scholar] [CrossRef]
- Randazzo, B.; Rolla, L.; Ofelio, C.; Planas, M.; Gioacchini, G.; Vargas, A.; Giorgini, E.; Olivotto, I. The influence of diet on the early development of two seahorse species (H. guttulatus and H. reidi): Traditional and innovative approaches. Aquaculture 2018, 490, 75–90. [Google Scholar] [CrossRef]
- Olivotto, I.; Mosconi, G.; Maradonna, F.; Cardinali, M.; Carnevali, O. Diplodus sargus interrenal-pituitary response: Chemical communication in stressed fish. Gen. Comp. Endocrinol. 2002, 127, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Olivotto, I.; Yasumasu, S.; Gioacchini, G.; Maradonna, F.; Cionna, C.; Carnevali, O. Cloning and expression of high choriolytic enzyme, a component of the hatching enzyme system, during embryonic development of the marine ornamental fish Chrysiptera parasema. Mar. Biol. 2004, 145, 1235–1241. [Google Scholar] [CrossRef]
- Gao, X.; Zhou, Y.; Zhu, X.; Tang, H.; Li, X.; Jiang, Q.; Wei, W.; Zhang, X. Enterobacter cloacae: A probable etiological agent associated with slow growth in the giant freshwater prawn Macrobrachium rosenbergii. Aquaculture 2021, 530, 735826. [Google Scholar] [CrossRef]
- Ding, Z.; Zhang, Y.; Ye, J.; Du, Z.; Kong, Y. An evaluation of replacing fish meal with fermented soybean meal in the diet of Macrobrachium nipponense: Growth, nonspecific immunity, and resistance to Aeromonas hydrophila. Fish Shellfish Immunol. 2015, 44, 295–301. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Srinivasan, S.; Xiong, Y.L.; Decker, E.A. Inhibition of protein and lipid oxidation in beef heart surimi-like material by antioxidants and combinations of pH, NaCl, and buffer type in the washing media. J. Agric. Food Chem. 1996, 44, 119–125. [Google Scholar] [CrossRef]
- Vyncke, W. Direct determination of the thiobarbituric acid value in trichloracetic acid extracts of fish as a measure of oxidative rancidity. Fette Seifen Anstrichm. 1970, 72, 1084–1087. [Google Scholar] [CrossRef]
- Hahn, T.; Roth, A.; Febel, E.; Fijalkowska, M.; Schmitt, E.; Arsiwalla, T.; Zibek, S. New methods for high-accuracy insect chitin measurement. J. Sci. Food Agric. 2018, 98, 5069–5073. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Zarantoniello, M.; Randazzo, B.; Secci, G.; Notarstefano, V.; Giorgini, E.; Lock, E.J.; Parisi, G.; Olivotto, I. Application of laboratory methods for understanding fish responses to black soldier fly (Hermetia illucens) based diets. J. Insects Food Feed 2022, 8, 1173–1195. [Google Scholar] [CrossRef]
- He, Y.; Zhang, N.; Wang, A.; Wang, S.; Che, Y.; Huang, S.; Yi, Q.; Ma, Y.; Jiang, Y. Positive effects of replacing commercial feeds by fresh black soldier fly (Hermetia illucens) larvae in the diets of Pacific white shrimp (Litopenaeus vannamei): Immune enzyme, water quality, and intestinal microbiota. Front. Mar. Sci. 2022, 9, 987363. [Google Scholar] [CrossRef]
- He, Y.; Liu, X.; Zhang, N.; Wang, S.; Wang, A.; Zuo, R.; Jiang, Y. Replacement of commercial feed with fresh black soldier fly (Hermetia illucens) larvae in Pacific white shrimp (Litopenaeus vannamei). Aquac. Nutr. 2022, 2022, 9130400. [Google Scholar] [CrossRef]
- Wang, G.; Peng, K.; Hu, J.; Mo, W.; Wei, Z.; Huang, Y. Evaluation of defatted Hermetia illucens larvae meal for Litopenaeus vannamei: Effects on growth performance, nutrition retention, antioxidant and immune response, digestive enzyme activity and hepatic morphology. Aquac. Nutr. 2021, 27, 986–997. [Google Scholar] [CrossRef]
- Kledal, P.R.; Thorarinsdottir, R. Aquaponics: A commercial niche for sustainable modern aquaculture. In Sustainable Aquaculture; Springer International Publishing: Cham, Switzerland, 2018; pp. 173–190. [Google Scholar]
- Kralik, B.; Weisstein, F.; Meyer, J.; Neves, K.; Anderson, D.; Kershaw, J. From water to table: A multidisciplinary approach comparing fish from aquaponics with traditional production methods. Aquaculture 2022, 552, 737953. [Google Scholar] [CrossRef]
- National Research Council. Nutrient Requirements of Fish and Shrimp; The National Academies Press: Washington, DC, USA, 2011. [CrossRef]
- Méndez-Martínez, Y.; Yamasaki-Granados, S.; García-Guerrero, M.U.; Martínez-Córdova, L.R.; Rivas-Vega, M.E.; Arcos-Ortega, F.G.; Cortés-Jacinto, E. Effect of dietary protein content on growth rate, survival and body composition of juvenile cauque river prawn, Macrobrachium americanum (Bate 1868). Aquac. Res. 2017, 48, 741–751. [Google Scholar] [CrossRef]
- Cummins, V.C.; Rawles, S.D.; Thompson, K.R.; Velasquez, A.; Kobayashi, Y.; Hager, J.; Webster, C.D. Evaluation of black soldier fly (Hermetia illucens) larvae meal as partial or total replacement of marine fish meal in practical diets for Pacific white shrimp (Litopenaeus vannamei). Aquaculture 2017, 473, 337–344. [Google Scholar] [CrossRef]
- Chen, Y.; Chi, S.; Zhang, S.; Dong, X.; Yang, Q.; Liu, H.; Tan, B.; Xie, S. Effect of black soldier fly (Hermetia illucens) larvae meal on lipid and glucose metabolism of Pacific white shrimp Litopenaeus vannamei. Br. J. Nutr. 2022, 128, 1674–1688. [Google Scholar] [CrossRef]
- Zhao, M.; Yao, D.; Li, S.; Zhang, Y.; Aweya, J.J. Effects of ammonia on shrimp physiology and immunity: A review. Rev. Aquac. 2020, 12, 2194–2211. [Google Scholar] [CrossRef]
- De Lourdes Cobo, M.; Sonnenholzner, S.; Wille, M.; Sorgeloos, P. Ammonia tolerance of Litopenaeus vannamei (Boone) larvae. Aquac. Res. 2014, 45, 470–475. [Google Scholar] [CrossRef]
- Sun, Z.; Hao, S.; Gong, Y.; Zhang, M.; Aweya, J.J.; Tran, N.T.; Zhang, Y.; Ma, H.; Li, S. Dual oxidases participate in the regulation of hemolymph microbiota homeostasis in mud crab Scylla paramamosain. Dev. Comp. Immunol. 2018, 89, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Orlando, P.; Giardinieri, A.; Lucci, P.; Nartea, A.; Balzano, M.; Pacetti, D.; Frega, N.G.; Silvestri, S.; Tiano, L. Impact of traditional and mild oven cooking treatments on antioxidant compounds levels and oxidative status of Atlantic salmon (Salmo salar) fillets. LWT 2020, 134, 110011. [Google Scholar] [CrossRef]
- Randazzo, B.; Zarantoniello, M.; Gioacchini, G.; Giorgini, E.; Truzzi, C.; Notarstefano, V.; Cardinaletti, G.; Huyen, K.T.; Carnevali, O.; Olivotto, I. Can insect-based diets affect zebrafish (Danio rerio) reproduction? A multidisciplinary study. Zebrafish 2020, 17, 287–304. [Google Scholar] [CrossRef] [PubMed]
- Glencross, B.D.; Smith, D.M.; Thomas, M.R.; Williams, K.C. The effect of dietary n-3 and n-6 fatty acid balance on the growth of the prawn Penaeus monodon. Aquac. Nutr. 2002, 8, 43–51. [Google Scholar] [CrossRef]
- Ding, Z.; Chen, L.; Du, Z.; Jiang, H.; Sun, S.; Li, E. A mixture of fish oil and soybean oil as a dietary lipid source prevents precocity and promotes growth in juvenile Macrobrachium nipponense (De Haan). Aquac. Res. 2014, 45, 1567–1572. [Google Scholar] [CrossRef]
- Cavalli, R.O.; Lavens, P.; Sorgeloos, P. Performance of Macrobrachium rosenbergii broodstock fed diets with different fatty acid composition. Aquaculture 1999, 179, 387–402. [Google Scholar] [CrossRef]
- D’Abramo, L.R.; Sheen, S.S. Polyunsaturated fatty acid nutrition in juvenile freshwater prawn Macrobrachium rosenbergii. Aquaculture 1993, 115, 63–86. [Google Scholar] [CrossRef]
- Deering, M.J.; Fielder, D.R.; Hewitt, D.R. Growth and fatty acid composition of juvenile leader prawns, Penaeus monodon, fed different lipids. Aquaculture 1997, 151, 131–141. [Google Scholar] [CrossRef]
- New, M.B.; Valenti, W.C. Freshwater Prawn Culture: The Farming of Macrobrachium Rosenbergii; Wiley-Blackwell: New York, NY, USA, 2007; ISBN 9780470999554. [Google Scholar]
- Johnston, D.J.; Calvert, K.A.; Crear, B.J.; Carter, C.G. Dietary carbohydrate/lipid ratios and nutritional condition in juvenile southern rock lobster, Jasus edwardsii. Aquaculture 2003, 220, 667–682. [Google Scholar] [CrossRef]
- Vogt, G.; Quinitio, E.T.; Pascual, F.P. Leucaena leucocephala leaves in formulated feed for Penaeus monodon: A concrete example of the application of histology in nutrition research. Aquaculture 1986, 59, 209–234. [Google Scholar] [CrossRef]
- Pourmozaffar, S.; Hajimoradloo, A.; Paknejad, H.; Rameshi, H. Effect of dietary supplementation with apple cider vinegar and propionic acid on hemolymph chemistry, intestinal microbiota and histological structure of hepatopancreas in white shrimp, Litopenaeus vannamei. Fish Shellfish Immunol. 2019, 86, 900–905. [Google Scholar] [CrossRef]
- Wang, Y.; Li, M.; Filer, K.; Xue, Y.; Ai, Q.; Mai, K. Replacement of fish oil with a DHA-rich Schizochytrium meal on growth performance, activities of digestive enzyme and fatty acid profile of Pacific white shrimp (Litopenaeus vannamei) larvae. Aquac. Nutr. 2017, 23, 1113–1120. [Google Scholar] [CrossRef]
- Franceschini-Vicentini, I.B.; Ribeiro, K.; Papa, L.P.; Marques Junior, J.; Vicentini, C.A.; Valenti, P.M.C.M. Histoarchitectural features of the hepatopancreas of the Amazon River prawn Macrobrachium amazonicum. Int. J. Morphol. 2009, 27, 121–128. [Google Scholar] [CrossRef]
- Purna, G.; Nagaraju, C. Is methyl farnesoate a crustacean hormone? Aquaculture 2007, 272, 39–54. [Google Scholar] [CrossRef]
- Homola, E.; Chang, E.S. Methyl Farnesoate: Crustacean juvenile hormone in search of functions. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 1997, 117, 347–356. [Google Scholar] [CrossRef]
- Lee, K.J.; Watson, R.D.; Roer, R.D. Molt-inhibiting hormone mRNA levels and ecdysteroid titer during a molt cycle of the blue crab, Callinectes sapidus. Biochem. Biophys. Res. Commun. 1998, 249, 624–627. [Google Scholar] [CrossRef]
- Chen, X.; Gao, Q.; Cheng, H.; Peng, F.; Wang, C.; Xu, B. Molecular cloning and expression pattern of the juvenile hormone epoxide hydrolase gene from the giant freshwater prawn Macrobrachium rosenbergii during larval development and the moult cycle. Aquac. Res. 2021, 52, 3890–3899. [Google Scholar] [CrossRef]
- Loizzi, R.F. Interpretation of crayfish hepatopancreatic function based on fine structural analysis of epithelial cell lines and muscle network. Z. Für Zellforsch. Und Mikrosk. Anat. 1971, 113, 420–440. [Google Scholar] [CrossRef]
- Hu, K.J.; Leung, P.C. Food digestion by cathepsin L and digestion-related rapid cell differentiation in shrimp hepatopancreas. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2007, 146, 69–80. [Google Scholar] [CrossRef]
- Hopkin, S.P.; Nott, J.A. Studies on the digestive cycle of the shore crab Carcinus maenas (L.) with special reference to the b cells in the hepatopancreas. J. Mar. Biol. Assoc. United Kingd. 1980, 60, 891–907. [Google Scholar] [CrossRef]
- Xiao, X.; Han, D.; Zhu, X.; Yang, Y.; Xie, S.; Huang, Y. Effect of dietary cornstarch levels on growth performance, enzyme activity and hepatopancreas histology of juvenile red swamp crayfish, Procambarus clarkii (Girard). Aquaculture 2014, 426–427, 112–119. [Google Scholar] [CrossRef]
- Jahromi, S.T.; Pourmozaffar, S.; Jahanbakhshi, A.; Rameshi, H.; Gozari, M.; Khodadadi, M.; Sohrabipour, J.; Behzadi, S.; Bazrkar, N.; Nahavandi, R.; et al. Effect of different levels of dietary Sargassum cristaefolium on growth performance, hematological parameters, histological structure of hepatopancreas and intestinal microbiota of Litopenaeus vannamei. Aquaculture 2021, 533, 736130. [Google Scholar] [CrossRef]
- Kroeckel, S.; Harjes, A.G.E.; Roth, I.; Katz, H.; Wuertz, S.; Susenbeth, A.; Schulz, C. When a turbot catches a fly: Evaluation of a pre-pupae meal of the Black Soldier Fly (Hermetia illucens) as fish meal substitute-Growth performance and chitin degradation in juvenile turbot (Psetta maxima). Aquaculture 2012, 364–365, 345–352. [Google Scholar] [CrossRef]
- Renna, M.; Schiavone, A.; Gai, F.; Dabbou, S.; Lussiana, C.; Malfatto, V.; Prearo, M.; Capucchio, M.T.; Biasato, I.; Biasibetti, E.; et al. Evaluation of the suitability of a partially defatted black soldier fly (Hermetia illucens L.) larvae meal as ingredient for rainbow trout (Oncorhynchus mykiss Walbaum) diets. J. Anim. Sci. Biotechnol. 2017, 8, 57. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, R.; Sánchez-López, A.; Leal, R.S.; Martínez-Llorens, S.; Oliva-Teles, A.; Peres, H. Black soldier fly (Hermetia illucens) pre-pupae meal as a fish meal replacement in diets for European seabass (Dicentrarchus labrax). Aquaculture 2017, 476, 79–85. [Google Scholar] [CrossRef]
- Kumar, P.; Sahu, N.P.; Saharan, N.; Reddy, A.K.; Kumar, S. Effect of dietary source and level of chitin on growth and survival of post-larvae Macrobrachium rosenbergii. J. Appl. Ichthyol. 2006, 22, 363–368. [Google Scholar] [CrossRef]
- Zhang, X.; Yuan, J.; Li, F.; Xiang, J. Chitin synthesis and degradation in crustaceans: A genomic view and application. Mar. Drugs 2021, 19, 153. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Martínez, Y.; García-Guerrero, M.U.; Arcos-Ortega, F.G.; Martínez-Córdova, L.R.; Yamasaki-Granados, S.; Pérez-Rodríguez, J.C.; Cortés-Jacinto, E. Effect of different ratios of dietary protein-energy on growth, body proximal composition, digestive enzyme activity, and hepatopancreas histology in Macrobrachium americanum (Bate, 1868) prawn juveniles. Aquaculture 2018, 485, 1–11. [Google Scholar] [CrossRef]
- Zhang, N.N.; Ma, Q.Q.; Fan, W.J.; Xing, Q.; Zhao, Y.L.; Chen, L.Q.; Ye, J.Y.; Zhang, M.L.; Du, Z.Y. Effects of the dietary protein to energy ratio on growth, feed utilization and body composition in Macrobrachium nipponense. Aquac. Nutr. 2017, 23, 313–321. [Google Scholar] [CrossRef]
- Rőszer, T. The invertebrate midintestinal gland (“hepatopancreas”) is an evolutionary forerunner in the integration of immunity and metabolism. Cell Tissue Res. 2014, 358, 685–695. [Google Scholar] [CrossRef]
- Ribeiro, K.; Papa, L.P.; Vicentini, C.A.; Franceschini-Vicentini, I.B. The ultrastructural evaluation of digestive cells in the hepatopancreas of the Amazon River prawn, Macrobrachium amazonicum. Aquac. Res. 2016, 47, 1251–1259. [Google Scholar] [CrossRef]
- Bouallegui, Y. A comprehensive review on crustaceans’ immune system with a focus on freshwater crayfish in relation to crayfish plague disease. Front. Immunol. 2021, 12, 1753. [Google Scholar] [CrossRef]
- Young Lee, S.; Soüderhaäll, K. Early events in crustacean innate immunity. Fish Shellfish Immunol. 2002, 12, 421–437. [Google Scholar] [CrossRef]
- Pan, D.; He, N.; Yang, Z.; Liu, H.; Xu, X. Differential gene expression profile in hepatopancreas of WSSV-resistant shrimp (Penaeus japonicus) by suppression subtractive hybridization. Dev. Comp. Immunol. 2005, 29, 103–112. [Google Scholar] [CrossRef]
- Zhao, Z.Y.; Yin, Z.X.; Weng, S.P.; Guan, H.J.; Li, S.D.; Xing, K.; Chan, S.M.; He, J.G. Profiling of differentially expressed genes in hepatopancreas of white spot syndrome virus-resistant shrimp (Litopenaeus vannamei) by suppression subtractive hybridisation. Fish Shellfish Immunol. 2007, 22, 520–534. [Google Scholar] [CrossRef]
- Kumar, V.; Roy, S.; Behera, B.K.; Das, B.K. Heat shock proteins (Hsps) in cellular homeostasis: A promising tool for health management in crustacean aquaculture. Life 2022, 12, 1777. [Google Scholar] [CrossRef]


| HM0 | HM3 | HM20 | |
|---|---|---|---|
| Ingredients | |||
| Fish meal | 400 | 388 | 317 |
| Soy protein concentrate | 60 | 60 | 60 |
| Hermetia illucens meal | - | 13 | 88 |
| Wheat bran | 190 | 190 | 190 |
| Wheat meal | 150 | 150 | 150 |
| Rice bran | 115 | 115 | 115 |
| Fish oil | 40 | 39 | 35 |
| Soya lecithin | 10 | 10 | 10 |
| Mineral and vitamin premix # | 15 | 15 | 15 |
| Binder | 20 | 20 | 10 |
| Proximate composition (% DM) | |||
| Dry matter | 92.3 | 93.8 | 92.5 |
| Crude protein | 36.1 | 36.0 | 35.8 |
| Crude lipid | 9.2 | 9.7 | 9.6 |
| Ash | 14.2 | 13.4 | 13.2 |
| Gross energy (MJ/kg) | 18.8 | 18.9 | 18.9 |
| FA profile (g/100g of total FAME) | |||
| C12:0 | 0.16 | 0.80 | 4.67 |
| C14:0 | 5.03 | 5.34 | 5.33 |
| C16:0 | 18.97 | 20.78 | 19.49 |
| C16:1n7 | 5.60 | 5.63 | 6.02 |
| C18:0 | 3.98 | 4.36 | 3.86 |
| C18:1n9 | 14.09 | 14.11 | 14.00 |
| C18:1n7 | 2.38 | 2.38 | 2.29 |
| C18:2n6, LNA | 11.15 | 11.23 | 11.72 |
| C18:3n3, ALA | 1.58 | 1.50 | 1.42 |
| C18:4n3 | 1.81 | 1.63 | 1.29 |
| C20:1n9 | 2.79 | 2.71 | 3.30 |
| C20:5n3, EPA | 7.44 | 6.53 | 5.21 |
| C22:1n11 | 3.68 | 3.50 | 4.69 |
| C22:6n3, DHA | 12.67 | 11.14 | 8.23 |
| ΣSFA | 30.02 | 33.46 | 36.15 |
| ΣMUFA | 29.72 | 29.50 | 31.60 |
| Σn6 PUFA | 12.78 | 12.67 | 13.07 |
| Σn3 PUFA | 25.15 | 22.23 | 17.30 |
| Σn4 PUFA | 1.39 | 1.39 | 1.26 |
| α-tocopherol and carotenoids content (mg/kg) | |||
| α-tocopherol | <LOD | 2.22 | 12.24 |
| β-carotene | <LOD | <LOD | 6.58 |
| zeaxanthin | <LOD | <LOD | 2.48 |
| Parameter | Score | Description |
|---|---|---|
| + | Scarce | |
| B cells’ relative abundance | ++ | Diffuse |
| +++ | Highly abundant | |
| + | Scarce | |
| R cells’ relative abundance | ++ | Diffuse |
| +++ | Highly abundant | |
| + | Scarce | |
| R cells’ lipid droplets abundance | ++ | Diffuse |
| +++ | Highly abundant |
| Gene | Forward Primer (5′-3′) | Reverse Primer (5′-3′) | AT (°C) | ID Number | References |
|---|---|---|---|---|---|
| jheh | GCCATTGTTGACGAAGCC | GCCACCCTGGAGGTAGAA | 58 | KM886343 | [56] |
| chit3 | GGGCTTGGCTGGTTGTAT | GGTGGAGGTGGAGTTGGA | 58 | LT574899 | [56] |
| catL | CCTCTTGGTCGTCGCCTTAG | CCTCTTGGTCGTCGCCTTAG | 59 | KY474042 | [56] |
| hsp90 | GAAGGAAAGGGACAAGGA | GGTCCATAAAGGCTTGGT | 58 | GU319963.1 | [57] |
| α2m | CTCGGCCATCTTATCCGTATG | GGGAGCGAAGTTGAGCATGT | 58 | ABK60046 | [24] |
| 18s | TCCGTAAGGACCTGTATGCC | TCGGGAGGTGCGATGATTTT | 57 | AY651918.2 | [16] |
| β-actin | CTGTTACGGGTGACGGAGAA | TCGGAAGAGTCCCGCATT | 57 | GQ131934 | [56] |
| HM0 | HM3 | HM20 | p Value | |
|---|---|---|---|---|
| SR (%) | 82.8 ± 0.9 | 82.2 ± 0.9 | 81.3 ± 1.1 | 0.242 |
| IBW (g/prawn) | 0.10 ± 0.01 | 0.10 ± 0.02 | 0.10 ± 0.02 | 0.368 |
| FBW (g/prawn) | 0.34 ± 0.03 | 0.35 ± 0.03 | 0.33 ± 0.01 | 0.606 |
| RGR (%) | 245.0 ± 12.1 | 253.7 ± 14.51 | 235.4 ± 5.78 | 0.226 |
| SGR (% day−1) | 2.53 ± 0.10 | 2.69 ± 0.09 | 2.52 ± 0.03 | 0.031 |
| MW (%) | 39.5 ± 2.4 | 39.6 ± 3.1 | 40.3 ± 3.0 | 0.053 |
| HEW (%) | 53.0 ± 4.0 a | 52.1 ± 3.5 b | 55.5 ± 3.8 b | <0.001 |
| K | 1.29 ± 0.2 | 1.27 ± 0.2 | 1.26 ± 0.2 | 0.701 |
| HM0 | HM3 | HM20 | |
|---|---|---|---|
| B cells’ relative abundance | ++ | +++ | +++ |
| R cells’ relative abundance | ++ | ++ | ++ |
| R cells’ lipid droplets abundance | +++ | +++ | +++ |
| Tubule diameter | 135.6 ± 6.1 | 134.6 ± 5.2 | 136.4 ± 6.3 |
| Epithelium height | 63.9 ± 3.1 | 64.2 ± 2.1 | 67.1 ± 2.2 |
| HM0 | HM3 | HM20 | p Value | |
|---|---|---|---|---|
| pH | 6.72 ± 0.07 | 6.63 ± 0.07 | 6.60 ± 0.05 | 0.189 |
| Moisture | 76.9 ± 0.4 | 76.7 ± 1.0 | 77.2 ± 0.6 | 0.550 |
| Chitin | 12.2 ± 0.7 | 12.2 ± 0.7 | 13.3 ± 0.5 | 0.056 |
| HM0 | HM3 | HM20 | p Value | |
|---|---|---|---|---|
| Total lipids | 5.41 ± 0.06 | 4.87 ± 0.32 | 5.19 ± 0.33 | 0.050 |
| Fatty acids | ||||
| C14:0 | 1.63 ± 0.06 | 1.58 ± 0.03 | 1.66 ± 0.09 | 0.258 |
| C16:0 | 23.4 ± 0.39 b | 22.56 ± 0.18 a | 22.14 ± 0.21 a | <0.001 |
| C16:1n7 | 2.07 ± 0.04 a | 2.32 ± 0.12 b | 2.22 ± 0.10 ab | 0.0141 |
| C18:0 | 9.13 ± 0.11 a | 9.04 ± 0.08 a | 9.48 ± 0.10 b | <0.001 |
| C18:1n9 | 15.60 ± 0.14 b | 15.34 ± 0.21 b | 14.80 ± 0.26 a | <0.001 |
| C18:1n7 | 3.72 ± 0.05 a | 3.93 ± 0.04 b | 4.03 ± 0.09 b | <0.001 |
| C18:2n6, LNA | 8.47 ± 0.17 a | 8.20 ± 0.17 a | 9.02 ± 0.12 b | <0.001 |
| C18:3n3, ALA | 0.77 ± 0.01 a | 1.03 ± 0.02 c | 0.82 ± 0.01 b | <0.001 |
| C20:1n9 | 1.02 ± 0.03 b | 0.88 ± 0.01 a | 1.19 ± 0.04 c | <0.001 |
| C20:4n6, ARA | 3.65 ± 0.10 a | 4.64 ± 0.25 b | 3.89 ± 0.19 a | <0.001 |
| C20:5n3, EPA | 16.63 ± 0.33 | 16.49 ± 0.06 | 16.99 ± 0.47 | 0.141 |
| C22:6n3, DHA | 7.89 ± 0.20 b | 7.55 ± 0.08 ab | 7.19 ± 0.22 a | 0.00125 |
| ΣSFA | 37.10 ± 0.54 | 36.42 ± 0.22 | 36.57 ± 0.29 | 0.0694 |
| ΣMUFA | 23.23 ± 0.25 | 23.38 ± 0.14 | 23.19 ± 0.40 | 0.61 |
| Σn3 PUFA | 26.15 ± 0.50 | 25.82 ± 0.24 | 25.81 ± 0.73 | 0.613 |
| Σn6 PUFA | 13.08 ± 0.10 a | 13.95 ± 0.13 b | 14.03 ± 0.15 b | <0.001 |
| HM0 | HM3 | HM20 | p Value | |
|---|---|---|---|---|
| CD | 0.21 ± 0.02 | 0.23 ± 0.04 | 0.21 ± 0.04 | 0.71 |
| TBARS | 0.56 ± 0.19 b | 0.50 ± 0.10 ab | 0.28 ± 0.06 a | 0.0321 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zarantoniello, M.; Chemello, G.; Ratti, S.; Pulido-Rodríguez, L.F.; Daniso, E.; Freddi, L.; Salinetti, P.; Nartea, A.; Bruni, L.; Parisi, G.; et al. Growth and Welfare Status of Giant Freshwater Prawn (Macrobrachium rosenbergii) Post-Larvae Reared in Aquaponic Systems and Fed Diets including Enriched Black Soldier Fly (Hermetia illucens) Prepupae Meal. Animals 2023, 13, 715. https://doi.org/10.3390/ani13040715
Zarantoniello M, Chemello G, Ratti S, Pulido-Rodríguez LF, Daniso E, Freddi L, Salinetti P, Nartea A, Bruni L, Parisi G, et al. Growth and Welfare Status of Giant Freshwater Prawn (Macrobrachium rosenbergii) Post-Larvae Reared in Aquaponic Systems and Fed Diets including Enriched Black Soldier Fly (Hermetia illucens) Prepupae Meal. Animals. 2023; 13(4):715. https://doi.org/10.3390/ani13040715
Chicago/Turabian StyleZarantoniello, Matteo, Giulia Chemello, Stefano Ratti, Lina Fernanda Pulido-Rodríguez, Enrico Daniso, Lorenzo Freddi, Pietro Salinetti, Ancuta Nartea, Leonardo Bruni, Giuliana Parisi, and et al. 2023. "Growth and Welfare Status of Giant Freshwater Prawn (Macrobrachium rosenbergii) Post-Larvae Reared in Aquaponic Systems and Fed Diets including Enriched Black Soldier Fly (Hermetia illucens) Prepupae Meal" Animals 13, no. 4: 715. https://doi.org/10.3390/ani13040715
APA StyleZarantoniello, M., Chemello, G., Ratti, S., Pulido-Rodríguez, L. F., Daniso, E., Freddi, L., Salinetti, P., Nartea, A., Bruni, L., Parisi, G., Riolo, P., & Olivotto, I. (2023). Growth and Welfare Status of Giant Freshwater Prawn (Macrobrachium rosenbergii) Post-Larvae Reared in Aquaponic Systems and Fed Diets including Enriched Black Soldier Fly (Hermetia illucens) Prepupae Meal. Animals, 13(4), 715. https://doi.org/10.3390/ani13040715






