Efficient Topical Treatment of Canine Nodular Sebaceous Hyperplasia with a Nitric Acid and Zinc Complex Solution
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Raghav, S.; Uppal, V. Gupta A Comparative Study on Distribution of Sebaceous and Sweat Glands in Skin of Different Domestic Animals. Indian J. Anim. Res. 2022, 56, 1356–1360. [Google Scholar] [CrossRef]
- Bongiovanni, L.; Suter, M.M.; Malatesta, D.; Ordinelli, A.; Ciccarelli, A.; Romanucci, M.; Brenner, O.; Della Salda, L. Nuclear survivin expression as a potentially useful tool for the diagnosis of canine cutaneous sebaceous lesions. Vet. Dermatol. 2012, 23, 394-e73. [Google Scholar] [CrossRef]
- Ginel, P.J.; Lucena, R.; Millán, Y.; González-Medina, S.; Guil, S.; García-Monterde, J.; de los Monteros, A.E.; de las Mulas, J.M. Expression of oestrogen and progesterone receptors in canine sebaceous gland tumours. Vet. Dermatol. 2010, 21, 297–302. [Google Scholar] [CrossRef]
- Goldschmidt, M.H.; Hendrick, M.J. Tumors of the skin and soft tissues. In Tumors in Domestic Animals, 4th ed.; Meuten, D.J., Ed.; Iowa State Press: Ames, IA, USA, 2002; pp. 45–117. [Google Scholar] [CrossRef]
- Scarampella, F.; Colombo, S.; Dehesa, A.; Godizzi, F.; Cavicchini, S.; Fabbri, E.; Roccabianca, P. Dermoscopic features of benign sebaceous proliferations in dogs: Description, assessment and inter-observer agreement. Vet. Dermatol. 2023, 34, 597–607. [Google Scholar] [CrossRef] [PubMed]
- Chikweto, A.; McNeil, P.; Bhaiyat, M.I.; Stone, D.; Sharma, R.N. Neoplastic and nonneoplastic cutaneous tumors of dogs in grenada, west indies. ISRN Vet. Sci. 2011, 2011, 416435. [Google Scholar] [CrossRef] [PubMed]
- Pakhrin, B.; Kang, M.S.; Bae, I.H.; Park, M.S.; Jee, H.; You, M.H.; Kim, J.H.; Yoon, B.I.; Choi, Y.K.; Kim, D.Y. Retrospective study of canine cutaneous tumors in Korea. J. Vet. Sci. 2007, 8, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Kaldrymidou, H.; Leontides, L.; Koutinas, A.F.; Saridomichelakis, M.N.; Karayannopoulou, M. Prevalence, distribution and factors associated with the presence and the potential for malignancy of cutaneous neoplasms in 174 dogs admitted to a clinic in northern Greece. J. Vet. Med. Ser. 2002, 49, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Rothwell, T.L.; Howlett, C.R.; Middleton, D.J.; Griffiths, D.A.; Duff, B.C. Skin neoplasms of dogs in Sydney. Aust. Vet. J. 1987, 64, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Baba, A.I.; Câtoi, C. Epithelial and melanocytic tumors of the skin. In Comparative Oncology; The Publishing House of the Romanian Academy: Bucharest, Romania, 2007. [Google Scholar]
- Sabattini, S.; Bassi, P.; Bettini, G. Histopathological findings and proliferative activity of canine sebaceous gland tumours with a predominant reserve cell population. J. Comp. Pathol. 2015, 152, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.H.; Griffin, C.E.; Campbell, K.L.; Muller, G.H. Muller and Kirk’s Small Animal Dermatology, 7th ed.; Elsevier Inc.: St. Louis, MO, USA, 2013. [Google Scholar]
- Yager, J.A.; Wilcock, B.P. Surgical Pathology of the Dog and Cat—Dermatopathology and Skin Tumors; Wolfe Publishing, Imprint of Mosby-Year Book Europe Limited: London, UK, 1994; Volume 1, pp. 262–264. [Google Scholar]
- Gross, T.L.; Ihrke, P.J.; Walder, E.J.; Affolter, V.K. Sebaceous tumors. In Skin Diseases of the Dog and Cat. Clinical and Histopathologic Diagnosis, 2nd ed.; Blackwell Publishing: Oxford, UK, 2005; pp. 641–644. [Google Scholar]
- Goldschmidt, M.H. Sebaceous and hepatoid gland neoplasms of dogs and cats. Am. J. Dermatopathol. 1984, 6, 287–293. [Google Scholar] [CrossRef]
- Bettini, G.; Morini, M.; Mandrioli, L.; Capitani, O.; Gandini, G. CNS and lung metastasis of sebaceous epithelioma in a dog. Vet. Dermatol. 2009, 20, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Misago, N.; Mihara, I.; Ansai, S.-I.; Narisawa, Y. Sebaceoma and Related Neoplasms With Sebaceous Differentiation. Am. J. Dermatopathol. 2002, 24, 294–304. [Google Scholar] [CrossRef] [PubMed]
- Withrow, S.J.; Vail, D.M.; Page, R.L. Withrow & MacEwen’s Small Animal Clinical Oncology, 5th ed.; Elsevier Saunders: St. Louis, MO, USA, 2013. [Google Scholar]
- Papadimitriou, I.; Vakirlis, E.; Sotiriou, E.; Bakirtzi, K.; Lallas, A.; Ioannides, D. Sebaceous Neoplasms. Diagnostics 2023, 9, 1676. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, R.M.; Young, K.M. Cutaneous mass aspirate from a Golden Retriever: “glandular guile”. Vet. Clin. Pathol. 2005, 34, 421–424. [Google Scholar] [CrossRef]
- Gautam, H.; Masand, A.; Asrani, R.K. Sebaceous carcinoma in a dog. Haryana Vet. 2017, 56, 111–112. [Google Scholar]
- Oztas, P.; Polat, M.; Oztas, M.; Alli, N.; Ustun, H. Bonbon toffee sign: A new dermatoscopic feature for sebaceous hyperplasia. J. Eur. Acad. Dermatol. Venereol. 2008, 22, 1200–1202. [Google Scholar] [CrossRef]
- Graf, R.; Pospischil, A.; Guscetti, F.; Meier, D.; Welle, M.; Dettwiler, M. Cutaneous Tumors in Swiss Dogs: Retrospective Data From the Swiss Canine Cancer Registry, 2008–2013. Vet. Pathol. 2018, 55, 809–820. [Google Scholar] [CrossRef]
- Vetscalpel. Removal of Canine Benign Cutaneous Growths with a Flexible Hollow Waveguide CO2 Laser. Available online: https://www.vetscalpel.com/wp-content/uploads/2018/02/72-01-2018_Duclos.pdf (accessed on 1 November 2023).
- Angileri, M.; Furlanello, T.; De Lucia, M. Cryotherapy to treat benign skin tumours in conscious dogs. Vet. Dermatol. 2020, 31, 163–166. [Google Scholar] [CrossRef]
- Salim, A.; Reece, S.M.; Smith, A.G.; Harrison, D.; Ramsay, H.M.; Harden, P.N.; Fryer, A.A. Sebaceous hyperplasia and skin cancer in patients undergoing renal transplant. J. Am. Acad. Dermatol. 2006, 55, 878–881. [Google Scholar] [CrossRef]
- Hussein, L.; Perrett, C.M. Treatment of sebaceous gland hyperplasia: A review of the literature. J. Dermatol. Treat. 2021, 32, 866–877. [Google Scholar] [CrossRef] [PubMed]
- McDonald, S.K.; Goh, M.S.; Chong, A.H. Successful treatment of cyclosporine-induced sebaceous hyperplasia with oral isotretinoin in two renal transplant recipients. Australas. J. Dermatol. 2011, 52, 227–230. [Google Scholar] [CrossRef]
- Shamloul, G.; Khachemoune, A. An updated review of the sebaceous gland and its role in health and diseases Part 2: Pathophysiological clinical disorders of sebaceous glands. Dermatol. Ther. 2021, 34, e14862. [Google Scholar] [CrossRef]
- Power, H.T.; Ihrke, P.J. Synthetic retinoids in veterinary dermatology. Vet. Clin. N. Am. Small Anim. Pr. 1990, 20, 1525–1539. [Google Scholar] [CrossRef] [PubMed]
- Viennet, C.; Gheit, T.; Muret, P.; Aubin, F.; Cabou, J.; Marchal, A.; Tommasino, M.; Humbert, P. Assessment of the efficacy of a new formulation for plantar wart mummification: New experimental design and human papillomavirus identification. Clin. Exp. Dermatol. 2013, 38, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Cusini, M.; Micali, G.; Lacarrubba, F.; Puviani, M.; Barcella, A.; Milani, M. Efficacy and tolerability of nitric-zinc complex in the treatment of external genital warts and “difficult-to-treat” warts: A “proof of concept”, prospective, multicentre, open study. G Ital. Dermatol. Venereol. 2015, 150, 643–648. [Google Scholar] [PubMed]
- Verrutop—Topical Treatment of Difficult Warts. Isdin. Available online: https://www.isdin.com/en/verrutop-difficult-warts-treatment/ (accessed on 24 January 2024).
- Pontini, P.; Mastorino, L.; Gaspari, V.; Granger, C.; Ramoni, S.; Delmonte, S.; Evangelista, V.; Cusini, M. A Multicentre, Randomised Clinical Trial to Compare a Topical Nitrizinc® Complex Solution Versus Cryotherapy for the Treatment of Anogenital Warts. Dermatol. Ther. 2020, 10, 1063–1073. [Google Scholar] [CrossRef] [PubMed]
- García-Oreja, S.; Álvaro-Afonso, F.J.; García-Madrid, M.; López-Moral, M.; García-Álvarez, Y.; Lázaro-Martínez, J.L. Cryotherapy versus topical nitric-zinc complex solution for the treatment of plantar warts: A randomized controlled trial. J. Med. Virol. 2023, 96, e29212. [Google Scholar] [CrossRef] [PubMed]
- Giacaman, A.; Granger, C.; Aladren, S.; Bauzá, A.; Alomar Torrens, B.; Riutort Mercant, M.; Martin-Santiago, A. Use of Topical Nitric-Zinc Complex Solution to Treat Palmoplantar and Periungual Warts in a Pediatric Population. Dermatol. Ther. 2019, 9, 755–760. [Google Scholar] [CrossRef]
- Rogsmo Spiro, E. Utvärdering av Topikal Behandling med Oxalic vid Benigna Talgkörtelnoduler hos Hund. Master’s Thesis, Swedish University of Acricultural Scienes, Uppsala, Sweden, 2015; p. 23. Available online: https://stud.epsilon.slu.se/9592/1/rogsmo_spiro_e_%20160928.pdf (accessed on 28 February 2023).
- Ngo, J.; Beco, L.; Calinot, C.; Floran, E.; Jamet, J.-F.; Muller AFontaine, J. A pilot uncontrolled open study on the use of Oxalic® (Medeor International) for treatment of sebaceous gland adenoma/hyperplasia in dogs. Abstracts of the 26th Annual Congress of the ECVD-ESVD, 19–21 September 2013, Valencia, Spain. Vet. Dermatol. 2013, 24, 377–397. [Google Scholar] [CrossRef]
- Ghisleni, G.; Roccabianca, P.; Ceruti, R.; Stefanello, D.; Bertazzolo, W.; Bonfanti, U.; Caniatti, M. Correlation between fine-needle aspiration cytology and histopathology in the evaluation of cutaneous and subcutaneous masses from dogs and cats. Vet. Clin. Pathol. 2006, 35, 24–30. [Google Scholar] [CrossRef]
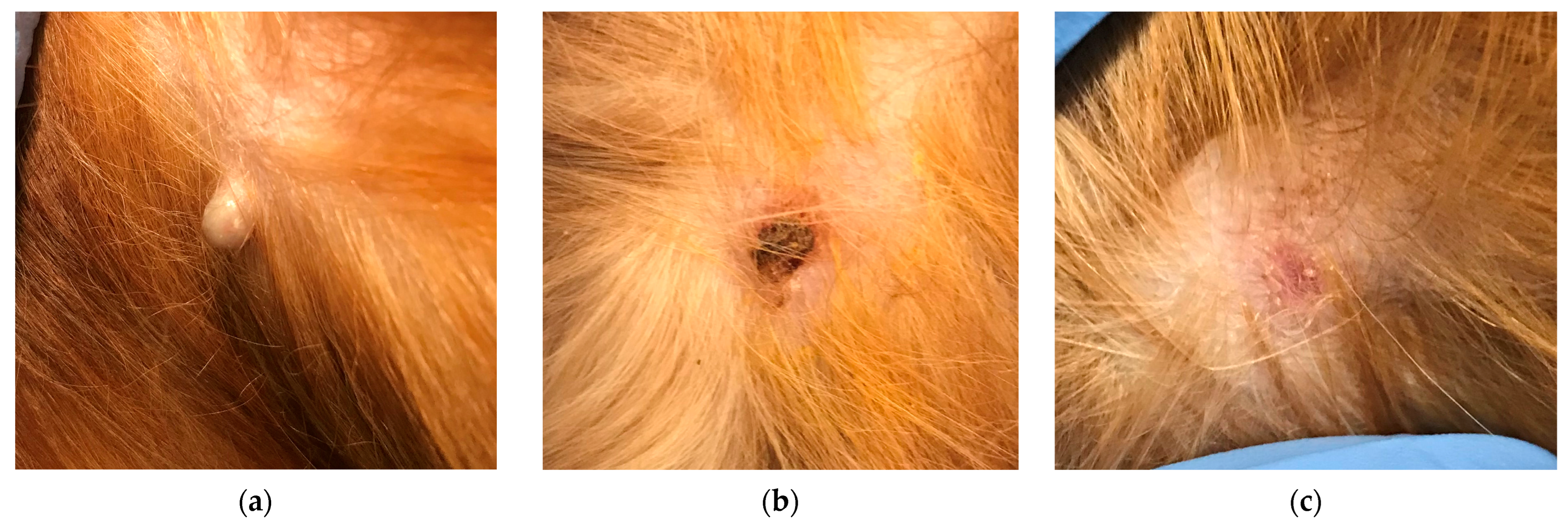

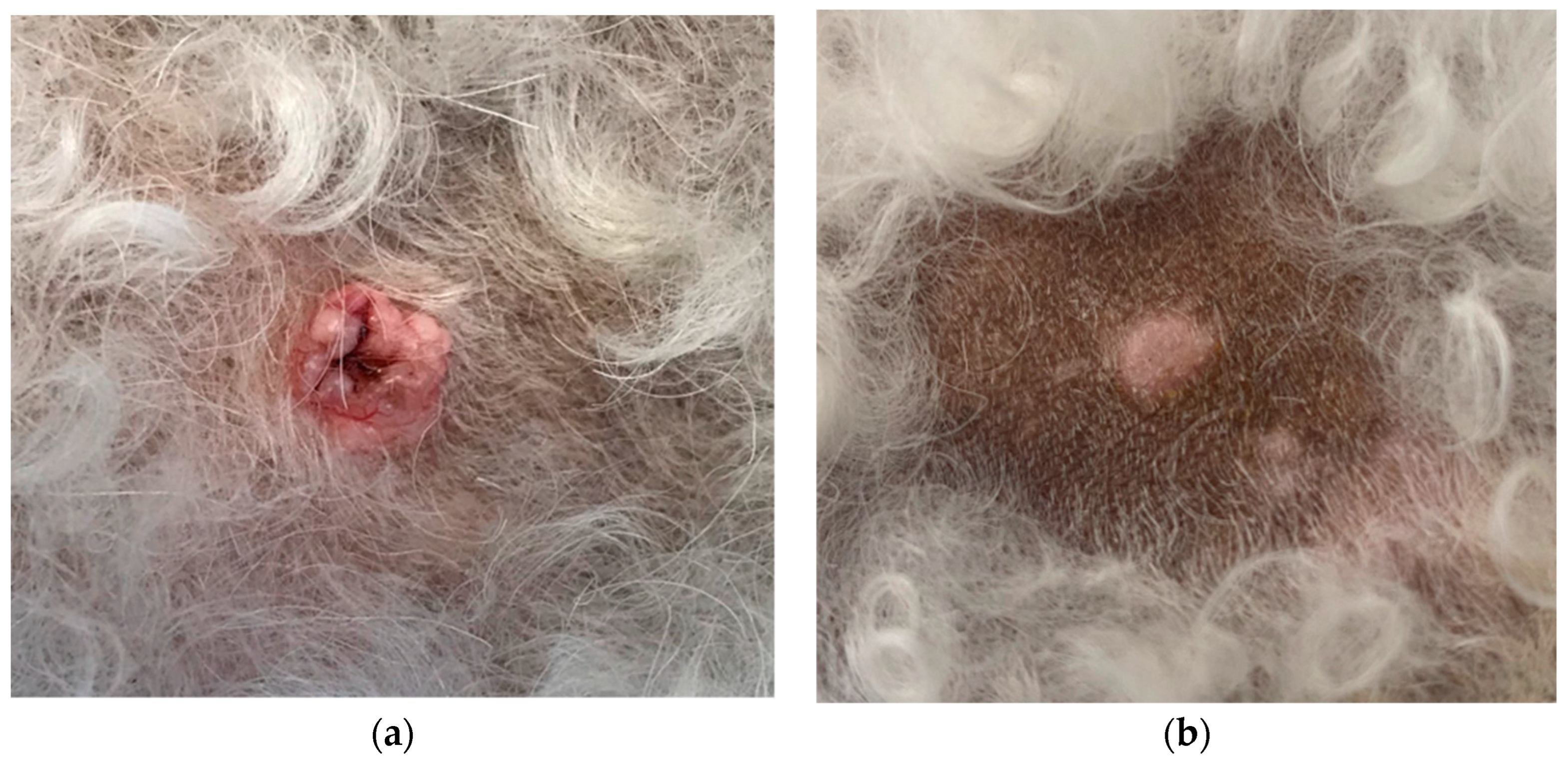

| Breed | Sex | Age | Weight | Concurrent Disease | Systemic Treatment | Treated (n = 18) | In Remission at Day 28 Total: 17/18 | Untreated (n = 11) | In Remission at Day 28 Total: 0/11 |
|---|---|---|---|---|---|---|---|---|---|
| Bichon frisé | NM | 7 y | 9.3 kg | None | None | 2 | 1/2 | 2 | 0/2 |
| Miniature bullterrier | NM | 10 y | 20.2 kg | cAD; pododermatitis; overweight | Methylprednisolone (0.2 mg/kg) every other day | 1 | 1/1 | 0 | NA |
| Bichon havanais | F | 8 y | 6.9 kg | cAD; mammary tumor | Lokivetmab (1.4 mg/kg) in irregular intervals; subcutaneous allergen specific immunotherapy | 2 | 2/2 | 2 | 0/2 |
| Border collie | NM | 4 y | 19.5 kg | None | None | 2 | 2/2 | 2 | 0/2 |
| Australian shepherd | NM | 8 y | 26.2 kg | Idiopathic epilepsy | Potassium bromide (31 mg/kg) q 24 h; phenobarbital (3.8 mg/kg) q 12 h | 1 | 1/1 | 0 | NA |
| Cavalier King Charles Spaniel | F | 9 y | 14.5 kg | cAD, food allergy; osteoarthritis | Lokivetmab (2.1 mg/kg) every 4–5 months; bedinvetmab (0.69 mg/kg) monthly; firocoxib (5.9 mg/kg) daily | 1 | 1/1 | 0 | NA |
| Mixed breed | NM | 9 y | 9.4 kg | cAD | Subcutaneous allergen specific immunotherapy | 2 | 2/2 | 1 | 0/1 |
| Mixed breed | F | 10 y | 7.9 kg | cAD; food allergy | Oclacitinib (0.45 mg/kg) q 24 h | 1 | 1/1 | 0 | NA |
| Miniature poodle | M | 14 y | 6 kg | Hypothyroid, chronic GI disease; tracheal collapse; MMVD | Levothyroxine (dose unknown) | 1 | 1/1 | 1 | 0/1 |
| Mixed breed | SF | 11 y | 19 kg | Transient lameness and neck pain | Robenacoxib (1 mg/kg), stopped 3 days before inclusion | 1 | 1/1 | 1 | 0/1 |
| Mixed breed | M | 12 y | 24.3 kg | Pyoderma | Amoxicillin (8 mg/kg) q 12 h; cetirizine (2 mg/kg) q 24 h | 4 | 4/4 | 2 | 0/2 |
| Volume Change | −100 | −83 | −68 | −56 | 0 | +7 | +50 | Total |
|---|---|---|---|---|---|---|---|---|
| Treated | 17 | 1 | 18 | |||||
| Untreated | 1 | 1 | 6 | 1 | 2 | 11 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gustafsson, L.; Wilson, A.; Bergvall, K. Efficient Topical Treatment of Canine Nodular Sebaceous Hyperplasia with a Nitric Acid and Zinc Complex Solution. Animals 2024, 14, 570. https://doi.org/10.3390/ani14040570
Gustafsson L, Wilson A, Bergvall K. Efficient Topical Treatment of Canine Nodular Sebaceous Hyperplasia with a Nitric Acid and Zinc Complex Solution. Animals. 2024; 14(4):570. https://doi.org/10.3390/ani14040570
Chicago/Turabian StyleGustafsson, Lina, Alison Wilson, and Kerstin Bergvall. 2024. "Efficient Topical Treatment of Canine Nodular Sebaceous Hyperplasia with a Nitric Acid and Zinc Complex Solution" Animals 14, no. 4: 570. https://doi.org/10.3390/ani14040570






