1. Introduction
The setting of dietary protein levels is critical and directly affects the growth, health, and overall production performance of aquatic animals, which, in turn, determines whether or not aquaculture practices are profitable [
1,
2]. A balanced protein diet ensures efficient energy utilization and facilitates essential biochemical reactions. Conversely, insufficient protein levels may result in stunted growth, delayed molting, and reduced health [
3]. The optimal protein content in feed can vary based on the species, life stage, and environmental conditions [
4]. For instance, juvenile shrimp generally require greater protein intakes than adult shrimp [
5]. Appropriate protein levels in feed maximize its benefits, while inappropriate protein levels may cause indigestion. Additionally, low-quality proteins may result in nutrient waste and deficiencies, while excessive protein contents in feed can also deteriorate the quality of water due to the breakdown of surplus proteins, negatively impacting the overall health of the aquatic environment [
6]. However, because proteins often represent a substantial expense in aquafeed production, it is crucial to balance their levels with other nutrients for cost-effectiveness [
7]. Martinez-Cordova et al. conducted a cost analysis, revealing that employing a high protein feed during breeding was not the most optimal feeding strategy for
Litopenaeus vannamei [
8]. This demonstrated that feeding high protein levels is not optimal under certain circumstances.
L. vannamei has a significant economic importance in global aquaculture. Its prolific breeding yield and substantial economic returns have prompted extensive research into understanding and enhancing its production efficiency [
9,
10]. Variations in dietary protein levels can have multifaceted effects on
L. vannamei. Xu et al. demonstrated that alterations in protein content impacted the plasma and hepatopancreas total antioxidant capacity as well as decreased the glutathione/oxidized glutathione ratios in plasma, potentially disrupting both the antioxidant capacity and immune responses of the shrimp [
11]. In a study examining the influence of protein levels on trypsin activity, it was observed that changes in dietary protein content coupled with induced stress-affected trypsin activity in juvenile
L. vannamei [
12]. The dynamic interplay between the dietary protein content and environmental factors has also been investigated from various angles. Jang et al. investigated the impact of dietary protein levels on whole-body proximate amino acid composition and key waste nitrogen excretion parameters, including ammonia, nitrite, and nitrate nitrogen. The emission of these metabolic by-products into the environment induced environmental stress [
13]. In another study, Jatobá et al. reported that utilizing a feed with 30.3% crude protein in a biological flocculation system reduced the feed costs for shrimp, offering potential savings in raw feed materials [
14]. Limited studies have examined the impact of protein levels on
L. vannamei at the molecular level. The effects of supplementing juvenile shrimp diets with 484.6 g/kg of protein were examined by Xie et al. in 2020. They observed higher mRNA expression levels of the target of rapamycin protein (TOR) and eukaryotic translation initiation factor 4e-binding protein (4EBP), along with significant alterations in the expression of two pivotal translation initiation factors, eukaryotic translation initiation factor 4E (eif4e) and eukaryotic translation initiation factor 3k (eif3k). Additionally, three endoplasmic reticulum (ER) stress-associated genes (eif2α, xbp1, and ayf4) were found to be influenced by changes in protein levels [
5].
However, while previous studies on the effects of dietary protein levels on
L. vannamei focused on macro-indicators such as growth performance and digestive enzyme activity, there have been few studies on these effects at the microbial and molecular levels. With the increasing sophistication of sequencing technologies, microbial and transcriptome sequencing tools have been widely utilized in many studies for various purposes, including examining the diversity and composition of the intestinal microbiota in response to varying proteins in the diet [
15,
16]; identifying the specific genes and pathways that are differentially expressed in response to differential feeding, shedding light on the molecular mechanisms underlying growth or physiological changes [
17]; and, integrating data from both microbial sequencing and transcriptomics to gain a holistic view of the relationship between feed, intestinal microbiota, and host gene expression [
18].
The association between dietary nutrition and the intestinal microbial community’s structural composition of the host has garnered increasing research attention. The intestinal tract serves as a site for nutrient absorption and accommodates a significant microbial population. The composition of the microbial communities plays a pivotal role in shaping host health and preserving homeostasis [
19,
20]. Furthermore, the diversity of the intestinal microbiota facilitates the presence of latent pathogens while concurrently hosting beneficial bacteria whose metabolites can be absorbed, thereby influencing the internal environment of the host [
21]. Transcriptomics analysis is a potent diagnostic tool that is frequently employed in shrimp nutrition and immunity research, offering a comprehensive understanding of gene expression and its implications [
22]. Zhen et al. explored the alterations in the whole transcriptome of
L. vannamei induced by chronic imidacloprid exposure, providing valuable insights [
23]. In another study, the authors identified that differentially expressed genes (DEGs) related to detoxification were associated with the intestinal microbiota, which sheds light on the competitive dynamics among specific bacteria [
24]. Jia et al. utilized transcriptomic analysis to probe into the adverse effects of low fishmeal on the metabolic response of
L. vannamei [
25]. Thus, this technology empowers researchers to dissect the molecular mechanisms underlying the response of
L. vannamei to endogenous and exogenous environmental factors.
This study aimed to shed light on the impact of varying the protein feed level, using fishmeal as the sole protein source, on the structure of the intestinal microbiota and the transcriptome profile of L. vannamei. This provided a deeper understanding of the protein requirements of species, furnishing valuable insights to further advance feed research and establish a theoretical framework for the optimization of feed usage.
2. Materials and Methods
2.1. Experimental Diets
The feed formulations utilized are detailed in
Table 1, where fishmeal served as the exclusive protein source in five feeds denoted as P32, P36, P40, P44, and P48, representing protein levels of 32%, 36%, 40%, 44%, and 48%, respectively. Raw materials underwent crushing and screening through an 80-mesh screen. Subsequently, they were meticulously mixed using a step-by-step expanding method and homogenized in a V-type mixer (JS-14S, Zhejiang Chint Electrics Co., Ltd., Yueqing, China). Additional ingredients like fish oil, corn oil, and soybean lecithin were incorporated and thoroughly blended before extrusion through a twin-screw extruder (M-256, South China University of Technology, Guangzhou, China). The resulting feed particles were cooked for 20 min in a drying box (Shanghai Yiheng Scientific Instrument Co., Ltd., Shanghai, China, DHG-9240A) at 75 °C and stored in a refrigerator at 20 °C after being air-dried at natural temperatures.
2.2. Shrimp and Feeding Management
L. vannamei larvae were sourced from Zhanjiang Yuehai Aquatic Fry Co., Ltd. (Zhanjiang, China) consisting of 600 individuals with an initial weight averaging 0.63 ± 0.02 g. These were randomly allocated into five equal groups and fed with P32, P36, P40, P44, and P48 diets. Each subgroup comprised three biological replicates, with 40 individuals placed in separate 300-L fiberglass tanks. The stocking density of the experimental shrimp was about 133/m3, using indoor recycled water systems. Feeding occurred four times daily at 7:00, 11:00, 17:00, and 21:00. Initially, L. vannamei shrimp were fed several feeds equivalent to 10% of their body weight. Feeding was continued until satiety was reached, and the amount fed each day after that was determined using the amount consumed the day before. Implementing a daily water change of 50% while closely monitoring and regulating the temperature (by measuring the water temperature prior to each change and adjusting the percentage of water change accordingly, maintaining it within the range of 27–30 °C), ammonia nitrogen (≤0.05 mg/L), salinity (27–30 ppt), dissolved oxygen (>6.0 mg/L), and pH (7.7–8.0) were all carried out to guarantee ideal circumstances. Water quality was assessed after routine alterations. AR8210, PH828, and AR8212 from Dongguan SMART SENSOR Instrument Co., Ltd. (Dongguan, China), and the RB-103C Portable Ammonia Nitrogen Rapid Measuring Instrument from Guangzhou Ruibin Technology Co., Ltd. (Guangzhou, China), were used to measure the levels of dissolved oxygen salinity, ph and ammonia nitrogen.
2.3. Growth Performance Analysis
At the end of the feeding trial, the shrimp were counted and weighed to determine their growth performance indicators, including their survival rate (SR), final body weight (FW), weight gain rate (WGR), specific growth rate (SGR), feed conversion ratio (FCR), and protein efficiency ratio (PER). They were computed based on these parameters.
2.4. Sample Collection
The shrimp were sampled after 8 weeks of culture and were fasted for 24 h before sampling. Six randomly selected shrimp from each tank were sampled. Hemolymph was extracted from the pericardial cavity using a 1-mL sterile syringe, while the muscle, hepatopancreas, and intestinal tissues were carefully separated. Tissues from three shrimp were combined into a single sample and placed in separate 1.5-mL centrifuge tubes. One tube containing the hemolymph was centrifuged at 3000× g for 5 min at 4 °C, precipitating it. The supernatant was removed, and 1 mL of RNAlater™ (Thermo Fisher, Waltham, MA, USA) was added to resuspend the hemolymph, preserving it at −80 °C for subsequent transcriptome sequencing and gene expression analyses. The other tube containing tissue samples was promptly frozen in liquid nitrogen and stored at −80 °C for subsequent enzyme activity assays, body composition analysis, and intestinal microbiome sequencing.
2.5. Challenge Tests
White spot syndrome virus (WSSV) isolates and
Vibrio parahaemolyticus strains were provided by the Crustacean Laboratory of Guangdong Ocean University (Zhanjiang, China). Preparations of WSSV and
V. parahaemolyticus were carried out, and the challenge concentrations of them were determined according to our previous studies [
26,
27]. The WSSV isolates and
V. parahaemolyticus strains were provided by the Crustacean Laboratory of Guangdong Ocean University (Zhanjiang, China). After the completion of sample collection, 40 shrimp from a group were transferred to a 250-L experimental drum for one week to reach a steady state. After feeding was stopped for 24 h, they were then divided into two groups for the challenge tests with WSSV and
V. parahaemolyticus. For the challenge test by WSSV, a total of 20 shrimp were chosen at random from each group and subjected to an exposure of WSSV at a concentration of 1 × 10
5 copies/g shrimp. The shrimp mortality rate was closely observed and documented every 4 h for 120 h. For the challenge test by
V. parahaemolyticus, the remaining 20 shrimp were chosen at random from each group and subjected to an exposure of
V. parahaemolyticus at a concentration of 1 × 10
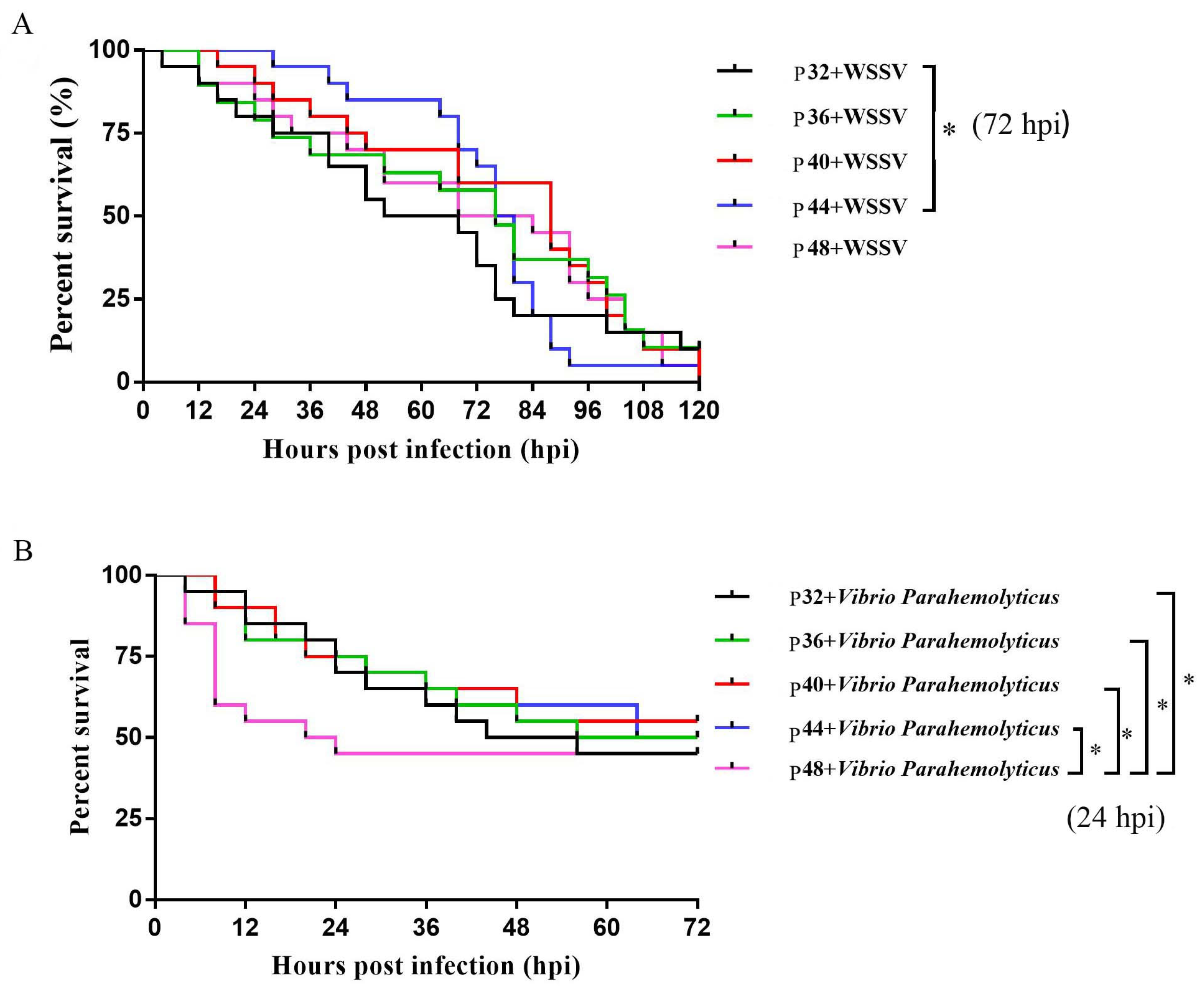
7 CFU/g shrimp. The cumulative mortality rate was recorded every 4 h for 72 h. The water quality conditions during the respite and testing periods remained consistent with those of the feeding trial experiment. No feed was provided during the experiments. The differences among groups were assessed using the Mantel–Cox (log-rank χ
2 test) method in GraphPad Prism 9.0.
2.6. Non-Specific Immune Indices
The stored hepatopancreas samples maintained at −80 °C were thawed, weighed, and ground, and then diluted with 0.90% saline at 4 °C in a ratio of 1:9. The resulting mixtures underwent centrifugation at 4000 rpm for 15 min at 4 °C, followed by the removal of the supernatant. Subsequently, the activities of lysozyme (LZM), superoxide dismutase (SOD), catalase (CAT), and phenoloxidase (PO), and the content of malondialdehyde (MDA) were determined with the help of a Multiskan Spectrum Microplate Spectrophotometer (Thermo, MultiskanGO1510) using the enzyme activity assay kits A050-1-1, A001-3-2, A007-2-1, H247, and A003-1-2, respectively. The enzyme activity assay kits were all brought from the Nanjing Jiancheng Institute of Bioengineering (Nanjing, China), and the experimental schemes were strictly following the manufacturer’s instructions.
2.7. Digestive Enzyme Activity
The intestinal samples stored in a refrigerator at a −80 °C were thawed and weighed. A total of 0.5 g of intestine were weighed, homogenized, and diluted with 0.9% saline at a ratio of 1:9 at 4 °C. The sample was then centrifuged at 4 °C (4000 rpm) for 15 min, and the supernatant was extracted to determine the amylase (AMS), trypsin, and lipase activities of the intestines using the enzyme activity assay kits C016-1-1, A080-2-2, and A054-2-1, respectively, with the help of a Multiskan Spectrum Microplate Spectrophotometer (Thermo, MultiskanGO1510). The enzyme activity determination kits were all obtained from the Nanjing Jiancheng Institute of Bioengineering, and the experimental scheme was strictly following the manufacturer’s instructions.
2.8. Intestinal Microbial Analysis
The genomic DNA extraction of microorganisms from the intestinal samples followed the manufacturer’s protocol using HiPure Soil DNA Kits (Magen, Guangzhou, China). The amplification of the V3–V4 region of the 16S rDNA gene was achieved using the primers 341F (CCTACGGGNGGCWGCAG) and 806R (GGACTACHVGGGTATCTAAT). The PCR program involved an initial denaturation step at 95 °C for 5 min, followed by 30 cycles at 95 °C for 1 min, 60 °C for 1 min, 72 °C for 1 min, and a final extension at 72 °C for 7 min. PCR reactions were conducted in triplicate 50-μL mixtures consisting of 10 μL of 5 × Q5@ Reaction Buffer, 10 μL of 5 × Q5@ High GC Enhancer, 1.5 μL of 2.5-mM dNTPs, 1.5 μL of each primer (10 μM), 0.2 μL of Q5@ High-Fidelity DNA Polymerase, and 50 ng of template DNA. PCR reagents were sourced from New England Biolabs, USA. Subsequently, amplicons were purified using the AxyPrep DNA gel extraction kit (Axygen Biosciences, Union City, CA, USA), pooled in equimolar concentrations, and sequenced via a Hiseq2500 PE250 machine (Illumina, San Diego, CA, USA) at Guangzhou Genedenovo Biotechnology Co., Ltd. (Guangzhou, China) The raw sequencing data were deposited in NCBI GenBank (
http://www.ncbi.nlm.nih.gov/genbank/, accessed on 12 December 2023).
FASTP [
15] was employed to filter the raw reads further and eliminate noise sequences under specific conditions to obtain high-quality, clean reads. This process generated high-quality clean tags. These clean tags underwent clustering into operational taxonomic units (OTUs) of ≥97% similarity using the UPARSE pipeline [
28]. The representative OTU sequences were classified using the RDP classifier based on the SILVA database [
29,
30], employing a confidence threshold of 0.8. Alpha diversity indexes, including OTUs, Chao1, ace, Shannon, Simpson, and Goods coverage, were computed using QIIME [
31,
32]. The sequencing information had the accession number PRJNA1061753 and could be found in the NCBI GenBank database.
2.9. Transcriptome Analysis
Total RNA extraction from the samples followed the Trizol method. As over 90% of RNA in typical species comprises rRNA, enriching mRNA was necessary by eliminating rRNA from the samples post-total RNA extraction using conventional kits. Oligo (dT) magnetic beads, followed by ultrasound-based blocking, achieved eukaryotic mRNA enrichment. The first cDNA strand was synthesized utilizing fragmented mRNA as a template and random oligonucleotides as primers within the M-MuLV reverse transcriptase system. Subsequently, the RNA strand was degraded using RNaseH, and the second cDNA strand was synthesized employing dNTPs in the DNA polymerase I system. The ru0bhaOAK2sulting double-stranded cDNA underwent purification, end-repair, A-tailing, and ligation to a sequence adapter. These constructed libraries were sequenced using Illumina HiSeqTM2000. These transcriptome data were assigned the accession number PRJNA1051837 and can be accessed in the NCBI GenBank database.
2.10. Statistical Analysis
All data were statistically validated using the one-way ANOVA with Tukey’s test for multiple comparisons. All statistical analyses were performed using GraphPad Prism 9.0. All data are expressed as the mean ± standard error (SEM), and p ≤ 0.05 indicates a significant difference.
4. Discussion
Growth performance is the most intuitive manifestation of an organism’s growth and development. Previous findings have investigated that
L. vannamei requires, at least, a 32% protein level for optimal growth, whereas a 48% protein level in feed can create better feed efficiencies [
33]. However, research by Gómez-Jiménez et al. and Xu et al. demonstrated no significant variations in the weight gain ratio (WGR) or digestibility of shrimp when protein levels in the feed ranged from 25% to 40% [
11,
34]. Given the considerable influence of environmental factors on nutritional requirements, the present study employed fish meal as the exclusive protein source to examine the effects of different protein levels on
L. vannamei. Shrimp fed with a 44% protein content displayed the best growth performance compared to other experimental groups, with a higher PER, WGR, and SGR and a lower FCR. Notably, this study indicated that while increasing the feed protein level positively impacted growth performance to a certain extent, reaching 40% seemed to represent a threshold beyond which there was no substantial improvement in the digestive or immune performances of the shrimp. This finding aligns with that of Jang et al. [
13].
Experiments were conducted under stress induced by V. parahaemolyticus and WSSV to assess the genuine impact of different protein levels on L. vannamei. The results showed that the P44 group was superior to the P32 group in terms of their WSSV stress response. Additionally, the P44 group showed a higher survival rate under V. parahaemolyticus infection than the P48 group. These findings further emphasized the importance of having the appropriate protein levels for enhancing immunity, with the shrimp being unable to maintain defense mechanism homeostasis in the face of a pathogenic infection when inappropriate protein levels were supplied.
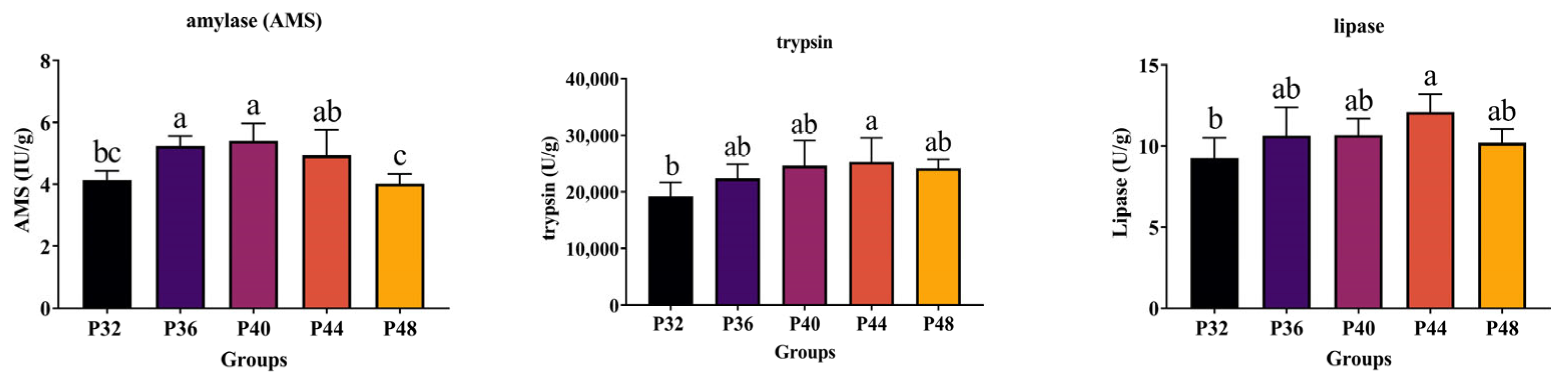
Talukdar et al. noted an increase in trypsin activity in
L. vannamei at higher dietary crude protein levels, which aligned with the present results [
35]. In this experiment, differences in digestive enzymes were observed in response to the dietary absorption of different protein levels, with an increase in digestive enzyme activity observed when higher protein levels were ingested. Similarly, Xia et al. found that the protease and amylase activities of the shrimp intestines changed significantly when their dietary protein levels were increased, and that protease activity was positively correlated with dietary protein levels [
36]. This was because as species grow, they consume more carbohydrates and lipids [
37].
Shrimp possess a comprehensive antioxidative stress system that is crucial for maintaining homeostasis and amplifies the antioxidant response in adverse environments [
38]. Inadequate protein levels can lead to malnutrition, sluggish growth, and increased vulnerability to bacterial disturbances. Conversely, excessive protein levels burden the hepatopancreas, inducing nutritional stress. Additionally, the release of surplus nutrients into the water can contribute to environmental pollution, and opportunistic pathogens within the body may damage vital organs such as the hepatopancreas [
39,
40]. LZM and PO are pivotal immune enzymes in the defense mechanism of
L. vannamei [
41,
42]. In the present study, the activities of LZM and PO were most pronounced in the P40 group. Nutritional imbalances can result in oxidative damage, whereas optimal dietary protein levels mitigate the extent of such damage [
5]. The content of MDA can often be indicative of the degree of lipid peroxidation and, by extension, the magnitude of cellular damage [
43]. CAT and SOD, with their similar functions in eliminating excessive reactive oxygen species (ROS) from aquaculture systems, also play vital roles [
44,
45]. In the present study, the P32 group exhibited the highest hepatopancreatic MDA levels, while the activities of SOD and CAT were most pronounced in the P44 group. These findings suggest that optimal dietary protein levels can significantly reduce the susceptibility of
L. vannamei to oxidative damage.
Utilizing 16S rDNA gene sequencing, the effects of three dietary protein levels (the P32, P40, and P48 groups) on the intestinal microbiome of
L. vannamei were explored. It is generally accepted that high diversity in the intestine benefits the host’s health [
46]. In this study, diets with different protein levels affected the abundance and diversity of intestinal microorganisms in the host, with inappropriate nutritional levels leading to an imbalance in the intestinal microbiota’s structure. Many previous studies have demonstrated that variations in dietary nutrient levels significantly affect the structure of the intestinal microbiota of shrimp. Liu et al. investigated the effects of two protein levels on silver pomfret larvae and observed significant effects on the structure of the intestinal microbiota [
47]. Fan et al. also observed that the composition of the intestinal microbiota was significantly affected after using cottonseed protein to de-substitute different levels of fish meal as a dietary protein [
48].
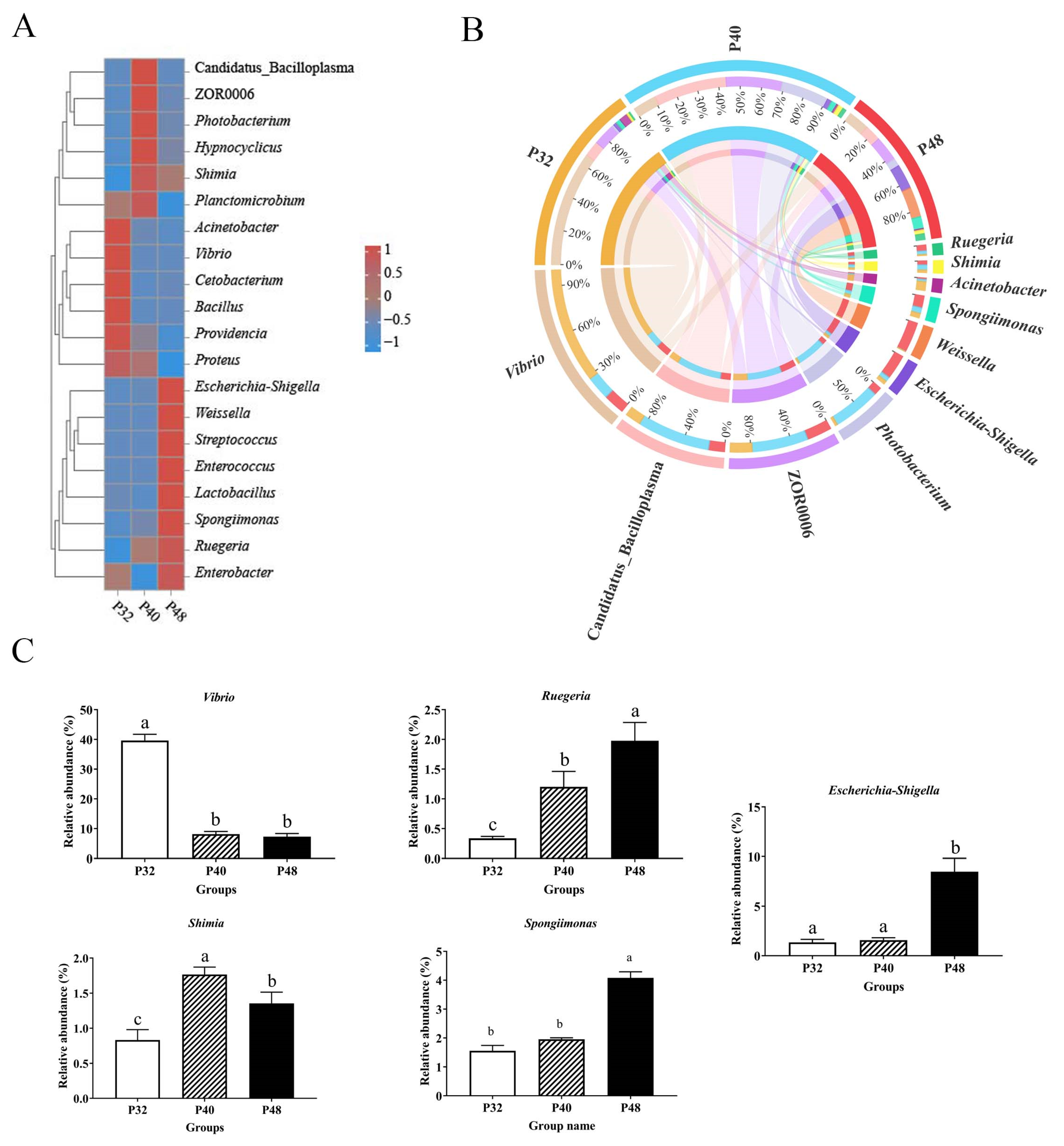
Firmicutes, Tenericutes, and Bacteroidetes are the most abundant phyla in prawns [
49]. Studies have found that higher numbers of Firmicutes/Bacteroidetes are linked to superior growth performance and nutrient uptake in shrimp [
50]. Proteobacteria are the largest bacterial phylum and the most extensively distributed in the marine environment, encompassing numerous harmful bacteria, and an increase in their abundance could lead to potential disease risks [
51]. In this study, the abundance of Firmicutes in the P40 group was, the abundance of Tenericutes/Bacteroidetes was high, and the abundance of Proteobacteria phyla was the lowest, which may have been mainly due to the change in the dietary protein level.
At the genus level, the typical genus of pathogenic bacteria,
Vibrio, contains several strains that can harm shrimp, such as
Vibrio harveyi and
V. parahaemolyticus. These can pose a significant threat to shrimp farming [
52,
53]. Additionally,
Acinetobacter has been recognized as a conditional pathogen in
L. vannamei [
54]. In the present study, the abundance of
Acinetobacter was significantly reduced when the protein level was increased, reducing the risk of disease in the shrimp. Furthermore, a significant boost in the abundance of two genera,
Ruegeria and
Shimia, was observed as the protein level increased. Most of the bacteria in these genera possess fundamental metabolic capabilities and partake in the host’s absorption and utilization of proteins [
55]. Additionally,
Ruegeria is antagonistic to some harmful bacteria and may be involved in the degradation of toxins and the production of beneficial metabolites [
56]. Thus, the findings of the present study highlight the relationship between dietary protein levels and the intestinal microbiome, showcasing how alterations in the diet can significantly impact the abundance and diversity of microbial communities and potentially influence the health and disease susceptibility of
L. vannamei shrimp.
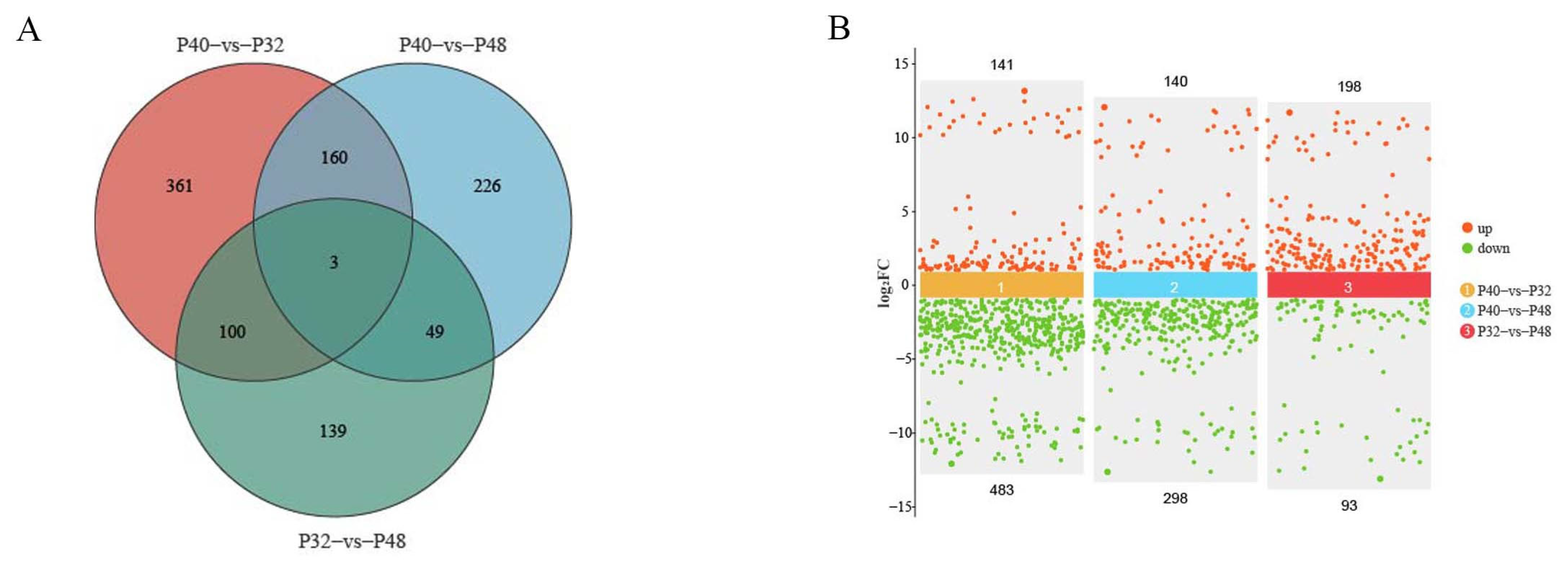
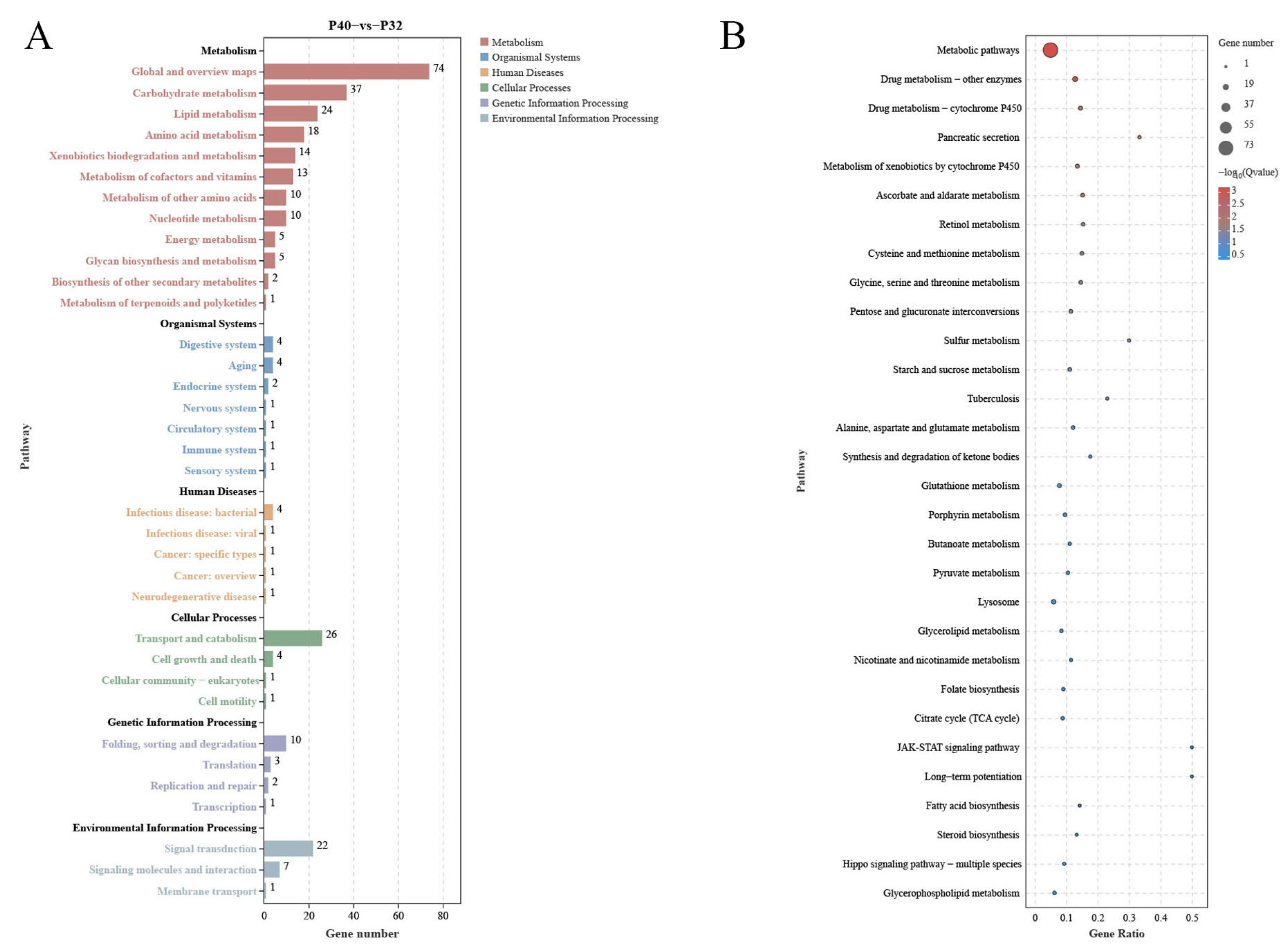
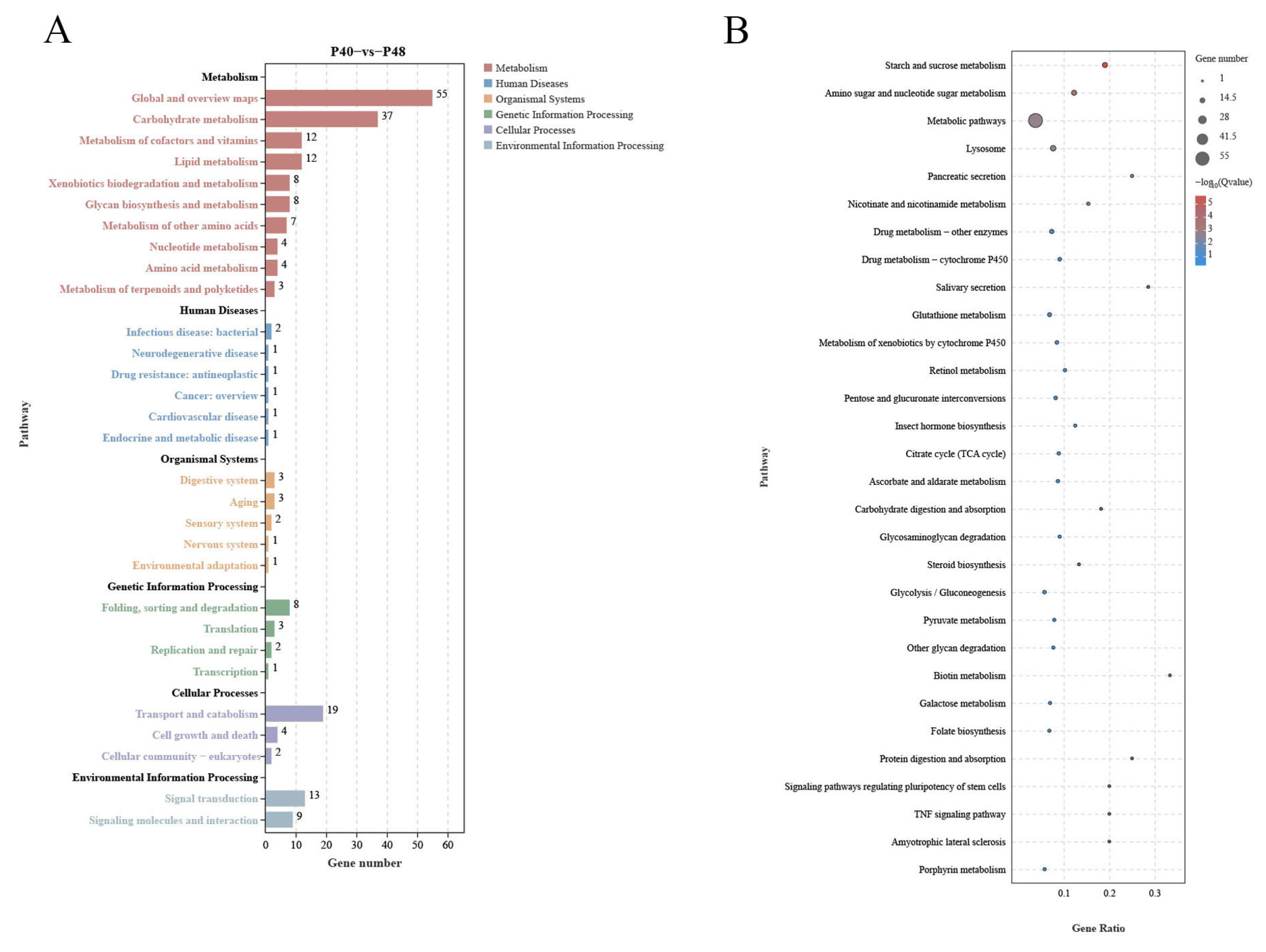
Comparative transcriptome analyses can provide comprehensive insights into the systemic gene expression and regulation mechanisms of organisms. In the present study, the changes in host DEGs induced by changes in dietary protein levels revealed a significant increase in down-regulated DEGs relative to the P40 control at the two other protein levels, which exceeded the number of up-regulated DEGs. This observation suggested that inappropriate dietary protein levels may suppress the expression of specific genes, potentially resulting in imbalances between crucial biological functions. Ensuring a stable level of energy metabolism is paramount for an organism’s survival, with carbohydrate metabolism as a vital means of energy acquisition. The predominant pathways annotated by KEGG across the three treatment groups were the Metabolic Pathways, indicative of the notable impact of dietary protein levels on this function. By comparing the levels of differential DEGs present and using the data annotated in the KEGG database, many DEGs were found in response to changes in protein levels; some affected nutrient absorption by the organism. The 28 co-occurring DEGs observed in this study were most highly expressed in the P40 group, demonstrating that this group had a greater activity of more metabolic functions and was the most active of all the groups. Metabolic Pathways involve three significant nutrients: sugars, lipids, and proteins, while some pathways are not directly related to nutrient metabolism, such as Drug Metabolism—Other Enzymes, Drug Metabolism—Cytochrome P450, and Glutathione Metabolism.
Glutathione Metabolism is commonly associated with performance in response to environmental stressors, which plays a vital role in maintaining physiological homeostasis in shrimp. Glutathione (GSH) is an important antioxidant that helps to protect cells from damage caused by ROS and other harmful molecules [
57]. In shrimp exposed to various environmental stressors, such as water quality, temperature, and salinity fluctuations, Glutathione Metabolism is essential for maintaining cellular health and protecting from oxidative stress [
58,
59]. The activity of this pathway in the P40 group and its relative inhibition in the P32 and P48 groups in the present study may be indicative of an enhancement in the antioxidant defense mechanism by the appropriate protein levels (P40 group). This meant that they could better neutralize ROS and other oxidative compounds, thereby reducing cellular damage and maintaining overall health. When Glutathione Metabolism in shrimp is optimal, shrimp may exhibit greater resistance to environmental stresses and diseases [
60]. In contrast, the inhibition of the glutathione pathway may lead to an increased susceptibility to oxidative stress, reducing the ability of shrimp to detoxify harmful compounds and resist oxidative damage, which may lead to impaired health, reduced immune function, and increased susceptibility to diseases or environmental stressors. Thus, maintaining a balance in Glutathione Metabolism is essential for shrimp health and adaptation under varying ecological conditions. Factors affecting this pathway can significantly influence the ability of shrimp to cope with stress and maintain overall health.
A pathway that is associated with immune factors, the Drug Metabolism—Cytochrome P450 pathway, is representative of the intricate biochemical processes that are responsible for the metabolism of a diverse array of compounds wherein cytochrome P450 enzymes play a pivotal role [
61]. Studies have shown that under environmental stressors, such as temperature fluctuations or exposure to pathogens, the expression and activity of cytochrome P450 enzymes may undergo alterations in organisms [
62,
63]. When the functionality of these enzymes is impaired, organisms may become more susceptible to environmental pollutants, rendering them more prone to the detrimental effects of pollution [
64]. In the present study, multiple DEGs within this pathway were observed to be down-regulated in both the P32 and P48 groups. This impairment in the in vivo homeostasis of this pathway could render organisms more susceptible to foreign pathogens. This may be one of the contributing factors to the lower survival rate observed in the P32 and P48 groups during the challenge tests, as well as the higher abundance of harmful bacteria in the intestinal environment.
Two extra Metabolic Pathways are worth noting: Lysosome and Pancreatic Secretion. The Lysosome pathway governs the acceptance and catabolism of macromolecules from the secretory, autophagic, and endocytic membrane transport pathways [
65,
66]. These processes are intricately involved in regulating various biological functions, including energy metabolism and cellular homeostasis. In the P32 and P48 groups, multiple DEGs related to this pathway were also significantly down-regulated. While invertebrates like shrimp lack a pancreas akin that of mammals, they possess a specialized digestive structure known as the hepatopancreas. Functionally, this organ is the liver and the pancreas, responsible for producing and secreting digestive enzymes into the stomach to facilitate food breakdown [
67]. Although the Pancreatic Secretion pathway defined in KEGG may not directly apply to invertebrates, particularly shrimp, KEGG annotations for these organisms emphasize specific physiological and biochemical processes that are unique to them. Notably, Jiang et al. observed that an elevated content of
Clostridium autoethanogenum protein adversely affected
L. vannamei by influencing pancreatic secretion [
68]. In our investigation, within the pancreatic secretory pathway, suppressed expression levels of
amy,
cpa1, and
cla2 in the P32 group and
amy and
cpa1 in the P48 group were observed compared to the P40 group. The genes
amy and
cpa1 are integral to the secretion of amylase and protease, respectively [
69,
70].
Cap1 encodes a member of the zinc metalloproteinase family of carboxypeptidases and is primarily synthesized in the pancreas, specializing in breaking C-terminal-branched chains and aromatic amino acids in dietary proteins [
71].
Amy is pivotal in starch and sucrose metabolism, metabolic pathways, salivary secretion, and pancreatic secretion, as well as the carbohydrate digestion and absorption pathways [
72]. Both enzymes are crucial for nutrient digestion and absorption, which may directly affect protein and starch absorption. Their down-regulation could trigger a series of malnutritional reactions, ultimately impacting the digestive function of the shrimp hepatopancreas; the observed reduction in digestive enzyme activity in the P32 and P48 groups may be attributed to this underlying mechanism. In the P32 group, inhibition of the gene
clca2 was observed, which also implicated the involvement of the Renin secretion pathway in vivo.
Clca2 encodes a member of the calcium-activated chloride channel regulatory protein (CLCR) family, known for its role in regulating the transport of chloride ions across the plasma membrane [
73,
74]. Additionally, it has been noted that
clca2 can moderately stimulate intracellular calcium pool release [
75]. However, the specific mechanisms behind this phenomenon remain to be elucidated. In summary, within the experimental framework of this study, which encompassed three different protein levels, it was observed that inappropriate protein levels may detrimentally impact
L. vannamei by disrupting its immune homeostasis.